These Highlights Do Not Include All The Information Needed To Use Zemdri™ Safely And Effectively. See Full Prescribing Information For Zemdri.
0b82ffed-27f4-4f5c-8135-670c148f0e12
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Nephrotoxicity has been reported with ZEMDRI. The risk of nephrotoxicity is greater in patients with impaired renal function, the elderly, and in those receiving concomitant nephrotoxic medications. Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy [see Dosage and Administration (2.2) and Warnings and Precautions (5.1) ] . Therapeutic Drug Monitoring (TDM) is recommended for complicated urinary tract infection (cUTI) patients with CLcr less than 90 mL/min to avoid potentially toxic levels [see Dosage and Administration (2.3 , 2.4) ] . Ototoxicity, manifested as hearing loss, tinnitus, and/or vertigo, has been reported with ZEMDRI. Symptoms of aminoglycoside-associated ototoxicity may be irreversible and may not become evident until after completion of therapy. Aminoglycoside-associated ototoxicity has been observed primarily in patients with a family history of hearing loss, patients with renal impairment, and in patients receiving higher doses and/or longer durations of therapy than recommended [see Warnings and Precautions (5.2) ] . Aminoglycosides have been associated with neuromuscular blockade. During therapy with ZEMDRI, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients, such as patients with underlying neuromuscular disorders (including myasthenia gravis) or in patients concomitantly receiving neuromuscular blocking agents [see Warning and Precautions (5.3) ] . Aminoglycosides, including ZEMDRI, can cause fetal harm when administered to a pregnant woman [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1) ] .
Indications and Usage
ZEMDRI is an aminoglycoside antibacterial indicated for the treatment of patients 18 years of age or older with Complicated Urinary Tract Infections (cUTI) including Pyelonephritis. ( 1.1 ) As only limited clinical safety and efficacy data are available, reserve ZEMDRI for use in patients who have limited or no alternative treatment options. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain effectiveness of ZEMDRI and other antibacterial drugs, ZEMDRI should be used only to treat infections that are proven or strongly suspected to be caused by susceptible microorganisms. ( 1.2 )
Dosage and Administration
Administer ZEMDRI 15 mg/kg every 24 hours by intravenous (IV) infusion over 30 minutes to patients 18 years of age or older with creatinine clearance greater than or equal to 90 mL/min. ( 2.1 ) Recommended duration of treatment is 4 to 7 days for cUTI, including pyelonephritis. ( 2.1 ) Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy. ( 2.2 ) Recommended initial dosage regimen for patients with renal impairment is shown in the table below. ( 2.3 ) Estimated CLcr CLcr estimated by the Cockcroft-Gault formula. ( 2.3 ) (mL/min) Recommended Dosage for ZEMDRI Calculate dosage using Total Body Weight (TBW). For patients with TBW greater than IBW by 25% or more, use adjusted body weight. ( 2.3 ) Dosing Interval Greater than or equal to 60 to less than 90 15 mg/kg Every 24 hours Greater than or equal to 30 to less than 60 10 mg/kg Every 24 hours Greater than or equal to 15 to less than 30 10 mg/kg Every 48 hours See Full Prescribing Information for subsequent dosage adjustment based on changes in renal function or Therapeutic Drug Monitoring (TDM). ( 2.3 , 2.4 ). See Full Prescribing Information for instructions on preparation of the solution, stability in intravenous fluids and drug compatibilities. ( 2.5 , 2.6 , 2.7 )
Warnings and Precautions
Hypersensitivity Reactions, including anaphylaxis: Reported for aminoglycosides. If an allergic reaction occurs, discontinue ZEMDRI. ( 5.4 ) Clostridium difficile -Associated Diarrhea : Reported for nearly all systemic antibacterial drugs. Evaluate if diarrhea occurs. ( 5.5 )
Contraindications
ZEMDRI is contraindicated in patients with known hypersensitivity to any aminoglycoside [see Warnings and Precautions (5.5) ] .
Adverse Reactions
The following important adverse reactions are discussed in greater detail in the Warnings and Precautions section: Nephrotoxicity [see Warnings and Precautions (5.1) ] Ototoxicity [see Warnings and Precautions (5.2) ] Neuromuscular Blockade [see Warnings and Precautions (5.3) ] Fetal Harm [see Warnings and Precautions (5.4) ] Hypersensitivity Reactions [see Warnings and Precautions (5.5) ] Clostridium difficile -Associated Diarrhea [see Warnings and Precautions (5.6) ]
Storage and Handling
Store ZEMDRI injection 500 mg/10 mL (50 mg/mL) refrigerated at 2°C to 8°C (36°F to 46°F).
How Supplied
ZEMDRI injection 500 mg/10 mL (50 mg/mL) is supplied in single-dose, 10-mL vials fitted with flip-off seals with royal blue polypropylene buttons as a clear, colorless to yellow, sterile solution. Each vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase at a concentration of 50 mg/mL plazomicin in Water for Injection. Each vial contains sodium hydroxide for pH adjustment to 6.5. The solution may become yellow in color; this does not indicate a decrease in potency. NDC number Package/Volume Units per carton Plazomicin content 71045-010-02 Single use, fliptop vial, 10-mL 10 500 mg in 10 mL (50 mg/mL)
Medication Information
Warnings and Precautions
Hypersensitivity Reactions, including anaphylaxis: Reported for aminoglycosides. If an allergic reaction occurs, discontinue ZEMDRI. ( 5.4 ) Clostridium difficile -Associated Diarrhea : Reported for nearly all systemic antibacterial drugs. Evaluate if diarrhea occurs. ( 5.5 )
Indications and Usage
ZEMDRI is an aminoglycoside antibacterial indicated for the treatment of patients 18 years of age or older with Complicated Urinary Tract Infections (cUTI) including Pyelonephritis. ( 1.1 ) As only limited clinical safety and efficacy data are available, reserve ZEMDRI for use in patients who have limited or no alternative treatment options. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain effectiveness of ZEMDRI and other antibacterial drugs, ZEMDRI should be used only to treat infections that are proven or strongly suspected to be caused by susceptible microorganisms. ( 1.2 )
Dosage and Administration
Administer ZEMDRI 15 mg/kg every 24 hours by intravenous (IV) infusion over 30 minutes to patients 18 years of age or older with creatinine clearance greater than or equal to 90 mL/min. ( 2.1 ) Recommended duration of treatment is 4 to 7 days for cUTI, including pyelonephritis. ( 2.1 ) Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy. ( 2.2 ) Recommended initial dosage regimen for patients with renal impairment is shown in the table below. ( 2.3 ) Estimated CLcr CLcr estimated by the Cockcroft-Gault formula. ( 2.3 ) (mL/min) Recommended Dosage for ZEMDRI Calculate dosage using Total Body Weight (TBW). For patients with TBW greater than IBW by 25% or more, use adjusted body weight. ( 2.3 ) Dosing Interval Greater than or equal to 60 to less than 90 15 mg/kg Every 24 hours Greater than or equal to 30 to less than 60 10 mg/kg Every 24 hours Greater than or equal to 15 to less than 30 10 mg/kg Every 48 hours See Full Prescribing Information for subsequent dosage adjustment based on changes in renal function or Therapeutic Drug Monitoring (TDM). ( 2.3 , 2.4 ). See Full Prescribing Information for instructions on preparation of the solution, stability in intravenous fluids and drug compatibilities. ( 2.5 , 2.6 , 2.7 )
Contraindications
ZEMDRI is contraindicated in patients with known hypersensitivity to any aminoglycoside [see Warnings and Precautions (5.5) ] .
Adverse Reactions
The following important adverse reactions are discussed in greater detail in the Warnings and Precautions section: Nephrotoxicity [see Warnings and Precautions (5.1) ] Ototoxicity [see Warnings and Precautions (5.2) ] Neuromuscular Blockade [see Warnings and Precautions (5.3) ] Fetal Harm [see Warnings and Precautions (5.4) ] Hypersensitivity Reactions [see Warnings and Precautions (5.5) ] Clostridium difficile -Associated Diarrhea [see Warnings and Precautions (5.6) ]
Storage and Handling
Store ZEMDRI injection 500 mg/10 mL (50 mg/mL) refrigerated at 2°C to 8°C (36°F to 46°F).
