These Highlights Do Not Include All The Information Needed To Use Vevye ®
0a60fccf-7e1b-44fe-e063-6394a90aadd5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.2 ) 9/2025
Indications and Usage
VEVYE indicated for the treatment of the signs and symptoms of dry eye disease.
Dosage and Administration
Instill one drop of VEVYE twice a day in each eye approximately 12 hours apart. ( 2 )
Warnings and Precautions
Care should be taken to not touch the eye or other surfaces with the bottle tip to avoid potential for eye injury and/or contamination. ( 5.1 )
Contraindications
None.
Adverse Reactions
The most common adverse reaction following the use of VEVYE were instillation site reactions (8%). ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Harrow at 1-833-4HARROW(427769) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
VEVYE is a sterile, clear, colorless non-aqueous ophthalmic solution packaged in multiple-dose eye drop bottles delivering single drops of approximately 0.01 mL volume. Each unit contains 2 mL of VEVYE in a 5 mL transparent squeezable polypropylene bottle with a transparent polyethylene tip and a white polyethylene cap with tamper-evident ring. NDC 82667-900-02
How Supplied
VEVYE is a sterile, clear, colorless non-aqueous ophthalmic solution packaged in multiple-dose eye drop bottles delivering single drops of approximately 0.01 mL volume. Each unit contains 2 mL of VEVYE in a 5 mL transparent squeezable polypropylene bottle with a transparent polyethylene tip and a white polyethylene cap with tamper-evident ring. NDC 82667-900-02
Medication Information
Warnings and Precautions
Care should be taken to not touch the eye or other surfaces with the bottle tip to avoid potential for eye injury and/or contamination. ( 5.1 )
Indications and Usage
VEVYE indicated for the treatment of the signs and symptoms of dry eye disease.
Dosage and Administration
Instill one drop of VEVYE twice a day in each eye approximately 12 hours apart. ( 2 )
Contraindications
None.
Adverse Reactions
The most common adverse reaction following the use of VEVYE were instillation site reactions (8%). ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Harrow at 1-833-4HARROW(427769) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Storage and Handling
VEVYE is a sterile, clear, colorless non-aqueous ophthalmic solution packaged in multiple-dose eye drop bottles delivering single drops of approximately 0.01 mL volume. Each unit contains 2 mL of VEVYE in a 5 mL transparent squeezable polypropylene bottle with a transparent polyethylene tip and a white polyethylene cap with tamper-evident ring. NDC 82667-900-02
How Supplied
VEVYE is a sterile, clear, colorless non-aqueous ophthalmic solution packaged in multiple-dose eye drop bottles delivering single drops of approximately 0.01 mL volume. Each unit contains 2 mL of VEVYE in a 5 mL transparent squeezable polypropylene bottle with a transparent polyethylene tip and a white polyethylene cap with tamper-evident ring. NDC 82667-900-02
Description
Dosage and Administration ( 2.2 ) 9/2025
Section 42229-5
Risk Summary
There are no adequate and well-controlled studies of VEVYE administration in pregnant women to inform a drug-associated risk. Oral administration of cyclosporine to pregnant rats or rabbits did not produce teratogenicity at clinically relevant doses VEVYE doses are approximately 4,700 times lower than recommended oral doses, with blood concentrations being undetectable after topical administration.
Section 43683-2
| Dosage and Administration ( 2.2) | 9/2025 |
Section 51945-4
Principal Display Panel - 2 mL Carton Label
NDC: 82667-900-02 Rx Only
vevye ®
(cyclosporine ophthalmic
soliton) 0.1%
For topical application
In the eye
Sterile, Non-Preserved
2 mL
11 Description
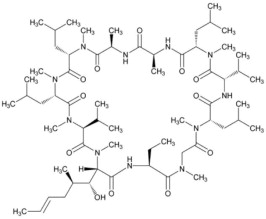
VEVYE (cyclosporine ophthalmic solution) 0.1% contains the immunomodulatory agent cyclosporine. Cyclosporine's chemical name is Cyclo[[€-(2S,3R,4R)-3-hydroxy-4-methyl-2-(methylamino)-6-octenoyl]-L-2-aminobutyryl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucyl-L-alanyl-D-alanyl-N-methyl-L-leucyl-N-methyl-L-leucyl-N-methyl-L-valyl] and it has the following structure:
Structural Formula
Formula: C 62H 111N 11O 12, Molecular weight: 1202.6
Cyclosporine is a white powder that is solubilized in perfluorobutylpentane, a semi-fluorinated alkane vehicle. VEVYE is supplied as a 2 mL sterile, clear, colorless, non-aqueous ophthalmic solution for topical ophthalmic use. VEVYE ®contains:
- Active: cyclosporine 0.1%
- Inactives: perfluorobutylpentane, ethanol (anhydrous).
