These Highlights Do Not Include All The Information Needed To Use Korsuva Safely And Effectively. See Full Prescribing Information For Korsuva.
0c7b81f2-0fd3-47cb-8b0f-185e07f19c87
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
KORSUVA is indicated for the treatment of moderate-to-severe pruritus associated with chronic kidney disease (CKD-aP) in adults undergoing hemodialysis (HD).
Indications and Usage
KORSUVA is indicated for the treatment of moderate-to-severe pruritus associated with chronic kidney disease (CKD-aP) in adults undergoing hemodialysis (HD).
Dosage and Administration
Recommended dosage is 0.5 mcg/kg. ( 2.1 ) Administer by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD treatment. ( 2.1 ) Do not mix or dilute KORSUVA prior to administration. ( 2.2 ) Administer within 4 hours of syringe preparation. ( 2.3 ) See full prescribing information for additional recommendations on preparation and administration of KORSUVA. ( 2.2 , 2.3 )
Warnings and Precautions
Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances: Dizziness, somnolence, mental status changes, and gait disturbances, including falls, have occurred. Centrally-acting depressant medications, sedating antihistamines, and opioid analgesics should be used with caution during treatment with KORSUVA. ( 5.1 ) Risk of Driving and Operating Machinery: May impair mental or physical abilities. Advise patients not to drive or operate dangerous machinery until the effect of KORSUVA on a patient's ability to drive or operate machinery is known. ( 5.2 )
Contraindications
None
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances [see Warnings and Precautions (5.1) ]
Medication Information
Warnings and Precautions
Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances: Dizziness, somnolence, mental status changes, and gait disturbances, including falls, have occurred. Centrally-acting depressant medications, sedating antihistamines, and opioid analgesics should be used with caution during treatment with KORSUVA. ( 5.1 ) Risk of Driving and Operating Machinery: May impair mental or physical abilities. Advise patients not to drive or operate dangerous machinery until the effect of KORSUVA on a patient's ability to drive or operate machinery is known. ( 5.2 )
Indications and Usage
KORSUVA is indicated for the treatment of moderate-to-severe pruritus associated with chronic kidney disease (CKD-aP) in adults undergoing hemodialysis (HD).
Dosage and Administration
Recommended dosage is 0.5 mcg/kg. ( 2.1 ) Administer by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD treatment. ( 2.1 ) Do not mix or dilute KORSUVA prior to administration. ( 2.2 ) Administer within 4 hours of syringe preparation. ( 2.3 ) See full prescribing information for additional recommendations on preparation and administration of KORSUVA. ( 2.2 , 2.3 )
Contraindications
None
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances [see Warnings and Precautions (5.1) ]
Description
KORSUVA is indicated for the treatment of moderate-to-severe pruritus associated with chronic kidney disease (CKD-aP) in adults undergoing hemodialysis (HD).
Section 42229-5
Limitations of Use
KORSUVA has not been studied in patients on peritoneal dialysis and is not recommended for use in this population.
Section 44425-7
Storage and Handling
Store vials at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Do not freeze.
KORSUVA injection must be administered within 4 hours of syringe preparation; prepared syringes can be stored at ambient temperature 20°C to 25°C (68°F to 77°F) until dosing [see Dosage and Administration (2.2, 2.3)] .
KORSUVA injection is supplied in a single-dose vial. Any unused drug remaining after injection must be discarded.
2.1 Dosage
- The recommended dosage of KORSUVA is 0.5 mcg/kg administered by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD treatment [see Dosage and Administration (2.3)].
- If a regularly scheduled HD treatment is missed, resume KORSUVA at the end of the next HD treatment.
10 Overdosage
Single doses of KORSUVA up to 12 times and multiple doses of KORSUVA up to 5 times the recommended dosage of 0.5 mcg/kg were administered in clinical studies in subjects undergoing HD. A dose-dependent increase in adverse reactions, including dizziness, somnolence, mental status changes, paresthesia, fatigue, hypertension, and vomiting, were observed.
In the event of overdosage, provide the appropriate medical attention based on patient's clinical status. Difelikefalin is primarily eliminated by the kidneys with a low plasma protein binding of approximately 23% to 28% in dialysis patients. Hemodialysis for 4 hours using a high-flux dialyzer effectively cleared approximately 70% to 80% of difelikefalin from plasma, and difelikefalin was not detectable in plasma at the end of the second of two dialysis cycles. [see Clinical Pharmacology (12.3)].