How Supplied
ZEMDRI injection 500 mg/10 mL (50 mg/mL) is supplied in single-dose, 10-mL vials fitted with flip-off seals with royal blue polypropylene buttons as a clear, colorless to yellow, sterile solution. Each vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase at a concentration of 50 mg/mL plazomicin in Water for Injection. Each vial contains sodium hydroxide for pH adjustment to 6.5. The solution may become yellow in color; this does not indicate a decrease in potency. NDC number Package/Volume Units per carton Plazomicin content 71045-010-02 Single use, fliptop vial, 10-mL 10 500 mg in 10 mL (50 mg/mL)
Description
Nephrotoxicity has been reported with ZEMDRI. The risk of nephrotoxicity is greater in patients with impaired renal function, the elderly, and in those receiving concomitant nephrotoxic medications. Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy [see Dosage and Administration (2.2) and Warnings and Precautions (5.1) ] . Therapeutic Drug Monitoring (TDM) is recommended for complicated urinary tract infection (cUTI) patients with CLcr less than 90 mL/min to avoid potentially toxic levels [see Dosage and Administration (2.3 , 2.4) ] . Ototoxicity, manifested as hearing loss, tinnitus, and/or vertigo, has been reported with ZEMDRI. Symptoms of aminoglycoside-associated ototoxicity may be irreversible and may not become evident until after completion of therapy. Aminoglycoside-associated ototoxicity has been observed primarily in patients with a family history of hearing loss, patients with renal impairment, and in patients receiving higher doses and/or longer durations of therapy than recommended [see Warnings and Precautions (5.2) ] . Aminoglycosides have been associated with neuromuscular blockade. During therapy with ZEMDRI, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients, such as patients with underlying neuromuscular disorders (including myasthenia gravis) or in patients concomitantly receiving neuromuscular blocking agents [see Warning and Precautions (5.3) ] . Aminoglycosides, including ZEMDRI, can cause fetal harm when administered to a pregnant woman [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1) ] .
Section 42229-5
Adverse Reactions Leading to Treatment Discontinuations in Trial 1
In Trial 1, treatment discontinuation from IV study drug due to an adverse reaction occurred in 2.0% of patients receiving ZEMDRI (6/303) and meropenem (6/301), respectively.
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEMDRI and other antibacterial drugs, ZEMDRI should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10. Overdosage
In the event of overdosage, ZEMDRI should be discontinued and supportive care is advised. Maintenance of glomerular filtration and careful monitoring of renal function is recommended. Hemodialysis may aid in the removal of ZEMDRI from the blood, especially if renal function is, or becomes, compromised. No clinical information is available on the use of hemodialysis to treat ZEMDRI overdosage.
15. References
- American Speech-Language-Hearing Association. (1994). Audiologic management of individuals receiving cochleotoxic drug therapy [Guidelines]. Available from www.asha.org/policy.
5.4 Fetal Harm
Aminoglycosides, including ZEMDRI, can cause fetal harm when administered to a pregnant woman. Aminoglycosides cross the placenta, and streptomycin has been associated with several reports of total, irreversible, bilateral congenital deafness in pediatric patients exposed in utero. Patients who use ZEMDRI during pregnancy, or become pregnant while taking ZEMDRI should be apprised of the potential hazard to the fetus [see Use in Specific Populations (8.1)].
11. Description
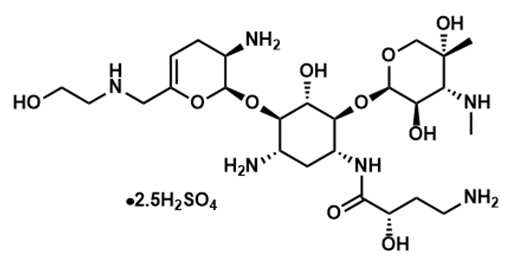
ZEMDRI contains plazomicin sulfate, a semi-synthetic aminoglycoside antibacterial derived from sisomicin. The chemical name of plazomicin sulfate is (2"R,3"R,4"R,5"R)-2"-[(1S,2S,3R,4S,6R)-4-amino-6-[(2'"S)-4'"-amino-2'"-hydroxybutanamido)amino]-3-[(2'S,3'R)-3'-amino-6'-((2-hydroxyethylamino)methyl)-3',4'-dihydro-2H-pyran-2'-yloxy]-2-hydroxycyclohexyloxy]-5''-methyl-4''-(methylamino)tetrahydro-2H-pyran-3'',5''-diol sulfate. Plazomicin sulfate contains a theoretical 2.5 molar equivalents of sulfate relative to the freebase, based on complete protonation. The molecular weight of plazomicin sulfate is calculated based on 1:2.5 stoichiometry. The corresponding empirical formula is C25H48N6O10∙2.5 H2SO4 (plazomicin sulfate) and the molecular weight of the plazomicin sulfate salt is 837.89 g/mol and the molecular weight of the freebase is 592.69 g/mol.
Figure 1: Chemical Structure of Plazomicin Sulfate
ZEMDRI injection 500 mg/10 mL is a sterile, clear, colorless-to-yellow liquid for intravenous administration supplied in 10-mL single-dose Type 1 glass vials. Each vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase at a concentration of 50 mg/mL adjusted to pH 6.5. Each vial also contains Water for Injection and sodium hydroxide for pH adjustment. This sterile, nonpyrogenic solution is formulated without preservatives.
5.2 Ototoxicity
Ototoxicity, manifested as hearing loss, tinnitus, and/or vertigo, has been reported with ZEMDRI. Symptoms of aminoglycoside-associated ototoxicity may be irreversible and may not become evident until after completion of therapy.
Regarding the incidence of adverse reactions associated with cochlear or vestibular function, in Trial 1, there was one case of reversible hypoacusis (1/303;0.3%) in ZEMDRI-treated patients and one case of tinnitus (1/301;0.3%) in meropenem-treated patients [see Adverse Reactions (6.1)]. In Trial 2, one case each of irreversible tinnitus and reversible vertigo was reported in ZEMDRI-treated patients, and one case of an abnormal audiogram occurred in a levofloxacin-treated patient [see Adverse Reactions (6.1)].
Aminoglycoside-associated ototoxicity has been observed primarily in patients with a family history of hearing loss (excluding age-related hearing loss), patients with renal impairment, and in patients receiving higher doses and/or for longer periods than recommended. In Trial 1 and Trial 2, patients with a history of hearing loss, with the exception of age-related hearing loss, were excluded. The benefit-risk of ZEMDRI therapy should be considered in these patients.
16.1 How Supplied
ZEMDRI injection 500 mg/10 mL (50 mg/mL) is supplied in single-dose, 10-mL vials fitted with flip-off seals with royal blue polypropylene buttons as a clear, colorless to yellow, sterile solution. Each vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase at a concentration of 50 mg/mL plazomicin in Water for Injection. Each vial contains sodium hydroxide for pH adjustment to 6.5. The solution may become yellow in color; this does not indicate a decrease in potency.
| NDC number | Package/Volume | Units per carton | Plazomicin content |
|---|---|---|---|
| 71045-010-02 | Single use, fliptop vial, 10-mL | 10 | 500 mg in 10 mL (50 mg/mL) |
8.5 Geriatric Use
Of the 425 patients treated with ZEMDRI in Trials 1 and 2, 40% (170/425) were 65 years of age and older, including 17.2% (73/425) patients 75 years of age and older. In Trial 1, for ZEMDRI- treated patients ≥ 65 years old, the incidence rate of adverse reactions was 27% (37/137) versus 18.9% (27/143) in the meropenem-treated patients ≥ 65 years old. For ZEMDRI- treated patients < 65 years old, the incidence rate of adverse reactions was 13.3% (22/166) versus 24.1% (38/158) in the meropenem-treated patients < 65 years old.
The rate of adverse reactions associated with renal function for the ZEMDRI-treated patients ≥ 65 years old was 6.6% (9/137) versus 2.8% (4/143) in the meropenem-treated patients. For ZEMDRI- treated patients < 65 years old, the incidence rate of adverse reactions associated with renal function was 1.2% (2/166), versus 0% (0/158) in the meropenem-treated patients [see Clinical Studies (14.1) and Adverse Reactions (6.1)].