The solution does not contain water or anti-microbial preservatives. As a water free product, there is no associated pH and no osmolarity.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients below the age of 18 years have not been established.
8.5 Geriatric Use
No overall difference in safety or effectiveness has been observed between elderly and younger patients.
14 Clinical Studies
The safety and efficacy of VEVYE were assessed in a total of 1369 patients with dry eye disease, of which 738 received VEVYE.
In two multicenter, randomized, adequate and well-controlled clinical studies, patients with dry eye disease (CYS-002: NCT02617667 and CYS-004: NCT04523129), treated with VEVYE ®were compared to patients treated with vehicle. At Day 29, there was a statistically significant higher percentage of eyes with increases of ≥ 10 mm from baseline in Schirmer wetting. This effect was seen in approximately 10% of VEVYE-treated patients versus approximately 6% of vehicle-treated patients.
| CYS-002 Day 29 | CYS-004 Day 29 | |||
|
VEVYE
N=51 |
Vehicle
N=51 |
VEVYE
N=409 |
Vehicle
N=395 |
|
| ≥ 10 mm increase in tear production (% patients) | 8% | 0% | 11% | 7% |
| Difference (95% CI) | 7.8% (0.5%, 15.%) | 3.9% (0.02%, 7.8%) | ||
| p-value versus vehicle | 0.04 | 0.05 |
4 Contraindications
None.
6 Adverse Reactions
The most common adverse reaction following the use of VEVYE were instillation site reactions (8%). ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact
Harrow at 1-833-4HARROW(427769) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
12.3 Pharmacokinetics
Following bilateral topical ocular dosing of one drop of VEVYE twice daily, the blood concentrations of cyclosporine were below the limit of quantification (0.1 ng/mL) at all timepoints.
2.1 Recommended Dosage
Instill one drop of VEVYE twice a day in each eye approximately 12 hours apart.
1 Indications and Usage
VEVYE indicated for the treatment of the signs and symptoms of dry eye disease.
12.1 Mechanism of Action
Cyclosporine, a calcineurin inhibitor, is a relatively selective immunomodulatory drug.
5 Warnings and Precautions
Care should be taken to not touch the eye or other surfaces with the bottle tip to avoid potential for eye injury and/or contamination. ( 5.1)
2 Dosage and Administration
Instill one drop of VEVYE twice a day in each eye approximately 12 hours apart. ( 2)
5.2 Use With Contact Lenses
VEVYE should not be administered while wearing contact lenses. If contact lenses are worn, they should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following administration of VEVYE ophthalmic solution.
3 Dosage Forms and Strengths
Clear, colorless non-preserved ophthalmic solution containing cyclosporine 0.1% (1 mg/mL), delivering 0.01 mg of cyclosporine per one drop (0.01 mL).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. In clinical trials with 738 subjects receiving at least 1 dose of VEVYE, the most common adverse reactions were instillation site reactions (8%) and temporary decreases in visual acuity (3%).
2.2 Administration Instructions
Wash hands before using.
Gently pull the lower eyelid downward. Tip the bottle upside down over the eye to allow one drop to dispense on its own into the eye. If a drop does not dispense after a few seconds, gently apply slight pressure to the sides of the bottle while holding over the eye.
Note:You may not feel the drop falling into your eye.
If VEVYE is used with other eye drops, a 15-minute interval between products should occur.
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Instructions for Use).
16 How Supplied/storage and Handling
VEVYE is a sterile, clear, colorless non-aqueous ophthalmic solution packaged in multiple-dose eye drop bottles delivering single drops of approximately 0.01 mL volume. Each unit contains 2 mL of VEVYE in a 5 mL transparent squeezable polypropylene bottle with a transparent polyethylene tip and a white polyethylene cap with tamper-evident ring.
NDC 82667-900-02
5.1 Potential for Eye Injury and Contamination
To avoid the potential for eye injury and/or contamination, patients should not touch the bottle tip to the eye or other surfaces.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There are no adequate and well-controlled studies of VEVYE administration in pregnant women to inform a drug-associated risk. Oral administration of cyclosporine to pregnant rats or rabbits did not produce teratogenicity at clinically relevant doses VEVYE doses are approximately 4,700 times lower than recommended oral doses, with blood concentrations being undetectable after topical administration.
