These Highlights Do Not Include All The Information Needed To Use Keytruda Qlex Safely And Effectively. See Full Prescribing Information For Keytruda Qlex.
097d166f-b73b-41d3-9b37-7653cd2a0c41
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 02/2026 Dosage and Administration ( 2 ) 02/2026
Indications and Usage
KEYTRUDA QLEX is a combination of pembrolizumab, a programmed death receptor-1 (PD-1)-blocking antibody, and berahyaluronidase alfa, an endoglycosidase, indicated: Melanoma for the treatment of adult patients with unresectable or metastatic melanoma. ( 1.1 ) for the adjuvant treatment of adult and pediatric patients 12 years and older with Stage IIB, IIC, or III melanoma following complete resection. ( 1.1 ) Non-Small Cell Lung Cancer (NSCLC) in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with metastatic nonsquamous NSCLC, with no EGFR or ALK genomic tumor aberrations. ( 1.2 ) in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, as first-line treatment of adult patients with metastatic squamous NSCLC. ( 1.2 ) as a single agent for the first-line treatment of adult patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-authorized test, with no EGFR or ALK genomic tumor aberrations, and is: Stage III where patients are not candidates for surgical resection or definitive chemoradiation, or metastatic. ( 1.2 , 2.1 ) as a single agent for the treatment of adult patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-authorized test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA QLEX. ( 1.2 , 2.1 ) for the treatment of adult patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. ( 1.2 ) as a single agent, for adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC. ( 1.2 ) Malignant Pleural Mesothelioma (MPM) in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with unresectable advanced or metastatic MPM. ( 1.3 ) Head and Neck Squamous Cell Cancer (HNSCC) for the treatment of adult patients with resectable locally advanced HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test, as a single agent as neoadjuvant treatment, continued as adjuvant treatment in combination with radiotherapy (RT) with or without cisplatin and then as a single agent. ( 1.4 ) in combination with platinum and FU for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC. ( 1.4 ) as a single agent for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test. ( 1.4 , 2.1 ) as a single agent for the treatment of adult patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy. ( 1.4 ) Urothelial Cancer in combination with enfortumab vedotin, for the treatment of adult patients with locally advanced or metastatic urothelial cancer. ( 1.5 ) as a single agent for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma who: are not eligible for any platinum-containing chemotherapy, or who have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. ( 1.5 ) in combination with enfortumab vedotin, as neoadjuvant treatment and then continued after cystectomy as adjuvant treatment of adult patients with muscle invasive bladder cancer (MIBC) who are ineligible for cisplatin-containing chemotherapy. ( 1.5 ) as a single agent for the treatment of adult patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy. ( 1.5 ) Microsatellite Instability-High or Mismatch Repair Deficient Cancer for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options. ( 1.6 , 2.1 ) Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer (CRC) for the treatment of adult patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-authorized test. ( 1.7 , 2.1 ) Gastric Cancer in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.8 ) in combination with fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.8 ) Esophageal Cancer for the treatment of adult patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either: in combination with platinum- and fluoropyrimidine-based chemotherapy for patients whose tumors express PD-L1 (CPS ≥1), or as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. ( 1.9 , 2.1 ) Cervical Cancer in combination with chemoradiotherapy, for the treatment of adult patients with locally advanced cervical cancer involving the lower third of the vagina, with or without extension to pelvic sidewall, or hydronephrosis/non-functioning kidney, or spread to adjacent pelvic organs (FIGO 2014 Stage III-IVA). ( 1.10 ) in combination with chemotherapy, with or without bevacizumab, for the treatment of adult patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.10 , 2.1 ) as a single agent for the treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.10 , 2.1 ) Hepatocellular Carcinoma (HCC) for the treatment of adult patients with HCC secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen. ( 1.11 ) Biliary Tract Cancer (BTC) in combination with gemcitabine and cisplatin, for the treatment of adult patients with locally advanced unresectable or metastatic biliary tract cancer. ( 1.12 ) Merkel Cell Carcinoma (MCC) for the treatment of adult and pediatric patients 12 years and older with recurrent locally advanced or metastatic Merkel cell carcinoma. ( 1.13 ) Renal Cell Carcinoma (RCC) in combination with axitinib, for the first-line treatment of adult patients with advanced RCC. ( 1.14 ) in combination with lenvatinib, for the first-line treatment of adult patients with advanced RCC. ( 1.14 ) for the adjuvant treatment of adult patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions. ( 1.14 ) Endometrial Carcinoma in combination with carboplatin and paclitaxel, followed by KEYTRUDA QLEX as a single agent, for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma. ( 1.15 ) in combination with lenvatinib, for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) or not MSI-H as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. ( 1.15 , 2.1 ) as a single agent, for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. ( 1.15 , 2.1 ) Tumor Mutational Burden-High (TMB-H) Cancer for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options. 1 ( 1.16 , 2.1 ) Limitations of Use : The safety and effectiveness of KEYTRUDA QLEX in pediatric patients 12 years and older with TMB-H central nervous system cancers have not been established. Cutaneous Squamous Cell Carcinoma (cSCC) for the treatment of adult patients with recurrent or metastatic cSCC or locally advanced cSCC that is not curable by surgery or radiation. ( 1.17 ) Triple-Negative Breast Cancer (TNBC) for the treatment of adult patients with high-risk early-stage TNBC in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. ( 1.18 ) in combination with chemotherapy, for the treatment of adult patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. ( 1.18 , 2.1 ) Ovarian Cancer in combination with paclitaxel, with or without bevacizumab, is indicated for the treatment of adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test, and who have received one or two prior systemic treatment regimens. ( 1.19 , 2.1 ) 1 This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Dosage and Administration
KEYTRUDA QLEX has different recommended dosage and administration instructions than intravenous pembrolizumab. To reduce the risk of medication errors, check the vial labels to ensure that the drug being prepared and administered is KEYTRUDA QLEX for subcutaneous use and not intravenous pembrolizumab. Do not substitute KEYTRUDA QLEX with intravenous pembrolizumab because they have different recommended dosages and routes of administration. Patients receiving intravenous pembrolizumab can switch to subcutaneous KEYTRUDA QLEX at their next scheduled dose. Patients receiving subcutaneous KEYTRUDA QLEX can switch to intravenous pembrolizumab at their next scheduled dose. Administer KEYTRUDA QLEX as a subcutaneous injection into the thigh or abdomen, avoiding the 5 cm area around the navel. Every 3-week dosing (395 mg/4,800 units) : inject 2.4 mL subcutaneously over 1 minute. Treatment duration is provided in Recommended Dosage (Table 1). Every 6-week dosing (790 mg/9,600 units): inject 4.8 mL subcutaneously over 2 minutes. Treatment duration is provided in Recommended Dosage (Table 1). Inject into healthy skin and never into areas where the skin is red, bruised, tender, or hard. Ensure the injection site is at least 2.5 cm from the previous injection site. During treatment with KEYTRUDA QLEX, do not administer other medications for subcutaneous use at the same site as KEYTRUDA QLEX. Do not administer KEYTRUDA QLEX intravenously. KEYTRUDA QLEX must be administered by a healthcare provider.
Warnings and Precautions
Immune-Mediated Adverse Reactions ( 5.1 ) Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and solid organ transplant rejection. Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. Withhold or permanently discontinue based on severity and type of reaction. Hypersensitivity and Administration-Related Reactions: Interrupt injection and resume upon symptom resolution, or permanently discontinue KEYTRUDA QLEX based on the severity of reaction. ( 5.2 ) Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. ( 5.3 ) Treatment of patients with multiple myeloma with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials. ( 5.4 ) Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective method of contraception. ( 5.5 , 8.1 , 8.3 )
Contraindications
KEYTRUDA QLEX is contraindicated in patients with known hypersensitivity to berahyaluronidase alfa, hyaluronidase or to any of its excipients.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling. Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1) ] . Hypersensitivity and Administration-Related Reactions [see Warnings and Precautions (5.2) ].