ZEMDRI is substantially excreted by the kidneys, and the risk of adverse reactions to ZEMDRI may be greater in patients with renal impairment. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and renal function should be monitored. Dosage adjustment in elderly patients should take into account renal function and plazomicin concentrations as appropriate [see Dosage and Administration (2.2, 2.3, 2.4) and Clinical Pharmacology (12.3)].
5.1 Nephrotoxicity
Nephrotoxicity has been reported with the use of ZEMDRI [see Adverse Reactions (6.1)]. Most serum creatinine increases were ≤ 1 mg/dL above baseline and reversible.
In Trial 1, the incidence of adverse reactions associated with renal function (acute kidney injury, serum creatinine increased, chronic kidney disease, creatinine clearance decreased, renal failure, renal impairment) was 3.6% (11/303) in ZEMDRI-treated patients compared with 1.3% (4/301) in meropenem-treated patients [see Adverse Reactions (6.1)].
Serum creatinine increases of 0.5 mg/dL or greater above baseline occurred in 7% (21/300) of ZEMDRI-treated patients compared with 4% (12/297) of meropenem-treated patients. These increases mainly occurred in patients with CLcr ≤ 90 mL/min and were associated with a plazomicin trough level (Cmin) greater than or equal to 3 mcg/mL [see Adverse Reactions (6.1) and Clinical Pharmacology (12.2)].
Assess CLcr in all patients prior to initiating therapy and daily during therapy with ZEMDRI, particularly in those at increased risk of nephrotoxicity, such as those with renal impairment, the elderly, and those receiving concomitant potentially nephrotoxic medications. In the setting of worsening renal function, the benefit of continuing ZEMDRI should be assessed [see Dosage and Administration (2.2, 2.4), Adverse Reactions (6.1) and Use in Specific Populations (8.5, 8.6)].
Adjust the initial dosage regimen in cUTI patients with CLcr ≥ 15 mL/min and < 60 mL/min [see Dosage and Administration (2.3)]. For subsequent doses, TDM is recommended for patients with CLcr ≥15 mL/min and < 90 mL/min [see Dosage and Administration (2.4)].
8.4. Pediatric Use
The safety and effectiveness of ZEMDRI in patients less than 18 years of age have not been established.
4. Contraindications
ZEMDRI is contraindicated in patients with known hypersensitivity to any aminoglycoside [see Warnings and Precautions (5.5)].
6. Adverse Reactions
The following important adverse reactions are discussed in greater detail in the Warnings and Precautions section:
- Nephrotoxicity [see Warnings and Precautions (5.1)]
- Ototoxicity [see Warnings and Precautions (5.2)]
- Neuromuscular Blockade [see Warnings and Precautions (5.3)]
- Fetal Harm [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.6)]
8.6 Renal Impairment
Plazomicin total body clearance was significantly decreased in patients with CLcr greater than or equal to 15 to less than 60 mL/min compared to patients with CLcr greater than or equal to 60 mL/min [see Clinical Pharmacology (12.3)]. Monitor CLcr daily and adjust ZEMDRI dosage accordingly [see Dosage and Administration (2.2)]. There is insufficient information to recommend a dosage regimen in patients with CLcr less than 15 mL/min or on renal replacement therapy, including hemodialysis or continuous renal replacement therapy.
For patients with CLcr greater than or equal to 15 mL/min and less than 90 mL/min, TDM is recommended. Monitor plazomicin trough concentrations and adjust ZEMDRI dosage accordingly [see Dosage and Administration (2.3, 2.4)].
12.2 Pharmacodynamics
The ratio of area under the plasma concentration-time curve to the minimum inhibitory concentration (AUC:MIC) for plazomicin has been shown to best correlate with efficacy in animal and in vitro models of infection against Enterobacteriaceae.
12.3 Pharmacokinetics
The pharmacokinetic (PK) parameters of plazomicin are similar for single- and multiple-dose administration of ZEMDRI in healthy subjects. No appreciable accumulation of plazomicin was observed following multiple IV infusions of 15 mg/kg administered every 24 hours in subjects with normal renal function. The AUC, maximum plasma concentration (Cmax), and Cmin increased in proportion to the dose over the dose range of 4 to 15 mg/kg. The plazomicin AUC, Cmax, and Cmin are summarized in Table 4.
| Healthy Subjects PK parameters following a single dose of 15 mg/kg; Based on non-compartmental analysis of PK data; AUC0-inf is reported; Cmin is concentration at 24 hours.
Geometric mean (±SD) N=54 |
cUTI Patients Day 1 PK parameters following administration of 15 mg/kg; Derived based on population PK model; AUC0-24h is reported.
Geometric mean (±SD) N=87 |
|
|---|---|---|
| AUC (mcg∙h/mL) | 257 (±67.0) | 226 (±113) |
| Cmax (mcg/mL) | 73.7 (±19.7) | 51.0 (±26.7) |
| Cmin (mcg/mL) | 0.3 (±0.2) | 0.5 (±1.2) |
2.1 Recommended Dosage
The recommended dosage regimen of ZEMDRI is 15 mg/kg administered every 24 hours by intravenous (IV) infusion over 30 minutes in patients 18 years of age or older and with creatinine clearance (CLcr) greater than or equal to 90 mL/min (Table 1). The duration of therapy should be guided by the severity of infection and the patient's clinical status for up to 7 days. During treatment, dosage adjustments may be required based on change in renal function [see Dosage and Administration (2.3, 2.4)].
| cUTI Infection | Dosage Regimen Calculate dosage using TBW. For patients with TBW greater than IBW by 25% or more, use adjusted body weight based on the equation: Adjusted body weight = IBW + 0.4 × [TBW – IBW].
|
Duration of Treatment |
|---|---|---|
| Complicated Urinary Tract Infections, including Pyelonephritis | 15 mg/kg every 24 hours | 4 to 7 days An appropriate oral therapy may be considered after 4 to 7 days of ZEMDRI therapy to complete a total duration of 7 to 10 days (IV plus oral). The maximum duration of ZEMDRI for cUTI is 7 days.
|
2.7 Drug Compatibility
Compatibility of ZEMDRI for administration with other drugs has not been established. ZEMDRI should not be mixed with other drugs or physically added to solutions containing other drugs. Other medications should not be infused simultaneously with ZEMDRI through the same IV line.
1. Indications and Usage
ZEMDRI is an aminoglycoside antibacterial indicated for the treatment of patients 18 years of age or older with Complicated Urinary Tract Infections (cUTI) including Pyelonephritis. (1.1)
As only limited clinical safety and efficacy data are available, reserve ZEMDRI for use in patients who have limited or no alternative treatment options. (1.1)
To reduce the development of drug-resistant bacteria and maintain effectiveness of ZEMDRI and other antibacterial drugs, ZEMDRI should be used only to treat infections that are proven or strongly suspected to be caused by susceptible microorganisms. (1.2)
12.1 Mechanism of Action
ZEMDRI is an antibacterial drug [see Microbiology (12.4)].
16.2 Storage and Handling
Store ZEMDRI injection 500 mg/10 mL (50 mg/mL) refrigerated at 2°C to 8°C (36°F to 46°F).
5.6 Clostridium Difficile
Clostridium difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial drugs and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial drugs alters the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B that contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial drugs.
If CDAD is suspected or confirmed, antibacterial drugs not directed against C. difficile may need to be discontinued. Manage fluid and electrolyte levels as appropriate, supplement protein intake, monitor antibacterial treatment of C. difficile, and institute surgical evaluation as clinically indicated.
5.3 Neuromuscular Blockade
Aminoglycosides have been associated with exacerbation of muscle weakness in patients with underlying neuromuscular disorders, or delay in recovery of neuromuscular function in patients receiving concomitant neuromuscular blocking agents.
During therapy with ZEMDRI, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients, such as patients with underlying neuromuscular disorders (including myasthenia gravis) or those patients concomitantly receiving neuromuscular blocking agents.
