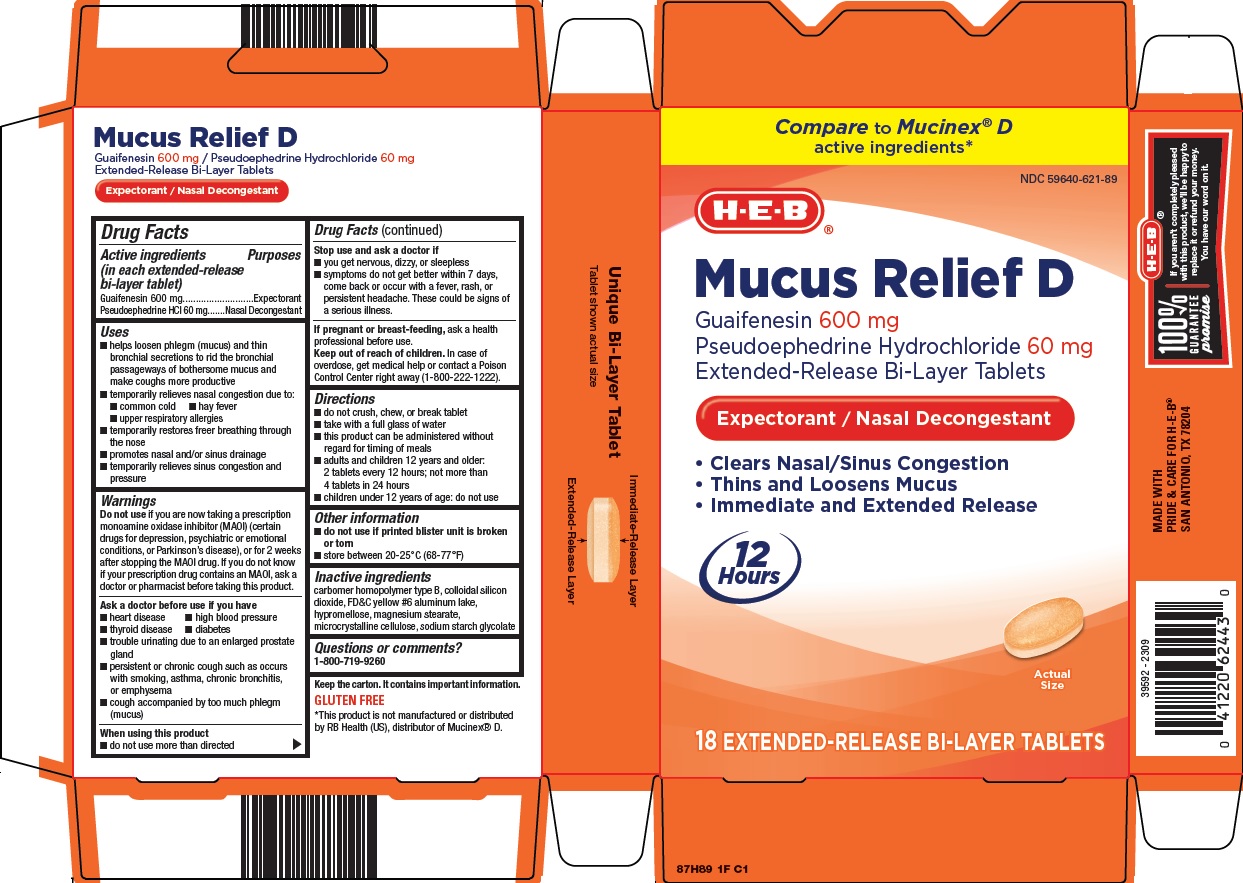
Heb Mucus Relief D Drug Facts
0a2a2822-ea30-4a50-ad45-d85fd0431f95
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Guaifenesin 600 mg Pseudoephedrine HCl 60 mg
Purpose
Expectorant Nasal Decongestant
Medication Information
Purpose
Expectorant Nasal Decongestant
Description
Guaifenesin 600 mg Pseudoephedrine HCl 60 mg
Uses
-
•helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
-
•temporarily relieves nasal congestion due to:
-
•common cold
-
•hay fever
-
•upper respiratory allergies
-
•temporarily restores freer breathing through the nose
-
•promotes nasal and/or sinus drainage
-
•temporarily relieves sinus congestion and pressure
Purposes
Expectorant
Nasal Decongestant
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Directions
-
•do not crush, chew, or break tablet
-
•take with a full glass of water
-
•this product can be administered without regard for timing of meals
-
•adults and children 12 years and older: 2 tablets every 12 hours; not more than 4 tablets in 24 hours
-
•children under 12 years of age: do not use
Other Information
-
•do not use if printed blister unit is broken or torn
-
•store between 20-25°C (68-77°F)
Inactive Ingredients
carbomer homopolymer type B, colloidal silicon dioxide, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, sodium starch glycolate
Questions Or Comments?