11 Description
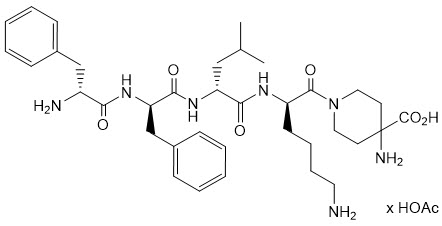
KORSUVA (difelikefalin) is a kappa opioid receptor agonist. Difelikefalin is a synthetic peptide with a single stereoisomer and is present as an acetate salt. Difelikefalin acetate is a white to off-white powder with a molecular formula of C36H53N7O6∙xAcOH (1.0≤ × ≤2.0) and a molecular weight of 679.4 g/mol (mono- isotopic; free base). It is soluble in water. The chemical name of difelikefalin acetate is 4-amino-1-(D-phenylalanyl-D-phenylalanyl-D-leucyl-D-lysyl)piperidine-4-carboxylic acid, acetate salt.
The chemical structure is:
KORSUVA (difelikefalin) injection is supplied in a single-dose vial containing 65 mcg/1.3 mL (50 mcg/mL) of difelikefalin as a sterile, preservative-free, clear and colorless solution for intravenous injection.
KORSUVA is formulated as an isotonic 40 mM acetate buffer solution with an osmolality of 250 to 350 mOsm and a pH of 4.5.
Each milliliter of KORSUVA injection contains 50 mcg of difelikefalin (equivalent to an average of 58.3 mcg of difelikefalin acetate), 1.3 mg of acetic acid, 2.5 mg of sodium acetate trihydrate, 7.2 mg of sodium chloride (to adjust tonicity), and water for injection.
8.4 Pediatric Use
The safety and effectiveness of KORSUVA in pediatric patients have not been established.
8.5 Geriatric Use
Of the 848 subjects in the placebo-controlled studies who received KORSUVA, 278 subjects (32.8%) were 65 years of age and older and 98 subjects (11.6%) were 75 years of age and older. No overall differences in safety or effectiveness of KORSUVA have been observed between patients 65 years of age and older and younger adult subjects, with the exception of the incidence of somnolence which was higher in KORSUVA-treated subjects 65 years of age and older (7.0%) than in KORSUVA-treated subjects less than 65 years of age (2.8%), and was comparable in both placebo age groups (3.0% and 2.1%, respectively). No differences in plasma concentrations of KORSUVA were observed between subjects 65 years of age and older and younger adult subjects [see Clinical Pharmacology (12.3)].
14 Clinical Studies
The efficacy of KORSUVA was evaluated in two randomized, multicenter, double-blind, placebo-controlled trials (Trial 1 [NCT03422653] and Trial 2 [NCT03636269]) that enrolled a total of 851 subjects 18 years of age and older undergoing HD who had moderate-to-severe pruritus. In both trials, subjects received intravenous bolus injections of KORSUVA 0.5 mcg per kilogram of dry body weight into the venous line of the hemodialysis circuit at the end of each hemodialysis session or placebo three times per week for 12 weeks. In both trials, a 7-day run-in period prior to randomization was used to confirm that each subject had moderate-to-severe pruritus and to establish a baseline itch intensity, as measured by the patient-reported daily 24-hour Worst Itching Intensity Numerical Rating Scale (WI-NRS) scores (0 "no itch" to 10 "worst itch imaginable").
The mean (SD) baseline WI-NRS score was 7.1 (1.5) in Trial 1 and 7.2 (1.4) in Trial 2. At baseline in Trial 1, 61% of subjects were male, 49% were White, 42% were Black or African American, the mean age was 57 years (range 22 to 88 years), and 40% of subjects were using prior anti-pruritic medications (including sedating antihistamines) and continued the use throughout the trial. At baseline in Trial 2, 58% of subjects were male, 70% were White, 19% were Black or African American, the mean age was 60 years (range 23 to 90 years), and 36% of subjects were using prior anti-pruritic medications (including sedating antihistamines) and continued the use throughout the trial.
In each trial, efficacy was assessed based on the proportion of subjects achieving a 4-point or greater improvement (reduction) from baseline in the weekly mean of the daily 24-hour WI-NRS score at Week 12.