5. Warnings and Precautions
2. Dosage and Administration
- Administer ZEMDRI 15 mg/kg every 24 hours by intravenous (IV) infusion over 30 minutes to patients 18 years of age or older with creatinine clearance greater than or equal to 90 mL/min. (2.1)
- Recommended duration of treatment is 4 to 7 days for cUTI, including pyelonephritis. (2.1)
- Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy. (2.2)
- Recommended initial dosage regimen for patients with renal impairment is shown in the table below. (2.3)
| Estimated CLcr CLcr estimated by the Cockcroft-Gault formula. (2.3) (mL/min) |
Recommended Dosage for ZEMDRI Calculate dosage using Total Body Weight (TBW). For patients with TBW greater than IBW by 25% or more, use adjusted body weight. (2.3)
|
Dosing Interval |
|---|---|---|
| Greater than or equal to 60 to less than 90 | 15 mg/kg | Every 24 hours |
| Greater than or equal to 30 to less than 60 |
10 mg/kg | Every 24 hours |
| Greater than or equal to 15 to less than 30 |
10 mg/kg | Every 48 hours |
- See Full Prescribing Information for subsequent dosage adjustment based on changes in renal function or Therapeutic Drug Monitoring (TDM). (2.3, 2.4).
- See Full Prescribing Information for instructions on preparation of the solution, stability in intravenous fluids and drug compatibilities. (2.5, 2.6, 2.7)
3. Dosage Forms and Strengths
ZEMDRI injection 500 mg/10 mL (50 mg/mL) is a sterile, clear, colorless to yellow solution supplied in a single-dose vial. Each single-dose vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be compared directly to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
ZEMDRI was evaluated in two comparator-controlled clinical trials (Trial 1, NCT02486627 and Trial 2, NCT01096849) in patients with cUTI, including pyelonephritis. In both trials, patients with CLcr greater than 60 mL/min received ZEMDRI 15 mg/kg IV once daily as a 30-minute infusion [see Clinical Studies (14.1)].
Trial 1 included 303 patients treated with ZEMDRI and 301 patients treated with meropenem. Patients were to receive 4 to 7 days of ZEMDRI (mean duration of 5.1 days). In some patients, parenteral therapy was followed by a switch to an oral antibacterial drug.
The median age of patients treated with ZEMDRI in Trial 1 was 62 years (range 18 to 90 years) and 45.2% of patients were 65 years of age or older. Patients treated with ZEMDRI were predominantly female (56.1%) and White (99.3%). A majority of patients (68.0%) had mild or moderate renal impairment (CLcr >30 to 90 mL/min) at baseline. Patients with CLcr of 30 mL/min or less were excluded.
5.5 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving aminoglycoside antibacterial drugs. Before therapy with ZEMDRI is instituted, careful inquiry about previous hypersensitivity reactions to other aminoglycosides should be made. A history of hypersensitivity to other aminoglycosides is a contraindication to the use of ZEMDRI, because cross-sensitivity among aminoglycoside antibacterial drugs has been established. Discontinue ZEMDRI if an allergic reaction occurs.
2.2 Monitoring of Renal Function
Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy with ZEMDRI [see Dosage and Administration (2.3),Warnings and Precautions (5.1) and Use in Specific Populations (8.6)].
5.7 Development of Drug Resistant Bacteria
Prescribing ZEMDRI in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Principal Display Panel 10 Ml Vial Carton
NDC 71045-010-02
Rx ONLY
ZEMDRI™
(plazomicin) Injection
10 (10 mL) Single-dose vials
500 mg/10 mL per vial (50 mg/mL)
For Intravenous Infusion Only
Must dilute before use
2.4 Tdm in Cuti Patients With Renal Impairment
For cUTI patients with CLcr greater than or equal to 15 mL/min and less than 90 mL/min, TDM is recommended to maintain plasma trough concentrations below 3 mcg/mL. Measure plazomicin plasma trough concentration within approximately 30 minutes before administration of the second dose of ZEMDRI. Adjustment of the ZEMDRI dosage regimen based on TDM involves extending ZEMDRI dosing interval by 1.5 fold (i.e., from every 24 hours to every 36 hours or from every 48 hours to every 72 hours) for patients with plasma trough concentrations greater than or equal to 3 mcg/mL [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2)].
2.5 Preparation of Diluted Solutions of Zemdri
ZEMDRI is supplied as a single-dose fliptop 10-mL vial that contains plazomicin sulfate equivalent to 500 mg plazomicin freebase in 10 mL Water for Injection (concentration of 50 mg/mL). The appropriate volume of ZEMDRI solution (50 mg/mL) for the required dose should be diluted in 0.9% Sodium Chloride Injection, USP or Lactated Ringer's Injection, USP to achieve a final volume of 50 mL for intravenous infusion. The stability of ZEMDRI solution in the compatible diluents is described below [see Dosage and Administration (2.7)].
ZEMDRI does not contain preservatives. Aseptic technique must be followed in preparing the infusion solution. Discard unused portion of the ZEMDRI vial.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
2.3 Dosage in Adult Patients With Renal Impairment
The recommended initial dosage regimen of ZEMDRI in adult patients with CLcr greater than or equal to 15 and less than 90 mL/min, estimated by the Cockcroft-Gault formula, is described in Table 2.
Patients with CLcr greater than or equal to 15 and less than 90 mL/min receiving ZEMDRI may require subsequent dosage adjustments based on change in renal function and/or Therapeutic Drug Monitoring (TDM) as appropriate [see Dosage and Administration (2.4)].
| Estimated CLcr CLcr estimated by the Cockcroft-Gault formula using total body weight (TBW). For patients with TBW greater than ideal body weight (IBW) by 25% or more, use IBW. (mL/min) |
Dosage Calculate dosage using TBW. For patients with TBW greater than IBW by 25% or more, use adjusted body weight based on the equation: Adjusted body weight = IBW + 0.4 × [TBW – IBW].
|
Dosing Interval |
|---|---|---|
| Greater than or equal to 60 to less than 90 | 15 mg/kg | Every 24 hours |
| Greater than or equal to 30 to less than 60 | 10 mg/kg | Every 24 hours |
| Greater than or equal to 15 to less than 30 | 10 mg/kg | Every 48 hours |
There is insufficient information to recommend a dosage regimen in patients with CLcr less than 15 mL/min or on renal replacement therapy, including hemodialysis or continuous renal replacement therapy.
2.6 Stability of Zemdri Solution in Intravenous Fluids
After dilution, ZEMDRI solution for administration is stable for 24 hours at room temperature at concentrations of 2.5 mg/mL to 45 mg/mL in the following solutions:
- 0.9% Sodium Chloride Injection, USP
- Lactated Ringer's Injection, USP
14.1 Complicated Urinary Tract Infections, Including Pyelonephritis
A total of 609 adults hospitalized with cUTI (including pyelonephritis) were randomized in a multinational, double-blind, noninferiority trial comparing ZEMDRI (15 mg/kg IV once daily as a 30-minute infusion) to meropenem (1 g intravenously every 8 hours as a 30-minute infusion) (Trial 1, NCT02486627). Switch to an oral antibacterial drug, such as levofloxacin, was allowed after a minimum of 4 and maximum of 7 days of IV therapy for a total of 7 to 10 days of treatment.
Efficacy was assessed in the microbiological modified intent-to-treat (mMITT) population, which included all patients who received study medication and had at least 1 baseline uropathogen. The mMITT population excluded patients with organisms resistant to study drugs. Patient demographic and baseline characteristics were balanced between treatment groups in the mMITT population. The mMITT population consisted of 388 patients with cUTI, including 162 (41.8%) with pyelonephritis. The median age was 64 years, 52.8% were female and 99.5% were White. The majority of the patients (99%) were from Eastern Europe; 3 patients were from the United States. Concomitant bacteremia was identified in 25 (13.1%) and 23 (11.7%) patients at baseline in the ZEMDRI and meropenem groups, respectively. The median treatment duration of IV study drug was 6 days in both groups.