1-800-719-9260
When Using This Product
-
•do not use more than directed
Stop Use and Ask A Doctor If
-
•you get nervous, dizzy, or sleepless
-
•symptoms do not get better within 7 days, come back or occur with a fever, rash, or persistent headache. These could be signs of a serious illness.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Before Use If You Have
-
•heart disease
-
•high blood pressure
-
•thyroid disease
-
•diabetes
-
•trouble urinating due to an enlarged prostate gland
-
•persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
-
•cough accompanied by too much phlegm (mucus)
Package/label Principal Display Panel
Compare to Mucinex® D
active ingredients
H-E-B®
Mucus Relief D
Guaifenesin 600 mg
Pseudoephedrine Hydrochloride 60 mg
Extended-Release Bi-Layer Tablets
Expectorant / Nasal Decongestant
• Clears Nasal/Sinus Congestion
• Thins and Loosens Mucus
• Immediate and Extended Release
12 Hours
Actual Size
18 EXTENDED-RELEASE BI-LAYER TABLETS
Active Ingredients (in Each Extended Release Bi Layer Tablet)
Guaifenesin 600 mg
Pseudoephedrine HCl 60 mg
Structured Label Content
Uses
-
•helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
-
•temporarily relieves nasal congestion due to:
-
•common cold
-
•hay fever
-
•upper respiratory allergies
-
•temporarily restores freer breathing through the nose
-
•promotes nasal and/or sinus drainage
-
•temporarily relieves sinus congestion and pressure
Purposes
Expectorant
Nasal Decongestant
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Directions
-
•do not crush, chew, or break tablet
-
•take with a full glass of water
-
•this product can be administered without regard for timing of meals
-
•adults and children 12 years and older: 2 tablets every 12 hours; not more than 4 tablets in 24 hours
-
•children under 12 years of age: do not use
Other Information (Other information)
-
•do not use if printed blister unit is broken or torn
-
•store between 20-25°C (68-77°F)
Inactive Ingredients (Inactive ingredients)
carbomer homopolymer type B, colloidal silicon dioxide, FD&C yellow #6 aluminum lake, hypromellose, magnesium stearate, microcrystalline cellulose, sodium starch glycolate
Questions Or Comments? (Questions or comments?)
1-800-719-9260
When Using This Product (When using this product)
-
•do not use more than directed
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
-
•you get nervous, dizzy, or sleepless
-
•symptoms do not get better within 7 days, come back or occur with a fever, rash, or persistent headache. These could be signs of a serious illness.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
-
•heart disease
-
•high blood pressure
-
•thyroid disease
-
•diabetes
-
•trouble urinating due to an enlarged prostate gland
-
•persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
-
•cough accompanied by too much phlegm (mucus)
Package/label Principal Display Panel (Package/Label Principal Display Panel)
Compare to Mucinex® D
active ingredients
H-E-B®
Mucus Relief D
Guaifenesin 600 mg
Pseudoephedrine Hydrochloride 60 mg
Extended-Release Bi-Layer Tablets
Expectorant / Nasal Decongestant
• Clears Nasal/Sinus Congestion
• Thins and Loosens Mucus
• Immediate and Extended Release
12 Hours
Actual Size
18 EXTENDED-RELEASE BI-LAYER TABLETS
Active Ingredients (in Each Extended Release Bi Layer Tablet) (Active ingredients (in each extended-release bi-layer tablet))
Guaifenesin 600 mg
Pseudoephedrine HCl 60 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:42.474409 · Updated: 2026-03-14T23:02:24.469183