The results of the KORSUVA trials (Trials 1 and 2) are presented in Table 3 and Figure 1.
| Trial 1 | Trial 2 | |||
|---|---|---|---|---|
| KORSUVA 0.5 mcg/kg 3 times weekly N=189 |
Placebo N=189 |
KORSUVA 0.5 mcg/kg 3 times weekly N=237 |
Placebo N=236 |
|
| Percentage of subjects with ≥4-point improvement from baseline in WI-NRS score | 40% | 21% | 37% | 26% |
| Difference from Placebo (95% CI) | 19% (9%, 28%) | 12% (3%, 20%) |
Figure 1: Percentage of Subjects with Moderate-to-Severe CKD-aP Undergoing HD with a ≥4-point Improvement from Baseline on the WI-NRS in Trial 1 and Trial 2
Itch reduction was seen by Week 4 and sustained through Week 12.
4 Contraindications
None
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances [see Warnings and Precautions (5.1)]
12.2 Pharmacodynamics
Difelikefalin exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
The pharmacokinetics of difelikefalin is dose proportional over a single dosage range from 1 to 3 mcg/kg (2 to 6 times the recommended dosage) and multiple intravenous dosage range from 0.5 to 2.5 mcg/kg (1 to 5 times the recommended dosage) in chronic kidney disease patients undergoing HD. Steady-state was reached after the second administered dosage and the mean accumulation ratio was up to 1.6.
8.6 Hepatic Impairment
The influence of mild-to-moderate hepatic impairment on the pharmacokinetics of KORSUVA was evaluated in a population pharmacokinetic analysis which concluded that no KORSUVA dosage adjustments are needed in these populations [see Clinical Pharmacology (12.3)]. The influence of severe hepatic impairment on the pharmacokinetics of KORSUVA in subjects undergoing HD has not been evaluated; therefore, use of KORSUVA in this population is not recommended.
1 Indications and Usage
KORSUVA is indicated for the treatment of moderate-to-severe pruritus associated with chronic kidney disease (CKD-aP) in adults undergoing hemodialysis (HD).
12.1 Mechanism of Action
KORSUVA is a kappa opioid receptor (KOR) agonist. The relevance of KOR activation to therapeutic effectiveness is not known.
5 Warnings and Precautions
- Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances: Dizziness, somnolence, mental status changes, and gait disturbances, including falls, have occurred. Centrally-acting depressant medications, sedating antihistamines, and opioid analgesics should be used with caution during treatment with KORSUVA. (5.1)
- Risk of Driving and Operating Machinery: May impair mental or physical abilities. Advise patients not to drive or operate dangerous machinery until the effect of KORSUVA on a patient's ability to drive or operate machinery is known. (5.2)
2 Dosage and Administration
- Recommended dosage is 0.5 mcg/kg. (2.1)
- Administer by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD treatment. (2.1)
- Do not mix or dilute KORSUVA prior to administration. (2.2)
- Administer within 4 hours of syringe preparation. (2.3)
- See full prescribing information for additional recommendations on preparation and administration of KORSUVA. (2.2, 2.3)
2.2 Preparation Instructions
- Do not mix or dilute KORSUVA prior to administration.
- Inspect KORSUVA for particulate matter and discoloration prior to administration. The solution should be clear and colorless. Do not use KORSUVA vials if particulate matter or discoloration is observed.
- KORSUVA is supplied in a single-dose vial. Discard any unused product.
- Injection volume to be administered is determined by patient's target dry body weight in kilograms (one patient may use less than the full contents of the vial or use more than one vial). See Table 1.
| Target Dry Body Weight Range (kg) | Injection Volume (mL) Total Injection Volume (mL) = Patient Target Dry Body Weight (kg) x 0.01, rounded to the nearest tenth (0.1 mL). For patient target dry body weight outside of the ranges in Table 1, use this formula.
|
|---|---|
| 36 – 44 | 0.4 |
| 45 – 54 | 0.5 |
| 55 – 64 | 0.6 |
| 65 – 74 | 0.7 |
| 75 – 84 | 0.8 |
| 85 – 94 | 0.9 |
| 95 – 104 | 1 |
| 105 – 114 | 1.1 |
| 115 – 124 | 1.2 |
| 125 – 134 | 1.3 |
| 135 – 144 | 1.4 |
| 145 – 154 | 1.5 |
| 155 – 164 | 1.6 |
| 165 – 174 | 1.7 |
| 175 – 184 | 1.8 |
| 185 – 194 | 1.9 |
| 195 – 204 | 2 |
3 Dosage Forms and Strengths
Injection: 65 mcg/1.3 mL (50 mcg/mL) of difelikefalin as a clear, colorless solution in a single-dose glass vial.