Storage and Handling
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration. Each carton contains one single-dose vial either as: 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/ 2,000 units per mL), NDC 0006-3083-01 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/ 2,000 units per mL), NDC 0006-5083-01
How Supplied
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration. Each carton contains one single-dose vial either as: 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/ 2,000 units per mL), NDC 0006-3083-01 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/ 2,000 units per mL), NDC 0006-5083-01
Medication Information
Warnings and Precautions
Immune-Mediated Adverse Reactions ( 5.1 ) Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and solid organ transplant rejection. Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. Withhold or permanently discontinue based on severity and type of reaction. Hypersensitivity and Administration-Related Reactions: Interrupt injection and resume upon symptom resolution, or permanently discontinue KEYTRUDA QLEX based on the severity of reaction. ( 5.2 ) Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. ( 5.3 ) Treatment of patients with multiple myeloma with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials. ( 5.4 ) Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective method of contraception. ( 5.5 , 8.1 , 8.3 )
Indications and Usage
KEYTRUDA QLEX is a combination of pembrolizumab, a programmed death receptor-1 (PD-1)-blocking antibody, and berahyaluronidase alfa, an endoglycosidase, indicated: Melanoma for the treatment of adult patients with unresectable or metastatic melanoma. ( 1.1 ) for the adjuvant treatment of adult and pediatric patients 12 years and older with Stage IIB, IIC, or III melanoma following complete resection. ( 1.1 ) Non-Small Cell Lung Cancer (NSCLC) in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with metastatic nonsquamous NSCLC, with no EGFR or ALK genomic tumor aberrations. ( 1.2 ) in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, as first-line treatment of adult patients with metastatic squamous NSCLC. ( 1.2 ) as a single agent for the first-line treatment of adult patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-authorized test, with no EGFR or ALK genomic tumor aberrations, and is: Stage III where patients are not candidates for surgical resection or definitive chemoradiation, or metastatic. ( 1.2 , 2.1 ) as a single agent for the treatment of adult patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-authorized test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA QLEX. ( 1.2 , 2.1 ) for the treatment of adult patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. ( 1.2 ) as a single agent, for adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC. ( 1.2 ) Malignant Pleural Mesothelioma (MPM) in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with unresectable advanced or metastatic MPM. ( 1.3 ) Head and Neck Squamous Cell Cancer (HNSCC) for the treatment of adult patients with resectable locally advanced HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test, as a single agent as neoadjuvant treatment, continued as adjuvant treatment in combination with radiotherapy (RT) with or without cisplatin and then as a single agent. ( 1.4 ) in combination with platinum and FU for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC. ( 1.4 ) as a single agent for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test. ( 1.4 , 2.1 ) as a single agent for the treatment of adult patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy. ( 1.4 ) Urothelial Cancer in combination with enfortumab vedotin, for the treatment of adult patients with locally advanced or metastatic urothelial cancer. ( 1.5 ) as a single agent for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma who: are not eligible for any platinum-containing chemotherapy, or who have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. ( 1.5 ) in combination with enfortumab vedotin, as neoadjuvant treatment and then continued after cystectomy as adjuvant treatment of adult patients with muscle invasive bladder cancer (MIBC) who are ineligible for cisplatin-containing chemotherapy. ( 1.5 ) as a single agent for the treatment of adult patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy. ( 1.5 ) Microsatellite Instability-High or Mismatch Repair Deficient Cancer for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options. ( 1.6 , 2.1 ) Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer (CRC) for the treatment of adult patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-authorized test. ( 1.7 , 2.1 ) Gastric Cancer in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.8 ) in combination with fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.8 ) Esophageal Cancer for the treatment of adult patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either: in combination with platinum- and fluoropyrimidine-based chemotherapy for patients whose tumors express PD-L1 (CPS ≥1), or as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. ( 1.9 , 2.1 ) Cervical Cancer in combination with chemoradiotherapy, for the treatment of adult patients with locally advanced cervical cancer involving the lower third of the vagina, with or without extension to pelvic sidewall, or hydronephrosis/non-functioning kidney, or spread to adjacent pelvic organs (FIGO 2014 Stage III-IVA). ( 1.10 ) in combination with chemotherapy, with or without bevacizumab, for the treatment of adult patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.10 , 2.1 ) as a single agent for the treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. ( 1.10 , 2.1 ) Hepatocellular Carcinoma (HCC) for the treatment of adult patients with HCC secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen. ( 1.11 ) Biliary Tract Cancer (BTC) in combination with gemcitabine and cisplatin, for the treatment of adult patients with locally advanced unresectable or metastatic biliary tract cancer. ( 1.12 ) Merkel Cell Carcinoma (MCC) for the treatment of adult and pediatric patients 12 years and older with recurrent locally advanced or metastatic Merkel cell carcinoma. ( 1.13 ) Renal Cell Carcinoma (RCC) in combination with axitinib, for the first-line treatment of adult patients with advanced RCC. ( 1.14 ) in combination with lenvatinib, for the first-line treatment of adult patients with advanced RCC. ( 1.14 ) for the adjuvant treatment of adult patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions. ( 1.14 ) Endometrial Carcinoma in combination with carboplatin and paclitaxel, followed by KEYTRUDA QLEX as a single agent, for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma. ( 1.15 ) in combination with lenvatinib, for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) or not MSI-H as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. ( 1.15 , 2.1 ) as a single agent, for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. ( 1.15 , 2.1 ) Tumor Mutational Burden-High (TMB-H) Cancer for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options. 1 ( 1.16 , 2.1 ) Limitations of Use : The safety and effectiveness of KEYTRUDA QLEX in pediatric patients 12 years and older with TMB-H central nervous system cancers have not been established. Cutaneous Squamous Cell Carcinoma (cSCC) for the treatment of adult patients with recurrent or metastatic cSCC or locally advanced cSCC that is not curable by surgery or radiation. ( 1.17 ) Triple-Negative Breast Cancer (TNBC) for the treatment of adult patients with high-risk early-stage TNBC in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. ( 1.18 ) in combination with chemotherapy, for the treatment of adult patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. ( 1.18 , 2.1 ) Ovarian Cancer in combination with paclitaxel, with or without bevacizumab, is indicated for the treatment of adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test, and who have received one or two prior systemic treatment regimens. ( 1.19 , 2.1 ) 1 This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Dosage and Administration
KEYTRUDA QLEX has different recommended dosage and administration instructions than intravenous pembrolizumab. To reduce the risk of medication errors, check the vial labels to ensure that the drug being prepared and administered is KEYTRUDA QLEX for subcutaneous use and not intravenous pembrolizumab. Do not substitute KEYTRUDA QLEX with intravenous pembrolizumab because they have different recommended dosages and routes of administration. Patients receiving intravenous pembrolizumab can switch to subcutaneous KEYTRUDA QLEX at their next scheduled dose. Patients receiving subcutaneous KEYTRUDA QLEX can switch to intravenous pembrolizumab at their next scheduled dose. Administer KEYTRUDA QLEX as a subcutaneous injection into the thigh or abdomen, avoiding the 5 cm area around the navel. Every 3-week dosing (395 mg/4,800 units) : inject 2.4 mL subcutaneously over 1 minute. Treatment duration is provided in Recommended Dosage (Table 1). Every 6-week dosing (790 mg/9,600 units): inject 4.8 mL subcutaneously over 2 minutes. Treatment duration is provided in Recommended Dosage (Table 1). Inject into healthy skin and never into areas where the skin is red, bruised, tender, or hard. Ensure the injection site is at least 2.5 cm from the previous injection site. During treatment with KEYTRUDA QLEX, do not administer other medications for subcutaneous use at the same site as KEYTRUDA QLEX. Do not administer KEYTRUDA QLEX intravenously. KEYTRUDA QLEX must be administered by a healthcare provider.
Contraindications
KEYTRUDA QLEX is contraindicated in patients with known hypersensitivity to berahyaluronidase alfa, hyaluronidase or to any of its excipients.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling. Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1) ] . Hypersensitivity and Administration-Related Reactions [see Warnings and Precautions (5.2) ].
Storage and Handling
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration. Each carton contains one single-dose vial either as: 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/ 2,000 units per mL), NDC 0006-3083-01 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/ 2,000 units per mL), NDC 0006-5083-01
How Supplied
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration. Each carton contains one single-dose vial either as: 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/ 2,000 units per mL), NDC 0006-3083-01 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/ 2,000 units per mL), NDC 0006-5083-01
Description
Indications and Usage ( 1 ) 02/2026 Dosage and Administration ( 2 ) 02/2026
Section 42229-5
KEYTRUDA QLEX is a ready-to-use solution. Do not dilute KEYTRUDA QLEX.