ZEMDRI demonstrated efficacy for composite cure at Day 5 and the Test of Cure (TOC) visit (Table 5). Composite cure at Day 5 was defined as resolution or improvement of clinical cUTI symptoms and a microbiological outcome of eradication (all baseline uropathogens reduced to <104 colony-forming units [CFU]/mL). Composite cure at the TOC visit (Day 17 ± 2 from the first dose of study drug) was defined as resolution of clinical cUTI symptoms and a microbiological outcome of eradication.
| Analysis Visit | ZEMDRI n/N (%) |
Meropenem n/N (%) |
Treatment Difference Treatment difference is ZEMDRI – meropenem.
(95% CI) |
|---|---|---|---|
| Abbreviations: CI=confidence interval; TOC=test-of-cure; CI=95% confidence interval based on Newcombe method with continuity correction. | |||
| Day 5 | 168/191 (88.0) | 180/197 (91.4) | -3.4 (-10.0, 3.1) |
| Clinical cure or improvement | 171/191 (89.5) | 182/197 (92.4) | |
| Microbiological eradication | 188/191 (98.4) | 193/197 (98.0) | |
| TOC | 156/191 (81.7) | 138/197 (70.1) | 11.6 (2.7, 20.3) |
| Clinical Cure | 170/191 (89.0) | 178/197 (90.4) | |
| Microbiological eradication | 171/191 (89.5) | 147/197 (74.6) |
Microbiological eradication rates at the TOC visit by baseline uropathogen in the mMITT population are presented in Table 6. Composite Cure at the TOC visit in individuals with concomitant bacteremia at baseline was achieved in 72.0% (18/25) of patients in the ZEMDRI group and 56.5% (13/23) of patients in the meropenem group.
| Pathogen | ZEMDRI n/N (%) |
Meropenem n/N (%) |
|---|---|---|
| All Enterobacteriaceae | 177/198 (89.4) | 157/208 (75.5) |
| Escherichia coli | 120/128 (93.8) | 106/142 (74.6) |
| Klebsiella pneumoniae | 27/33 (81.8) | 32/43 (74.4) |
| Proteus mirabilis | 9/11 (81.8) | 4/7 (57.1) |
| Enterobacter cloacae | 13/16 (81.3) | 3/3 (100.0) |
There were 52 baseline Enterobacteriaceae isolates in 51/189 (27%) patients in the ZEMDRI group that were non-susceptible (defined as intermediate or resistant) to gentamicin, or tobramycin or both. All of these isolates were susceptible to plazomicin and all but one was susceptible to amikacin (one isolate was intermediate to amikacin). The microbiological eradication rate at the TOC visit in this subset was 78.9% (41/52) in the ZEMDRI group. Note that certain resistance mechanisms can confer resistance to all aminoglycosides, including plazomicin [see Microbiology (12.4)].
1.1 Complicated Urinary Tract Infections (cuti), Including Pyelonephritis
ZEMDRI is indicated in patients 18 years of age or older for the treatment of complicated urinary tract infections (cUTI), including pyelonephritis caused by the following susceptible microorganism(s): Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, and Enterobacter cloacae.
As only limited clinical safety and efficacy data for ZEMDRI are currently available, reserve ZEMDRI for use in cUTI patients who have limited or no alternative treatment options [see Clinical Studies (14.1)].
Warning: Nephrotoxicity, Ototoxicity, Neuromuscular Blockade and Fetal Harm
- Nephrotoxicity has been reported with ZEMDRI. The risk of nephrotoxicity is greater in patients with impaired renal function, the elderly, and in those receiving concomitant nephrotoxic medications. Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy [see Dosage and Administration (2.2) and Warnings and Precautions (5.1)] . Therapeutic Drug Monitoring (TDM) is recommended for complicated urinary tract infection (cUTI) patients with CLcr less than 90 mL/min to avoid potentially toxic levels [see Dosage and Administration (2.3, 2.4)] .
- Ototoxicity, manifested as hearing loss, tinnitus, and/or vertigo, has been reported with ZEMDRI. Symptoms of aminoglycoside-associated ototoxicity may be irreversible and may not become evident until after completion of therapy. Aminoglycoside-associated ototoxicity has been observed primarily in patients with a family history of hearing loss, patients with renal impairment, and in patients receiving higher doses and/or longer durations of therapy than recommended [see Warnings and Precautions (5.2)].
- Aminoglycosides have been associated with neuromuscular blockade. During therapy with ZEMDRI, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients, such as patients with underlying neuromuscular disorders (including myasthenia gravis) or in patients concomitantly receiving neuromuscular blocking agents [see Warning and Precautions (5.3)] .
- Aminoglycosides, including ZEMDRI, can cause fetal harm when administered to a pregnant woman [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)] .
Structured Label Content
Section 42229-5 (42229-5)
Adverse Reactions Leading to Treatment Discontinuations in Trial 1
In Trial 1, treatment discontinuation from IV study drug due to an adverse reaction occurred in 2.0% of patients receiving ZEMDRI (6/303) and meropenem (6/301), respectively.
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ZEMDRI and other antibacterial drugs, ZEMDRI should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10. Overdosage (10. OVERDOSAGE)
In the event of overdosage, ZEMDRI should be discontinued and supportive care is advised. Maintenance of glomerular filtration and careful monitoring of renal function is recommended. Hemodialysis may aid in the removal of ZEMDRI from the blood, especially if renal function is, or becomes, compromised. No clinical information is available on the use of hemodialysis to treat ZEMDRI overdosage.
15. References (15. REFERENCES)
- American Speech-Language-Hearing Association. (1994). Audiologic management of individuals receiving cochleotoxic drug therapy [Guidelines]. Available from www.asha.org/policy.
5.4 Fetal Harm
Aminoglycosides, including ZEMDRI, can cause fetal harm when administered to a pregnant woman. Aminoglycosides cross the placenta, and streptomycin has been associated with several reports of total, irreversible, bilateral congenital deafness in pediatric patients exposed in utero. Patients who use ZEMDRI during pregnancy, or become pregnant while taking ZEMDRI should be apprised of the potential hazard to the fetus [see Use in Specific Populations (8.1)].
11. Description (11. DESCRIPTION)
ZEMDRI contains plazomicin sulfate, a semi-synthetic aminoglycoside antibacterial derived from sisomicin. The chemical name of plazomicin sulfate is (2"R,3"R,4"R,5"R)-2"-[(1S,2S,3R,4S,6R)-4-amino-6-[(2'"S)-4'"-amino-2'"-hydroxybutanamido)amino]-3-[(2'S,3'R)-3'-amino-6'-((2-hydroxyethylamino)methyl)-3',4'-dihydro-2H-pyran-2'-yloxy]-2-hydroxycyclohexyloxy]-5''-methyl-4''-(methylamino)tetrahydro-2H-pyran-3'',5''-diol sulfate. Plazomicin sulfate contains a theoretical 2.5 molar equivalents of sulfate relative to the freebase, based on complete protonation. The molecular weight of plazomicin sulfate is calculated based on 1:2.5 stoichiometry. The corresponding empirical formula is C25H48N6O10∙2.5 H2SO4 (plazomicin sulfate) and the molecular weight of the plazomicin sulfate salt is 837.89 g/mol and the molecular weight of the freebase is 592.69 g/mol.
Figure 1: Chemical Structure of Plazomicin Sulfate
ZEMDRI injection 500 mg/10 mL is a sterile, clear, colorless-to-yellow liquid for intravenous administration supplied in 10-mL single-dose Type 1 glass vials. Each vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase at a concentration of 50 mg/mL adjusted to pH 6.5. Each vial also contains Water for Injection and sodium hydroxide for pH adjustment. This sterile, nonpyrogenic solution is formulated without preservatives.
5.2 Ototoxicity
Ototoxicity, manifested as hearing loss, tinnitus, and/or vertigo, has been reported with ZEMDRI. Symptoms of aminoglycoside-associated ototoxicity may be irreversible and may not become evident until after completion of therapy.
Regarding the incidence of adverse reactions associated with cochlear or vestibular function, in Trial 1, there was one case of reversible hypoacusis (1/303;0.3%) in ZEMDRI-treated patients and one case of tinnitus (1/301;0.3%) in meropenem-treated patients [see Adverse Reactions (6.1)]. In Trial 2, one case each of irreversible tinnitus and reversible vertigo was reported in ZEMDRI-treated patients, and one case of an abnormal audiogram occurred in a levofloxacin-treated patient [see Adverse Reactions (6.1)].