8 Use in Specific Populations
Severe Hepatic Impairment: Not recommended in patients with severe hepatic impairment. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 1306 subjects undergoing HD who had moderate-to-severe pruritus were treated with KORSUVA in placebo-controlled and uncontrolled Phase 3 clinical trials. Of these, 711 were treated for at least 6 months and 400 were treated for at least one year.
Two placebo-controlled Phase 3 trials (Trial 1 and Trial 2), in subjects undergoing HD who had moderate-to-severe pruritus were pooled to evaluate the safety of KORSUVA in comparison to placebo up to 12 weeks. In total, 848 subjects were evaluated (424 in KORSUVA group and 424 in placebo group). The mean age of the subjects was 59 years (range 22 to 88 years), and 59% of the subjects were male. Of the total subjects, 61% were White, 29% were Black or African American, and 5% were Asian.
Table 2 summarizes the adverse reactions that occurred at a rate of ≥2% in the KORSUVA group and ≥1% higher than that of the placebo group during the 12-week placebo-controlled period of Trials 1 and 2. The percentage of subjects who discontinued treatment due to any adverse reaction was 2.6% for subjects taking KORSUVA and 0.7% for subjects taking placebo. The most common adverse reactions (≥0.5% of subjects) leading to discontinuation were dizziness (0.9% for KORSUVA and 0.2% for placebo), mental status change (0.7% and 0.2%, respectively), nausea (0.5% and 0%, respectively), and headache (0.5% and 0%, respectively). The percentage of subjects who developed serious adverse reactions was 4.5% in the KORSUVA group and 2.8% in the placebo group.
| Adverse Reactions | Placebo (N=424) n (%) |
KORSUVA (N=424) n (%) |
|---|---|---|
| Diarrhea | 24 (5.7) | 38 (9.0) |
| Dizziness | 16 (3.8) | 29 (6.8) |
| Nausea | 19 (4.5) | 28 (6.6) |
| Gait Disturbances Gait disturbances includes: preferred terms of falls and gait disturbances
|
23 (5.4) | 28 (6.6) |
| Hyperkalemia | 15 (3.5) | 20 (4.7) |
| Headache | 11 (2.6) | 19 (4.5) |
| Somnolence | 10 (2.4) | 18 (4.2) |
| Mental Status Change Mental Status Change includes: preferred terms of confusional state and mental status change.
|
6 (1.4) | 14 (3.3) |
2.3 Administration Instructions
- KORSUVA is removed by the dialyzer membrane and must be administered after blood is no longer circulating through the dialyzer.
- Administer KORSUVA by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD session.
- The dose may be given either during or after rinse back of the dialysis circuit.
- If the dose is given after rinse back, administer KORSUVA into the venous line followed by at least 10 mL of normal saline flush.
- If the dose is given during rinse back, no additional normal saline is needed to flush the line.
- The dose must be administered within 4 hours of the syringe preparation. Discard any unused product.
5.2 Risk of Driving and Operating Machinery
Dizziness, somnolence, and mental status changes have occurred in patients taking KORSUVA. KORSUVA may impair the mental or physical abilities needed to perform potentially hazardous activities such as driving a car and operating machinery. Advise patients not to drive or operate dangerous machinery until the effect of KORSUVA on a patient's ability to drive or operate machinery is known.
Principal Display Panel 1.3 Ml Vial Label
NDC 59353-065-01
Store at 20°C to 25°C (68°F to 77°F)
Rx only
KORSUVA™
(difelikefalin) Injection
65 mcg/1.3 mL
(50 mcg/mL)
For Intravenous Use Only
1.3 mL Single-dose Vial. Discard unused portion
Mfd. for Vifor (International) Inc., Rechenstrasse 37,
9014 St. Gallen, Switzerland. Made in France.
3102942-01 48307/16/25
Principal Display Panel 1.3 Ml Tray Carton
NDC 59353-065-12
Rx only
KORSUVA™
(difelikefalin) Injection
65 mcg/1.3 mL
(50 mcg/mL)
For Intravenous Use after Hemodialysis
Single-dose vial. Discard unused portion.
Store at 20°C to 25°C (68°F to 77°F), excursions
permitted to 15°C to 30°C (59°F to 86°F) [see USP
Controlled Room Temperature]. Do not freeze.
Sterile, preservative-free solution.
12 x 1.3 mL
Single-dose Vials
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year carcinogenicity study in rats, difelikefalin was not carcinogenic when administered via subcutaneous injection at doses up to 1.0 mg/kg/day (597 times the MRHD based on AUC comparison). Difelikefalin was not carcinogenic in a 6-month carcinogenicity study in transgenic rasH2 mice at subcutaneous doses up to 30 mg/kg/day.