Do not shake.
Preparation of the Syringe
- Remove KEYTRUDA QLEX vial from refrigerated storage [2°C to 8°C (36°F to 46°F)] and allow it to equilibrate to room temperature [20°C to 25°C (68°F to 77°F)] for at least 30 minutes.
- Prior to preparation for administration, if needed, the unpunctured vial may be stored at room temperature for up to 24 hours.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. The solution is clear to slightly opalescent, colorless to slightly yellow. Discard the vial if visible particles are observed.
- Use a sterile, polypropylene or polycarbonate syringe and a stainless steel transfer needle (18 to 21 gauge) to withdraw KEYTRUDA QLEX from the vial.
- Every 3-week dosing (395 mg pembrolizumab/4,800 units berahyaluronidase alfa): withdraw 2.4 mL into the syringe.
- Every 6-week dosing (790 mg pembrolizumab/9,600 units berahyaluronidase alfa): withdraw 4.8 mL into the syringe.
- To avoid needle clogging, change the needle to a 25 to 30 gauge, ½-inch, stainless steel hypodermic injection needle immediately prior to subcutaneous injection.
- Discard any unused portion left in the vial.
Storage of Prepared Syringe
The product does not contain a preservative and should be used immediately after withdrawing from the vial. If not used immediately, store the syringe containing KEYTRUDA QLEX with the transfer needle and cap in place:
- At room temperature 20°C to 25°C (68°F to 77°F) for up to 8 hours, or
- In the refrigerator at 2°C to 8°C (36°F to 46°F) for up to 24 hours. The 24-hour period may include up to 8 hours at room temperature.
Discard if storage time exceeds these limits.
If refrigerated, allow the filled syringe to come to room temperature for at least 30 minutes prior to administration.
Do not freeze.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Issued: 02/2026 | ||
|
MEDICATION GUIDE KEYTRUDA QLEX™ (key-true-duh Q-lex) (pembrolizumab and berahyaluronidase alfa-pmph) injection, for subcutaneous use |
|||
|
What is the most important information I should know about KEYTRUDA QLEX? |
|||
| KEYTRUDA QLEX is a medicine that may treat certain cancers by working with your immune system. KEYTRUDA QLEX can cause your immune system to attack normal organs and tissues in any area of your body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death. You can have more than one of these problems at the same time. These problems may happen anytime during treatment or even after your treatment has ended. | |||
| Call or see your healthcare provider right away if you develop any new or worsening signs or symptoms, including: | |||
| Lung problems | |||
|
|
|
|
| Intestinal problems | |||
|
|||
| Liver problems | |||
|
|
||
| Hormone gland problems | |||
|
|
||
| Kidney problems | |||
|
|
||
| Skin problems | |||
|
|
||
| Problems can also happen in other organs and tissues. These are not all of the signs and symptoms of immune system problems that can happen with KEYTRUDA QLEX. Call or see your healthcare provider right away for any new or worsening signs or symptoms, which may include: | |||
|
|||
| Allergic and injection-related reactions that can sometimes be severe or life-threatening, can happen during treatment with KEYTRUDA QLEX. Tell your healthcare provider right away if you get any signs or symptoms, including: | |||
|
|
||
| Rejection of a transplanted organ or tissue. Your healthcare provider should tell you what signs and symptoms you should report and monitor you depending on the type of organ or tissue transplant that you have had. | |||
| Complications, including graft-versus-host-disease (GVHD), in people who have received a bone marrow (stem cell) transplant that uses donor stem cells (allogeneic). These complications can be serious and can lead to death. These complications may happen if you underwent transplantation either before or after being treated with KEYTRUDA QLEX. Your healthcare provider will monitor you for these complications. | |||
|
Getting medical treatment right away may help keep these problems from becoming more serious. Your healthcare provider will check you for these problems during treatment with KEYTRUDA QLEX. Your healthcare provider may treat you with corticosteroid or hormone replacement medicines. Your healthcare provider may also need to delay or completely stop treatment with KEYTRUDA QLEX if you have severe side effects. |
|||
| What is KEYTRUDA QLEX? | |||
| KEYTRUDA QLEX is a prescription medicine used to treat: | |||
|
|||
|
Who should not receive KEYTRUDA QLEX?
Do not receive KEYTRUDA QLEX if you are allergic to berahyaluronidase, hyaluronidase or any of the inactive ingredients in KEYTRUDA QLEX. See the end of this Medication Guide for a complete list of ingredients in KEYTRUDA QLEX. |
|||
Before receiving KEYTRUDA QLEX, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| How will I receive KEYTRUDA QLEX? | |||
|
|||
| What are the possible side effects of KEYTRUDA QLEX? | |||
| KEYTRUDA QLEX can cause serious side effects. See “What is the most important information I should know about KEYTRUDA QLEX?” | |||
| The most common side effects of KEYTRUDA QLEX when given with certain chemotherapy medicines include: nausea, tiredness, and muscle, bone, and joint pain. | |||
| The most common side effects seen with pembrolizumab given into the vein (intravenous pembrolizumab), which may happen with KEYTRUDA QLEX, are shown below: | |||
|
|||
| These are not all of the possible side effects of KEYTRUDA QLEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
| General information about the safe and effective use of KEYTRUDA QLEX | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about KEYTRUDA QLEX that is written for health professionals. | |||
| What are the ingredients in KEYTRUDA QLEX? | |||
| Active ingredients: pembrolizumab and berahyaluronidase alfa-pmph | |||
| Inactive ingredients: histidine, histidine hydrochloride monohydrate, methionine, polysorbate 80, sucrose, and Water for Injection. | |||
| Manufactured by: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA U.S. License No. 0002 |
For patent information: www.msd.com/research/patent
Copyright © 2026 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved. usmg-mk3475a-i-2602r003 For more information, go to www.keytruda.com/qlex . |
Section 44425-7
Store vials refrigerated at 2°C to 8°C (36°F to 46°F) in original carton to protect from light. Do not freeze. Do not shake.
1.1 Melanoma
KEYTRUDA QLEX™ is indicated for the treatment of adult patients with unresectable or metastatic melanoma.
KEYTRUDA QLEX is indicated for the adjuvant treatment of adult and pediatric patients 12 years and older with Stage IIB, IIC, or III melanoma following complete resection.
11 Description
KEYTRUDA QLEX is a fixed-combination drug product containing pembrolizumab and berahyaluronidase alfa.
Pembrolizumab is a programmed death receptor-1 (PD 1)-blocking antibody. Pembrolizumab is a humanized monoclonal IgG4 kappa antibody with an approximate molecular weight of 149 kDa. Pembrolizumab is produced in recombinant Chinese hamster ovary (CHO) cells.
Berahyaluronidase alfa is an endoglycosidase used to enhance dispersion and permeation, which facilitates delivery of increased volume of pembrolizumab that is co-administered subcutaneously. It is produced by mammalian CHO (Chinese Hamster Ovary) cells containing a DNA plasmid encoding a variant of human hyaluronidase PH20. It is a glycosylated protein with an approximate molecular weight of 49 kDa under nonreducing, deglycosylated conditions.
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration.
KEYTRUDA QLEX is supplied as two different configurations:
- Each KEYTRUDA QLEX 2.4 mL single-dose vial contains 395 mg of pembrolizumab and 4,800 units of berahyaluronidase alfa, and histidine (0.7 mg), histidine hydrochloride monohydrate (4.1 mg), methionine (3.6 mg), polysorbate 80 (0.5 mg), sucrose (168 mg), and Water for Injection, USP. The pH is 5.3-5.9.
- Each KEYTRUDA QLEX 4.8 mL single-dose vial contains 790 mg of pembrolizumab and 9,600 units of berahyaluronidase alfa, and histidine (1.4 mg), histidine hydrochloride monohydrate (8.2 mg), methionine (7.2 mg), polysorbate 80 (1 mg), sucrose (336 mg), and Water for Injection, USP. The pH is 5.3-5.9.