Aminoglycoside-associated ototoxicity has been observed primarily in patients with a family history of hearing loss (excluding age-related hearing loss), patients with renal impairment, and in patients receiving higher doses and/or for longer periods than recommended. In Trial 1 and Trial 2, patients with a history of hearing loss, with the exception of age-related hearing loss, were excluded. The benefit-risk of ZEMDRI therapy should be considered in these patients.
16.1 How Supplied
ZEMDRI injection 500 mg/10 mL (50 mg/mL) is supplied in single-dose, 10-mL vials fitted with flip-off seals with royal blue polypropylene buttons as a clear, colorless to yellow, sterile solution. Each vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase at a concentration of 50 mg/mL plazomicin in Water for Injection. Each vial contains sodium hydroxide for pH adjustment to 6.5. The solution may become yellow in color; this does not indicate a decrease in potency.
| NDC number | Package/Volume | Units per carton | Plazomicin content |
|---|---|---|---|
| 71045-010-02 | Single use, fliptop vial, 10-mL | 10 | 500 mg in 10 mL (50 mg/mL) |
8.5 Geriatric Use
Of the 425 patients treated with ZEMDRI in Trials 1 and 2, 40% (170/425) were 65 years of age and older, including 17.2% (73/425) patients 75 years of age and older. In Trial 1, for ZEMDRI- treated patients ≥ 65 years old, the incidence rate of adverse reactions was 27% (37/137) versus 18.9% (27/143) in the meropenem-treated patients ≥ 65 years old. For ZEMDRI- treated patients < 65 years old, the incidence rate of adverse reactions was 13.3% (22/166) versus 24.1% (38/158) in the meropenem-treated patients < 65 years old.
The rate of adverse reactions associated with renal function for the ZEMDRI-treated patients ≥ 65 years old was 6.6% (9/137) versus 2.8% (4/143) in the meropenem-treated patients. For ZEMDRI- treated patients < 65 years old, the incidence rate of adverse reactions associated with renal function was 1.2% (2/166), versus 0% (0/158) in the meropenem-treated patients [see Clinical Studies (14.1) and Adverse Reactions (6.1)].
ZEMDRI is substantially excreted by the kidneys, and the risk of adverse reactions to ZEMDRI may be greater in patients with renal impairment. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and renal function should be monitored. Dosage adjustment in elderly patients should take into account renal function and plazomicin concentrations as appropriate [see Dosage and Administration (2.2, 2.3, 2.4) and Clinical Pharmacology (12.3)].
5.1 Nephrotoxicity
Nephrotoxicity has been reported with the use of ZEMDRI [see Adverse Reactions (6.1)]. Most serum creatinine increases were ≤ 1 mg/dL above baseline and reversible.
In Trial 1, the incidence of adverse reactions associated with renal function (acute kidney injury, serum creatinine increased, chronic kidney disease, creatinine clearance decreased, renal failure, renal impairment) was 3.6% (11/303) in ZEMDRI-treated patients compared with 1.3% (4/301) in meropenem-treated patients [see Adverse Reactions (6.1)].
Serum creatinine increases of 0.5 mg/dL or greater above baseline occurred in 7% (21/300) of ZEMDRI-treated patients compared with 4% (12/297) of meropenem-treated patients. These increases mainly occurred in patients with CLcr ≤ 90 mL/min and were associated with a plazomicin trough level (Cmin) greater than or equal to 3 mcg/mL [see Adverse Reactions (6.1) and Clinical Pharmacology (12.2)].
Assess CLcr in all patients prior to initiating therapy and daily during therapy with ZEMDRI, particularly in those at increased risk of nephrotoxicity, such as those with renal impairment, the elderly, and those receiving concomitant potentially nephrotoxic medications. In the setting of worsening renal function, the benefit of continuing ZEMDRI should be assessed [see Dosage and Administration (2.2, 2.4), Adverse Reactions (6.1) and Use in Specific Populations (8.5, 8.6)].
Adjust the initial dosage regimen in cUTI patients with CLcr ≥ 15 mL/min and < 60 mL/min [see Dosage and Administration (2.3)]. For subsequent doses, TDM is recommended for patients with CLcr ≥15 mL/min and < 90 mL/min [see Dosage and Administration (2.4)].
8.4. Pediatric Use
The safety and effectiveness of ZEMDRI in patients less than 18 years of age have not been established.
4. Contraindications (4. CONTRAINDICATIONS)
ZEMDRI is contraindicated in patients with known hypersensitivity to any aminoglycoside [see Warnings and Precautions (5.5)].
6. Adverse Reactions (6. ADVERSE REACTIONS)
The following important adverse reactions are discussed in greater detail in the Warnings and Precautions section:
- Nephrotoxicity [see Warnings and Precautions (5.1)]
- Ototoxicity [see Warnings and Precautions (5.2)]
- Neuromuscular Blockade [see Warnings and Precautions (5.3)]
- Fetal Harm [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
- Clostridium difficile-Associated Diarrhea [see Warnings and Precautions (5.6)]
8.6 Renal Impairment
Plazomicin total body clearance was significantly decreased in patients with CLcr greater than or equal to 15 to less than 60 mL/min compared to patients with CLcr greater than or equal to 60 mL/min [see Clinical Pharmacology (12.3)]. Monitor CLcr daily and adjust ZEMDRI dosage accordingly [see Dosage and Administration (2.2)]. There is insufficient information to recommend a dosage regimen in patients with CLcr less than 15 mL/min or on renal replacement therapy, including hemodialysis or continuous renal replacement therapy.
For patients with CLcr greater than or equal to 15 mL/min and less than 90 mL/min, TDM is recommended. Monitor plazomicin trough concentrations and adjust ZEMDRI dosage accordingly [see Dosage and Administration (2.3, 2.4)].
12.2 Pharmacodynamics
The ratio of area under the plasma concentration-time curve to the minimum inhibitory concentration (AUC:MIC) for plazomicin has been shown to best correlate with efficacy in animal and in vitro models of infection against Enterobacteriaceae.
12.3 Pharmacokinetics
The pharmacokinetic (PK) parameters of plazomicin are similar for single- and multiple-dose administration of ZEMDRI in healthy subjects. No appreciable accumulation of plazomicin was observed following multiple IV infusions of 15 mg/kg administered every 24 hours in subjects with normal renal function. The AUC, maximum plasma concentration (Cmax), and Cmin increased in proportion to the dose over the dose range of 4 to 15 mg/kg. The plazomicin AUC, Cmax, and Cmin are summarized in Table 4.
| Healthy Subjects PK parameters following a single dose of 15 mg/kg; Based on non-compartmental analysis of PK data; AUC0-inf is reported; Cmin is concentration at 24 hours.
Geometric mean (±SD) N=54 |
cUTI Patients Day 1 PK parameters following administration of 15 mg/kg; Derived based on population PK model; AUC0-24h is reported.
Geometric mean (±SD) N=87 |
|
|---|---|---|
| AUC (mcg∙h/mL) | 257 (±67.0) | 226 (±113) |
| Cmax (mcg/mL) | 73.7 (±19.7) | 51.0 (±26.7) |
| Cmin (mcg/mL) | 0.3 (±0.2) | 0.5 (±1.2) |
2.1 Recommended Dosage
The recommended dosage regimen of ZEMDRI is 15 mg/kg administered every 24 hours by intravenous (IV) infusion over 30 minutes in patients 18 years of age or older and with creatinine clearance (CLcr) greater than or equal to 90 mL/min (Table 1). The duration of therapy should be guided by the severity of infection and the patient's clinical status for up to 7 days. During treatment, dosage adjustments may be required based on change in renal function [see Dosage and Administration (2.3, 2.4)].
| cUTI Infection | Dosage Regimen Calculate dosage using TBW. For patients with TBW greater than IBW by 25% or more, use adjusted body weight based on the equation: Adjusted body weight = IBW + 0.4 × [TBW – IBW].
|
Duration of Treatment |
|---|---|---|
| Complicated Urinary Tract Infections, including Pyelonephritis | 15 mg/kg every 24 hours | 4 to 7 days An appropriate oral therapy may be considered after 4 to 7 days of ZEMDRI therapy to complete a total duration of 7 to 10 days (IV plus oral). The maximum duration of ZEMDRI for cUTI is 7 days.
|
2.7 Drug Compatibility
Compatibility of ZEMDRI for administration with other drugs has not been established. ZEMDRI should not be mixed with other drugs or physically added to solutions containing other drugs. Other medications should not be infused simultaneously with ZEMDRI through the same IV line.
