09835640-55c3-4bfa-9ad3-15d37f06e871
34391-3
HUMAN PRESCRIPTION DRUG LABEL
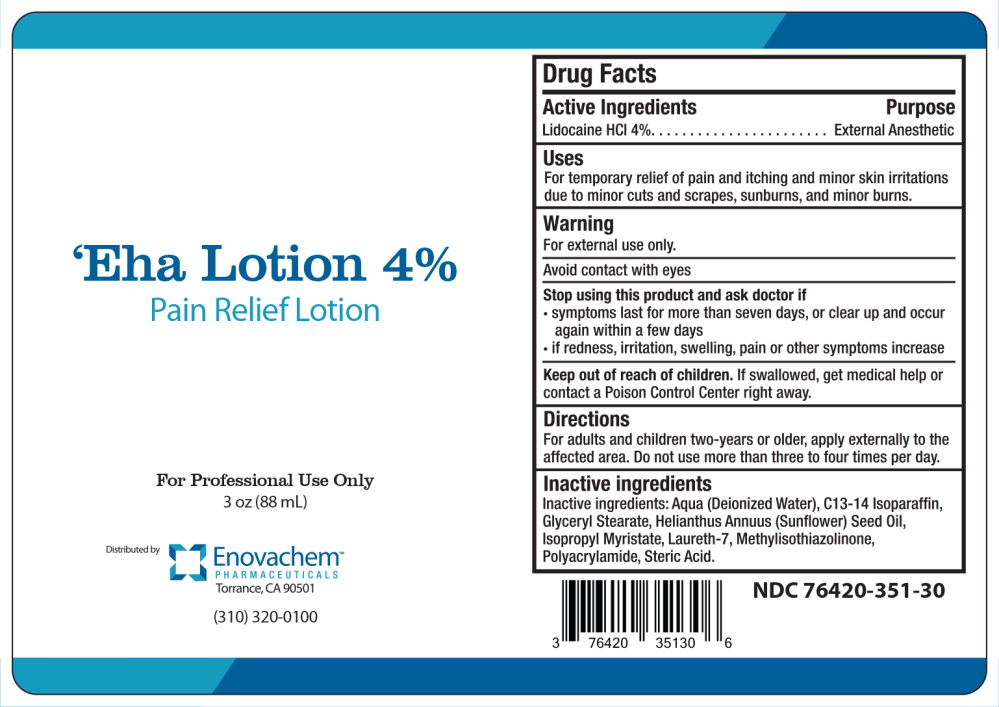
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
External Anesthetic
Medication Information
Purpose
External Anesthetic
Description
Drug Facts
Uses
For temporary relief of pain and itching and minor skin irritations due to minor cuts and scrapes, sunburns, and minor burns.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warning
For external use only.
Avoid contact with eyes
Directions
For adults and children two-years or older, apply externally to the affected area. Do not use more than three to four times per day.
Active Ingredients
Lidocaine HCI 4%
Inactive Ingredients
Inactive ingredients: Aqua (Deionized Water), C13-14 Isoparaffin, Glyceryl Stearate, Helianthus Annuus (Sunflower) Seed Oil, Isopropyl Myristate, Laureth-7, M Polyacrylamide, Steric Acid.
Stop Using This Product and Ask Doctor If
- symptoms last for more than seven days, or clear up and occur again within a few days
- if redness, irritation, swelling, pain or other symptoms increase
Principal Display Panel 88 Ml Bottle Label
‘Eha Lotion 4%
Pain Relief Lotion
For Professional Use Only
3 OZ (88 mL)
Distributed by
Enovachem ™
PHARMACEUTICALS
Torrance, CA 90501
(310) 320-0100
Structured Label Content
Uses
For temporary relief of pain and itching and minor skin irritations due to minor cuts and scrapes, sunburns, and minor burns.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
External Anesthetic
Warning
For external use only.
Avoid contact with eyes
Directions
For adults and children two-years or older, apply externally to the affected area. Do not use more than three to four times per day.
Active Ingredients
Lidocaine HCI 4%
Inactive Ingredients (Inactive ingredients)
Inactive ingredients: Aqua (Deionized Water), C13-14 Isoparaffin, Glyceryl Stearate, Helianthus Annuus (Sunflower) Seed Oil, Isopropyl Myristate, Laureth-7, M Polyacrylamide, Steric Acid.
Stop Using This Product and Ask Doctor If (Stop using this product and ask doctor if)
- symptoms last for more than seven days, or clear up and occur again within a few days
- if redness, irritation, swelling, pain or other symptoms increase
Principal Display Panel 88 Ml Bottle Label (Principal Display Panel - 88 mL Bottle Label)
‘Eha Lotion 4%
Pain Relief Lotion
For Professional Use Only
3 OZ (88 mL)
Distributed by
Enovachem ™
PHARMACEUTICALS
Torrance, CA 90501
(310) 320-0100
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:27.294504 · Updated: 2026-03-14T21:40:40.444869