Vagical
0ab35c03-942e-4ba5-ba5a-65c5a99830d7
34390-5
HUMAN OTC DRUG LABEL
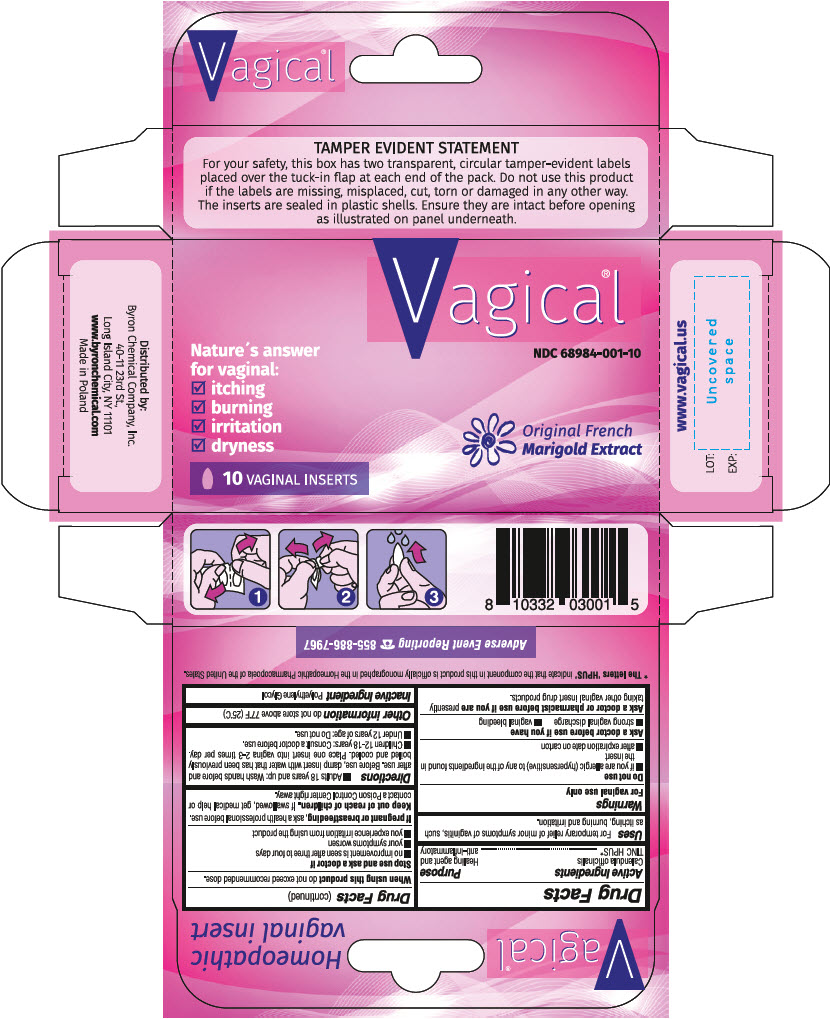
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Calendula officinalis TINC HPUS* *The letters 'HPUS' indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States.
Purpose
Healing agent and anti-inflammatory
Medication Information
Purpose
Healing agent and anti-inflammatory
Description
Calendula officinalis TINC HPUS* *The letters 'HPUS' indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States.
Uses
For temporary relief of minor symptoms of vaginitis, such as itching, burning and irritation.
Section 50567-7
When using this productdo not exceed recommended dose.
Section 50568-5
Ask a doctor or pharmacist before use if you arepresently taking other vaginal insert drug products
Section 53414-9
If pregnant or breastfeeding,ask a health professional before use.
Warnings
For vaginal use only
Directions
- Adults 18 years and up: Wash hands before and after use. Before use, damp insert with water that has been previously boiled and cooled. Place one insert into vagina 2-3 times per day.
- Children 12-18 years: Consult a doctor before use.
- Under 12 years of age: Do not use
Do Not Use
- if you are allergic (hypersensitive) to any of the ingredients found in the insert
- after expiration date on carton
Active Ingredient
Calendula officinalis TINC HPUS*
*The letters 'HPUS' indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States.
Other Information
do not store above 77 degrees F (25 degrees C)
Inactive Ingredient
Polyethylene Glycol.
Stop Use and Ask A Doctor If
- no improvement is seen after three to four days
- your symptoms worsen
- you experience irritation from using the product
Keep Out of the Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have
- strong vaginal discharge
- vaginal bleeding
Principal Display Panel 10 Insert Box
Vagical ®
NDC 68984-001-10
Nature's answer
for vaginal:
- itching
- burning
- irritation
- dryness
Original French
Marigold Extract
10 VAGINAL INSERTS
Structured Label Content
Uses
For temporary relief of minor symptoms of vaginitis, such as itching, burning and irritation.
Section 50567-7 (50567-7)
When using this productdo not exceed recommended dose.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you arepresently taking other vaginal insert drug products
Section 53414-9 (53414-9)
If pregnant or breastfeeding,ask a health professional before use.
Purpose
Healing agent and anti-inflammatory
Warnings
For vaginal use only
Directions
- Adults 18 years and up: Wash hands before and after use. Before use, damp insert with water that has been previously boiled and cooled. Place one insert into vagina 2-3 times per day.
- Children 12-18 years: Consult a doctor before use.
- Under 12 years of age: Do not use
Do Not Use (Do not use)
- if you are allergic (hypersensitive) to any of the ingredients found in the insert
- after expiration date on carton
Active Ingredient (Active ingredient)
Calendula officinalis TINC HPUS*
*The letters 'HPUS' indicate that the component in this product is officially monographed in the Homeopathic Pharmacopoeia of the United States.
Other Information (Other information)
do not store above 77 degrees F (25 degrees C)
Inactive Ingredient (Inactive ingredient)
Polyethylene Glycol.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- no improvement is seen after three to four days
- your symptoms worsen
- you experience irritation from using the product
Keep Out of the Reach of Children. (Keep out of the reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- strong vaginal discharge
- vaginal bleeding
Principal Display Panel 10 Insert Box (PRINCIPAL DISPLAY PANEL - 10 Insert Box)
Vagical ®
NDC 68984-001-10
Nature's answer
for vaginal:
- itching
- burning
- irritation
- dryness
Original French
Marigold Extract
10 VAGINAL INSERTS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:56.954132 · Updated: 2026-03-14T23:17:15.281880