8.4 Pediatric Use
The safety and effectiveness of KEYTRUDA QLEX for the treatment of pediatric patients 12 years and older who weigh greater than 40 kg have been established for:
- Stage IIB, IIC, or III melanoma following complete resection [see Indications and Usage (1.1)]
- Unresectable or metastatic microsatellite instability high (MSI-H) or mismatch repair deficient (dMMR) solid tumors [see Indications and Usage (1.6)]
- Recurrent locally advanced or metastatic Merkel cell carcinoma [see Indications and Usage (1.13)]
- Unresectable or metastatic tumor mutational burden high solid tumors (TMB-H) [see Indications and Usage (1.16)]
Use of KEYTRUDA QLEX in pediatric patients for these indications is supported by evidence from adequate and well-controlled studies of intravenous pembrolizumab in adults and additional pharmacokinetic and safety data for intravenous pembrolizumab in pediatric patients 12 years and older [see Adverse Reactions (6.1), Clinical Studies (14)]. Pembrolizumab exposures in pediatric patients 12 years and older who weigh greater than 40 kg are predicted to be within range of those observed in adults at the same dosage [see Clinical Pharmacology (12.3)].
The safety and effectiveness of KEYTRUDA QLEX have not been established in pediatric patients younger than 12 years of age for the treatment of melanoma, MCC, MSI-H or dMMR cancer, and TMB-H cancer.
The safety and effectiveness of KEYTRUDA QLEX have not been established in pediatric patients for other approved indications [see Indications and Usage (1)].
Intravenous pembrolizumab
In KEYNOTE-051, 173 pediatric patients (including 108 pediatric patients aged 12 to 17 years) with advanced melanoma, lymphoma, or PD-L1 positive or MSI-H solid tumors received intravenous pembrolizumab 2 mg/kg every 3 weeks. The median duration of exposure was 2.1 months (range: 1 day to 25 months). Adverse reactions that occurred at a ≥10% higher rate in pediatric patients when compared to adults included pyrexia (33%), vomiting (29%), headache (25%), abdominal pain (23%), decreased lymphocyte count (13%), and decreased white blood cell count (11%). Laboratory abnormalities that occurred at a ≥10% higher rate in pediatric patients when compared to adults were leukopenia (30%), neutropenia (28%), thrombocytopenia (22%), and Grade 3 anemia (17%).
8.5 Geriatric Use
Of the 251 patients treated with KEYTRUDA QLEX in combination with platinum doublet chemotherapy in Study MK-3745A-D77, 53% were 65 years and older and 16% were 75 years and older. No overall differences in safety or effectiveness of KEYTRUDA QLEX have been observed between patients aged 65 years or older and younger adult patients.
The safety of KEYTRUDA QLEX as monotherapy or in combination with other antineoplastic drugs for its approved indications [see Indications and Usage (1)] has been established in adequate and well-controlled studies of intravenous pembrolizumab as a single agent and in combination with other antineoplastic drugs. Below is a description of geriatric use information from the intravenous pembrolizumab studies.
- Of 3781 patients with melanoma, NSCLC, HNSCC, or urothelial carcinoma who were treated with intravenous pembrolizumab in clinical studies, 48% were 65 years and over and 17% were 75 years and over.
- Of 506 adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC following complete resection and platinum-based chemotherapy who were treated with intravenous pembrolizumab in KEYNOTE-091, 242 (48%) were 65 years and over.
- Of 596 adult patients with TNBC who were treated with intravenous pembrolizumab in combination with paclitaxel, paclitaxel protein-bound, or gemcitabine and carboplatin in KEYNOTE-355, 137 (23%) were 65 years and over.
- Of 406 adult patients with endometrial carcinoma who were treated with intravenous pembrolizumab in combination with lenvatinib in KEYNOTE-775, 201 (50%) were 65 years and over.
- Of the 564 patients with locally advanced or metastatic urothelial cancer treated with intravenous pembrolizumab in combination with enfortumab vedotin, 44% (n=247) were 65-74 years and 26% (n=144) were 75 years or older. No overall differences in effectiveness were observed between patients 65 years of age or older and younger patients. Patients 75 years of age or older treated with intravenous pembrolizumab in combination with enfortumab vedotin experienced a higher incidence of fatal adverse reactions than younger patients. The incidence of fatal adverse reactions was 4% in patients younger than 75 and 7% in patients 75 years or older.
- Of the 167 patients with MIBC treated with intravenous pembrolizumab in combination with enfortumab vedotin, 37% (n=61) were 65-74 years and 46% (n=77) were 75 years or older. Patients 75 years of age or older treated with intravenous pembrolizumab in combination with enfortumab vedotin experienced a higher incidence of fatal adverse reactions than younger patients. The incidence of fatal adverse reactions was 4% in patients younger than 75 and 12% in patients 75 years or older.
- Of the 432 patients randomized to intravenous pembrolizumab in combination with axitinib in the KEYNOTE-426 trial, 40% were 65 years or older.
- Of 294 adult patients with FIGO 2014 Stage III-IVA cervical cancer who were treated with intravenous pembrolizumab in combination with CRT in KEYNOTE-A18, 42 (14%) were 65 years and over.
- Of 643 adult patients with ovarian cancer who were treated with intravenous pembrolizumab in combination with paclitaxel with or without bevacizumab in KEYNOTE-B96, 236 (37%) were 65 years and over and 58 (9%) were 75 years and over.
No overall differences in safety or effectiveness were observed between intravenous pembrolizumab-treated patients aged 65 years or older and younger adult patients.
1.8 Gastric Cancer
KEYTRUDA QLEX, in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, in combination with fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥ 1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
14.1 Keytruda Qlex
NSCLC (Study MK-3475A-D77)
KEYTRUDA QLEX was evaluated in Study MK-3475A-D77 (NCT05722015), a randomized, multicenter, open-label, active-controlled trial conducted in patients with treatment-naïve metastatic NSCLC, in whom there were no EGFR, ALK, or ROS1 genomic tumor aberrations. Patients were excluded if they had a history of autoimmune disease that required systemic therapy within 2 years of treatment; had a medical condition that required immunosuppression; or had received more than 30 Gy of thoracic radiation within the prior 26 weeks.
A total of 377 patients were randomized (2:1) to receive either KEYTRUDA QLEX (containing 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa) administered subcutaneously every 6 weeks with platinum doublet chemotherapy (n=251) or pembrolizumab 400 mg intravenously every 6 weeks with platinum doublet chemotherapy (n=126).
The chemotherapy regimens were as follows:
- Non-squamous NSCLC: pemetrexed 500 mg/m2 and a platinum chemotherapy (cisplatin 75 mg/m2 or carboplatin AUC 5 mg/mL/min) intravenously every 3 weeks for 4 cycles, followed by pemetrexed 500 mg/m2 intravenously every 3 weeks.
- Squamous NSCLC: carboplatin AUC 6 mg/mL/min and a taxane (paclitaxel 200 mg/m2 on Day 1 of each 21-day cycle or paclitaxel protein-bound 100 mg/m2 on Days 1, 8 and 15 of each 21-day cycle) intravenously every 3 weeks for 4 cycles.
Randomization was stratified by ECOG performance status (0 vs. 1), histology (squamous vs. non-squamous), PD-L1 TPS (<50% vs. ≥50%), and geographic region (East Asia vs. North America/Western Europe/Australia/New Zealand vs. Rest of the World).
Treatment with KEYTRUDA QLEX or intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by the investigator, unacceptable toxicity, or a maximum of 18 cycles (approximately 24 months). Administration of KEYTRUDA QLEX or intravenous pembrolizumab was permitted beyond RECIST-defined disease progression by BICR or beyond discontinuation of pemetrexed if the patient was clinically stable and deriving clinical benefit as determined by the investigator.
The primary outcome measure was pembrolizumab exposure [Cycle 1 AUC0-6 weeks and Cycle 3 (i.e., Steady State) Ctrough] of subcutaneous KEYTRUDA QLEX as compared to intravenous pembrolizumab [see Clinical Pharmacology (12.3)]. Additional descriptive efficacy outcome measures were overall response rate (ORR) by blinded independent central review (BICR), progression-free survival (PFS) by BICR, and overall survival (OS).