Difelikefalin was negative for genotoxicity in a bacterial reverse mutation assay, an in vitro mammalian chromosomal aberration assay, and an in vivo mouse micronucleus assay.
Difelikefalin administered via intravenous injection caused a significant decrease in the number of estrous cycles per 14 days (i.e., prolonged diestrus) in female rats at doses greater than or equal to 2.5 mg/kg/day (56 times the MRHD based on AUC comparison). Difelikefalin had no effects on mating index, fertility index, or any ovarian or uterine parameters in female rats at doses up to 25 mg/kg/day (635 times the MRHD based on AUC comparison). Difelikefalin did not impair male fertility at doses up to 25 mg/kg/day (971 times the MRHD based on AUC comparison).
5.1 Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances
Dizziness, somnolence, mental status changes, and gait disturbances, including falls, have occurred in patients taking KORSUVA and may subside over time with continued treatment [see Adverse Reactions (6.1)]. In Trial 1 and Trial 2, 17.0% of patients randomized to receive KORSUVA reported at least one of these adverse reactions, compared to 12.0% of patients who received placebo. The incidence of somnolence was higher in KORSUVA-treated subjects 65 years of age and older (7.0%) than in KORSUVA-treated subjects less than 65 years of age (2.8%). Concomitant use of centrally-acting depressant medications, sedating antihistamines and opioid analgesics may increase the likelihood of these adverse reactions and should be used with caution during treatment with KORSUVA.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
KORSUVA has not been studied in patients on peritoneal dialysis and is not recommended for use in this population.
Section 44425-7 (44425-7)
Storage and Handling
Store vials at 20°C to 25°C (68°F to 77°F), excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Do not freeze.
KORSUVA injection must be administered within 4 hours of syringe preparation; prepared syringes can be stored at ambient temperature 20°C to 25°C (68°F to 77°F) until dosing [see Dosage and Administration (2.2, 2.3)] .
KORSUVA injection is supplied in a single-dose vial. Any unused drug remaining after injection must be discarded.
2.1 Dosage
- The recommended dosage of KORSUVA is 0.5 mcg/kg administered by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD treatment [see Dosage and Administration (2.3)].
- If a regularly scheduled HD treatment is missed, resume KORSUVA at the end of the next HD treatment.
10 Overdosage (10 OVERDOSAGE)
Single doses of KORSUVA up to 12 times and multiple doses of KORSUVA up to 5 times the recommended dosage of 0.5 mcg/kg were administered in clinical studies in subjects undergoing HD. A dose-dependent increase in adverse reactions, including dizziness, somnolence, mental status changes, paresthesia, fatigue, hypertension, and vomiting, were observed.
In the event of overdosage, provide the appropriate medical attention based on patient's clinical status. Difelikefalin is primarily eliminated by the kidneys with a low plasma protein binding of approximately 23% to 28% in dialysis patients. Hemodialysis for 4 hours using a high-flux dialyzer effectively cleared approximately 70% to 80% of difelikefalin from plasma, and difelikefalin was not detectable in plasma at the end of the second of two dialysis cycles. [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
KORSUVA (difelikefalin) is a kappa opioid receptor agonist. Difelikefalin is a synthetic peptide with a single stereoisomer and is present as an acetate salt. Difelikefalin acetate is a white to off-white powder with a molecular formula of C36H53N7O6∙xAcOH (1.0≤ × ≤2.0) and a molecular weight of 679.4 g/mol (mono- isotopic; free base). It is soluble in water. The chemical name of difelikefalin acetate is 4-amino-1-(D-phenylalanyl-D-phenylalanyl-D-leucyl-D-lysyl)piperidine-4-carboxylic acid, acetate salt.
The chemical structure is:
KORSUVA (difelikefalin) injection is supplied in a single-dose vial containing 65 mcg/1.3 mL (50 mcg/mL) of difelikefalin as a sterile, preservative-free, clear and colorless solution for intravenous injection.
KORSUVA is formulated as an isotonic 40 mM acetate buffer solution with an osmolality of 250 to 350 mOsm and a pH of 4.5.
Each milliliter of KORSUVA injection contains 50 mcg of difelikefalin (equivalent to an average of 58.3 mcg of difelikefalin acetate), 1.3 mg of acetic acid, 2.5 mg of sodium acetate trihydrate, 7.2 mg of sodium chloride (to adjust tonicity), and water for injection.
8.4 Pediatric Use
The safety and effectiveness of KORSUVA in pediatric patients have not been established.