Section 43683-2 (43683-2)
| Dosage and Administration ( 2.2) | 9/2025 |
Section 51945-4 (51945-4)
Principal Display Panel - 2 mL Carton Label
NDC: 82667-900-02 Rx Only
vevye ®
(cyclosporine ophthalmic
soliton) 0.1%
For topical application
In the eye
Sterile, Non-Preserved
2 mL
11 Description (11 DESCRIPTION)
VEVYE (cyclosporine ophthalmic solution) 0.1% contains the immunomodulatory agent cyclosporine. Cyclosporine's chemical name is Cyclo[[€-(2S,3R,4R)-3-hydroxy-4-methyl-2-(methylamino)-6-octenoyl]-L-2-aminobutyryl-N-methylglycyl-N-methyl-L-leucyl-L-valyl-N-methyl-L-leucyl-L-alanyl-D-alanyl-N-methyl-L-leucyl-N-methyl-L-leucyl-N-methyl-L-valyl] and it has the following structure:
Structural Formula
Formula: C 62H 111N 11O 12, Molecular weight: 1202.6
Cyclosporine is a white powder that is solubilized in perfluorobutylpentane, a semi-fluorinated alkane vehicle. VEVYE is supplied as a 2 mL sterile, clear, colorless, non-aqueous ophthalmic solution for topical ophthalmic use. VEVYE ®contains:
- Active: cyclosporine 0.1%
- Inactives: perfluorobutylpentane, ethanol (anhydrous).
The solution does not contain water or anti-microbial preservatives. As a water free product, there is no associated pH and no osmolarity.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients below the age of 18 years have not been established.
8.5 Geriatric Use
No overall difference in safety or effectiveness has been observed between elderly and younger patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of VEVYE were assessed in a total of 1369 patients with dry eye disease, of which 738 received VEVYE.
In two multicenter, randomized, adequate and well-controlled clinical studies, patients with dry eye disease (CYS-002: NCT02617667 and CYS-004: NCT04523129), treated with VEVYE ®were compared to patients treated with vehicle. At Day 29, there was a statistically significant higher percentage of eyes with increases of ≥ 10 mm from baseline in Schirmer wetting. This effect was seen in approximately 10% of VEVYE-treated patients versus approximately 6% of vehicle-treated patients.
| CYS-002 Day 29 | CYS-004 Day 29 | |||
|
VEVYE
N=51 |
Vehicle
N=51 |
VEVYE
N=409 |
Vehicle
N=395 |
|
| ≥ 10 mm increase in tear production (% patients) | 8% | 0% | 11% | 7% |
| Difference (95% CI) | 7.8% (0.5%, 15.%) | 3.9% (0.02%, 7.8%) | ||
| p-value versus vehicle | 0.04 | 0.05 |
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reaction following the use of VEVYE were instillation site reactions (8%). ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact
Harrow at 1-833-4HARROW(427769) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
12.3 Pharmacokinetics
Following bilateral topical ocular dosing of one drop of VEVYE twice daily, the blood concentrations of cyclosporine were below the limit of quantification (0.1 ng/mL) at all timepoints.
2.1 Recommended Dosage
Instill one drop of VEVYE twice a day in each eye approximately 12 hours apart.
1 Indications and Usage (1 INDICATIONS AND USAGE)
VEVYE indicated for the treatment of the signs and symptoms of dry eye disease.
12.1 Mechanism of Action
Cyclosporine, a calcineurin inhibitor, is a relatively selective immunomodulatory drug.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Care should be taken to not touch the eye or other surfaces with the bottle tip to avoid potential for eye injury and/or contamination. ( 5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Instill one drop of VEVYE twice a day in each eye approximately 12 hours apart. ( 2)
5.2 Use With Contact Lenses (5.2 Use with Contact Lenses)
VEVYE should not be administered while wearing contact lenses. If contact lenses are worn, they should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following administration of VEVYE ophthalmic solution.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Clear, colorless non-preserved ophthalmic solution containing cyclosporine 0.1% (1 mg/mL), delivering 0.01 mg of cyclosporine per one drop (0.01 mL).
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. In clinical trials with 738 subjects receiving at least 1 dose of VEVYE, the most common adverse reactions were instillation site reactions (8%) and temporary decreases in visual acuity (3%).
2.2 Administration Instructions
Wash hands before using.
Gently pull the lower eyelid downward. Tip the bottle upside down over the eye to allow one drop to dispense on its own into the eye. If a drop does not dispense after a few seconds, gently apply slight pressure to the sides of the bottle while holding over the eye.
Note:You may not feel the drop falling into your eye.
If VEVYE is used with other eye drops, a 15-minute interval between products should occur.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
VEVYE is a sterile, clear, colorless non-aqueous ophthalmic solution packaged in multiple-dose eye drop bottles delivering single drops of approximately 0.01 mL volume. Each unit contains 2 mL of VEVYE in a 5 mL transparent squeezable polypropylene bottle with a transparent polyethylene tip and a white polyethylene cap with tamper-evident ring.
NDC 82667-900-02
5.1 Potential for Eye Injury and Contamination
To avoid the potential for eye injury and/or contamination, patients should not touch the bottle tip to the eye or other surfaces.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:27.611159 · Updated: 2026-03-14T22:51:04.846555