The study population characteristics were: median age 65 years (range: 37 to 87); 71% male; 63% White, 29% Asian, 4% multiracial, 3% Black or African American, 2% American Indian or Alaska Native; 31% of Hispanic or Latino ethnicity; 35% ECOG performance status (PS) of 0 and 65% ECOG PS of 1. Nineteen percent had PD-L1 TPS ≥50%; 34% had tumors with squamous histology and 66% had tumors with non-squamous histology; and 9% had brain metastases at baseline.
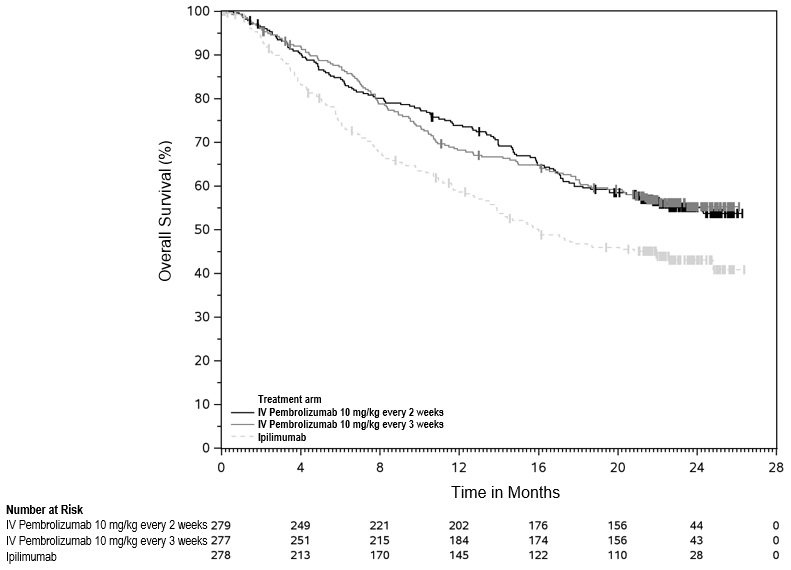
At the primary analysis, the confirmed ORR was 45% (95% CI: 39, 52) in the subcutaneous KEYTRUDA QLEX arm and 42% (95% CI: 33, 51) in the intravenous pembrolizumab arm. There were no notable differences in PFS or OS observed in patients who received KEYTRUDA QLEX compared to patients who received intravenous pembrolizumab.
Intravenous Pembrolizumab
The effectiveness of KEYTRUDA QLEX for its approved indications [see Indications and Usage (1)] has been established based upon the evidence from the adequate and well-controlled studies conducted with intravenous pembrolizumab [see Clinical Studies (14.2-14.20)] and additional data that demonstrated comparable pharmacokinetic, efficacy, and safety profiles between KEYTRUDA QLEX and intravenous pembrolizumab in Study MK-3475A-D77 [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
Below is a description of the efficacy results of these adequate and well-controlled studies of intravenous pembrolizumab in these patient populations.
1.19 Ovarian Cancer
KEYTRUDA QLEX, in combination with paclitaxel, with or without bevacizumab, is indicated for the treatment of adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)], and who have received one or two prior systemic treatment regimens.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described in this section with the incidence of ADA in other studies, including those of KEYTRUDA QLEX or of other pembrolizumab products or berahyaluronidase alfa products.
With a median (min, max) duration of treatment on KEYTRUDA QLEX of 6.9 months (1 day, 1 year) in Study MK-3475A-D77, 1.4% (3/211) of patients developed anti-pembrolizumab antibodies, and one ADA-positive patient developed neutralizing antibodies (NAb) against pembrolizumab. The incidence of anti-berahyaluronidase alfa antibodies was 1.5% (3/194). No analysis of neutralizing antibodies was performed for berahyaluronidase alfa ADA-positive samples. Because of the low occurrence of anti-pembrolizumab or anti-berahyaluronidase antibodies, the effect of these antibodies on the pharmacokinetics, safety and effectiveness of KEYTRUDA QLEX is unknown.
14.9 Gastric Cancer
First-line Treatment of Locally Advanced Unresectable or Metastatic HER2-Positive Gastric or Gastroesophageal Junction Adenocarcinoma for Tumors Expressing PD-L1 (CPS ≥1)
The efficacy of intravenous pembrolizumab in combination with trastuzumab plus fluoropyrimidine and platinum chemotherapy was investigated in KEYNOTE-811 (NCT03615326), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 698 patients with HER2-positive advanced gastric or gastroesophageal junction (GEJ) adenocarcinoma who had not previously received systemic therapy for metastatic disease. PD-L1 status was determined using the PD-L1 IHC 22C3 pharmDx assay. Patients with an autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by PD-L1 expression (CPS ≥1 or CPS <1), chemotherapy regimen (5-FU plus cisplatin [FP] or capecitabine plus oxaliplatin [CAPOX]), and geographic region (Europe/Israel/North America/Australia, Asia, or Rest of the World). Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg, trastuzumab 8 mg/kg on first infusion and 6 mg/kg in subsequent cycles, followed by investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 for up to 6 cycles and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 up to 6-8 cycles and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
- Placebo, trastuzumab 8 mg/kg on first infusion and 6 mg/kg in subsequent cycles, followed by investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 for up to 6 cycles and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 up to 6-8 cycles and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
All study medications, except oral capecitabine, were administered as an intravenous infusion every 3-week cycle. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by BICR, unacceptable toxicity, or a maximum of 24 months. The major outcome measures assessed were PFS by BICR using RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, and OS.
Additional outcome measures included ORR and DoR, based on BICR using RECIST 1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among the 698 patients randomized, 594 (85%) had tumors that expressed PD-L1 with a CPS ≥1. PD-L1 status was determined using the PD-L1 IHC 22C3 pharmDx assay. The population characteristics of these 594 patients were: median age of 63 years (range: 19 to 85), 43% age 65 or older; 80% male; 63% White, 33% Asian, and 0.7% Black; 42% ECOG PS of 0 and 58% ECOG PS of 1. Ninety-eight percent of patients had metastatic disease (Stage IV) and 2% had locally advanced unresectable disease. Ninety-five percent (n=562) had tumors that were not MSI-H, 1% (n=8) had tumors that were MSI-H, and in 4% (n=24) the status was not known. Eighty-five percent of patients received CAPOX.
A statistically significant improvement in OS and PFS was demonstrated in patients randomized to intravenous pembrolizumab in combination with trastuzumab and chemotherapy compared with placebo in combination with trastuzumab and chemotherapy; however, an exploratory analysis of OS in the PD-L1 CPS <1 population showed a HR of 1.10 (95% CI: 0.72, 1.68), indicating that the improvement in the ITT population was primarily attributed to the results observed in the subgroup of patients with PD-L1 CPS ≥1.