8.5 Geriatric Use
Of the 848 subjects in the placebo-controlled studies who received KORSUVA, 278 subjects (32.8%) were 65 years of age and older and 98 subjects (11.6%) were 75 years of age and older. No overall differences in safety or effectiveness of KORSUVA have been observed between patients 65 years of age and older and younger adult subjects, with the exception of the incidence of somnolence which was higher in KORSUVA-treated subjects 65 years of age and older (7.0%) than in KORSUVA-treated subjects less than 65 years of age (2.8%), and was comparable in both placebo age groups (3.0% and 2.1%, respectively). No differences in plasma concentrations of KORSUVA were observed between subjects 65 years of age and older and younger adult subjects [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of KORSUVA was evaluated in two randomized, multicenter, double-blind, placebo-controlled trials (Trial 1 [NCT03422653] and Trial 2 [NCT03636269]) that enrolled a total of 851 subjects 18 years of age and older undergoing HD who had moderate-to-severe pruritus. In both trials, subjects received intravenous bolus injections of KORSUVA 0.5 mcg per kilogram of dry body weight into the venous line of the hemodialysis circuit at the end of each hemodialysis session or placebo three times per week for 12 weeks. In both trials, a 7-day run-in period prior to randomization was used to confirm that each subject had moderate-to-severe pruritus and to establish a baseline itch intensity, as measured by the patient-reported daily 24-hour Worst Itching Intensity Numerical Rating Scale (WI-NRS) scores (0 "no itch" to 10 "worst itch imaginable").
The mean (SD) baseline WI-NRS score was 7.1 (1.5) in Trial 1 and 7.2 (1.4) in Trial 2. At baseline in Trial 1, 61% of subjects were male, 49% were White, 42% were Black or African American, the mean age was 57 years (range 22 to 88 years), and 40% of subjects were using prior anti-pruritic medications (including sedating antihistamines) and continued the use throughout the trial. At baseline in Trial 2, 58% of subjects were male, 70% were White, 19% were Black or African American, the mean age was 60 years (range 23 to 90 years), and 36% of subjects were using prior anti-pruritic medications (including sedating antihistamines) and continued the use throughout the trial.
In each trial, efficacy was assessed based on the proportion of subjects achieving a 4-point or greater improvement (reduction) from baseline in the weekly mean of the daily 24-hour WI-NRS score at Week 12.
The results of the KORSUVA trials (Trials 1 and 2) are presented in Table 3 and Figure 1.
| Trial 1 | Trial 2 | |||
|---|---|---|---|---|
| KORSUVA 0.5 mcg/kg 3 times weekly N=189 |
Placebo N=189 |
KORSUVA 0.5 mcg/kg 3 times weekly N=237 |
Placebo N=236 |
|
| Percentage of subjects with ≥4-point improvement from baseline in WI-NRS score | 40% | 21% | 37% | 26% |
| Difference from Placebo (95% CI) | 19% (9%, 28%) | 12% (3%, 20%) |
Figure 1: Percentage of Subjects with Moderate-to-Severe CKD-aP Undergoing HD with a ≥4-point Improvement from Baseline on the WI-NRS in Trial 1 and Trial 2
Itch reduction was seen by Week 4 and sustained through Week 12.
4 Contraindications (4 CONTRAINDICATIONS)
None
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances [see Warnings and Precautions (5.1)]
12.2 Pharmacodynamics
Difelikefalin exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
The pharmacokinetics of difelikefalin is dose proportional over a single dosage range from 1 to 3 mcg/kg (2 to 6 times the recommended dosage) and multiple intravenous dosage range from 0.5 to 2.5 mcg/kg (1 to 5 times the recommended dosage) in chronic kidney disease patients undergoing HD. Steady-state was reached after the second administered dosage and the mean accumulation ratio was up to 1.6.
8.6 Hepatic Impairment
The influence of mild-to-moderate hepatic impairment on the pharmacokinetics of KORSUVA was evaluated in a population pharmacokinetic analysis which concluded that no KORSUVA dosage adjustments are needed in these populations [see Clinical Pharmacology (12.3)]. The influence of severe hepatic impairment on the pharmacokinetics of KORSUVA in subjects undergoing HD has not been evaluated; therefore, use of KORSUVA in this population is not recommended.
1 Indications and Usage (1 INDICATIONS AND USAGE)
KORSUVA is indicated for the treatment of moderate-to-severe pruritus associated with chronic kidney disease (CKD-aP) in adults undergoing hemodialysis (HD).