1. Indications and Usage (1. INDICATIONS AND USAGE)
ZEMDRI is an aminoglycoside antibacterial indicated for the treatment of patients 18 years of age or older with Complicated Urinary Tract Infections (cUTI) including Pyelonephritis. (1.1)
As only limited clinical safety and efficacy data are available, reserve ZEMDRI for use in patients who have limited or no alternative treatment options. (1.1)
To reduce the development of drug-resistant bacteria and maintain effectiveness of ZEMDRI and other antibacterial drugs, ZEMDRI should be used only to treat infections that are proven or strongly suspected to be caused by susceptible microorganisms. (1.2)
12.1 Mechanism of Action
ZEMDRI is an antibacterial drug [see Microbiology (12.4)].
16.2 Storage and Handling
Store ZEMDRI injection 500 mg/10 mL (50 mg/mL) refrigerated at 2°C to 8°C (36°F to 46°F).
5.6 Clostridium Difficile (5.6 Clostridium difficile)
Clostridium difficile-associated diarrhea (CDAD) has been reported for nearly all systemic antibacterial drugs and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial drugs alters the normal flora of the colon and may permit overgrowth of C. difficile.
C. difficile produces toxins A and B that contribute to the development of CDAD. Hypertoxin-producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary because CDAD has been reported to occur more than 2 months after the administration of antibacterial drugs.
If CDAD is suspected or confirmed, antibacterial drugs not directed against C. difficile may need to be discontinued. Manage fluid and electrolyte levels as appropriate, supplement protein intake, monitor antibacterial treatment of C. difficile, and institute surgical evaluation as clinically indicated.
5.3 Neuromuscular Blockade
Aminoglycosides have been associated with exacerbation of muscle weakness in patients with underlying neuromuscular disorders, or delay in recovery of neuromuscular function in patients receiving concomitant neuromuscular blocking agents.
During therapy with ZEMDRI, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients, such as patients with underlying neuromuscular disorders (including myasthenia gravis) or those patients concomitantly receiving neuromuscular blocking agents.
5. Warnings and Precautions (5. WARNINGS AND PRECAUTIONS)
2. Dosage and Administration (2. DOSAGE AND ADMINISTRATION)
- Administer ZEMDRI 15 mg/kg every 24 hours by intravenous (IV) infusion over 30 minutes to patients 18 years of age or older with creatinine clearance greater than or equal to 90 mL/min. (2.1)
- Recommended duration of treatment is 4 to 7 days for cUTI, including pyelonephritis. (2.1)
- Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy. (2.2)
- Recommended initial dosage regimen for patients with renal impairment is shown in the table below. (2.3)
| Estimated CLcr CLcr estimated by the Cockcroft-Gault formula. (2.3) (mL/min) |
Recommended Dosage for ZEMDRI Calculate dosage using Total Body Weight (TBW). For patients with TBW greater than IBW by 25% or more, use adjusted body weight. (2.3)
|
Dosing Interval |
|---|---|---|
| Greater than or equal to 60 to less than 90 | 15 mg/kg | Every 24 hours |
| Greater than or equal to 30 to less than 60 |
10 mg/kg | Every 24 hours |
| Greater than or equal to 15 to less than 30 |
10 mg/kg | Every 48 hours |
- See Full Prescribing Information for subsequent dosage adjustment based on changes in renal function or Therapeutic Drug Monitoring (TDM). (2.3, 2.4).
- See Full Prescribing Information for instructions on preparation of the solution, stability in intravenous fluids and drug compatibilities. (2.5, 2.6, 2.7)
3. Dosage Forms and Strengths (3. DOSAGE FORMS AND STRENGTHS)
ZEMDRI injection 500 mg/10 mL (50 mg/mL) is a sterile, clear, colorless to yellow solution supplied in a single-dose vial. Each single-dose vial contains plazomicin sulfate equivalent to 500 mg plazomicin freebase.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be compared directly to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
ZEMDRI was evaluated in two comparator-controlled clinical trials (Trial 1, NCT02486627 and Trial 2, NCT01096849) in patients with cUTI, including pyelonephritis. In both trials, patients with CLcr greater than 60 mL/min received ZEMDRI 15 mg/kg IV once daily as a 30-minute infusion [see Clinical Studies (14.1)].
Trial 1 included 303 patients treated with ZEMDRI and 301 patients treated with meropenem. Patients were to receive 4 to 7 days of ZEMDRI (mean duration of 5.1 days). In some patients, parenteral therapy was followed by a switch to an oral antibacterial drug.
The median age of patients treated with ZEMDRI in Trial 1 was 62 years (range 18 to 90 years) and 45.2% of patients were 65 years of age or older. Patients treated with ZEMDRI were predominantly female (56.1%) and White (99.3%). A majority of patients (68.0%) had mild or moderate renal impairment (CLcr >30 to 90 mL/min) at baseline. Patients with CLcr of 30 mL/min or less were excluded.
5.5 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving aminoglycoside antibacterial drugs. Before therapy with ZEMDRI is instituted, careful inquiry about previous hypersensitivity reactions to other aminoglycosides should be made. A history of hypersensitivity to other aminoglycosides is a contraindication to the use of ZEMDRI, because cross-sensitivity among aminoglycoside antibacterial drugs has been established. Discontinue ZEMDRI if an allergic reaction occurs.
2.2 Monitoring of Renal Function
Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy with ZEMDRI [see Dosage and Administration (2.3),Warnings and Precautions (5.1) and Use in Specific Populations (8.6)].
5.7 Development of Drug Resistant Bacteria (5.7 Development of Drug-Resistant Bacteria)
Prescribing ZEMDRI in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Principal Display Panel 10 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton)
NDC 71045-010-02
Rx ONLY
ZEMDRI™
(plazomicin) Injection
10 (10 mL) Single-dose vials
500 mg/10 mL per vial (50 mg/mL)
For Intravenous Infusion Only
Must dilute before use
2.4 Tdm in Cuti Patients With Renal Impairment (2.4 TDM in cUTI Patients With Renal Impairment)
For cUTI patients with CLcr greater than or equal to 15 mL/min and less than 90 mL/min, TDM is recommended to maintain plasma trough concentrations below 3 mcg/mL. Measure plazomicin plasma trough concentration within approximately 30 minutes before administration of the second dose of ZEMDRI. Adjustment of the ZEMDRI dosage regimen based on TDM involves extending ZEMDRI dosing interval by 1.5 fold (i.e., from every 24 hours to every 36 hours or from every 48 hours to every 72 hours) for patients with plasma trough concentrations greater than or equal to 3 mcg/mL [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2)].
2.5 Preparation of Diluted Solutions of Zemdri (2.5 Preparation of Diluted Solutions of ZEMDRI)
ZEMDRI is supplied as a single-dose fliptop 10-mL vial that contains plazomicin sulfate equivalent to 500 mg plazomicin freebase in 10 mL Water for Injection (concentration of 50 mg/mL). The appropriate volume of ZEMDRI solution (50 mg/mL) for the required dose should be diluted in 0.9% Sodium Chloride Injection, USP or Lactated Ringer's Injection, USP to achieve a final volume of 50 mL for intravenous infusion. The stability of ZEMDRI solution in the compatible diluents is described below [see Dosage and Administration (2.7)].
ZEMDRI does not contain preservatives. Aseptic technique must be followed in preparing the infusion solution. Discard unused portion of the ZEMDRI vial.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
2.3 Dosage in Adult Patients With Renal Impairment
The recommended initial dosage regimen of ZEMDRI in adult patients with CLcr greater than or equal to 15 and less than 90 mL/min, estimated by the Cockcroft-Gault formula, is described in Table 2.