Efficacy results at the final analysis for the subgroup of patients whose tumors expressed PD-L1 with a CPS ≥1 are summarized in Table 82 and Figure 22.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks Trastuzumab Fluoropyrimidine and Platinum Chemotherapy n=298 |
Placebo Trastuzumab Fluoropyrimidine and Platinum Chemotherapy n=296 |
|---|---|---|
| + Denotes ongoing response | ||
| OS | ||
| Number (%) of patients with event | 226 (76%) | 244 (82%) |
| Median in months Based on Kaplan-Meier estimation (95% CI) |
20.1 (17.9, 22.9) | 15.7 (13.5, 18.5) |
| Hazard ratio Based on the unstratified Cox proportional hazard model (95% CI) |
0.79 (0.66, 0.95) | |
| PFS | ||
| Number (%) of patients with event | 221 (74%) | 226 (76%) |
| Median in months (95% CI) | 10.9 (8.5, 12.5) | 7.3 (6.8, 8.4) |
| Hazard ratio (95% CI) | 0.72 (0.60, 0.87) | |
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
73% (68, 78) | 58% (53, 64) |
| Complete response rate | 17% | 10% |
| Partial response rate | 56% | 48% |
| Duration of Response | n=218 | n=173 |
| Median in months (95% CI) Range in months |
11.3 (9.9, 13.7) 1.1+, 60.8+ |
9.6 (7.1, 11.2) 1.4+, 60.5+ |
|
|
First-line Treatment of Locally Unresectable or Metastatic HER2-Negative Gastric or Gastroesophageal Junction Adenocarcinoma for Tumors Expressing PD-L1 (CPS ≥1)
The efficacy of intravenous pembrolizumab in combination with fluoropyrimidine- and platinum-containing chemotherapy was investigated in KEYNOTE-859 (NCT03675737), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 1579 patients with HER2-negative advanced gastric or GEJ adenocarcinoma who had not previously received systemic therapy for metastatic disease. Patients with an autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by PD-L1 expression (CPS ≥1 or CPS <1), chemotherapy regimen (FP or CAPOX), and geographic region (Europe/Israel/North America/Australia, Asia, or Rest of the World). Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg, investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
- Placebo, investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
All study medications, except oral capecitabine, were administered as an intravenous infusion every 3-week cycle. Platinum agents could be administered for 6 or more cycles following local guidelines. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by BICR, unacceptable toxicity, or a maximum of 24 months. The major efficacy outcome measure was OS. Additional secondary efficacy outcome measures included PFS, ORR, and DoR as assessed by BICR using RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among 1,579 patients, 1,235 (78%) had tumors expressing PD-L1 CPS ≥ 1. The population characteristics in patients with PD-L1 CPS ≥ 1 expressing tumors were: median age of 62 years (range: 24 to 86), 40% age 65 or older; 70% male and 30% female; 55% White, 33% Asian, 4.6% Multiple, 4.3% American Indian or Alaskan Native, 1.3% Black, and 0.2% Native Hawaiian or other Pacific Islander; 76% Not Hispanic or Latino and 21% Hispanic or Latino; 37% ECOG PS of 0 and 63% ECOG PS of 1. Ninety-six percent of patients had metastatic disease (Stage IV) and 3% had locally advanced unresectable disease. Five percent (n=66) had tumors that were MSI-H. Eighty-six percent of patients received CAPOX.
A statistically significant improvement in OS, PFS, and ORR was demonstrated in patients randomized to intravenous pembrolizumab in combination with chemotherapy compared with placebo in combination with chemotherapy at the time of a pre-specified interim analysis of OS; however, an exploratory analysis of OS in the PD-L1 CPS <1 population showed a HR of 0.92 (95% CI 0.73, 1.17) indicating that the improvement in the ITT population was primarily attributed to the results observed in the subgroup of patients with PD-L1 CPS ≥1. Efficacy results for patients whose tumors expressed PD-L1 CPS ≥1 and CPS ≥10 are summarized in Table 83 and Figures 23 and 24.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and FP or CAPOX n=618 |
Placebo and FP or CAPOX n=617 |
Intravenous Pembrolizumab 200 mg every 3 weeks and FP or CAPOX n=279 |
Placebo and FP or CAPOX n=272 |
|---|---|---|---|---|
| CPS ≥1 | CPS ≥10 | |||
| + Denotes ongoing response | ||||
| OS | ||||
| Number (%) of patients with event | 464 (75) | 526 (85) | 188 (67) | 226 (83) |
| Median in months (95% CI) | 13.0 (11.6, 14.2) | 11.4 (10.5, 12.0) | 15.7 (13.8, 19.3) | 11.8 (10.3, 12.7) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.74 (0.65, 0.84) | 0.65 (0.53, 0.79) | ||
| p-Value (stratified log-rank) One-sided p-Value based on stratified log-rank test
|
<0.0001 | <0.0001 | ||
| PFS | ||||
| Number (%) of patients with event | 443 (72%) | 483 (78%) | 190 (68) | 210 (77) |
| Median in months (95% CI) | 6.9 (6.0, 7.2) | 5.6 (5.4, 5.7) | 8.1 (6.8, 8.5) | 5.6 (5.4, 6.7) |
| Hazard ratio (95% CI) | 0.72 (0.63, 0.82) | 0.62 (0.51, 0.76) | ||
| p-Value (stratified log-rank) | <0.0001 | <0.0001 | ||
| Objective Response Rate | ||||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
52% (48, 56) | 43% (39, 47) | 61% (55, 66) | 43% (37, 49) |
| Complete response rate | 10% | 6% | 13% | 5% |
| Partial response rate | 42% | 37% | 48% | 38% |
| p-Value One-sided p-Value based on stratified Miettinen & Nurminen method
|
0.0004 | <0.0001 | ||
| Duration of Response | n=322 | n=263 | n=169 | n=117 |
| Median in months Based on Kaplan-Meier estimates (95% CI) |
8.3 (7.0, 10.9) | 5.6 (5.4, 6.9) | 10.9 (8.0, 13.8) | 5.8 (5.3, 7.0) |
| Range in months | 1.2+, 41.5+ | 1.3+, 34.2+ | 1.2+, 41.5+ | 1.4+, 31.2+ |
|
|
|
|
An exploratory analysis of OS in the 74 patients with MSI-H tumors irrespective of PD-L1 status showed a HR of 0.34 (95% CI: 0.18, 0.66).
4 Contraindications
KEYTRUDA QLEX is contraindicated in patients with known hypersensitivity to berahyaluronidase alfa, hyaluronidase or to any of its excipients.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling.
- Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1)].
- Hypersensitivity and Administration-Related Reactions [see Warnings and Precautions (5.2)].
1.10 Cervical Cancer
KEYTRUDA QLEX, in combination with chemoradiotherapy (CRT), is indicated for the treatment of adult patients with locally advanced cervical cancer involving the lower third of the vagina, with or without extension to pelvic sidewall, or hydronephrosis/non-functioning kidney, or spread to adjacent pelvic organs (FIGO 2014 Stage III-IVA).
KEYTRUDA QLEX, in combination with chemotherapy, with or without bevacizumab, is indicated for the treatment of adult patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
14.20 Ovarian Cancer
The efficacy of intravenous pembrolizumab in combination with paclitaxel, with or without bevacizumab, was evaluated in KEYNOTE-B96 (NCT05116189), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 643 patients with platinum-resistant, epithelial ovarian, fallopian tube, or primary peritoneal carcinoma who received one or two prior lines of systemic therapy for ovarian carcinoma. Patients must have received at least one line of platinum-based chemotherapy for ovarian cancer with radiographic evidence of disease progression within 6 months after the last dose. Prior therapy with an anti-PD-1/PD-L1 inhibitor, PARP inhibitor, or bevacizumab was permitted. Patients were enrolled regardless of PD-L1 tumor expression status. Patients with autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by investigator decision to use bevacizumab, geographic region (U.S. or European Union or Rest of World), and PD-L1 status according to the PD-L1 IHC 22C3 pharmDx assay (CPS <1 or CPS 1 to <10 or CPS ≥10). Patients were randomized (1:1) to one of the two treatment groups:
- Intravenous pembrolizumab 400 mg every 6 weeks plus paclitaxel 80 mg/m2 with or without bevacizumab 10 mg/kg
- Placebo every 6 weeks plus paclitaxel 80 mg/m2 with or without bevacizumab 10 mg/kg
All study medications were administered as an intravenous infusion. Intravenous pembrolizumab 400 mg or placebo were administered on Day 1 of each 6-week treatment cycle and paclitaxel 80 mg/m2 was administered on Days 1, 8, and 15 of each 3-week treatment cycle. The option to use bevacizumab was by investigator choice prior to randomization. Bevacizumab 10 mg/kg was administered on Day 1 of a 2-week treatment cycle. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease, unacceptable toxicity, or a maximum of 24 months. Administration of intravenous pembrolizumab was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed every 9 weeks for the first year, followed by every 12 weeks thereafter. The major efficacy outcome measure was PFS as assessed by investigator according to RECIST v1.1. An additional efficacy outcome measure was OS.
Among the 643 patients randomized, 466 patients (72%) had tumors expressing PD-L1 with a CPS ≥1. The population characteristics of these 466 patients were: median age of 62 years (range: 37 to 85), 38% age 65 or older; 67% White, 20% Asian, 8% Missing, 3% Multiple, 2% Black, 0.4% Native Hawaiian or other Pacific Islander; 13% Hispanic or Latino; 55% and 44% ECOG performance status of 0 or 1, respectively; 73% received bevacizumab as study treatment; 36% of patients had received one prior line and 64% had received two prior lines of therapy; prior systemic therapy included: 46% with bevacizumab; 39% with a PARP inhibitor, or 3% with an anti-PD-1/PD-L1 inhibitor. The platinum-free interval following the most recent line of therapy was <3 months in 47% of patients, and 3 to 6 months in 53% of patients.