12.1 Mechanism of Action
KORSUVA is a kappa opioid receptor (KOR) agonist. The relevance of KOR activation to therapeutic effectiveness is not known.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances: Dizziness, somnolence, mental status changes, and gait disturbances, including falls, have occurred. Centrally-acting depressant medications, sedating antihistamines, and opioid analgesics should be used with caution during treatment with KORSUVA. (5.1)
- Risk of Driving and Operating Machinery: May impair mental or physical abilities. Advise patients not to drive or operate dangerous machinery until the effect of KORSUVA on a patient's ability to drive or operate machinery is known. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended dosage is 0.5 mcg/kg. (2.1)
- Administer by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD treatment. (2.1)
- Do not mix or dilute KORSUVA prior to administration. (2.2)
- Administer within 4 hours of syringe preparation. (2.3)
- See full prescribing information for additional recommendations on preparation and administration of KORSUVA. (2.2, 2.3)
2.2 Preparation Instructions
- Do not mix or dilute KORSUVA prior to administration.
- Inspect KORSUVA for particulate matter and discoloration prior to administration. The solution should be clear and colorless. Do not use KORSUVA vials if particulate matter or discoloration is observed.
- KORSUVA is supplied in a single-dose vial. Discard any unused product.
- Injection volume to be administered is determined by patient's target dry body weight in kilograms (one patient may use less than the full contents of the vial or use more than one vial). See Table 1.
| Target Dry Body Weight Range (kg) | Injection Volume (mL) Total Injection Volume (mL) = Patient Target Dry Body Weight (kg) x 0.01, rounded to the nearest tenth (0.1 mL). For patient target dry body weight outside of the ranges in Table 1, use this formula.
|
|---|---|
| 36 – 44 | 0.4 |
| 45 – 54 | 0.5 |
| 55 – 64 | 0.6 |
| 65 – 74 | 0.7 |
| 75 – 84 | 0.8 |
| 85 – 94 | 0.9 |
| 95 – 104 | 1 |
| 105 – 114 | 1.1 |
| 115 – 124 | 1.2 |
| 125 – 134 | 1.3 |
| 135 – 144 | 1.4 |
| 145 – 154 | 1.5 |
| 155 – 164 | 1.6 |
| 165 – 174 | 1.7 |
| 175 – 184 | 1.8 |
| 185 – 194 | 1.9 |
| 195 – 204 | 2 |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 65 mcg/1.3 mL (50 mcg/mL) of difelikefalin as a clear, colorless solution in a single-dose glass vial.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Severe Hepatic Impairment: Not recommended in patients with severe hepatic impairment. (8.6)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
A total of 1306 subjects undergoing HD who had moderate-to-severe pruritus were treated with KORSUVA in placebo-controlled and uncontrolled Phase 3 clinical trials. Of these, 711 were treated for at least 6 months and 400 were treated for at least one year.
Two placebo-controlled Phase 3 trials (Trial 1 and Trial 2), in subjects undergoing HD who had moderate-to-severe pruritus were pooled to evaluate the safety of KORSUVA in comparison to placebo up to 12 weeks. In total, 848 subjects were evaluated (424 in KORSUVA group and 424 in placebo group). The mean age of the subjects was 59 years (range 22 to 88 years), and 59% of the subjects were male. Of the total subjects, 61% were White, 29% were Black or African American, and 5% were Asian.
Table 2 summarizes the adverse reactions that occurred at a rate of ≥2% in the KORSUVA group and ≥1% higher than that of the placebo group during the 12-week placebo-controlled period of Trials 1 and 2. The percentage of subjects who discontinued treatment due to any adverse reaction was 2.6% for subjects taking KORSUVA and 0.7% for subjects taking placebo. The most common adverse reactions (≥0.5% of subjects) leading to discontinuation were dizziness (0.9% for KORSUVA and 0.2% for placebo), mental status change (0.7% and 0.2%, respectively), nausea (0.5% and 0%, respectively), and headache (0.5% and 0%, respectively). The percentage of subjects who developed serious adverse reactions was 4.5% in the KORSUVA group and 2.8% in the placebo group.
| Adverse Reactions | Placebo (N=424) n (%) |
KORSUVA (N=424) n (%) |
|---|---|---|
| Diarrhea | 24 (5.7) | 38 (9.0) |
| Dizziness | 16 (3.8) | 29 (6.8) |
| Nausea | 19 (4.5) | 28 (6.6) |
| Gait Disturbances Gait disturbances includes: preferred terms of falls and gait disturbances
|
23 (5.4) | 28 (6.6) |
| Hyperkalemia | 15 (3.5) | 20 (4.7) |
| Headache | 11 (2.6) | 19 (4.5) |
| Somnolence | 10 (2.4) | 18 (4.2) |
| Mental Status Change Mental Status Change includes: preferred terms of confusional state and mental status change.
|
6 (1.4) | 14 (3.3) |
2.3 Administration Instructions
- KORSUVA is removed by the dialyzer membrane and must be administered after blood is no longer circulating through the dialyzer.