Patients with CLcr greater than or equal to 15 and less than 90 mL/min receiving ZEMDRI may require subsequent dosage adjustments based on change in renal function and/or Therapeutic Drug Monitoring (TDM) as appropriate [see Dosage and Administration (2.4)].
| Estimated CLcr CLcr estimated by the Cockcroft-Gault formula using total body weight (TBW). For patients with TBW greater than ideal body weight (IBW) by 25% or more, use IBW. (mL/min) |
Dosage Calculate dosage using TBW. For patients with TBW greater than IBW by 25% or more, use adjusted body weight based on the equation: Adjusted body weight = IBW + 0.4 × [TBW – IBW].
|
Dosing Interval |
|---|---|---|
| Greater than or equal to 60 to less than 90 | 15 mg/kg | Every 24 hours |
| Greater than or equal to 30 to less than 60 | 10 mg/kg | Every 24 hours |
| Greater than or equal to 15 to less than 30 | 10 mg/kg | Every 48 hours |
There is insufficient information to recommend a dosage regimen in patients with CLcr less than 15 mL/min or on renal replacement therapy, including hemodialysis or continuous renal replacement therapy.
2.6 Stability of Zemdri Solution in Intravenous Fluids (2.6 Stability of ZEMDRI Solution in Intravenous Fluids)
After dilution, ZEMDRI solution for administration is stable for 24 hours at room temperature at concentrations of 2.5 mg/mL to 45 mg/mL in the following solutions:
- 0.9% Sodium Chloride Injection, USP
- Lactated Ringer's Injection, USP
14.1 Complicated Urinary Tract Infections, Including Pyelonephritis
A total of 609 adults hospitalized with cUTI (including pyelonephritis) were randomized in a multinational, double-blind, noninferiority trial comparing ZEMDRI (15 mg/kg IV once daily as a 30-minute infusion) to meropenem (1 g intravenously every 8 hours as a 30-minute infusion) (Trial 1, NCT02486627). Switch to an oral antibacterial drug, such as levofloxacin, was allowed after a minimum of 4 and maximum of 7 days of IV therapy for a total of 7 to 10 days of treatment.
Efficacy was assessed in the microbiological modified intent-to-treat (mMITT) population, which included all patients who received study medication and had at least 1 baseline uropathogen. The mMITT population excluded patients with organisms resistant to study drugs. Patient demographic and baseline characteristics were balanced between treatment groups in the mMITT population. The mMITT population consisted of 388 patients with cUTI, including 162 (41.8%) with pyelonephritis. The median age was 64 years, 52.8% were female and 99.5% were White. The majority of the patients (99%) were from Eastern Europe; 3 patients were from the United States. Concomitant bacteremia was identified in 25 (13.1%) and 23 (11.7%) patients at baseline in the ZEMDRI and meropenem groups, respectively. The median treatment duration of IV study drug was 6 days in both groups.
ZEMDRI demonstrated efficacy for composite cure at Day 5 and the Test of Cure (TOC) visit (Table 5). Composite cure at Day 5 was defined as resolution or improvement of clinical cUTI symptoms and a microbiological outcome of eradication (all baseline uropathogens reduced to <104 colony-forming units [CFU]/mL). Composite cure at the TOC visit (Day 17 ± 2 from the first dose of study drug) was defined as resolution of clinical cUTI symptoms and a microbiological outcome of eradication.
| Analysis Visit | ZEMDRI n/N (%) |
Meropenem n/N (%) |
Treatment Difference Treatment difference is ZEMDRI – meropenem.
(95% CI) |
|---|---|---|---|
| Abbreviations: CI=confidence interval; TOC=test-of-cure; CI=95% confidence interval based on Newcombe method with continuity correction. | |||
| Day 5 | 168/191 (88.0) | 180/197 (91.4) | -3.4 (-10.0, 3.1) |
| Clinical cure or improvement | 171/191 (89.5) | 182/197 (92.4) | |
| Microbiological eradication | 188/191 (98.4) | 193/197 (98.0) | |
| TOC | 156/191 (81.7) | 138/197 (70.1) | 11.6 (2.7, 20.3) |
| Clinical Cure | 170/191 (89.0) | 178/197 (90.4) | |
| Microbiological eradication | 171/191 (89.5) | 147/197 (74.6) |
Microbiological eradication rates at the TOC visit by baseline uropathogen in the mMITT population are presented in Table 6. Composite Cure at the TOC visit in individuals with concomitant bacteremia at baseline was achieved in 72.0% (18/25) of patients in the ZEMDRI group and 56.5% (13/23) of patients in the meropenem group.
| Pathogen | ZEMDRI n/N (%) |
Meropenem n/N (%) |
|---|---|---|
| All Enterobacteriaceae | 177/198 (89.4) | 157/208 (75.5) |
| Escherichia coli | 120/128 (93.8) | 106/142 (74.6) |
| Klebsiella pneumoniae | 27/33 (81.8) | 32/43 (74.4) |
| Proteus mirabilis | 9/11 (81.8) | 4/7 (57.1) |
| Enterobacter cloacae | 13/16 (81.3) | 3/3 (100.0) |
There were 52 baseline Enterobacteriaceae isolates in 51/189 (27%) patients in the ZEMDRI group that were non-susceptible (defined as intermediate or resistant) to gentamicin, or tobramycin or both. All of these isolates were susceptible to plazomicin and all but one was susceptible to amikacin (one isolate was intermediate to amikacin). The microbiological eradication rate at the TOC visit in this subset was 78.9% (41/52) in the ZEMDRI group. Note that certain resistance mechanisms can confer resistance to all aminoglycosides, including plazomicin [see Microbiology (12.4)].
1.1 Complicated Urinary Tract Infections (cuti), Including Pyelonephritis (1.1 Complicated Urinary Tract Infections (cUTI), including Pyelonephritis)
ZEMDRI is indicated in patients 18 years of age or older for the treatment of complicated urinary tract infections (cUTI), including pyelonephritis caused by the following susceptible microorganism(s): Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, and Enterobacter cloacae.
As only limited clinical safety and efficacy data for ZEMDRI are currently available, reserve ZEMDRI for use in cUTI patients who have limited or no alternative treatment options [see Clinical Studies (14.1)].
Warning: Nephrotoxicity, Ototoxicity, Neuromuscular Blockade and Fetal Harm (WARNING: NEPHROTOXICITY, OTOTOXICITY, NEUROMUSCULAR BLOCKADE and FETAL HARM)
- Nephrotoxicity has been reported with ZEMDRI. The risk of nephrotoxicity is greater in patients with impaired renal function, the elderly, and in those receiving concomitant nephrotoxic medications. Assess creatinine clearance in all patients prior to initiating therapy and daily during therapy [see Dosage and Administration (2.2) and Warnings and Precautions (5.1)] . Therapeutic Drug Monitoring (TDM) is recommended for complicated urinary tract infection (cUTI) patients with CLcr less than 90 mL/min to avoid potentially toxic levels [see Dosage and Administration (2.3, 2.4)] .
- Ototoxicity, manifested as hearing loss, tinnitus, and/or vertigo, has been reported with ZEMDRI. Symptoms of aminoglycoside-associated ototoxicity may be irreversible and may not become evident until after completion of therapy. Aminoglycoside-associated ototoxicity has been observed primarily in patients with a family history of hearing loss, patients with renal impairment, and in patients receiving higher doses and/or longer durations of therapy than recommended [see Warnings and Precautions (5.2)].
- Aminoglycosides have been associated with neuromuscular blockade. During therapy with ZEMDRI, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients, such as patients with underlying neuromuscular disorders (including myasthenia gravis) or in patients concomitantly receiving neuromuscular blocking agents [see Warning and Precautions (5.3)] .
- Aminoglycosides, including ZEMDRI, can cause fetal harm when administered to a pregnant woman [see Warnings and Precautions (5.4), Use in Specific Populations (8.1)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:17.960518 · Updated: 2026-03-14T21:40:11.544958