The study demonstrated statistically significant improvement in PFS and OS for patients randomized to intravenous pembrolizumab in combination with paclitaxel with or without bevacizumab compared to placebo in combination with paclitaxel with or without bevacizumab in patients whose tumors expressed PD-L1 CPS ≥1. Efficacy results are summarized in Table 104 and Figures 46 and 47.
| Endpoint | Intravenous pembrolizumab 400 mg every 6 weeks plus paclitaxel with or without bevacizumab n=234 |
Placebo plus paclitaxel with or without bevacizumab n=232 |
|---|---|---|
| PFS | ||
| Number of patients with event (%) | 162 (69) | 180 (78) |
| Median in months (95% CI) | 8.3 (7.0, 9.4) | 7.2 (6.2, 8.1) |
| Hazard ratio (95% CI) | 0.72 (0.58, 0.89) | |
| p-Value | 0.0014 Based on stratified log-rank test (p-Value [one-sided] is compared to an alpha boundary of 0.0116)
|
|
| OS | ||
| Number of patients with event (%) | 157 (67) | 175 (75) |
| Median in months (95% CI) | 18.2 (15.3, 21.0) | 14.0 (12.5, 16.1) |
| Hazard ratio (95% CI) | 0.76 (0.61, 0.94) | |
| p-Value | 0.0053 Based on stratified log-rank test (p-Value [one-sided] is compared to an alpha boundary of 0.0083)
|
| Figure 46: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-B96 (CPS ≥1) |
|
|
| Figure 47: Kaplan-Meier Curve for Overall Survival in KEYNOTE-B96 (CPS ≥1) |
|
|
1.5 Urothelial Cancer
KEYTRUDA QLEX, in combination with enfortumab vedotin, is indicated for the treatment of adult patients with locally advanced or metastatic urothelial cancer.
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma:
- who are not eligible for any platinum-containing chemotherapy, or
- who have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy.
KEYTRUDA QLEX, in combination with enfortumab vedotin, as neoadjuvant treatment and then continued after cystectomy as adjuvant treatment, is indicated for the treatment of adult patients with muscle invasive bladder cancer (MIBC) who are ineligible for cisplatin-containing chemotherapy.
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy.
1.9 Esophageal Cancer
KEYTRUDA QLEX is indicated for the treatment of adult patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either:
- in combination with platinum- and fluoropyrimidine-based chemotherapy for patients with tumors that express PD-L1 (CPS ≥ 1) [see Dosage and Administration (2.1)], or
- as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
12.2 Pharmacodynamics
There are no clinically significant exposure-response relationships for efficacy or safety for intravenous pembrolizumab across the approved dosing regimens, regardless of cancer type. The exposures from subcutaneous KEYTRUDA QLEX doses of 395 mg/4,800 units every 3 weeks or 790 mg/9,600 units every 6 weeks are within the range of exposures from intravenous pembrolizumab doses.
12.3 Pharmacokinetics
Pembrolizumab pharmacokinetics were characterized at Cycle 1 and at steady state in patients with advanced solid tumors at the approved recommended dosages and are presented as mean (CV%) unless otherwise specified.
When comparing pembrolizumab exposure following subcutaneous administration every 6 weeks to that of intravenous administration every 6 weeks in Study MK-3475A-D77 [see Clinical Studies (14.1)], the geometric mean ratio (GMR) for Cycle 1 AUC0-6wks was 1.14 (96% CI: 1.06, 1.22) and Cycle 3 Ctrough (i.e., steady state) was 1.67 (94% CI: 1.52, 1.84).
Pembrolizumab steady state was reached by 16 weeks. At steady state following subcutaneous administration, the mean pembrolizumab AUC0-6wks was 2,798 mcg•day/mL for the every 6 week dosing and pembrolizumab AUC0-3wks was 1,343 mcg•day/mL for the every 3 week dosing. Pembrolizumab Ctrough was 39 mcg/mL for the every 6 week dosing and 49 mcg/mL for the every 3 week dosing.
The systemic accumulation ratio was 1.6-fold following administration of KEYTRUDA QLEX 790 mg/9,600 units every 6 weeks and 2.5-fold following administration of KEYTRUDA QLEX 395 mg/4,800 units every 3 weeks.
Absorption
Pembrolizumab bioavailability (CV%) is approximately 60% (14%). Peak concentrations occurred by approximately 4 days.
Distribution
The volume of distribution is 6 L.
Elimination
Pembrolizumab clearance decreases over time, resulting in a steady state clearance (CV%) of - 195 mL/day (40%); this decrease in clearance with time is not considered clinically significant. The terminal half-life is 22 days.
Specific Populations
No clinically significant differences in the pharmacokinetics of pembrolizumab were observed based on age (37 to 87 years), race (63% White, 28% Asian, 3% Black), sex, body weight (37 to 144 kg), tumor type, injection site (thigh or abdomen), estimated glomerular filtration rate (eGFR) ≥15 mL/min/1.73 m2, and mild to moderate hepatic impairment (total bilirubin ≤3 times ULN and any AST). The effect of severe hepatic impairment (total bilirubin >3 times ULN and any AST) on pembrolizumab pharmacokinetics is unknown.
Pediatric Patients
Pembrolizumab exposures in pediatric patients 12 years and older who weigh greater than 40 kg are predicted to be within range of those observed in adult patients at the same dosage.
14.11 Cervical Cancer
FIGO 2014 Stage III-IVA Cervical Cancer with Chemoradiotherapy
The efficacy of intravenous pembrolizumab in combination with CRT (cisplatin and external beam radiation therapy [EBRT] followed by brachytherapy [BT]) was investigated in KEYNOTE-A18 (NCT04221945), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 1060 patients with cervical cancer who had not previously received any definitive surgery, radiation, or systemic therapy for cervical cancer. There were 599 patients with FIGO 2014 Stage III-IVA disease (tumor involves the lower third of the vagina or the pelvic sidewall, or there is hydronephrosis/non-functioning kidney or spread to adjacent pelvic organs, all without spread to distant organs), and 459 patients with FIGO 2014 Stage IB2-IIB disease (clinical lesion >4 cm confined to the cervix, or clinical lesion of any size with extension beyond the uterus, but which has not extended to the pelvic wall or lower third of the vagina) with positive nodes. Two patients had FIGO 2014 Stage IVB disease. Randomization was stratified by planned type of EBRT (Intensity-modulated radiation therapy [IMRT] or volumetric modulated arc therapy [VMAT] vs. non-IMRT and non-VMAT), stage at screening of cervical cancer (FIGO 2014 Stage IB2-IIB vs. FIGO 2014 Stage III-IVA), and planned total radiotherapy dose (EBRT + brachytherapy dose of <70 Gy vs. ≥70 Gy as per equivalent dose [EQD2]).
Patients were randomized (1:1) to one of two treatment arms:
- Intravenous pembrolizumab 200 mg IV every 3 weeks (5 cycles) concurrent with cisplatin 40 mg/m2 IV weekly (5 cycles, an optional sixth infusion could be administered per local practice), and radiotherapy (EBRT followed by BT), followed by intravenous pembrolizumab 400 mg IV every 6 weeks (15 cycles)
- Placebo IV every 3 weeks (5 cycles) concurrent with cisplatin 40 mg/m2 IV weekly (5 cycles, an optional sixth infusion could be administered per local practice), and radiotherapy (EBRT followed by BT), followed by placebo IV every 6 weeks (15 cycles)
Treatment continued until RECIST v1.1-defined progression of disease as determined by investigator or unacceptable toxicity.
Assessment of tumor status was performed every 12 weeks from completion of CRT for the first two years, followed by every 24 weeks in year 3, and then annually. The major efficacy outcome measures were PFS as assessed by investigator according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, or histopathologic confirmation, and OS.
Among the 599 patients with FIGO 2014 Stage III-IVA disease, the baseline characteristics were: median age of 52 years (range: 22 to 87), 17% age 65 or older; 36% White, 34% Asian, 2% Black; 38% Hispanic or Latino; 68% ECOG PS 0 and 32% ECOG PS 1; 93% with CPS ≥1; 71% had positive pelvic and/or positive para-aortic lymph node(s) and 29% had neither positive pelvic nor para-aortic lymph node(s); 83% had squamous cell carcinoma and 17% had non-squamous histology. Regarding radiation, 86% of patients received IMRT or VMAT EBRT, and the median EQD2 dose was 87 Gy (range: 7 to 114).