- Administer KORSUVA by intravenous bolus injection into the venous line of the dialysis circuit at the end of each HD session.
- The dose may be given either during or after rinse back of the dialysis circuit.
- If the dose is given after rinse back, administer KORSUVA into the venous line followed by at least 10 mL of normal saline flush.
- If the dose is given during rinse back, no additional normal saline is needed to flush the line.
- The dose must be administered within 4 hours of the syringe preparation. Discard any unused product.
5.2 Risk of Driving and Operating Machinery
Dizziness, somnolence, and mental status changes have occurred in patients taking KORSUVA. KORSUVA may impair the mental or physical abilities needed to perform potentially hazardous activities such as driving a car and operating machinery. Advise patients not to drive or operate dangerous machinery until the effect of KORSUVA on a patient's ability to drive or operate machinery is known.
Principal Display Panel 1.3 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 1.3 mL Vial Label)
NDC 59353-065-01
Store at 20°C to 25°C (68°F to 77°F)
Rx only
KORSUVA™
(difelikefalin) Injection
65 mcg/1.3 mL
(50 mcg/mL)
For Intravenous Use Only
1.3 mL Single-dose Vial. Discard unused portion
Mfd. for Vifor (International) Inc., Rechenstrasse 37,
9014 St. Gallen, Switzerland. Made in France.
3102942-01 48307/16/25
Principal Display Panel 1.3 Ml Tray Carton (PRINCIPAL DISPLAY PANEL - 1.3 mL Tray Carton)
NDC 59353-065-12
Rx only
KORSUVA™
(difelikefalin) Injection
65 mcg/1.3 mL
(50 mcg/mL)
For Intravenous Use after Hemodialysis
Single-dose vial. Discard unused portion.
Store at 20°C to 25°C (68°F to 77°F), excursions
permitted to 15°C to 30°C (59°F to 86°F) [see USP
Controlled Room Temperature]. Do not freeze.
Sterile, preservative-free solution.
12 x 1.3 mL
Single-dose Vials
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year carcinogenicity study in rats, difelikefalin was not carcinogenic when administered via subcutaneous injection at doses up to 1.0 mg/kg/day (597 times the MRHD based on AUC comparison). Difelikefalin was not carcinogenic in a 6-month carcinogenicity study in transgenic rasH2 mice at subcutaneous doses up to 30 mg/kg/day.
Difelikefalin was negative for genotoxicity in a bacterial reverse mutation assay, an in vitro mammalian chromosomal aberration assay, and an in vivo mouse micronucleus assay.
Difelikefalin administered via intravenous injection caused a significant decrease in the number of estrous cycles per 14 days (i.e., prolonged diestrus) in female rats at doses greater than or equal to 2.5 mg/kg/day (56 times the MRHD based on AUC comparison). Difelikefalin had no effects on mating index, fertility index, or any ovarian or uterine parameters in female rats at doses up to 25 mg/kg/day (635 times the MRHD based on AUC comparison). Difelikefalin did not impair male fertility at doses up to 25 mg/kg/day (971 times the MRHD based on AUC comparison).
5.1 Dizziness, Somnolence, Mental Status Changes, and Gait Disturbances
Dizziness, somnolence, mental status changes, and gait disturbances, including falls, have occurred in patients taking KORSUVA and may subside over time with continued treatment [see Adverse Reactions (6.1)]. In Trial 1 and Trial 2, 17.0% of patients randomized to receive KORSUVA reported at least one of these adverse reactions, compared to 12.0% of patients who received placebo. The incidence of somnolence was higher in KORSUVA-treated subjects 65 years of age and older (7.0%) than in KORSUVA-treated subjects less than 65 years of age (2.8%). Concomitant use of centrally-acting depressant medications, sedating antihistamines and opioid analgesics may increase the likelihood of these adverse reactions and should be used with caution during treatment with KORSUVA.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:09.268551 · Updated: 2026-03-14T22:32:10.275892