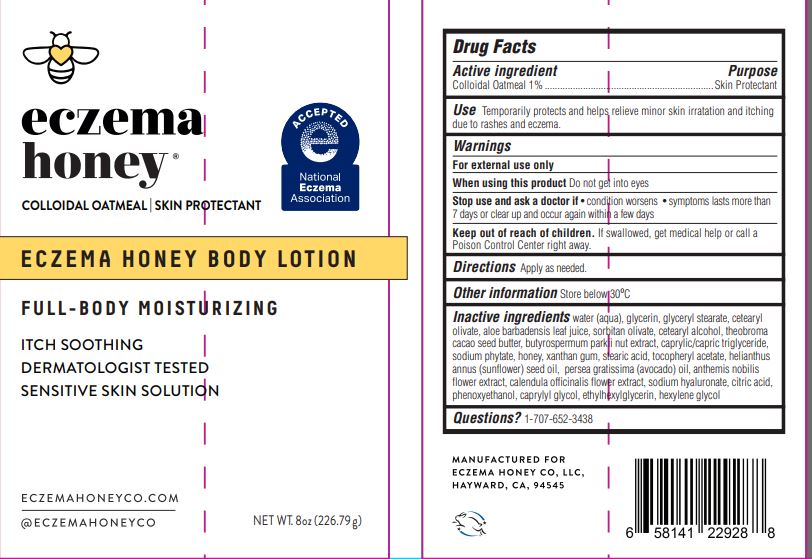
drug facts
0a710be0-562e-8368-e063-6294a90a3b1b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Colloidal Oatmeal 1%
Medication Information
Warnings and Precautions
For external use only
Active Ingredient
Active ingredient
Colloidal Oatmeal 1%
Indications and Usage
Use Temporarily protects and helps relieve minor skin irritation and itching due to rashes and eczema
Dosage and Administration
Directions Apply as needed.
Description
Active ingredient Colloidal Oatmeal 1%
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or call a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if • condition worsens • symptoms lasts more than 7 days or clear up and occur again within a few days
Section 50567-7
When using this product Do not get into eyes
Section 50570-1
Do not use if condition worsens.
Section 51727-6
Inactive ingredients
water (aqua), glycerin, glyceryl stearate, cetearyl olivate, aloe barbadensis leaf juice, soritan olivate, cetearyl alcohol, theobroma cacao seed butter, butyrospermum parkii nut extract, caprylic/capric triglyceride, sodium phytate, honey, xanthan gum, stearic acid, tocopheryl acetate, helianthus annus (sunflower) seed oil, persea gratissima (avocado) oil, anthemis nobilis flower extract, calendula officinalis flower extract, sodium hyaluronate, citric acid, phenoxyethanol, caprylyl glycol, ethylhexylglycerin, hexylene glycol
Section 51945-4
Section 55105-1
Purpose - Skin Protectant
Section 60561-8
Other information Store below 30°C
Structured Label Content
Warnings and Precautions (34071-1)
For external use only
Active Ingredient (55106-9)
Active ingredient
Colloidal Oatmeal 1%
Indications and Usage (34067-9)
Use Temporarily protects and helps relieve minor skin irritation and itching due to rashes and eczema
Dosage and Administration (34068-7)
Directions Apply as needed.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or call a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if • condition worsens • symptoms lasts more than 7 days or clear up and occur again within a few days
Section 50567-7 (50567-7)
When using this product Do not get into eyes
Section 50570-1 (50570-1)
Do not use if condition worsens.
Section 51727-6 (51727-6)
Inactive ingredients
water (aqua), glycerin, glyceryl stearate, cetearyl olivate, aloe barbadensis leaf juice, soritan olivate, cetearyl alcohol, theobroma cacao seed butter, butyrospermum parkii nut extract, caprylic/capric triglyceride, sodium phytate, honey, xanthan gum, stearic acid, tocopheryl acetate, helianthus annus (sunflower) seed oil, persea gratissima (avocado) oil, anthemis nobilis flower extract, calendula officinalis flower extract, sodium hyaluronate, citric acid, phenoxyethanol, caprylyl glycol, ethylhexylglycerin, hexylene glycol
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose - Skin Protectant
Section 60561-8 (60561-8)
Other information Store below 30°C
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:22.528710 · Updated: 2026-03-14T23:01:51.692931