The trial demonstrated statistically significant improvements in PFS and OS in the ITT population. Exploratory analyses of PFS and OS by the stratification factor of FIGO 2014 stage showed that the improvement in the ITT population was primarily attributed to the results seen in the patients with FIGO 2014 Stage III-IVA disease. Table 86 and Figures 28 and 29 summarize the results in exploratory subgroup analyses of 599 patients with FIGO 2014 Stage III-IVA disease.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and 400 mg every 6 weeks with CRT n=295 |
Placebo with CRT n=304 |
|---|---|---|
| CRT = Chemoradiotherapy NR = not reached |
||
|
OS Results at the time of pre-specified final analysis for OS
|
||
| Number of patients with event (%) | 61 (21) | 90 (30) |
| Hazard ratio Based on the unstratified Cox proportional hazard model (95% CI) |
0.65 (0.47, 0.90) | |
|
PFS by Investigator
Results at the time of first pre-specified interim analysis for PFS
|
||
| Number of patients with event (%) | 61 (21) | 94 (31) |
| Median in months (95% CI) | NR (NR, NR) | NR (18.8, NR) |
| 12-month PFS rate (95% CI) | 81% (75, 85) | 70% (64, 76) |
| Hazard ratio (95% CI) | 0.59 (0.43, 0.81) |
|
|
|
|
Persistent, Recurrent, or Metastatic Cervical Cancer for Tumors Expressing PD-L1 (CPS ≥1)
The efficacy of intravenous pembrolizumab in combination with paclitaxel and cisplatin or paclitaxel and carboplatin, with or without bevacizumab, was investigated in KEYNOTE-826 (NCT03635567), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 617 patients with persistent, recurrent, or first-line metastatic cervical cancer who had not been treated with chemotherapy except when used concurrently as a radio-sensitizing agent. Patients were enrolled regardless of tumor PD-L1 expression status. Patients with autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by metastatic status at initial diagnosis, investigator decision to use bevacizumab, and PD-L1 status (CPS <1 vs. CPS 1 to <10 vs. CPS ≥10). Patients were randomized (1:1) to one of the two treatment groups:
- Treatment Group 1: Intravenous pembrolizumab 200 mg plus chemotherapy with or without bevacizumab
- Treatment Group 2: Placebo plus chemotherapy with or without bevacizumab
The investigator selected one of the following four treatment regimens prior to randomization:
- Paclitaxel 175 mg/m2 + cisplatin 50 mg/m2
- Paclitaxel 175 mg/m2 + cisplatin 50 mg/m2 + bevacizumab 15 mg/kg
- Paclitaxel 175 mg/m2 + carboplatin AUC 5 mg/mL/min
- Paclitaxel 175 mg/m2 + carboplatin AUC 5 mg/mL/min + bevacizumab 15 mg/kg
All study medications were administered as an intravenous infusion. All study treatments were administered on Day 1 of each 3-week treatment cycle. Cisplatin could be administered on Day 2 of each 3-week treatment cycle. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease, unacceptable toxicity, or a maximum of 24 months. Administration of intravenous pembrolizumab was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed every 9 weeks for the first year, followed by every 12 weeks thereafter. The main efficacy outcome measures were OS and PFS as assessed by investigator according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ. Additional efficacy outcome measures were ORR and DoR, according to RECIST v1.1, as assessed by investigator.
Of the 617 enrolled patients, 548 patients (89%) had tumors expressing PD-L1 with a CPS ≥1. Among these 548 enrolled patients with tumors expressing PD-L1, 273 patients were randomized to intravenous pembrolizumab in combination with chemotherapy with or without bevacizumab, and 275 patients were randomized to placebo in combination with chemotherapy with or without bevacizumab. Sixty-three percent of the 548 patients received bevacizumab as part of study treatment. The baseline characteristics of the 548 patients were: median age of 51 years (range: 22 to 82), 16% age 65 or older; 59% White, 18% Asian, 6% American Indian or Alaska Native, and 1% Black; 37% Hispanic or Latino; 56% ECOG performance status 0 and 43% ECOG performance status 1. Seventy-five percent had squamous cell carcinoma, 21% adenocarcinoma, and 5% adenosquamous histology, and 32% of patients had metastatic disease at diagnosis. At study entry, 21% of patients had metastatic disease only and 79% had persistent or recurrent disease with or without distant metastases, of whom 39% had received prior chemoradiation only and 17% had received prior chemoradiation plus surgery.
A statistically significant improvement in OS and PFS was demonstrated in patients randomized to receive intravenous pembrolizumab compared with patients randomized to receive placebo. An updated OS analysis was conducted at the time of final analysis when 354 deaths in the CPS ≥1 population were observed. Table 87 and Figure 30 summarize the key efficacy measures for KEYNOTE-826 for patients with tumors expressing PD-L1 (CPS ≥1).
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and chemotherapy Chemotherapy (paclitaxel and cisplatin or paclitaxel and carboplatin) with or without bevacizumabn=273 |
Placebo and chemotherapy with or without bevacizumab n=275 |
|---|---|---|
| + Denotes ongoing response NR = not reached |
||
| OS | ||
| Number of patients with event (%) | 118 (43.2) | 154 (56.0) |
| Median in months (95% CI) | NR (19.8, NR) | 16.3 (14.5, 19.4) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.64 (0.50, 0.81) | |
| p-Value p-Value (one-sided) is compared with the allocated alpha of 0.0055 for this interim analysis (with 72% of the planned number of events for final analysis)
|
0.0001 | |
| Updated OS | ||
| Number of patients with event (%) | 153 (56.0%) | 201 (73.1%) |
| Median in months (95% CI) | 28.6 (22.1, 38.0) | 16.5 (14.5, 20.0) |
| Hazard ratio (95% CI) | 0.60 (0.49, 0.74) | |
| PFS | ||
| Number of patients with event (%) | 157 (57.5) | 198 (72.0) |
| Median in months (95% CI) | 10.4 (9.7, 12.3) | 8.2 (6.3, 8.5) |
| Hazard ratio (95% CI) | 0.62 (0.50, 0.77) | |
| p-Value p-Value (one-sided) is compared with the allocated alpha of 0.0014 for this interim analysis (with 82% of the planned number of events for final analysis)
|
< 0.0001 | |
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
68% (62, 74) | 50% (44, 56) |
| Complete response rate | 23% | 13% |
| Partial response rate | 45% | 37% |
| Duration of Response | ||
| Median in months (range) | 18.0 (1.3+, 24.2+) | 10.4 (1.5+, 22.0+) |
|
|
Previously Treated Recurrent or Metastatic Cervical Cancer for Tumors Expressing PD-L1 (CPS≥ 1)
The efficacy of intravenous pembrolizumab was investigated in 98 patients with recurrent or metastatic cervical cancer enrolled in a single cohort (Cohort E) in KEYNOTE-158 (NCT02628067), a multicenter, non-randomized, open-label, multi-cohort trial. The trial excluded patients with autoimmune disease or a medical condition that required immunosuppression. Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression. Patients with initial radiographic disease progression could receive additional doses of treatment during confirmation of progression unless disease progression was symptomatic, was rapidly progressive, required urgent intervention, or occurred with a decline in performance status. Patients without disease progression could be treated for up to 24 months. Assessment of tumor status was performed every 9 weeks for the first 12 months, and every 12 weeks thereafter. The major efficacy outcome measures were ORR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, as assessed by BICR, and DoR.
Among the 98 patients in Cohort E, 77 (79%) had tumors that expressed PD-L1 with a CPS ≥ 1 and received at least one line of chemotherapy in the metastatic setting. PD-L1 status was determined using the IHC 22C3 pharmDx assay. The baseline characteristics of these 77 patients were: median age of 45 years (range: 27 to 75); 81% White, 14% Asian, and 3% Black; 32% ECOG PS of 0 and 68% ECOG PS of 1; 92% had squamous cell carcinoma, 6% adenocarcinoma, and 1% adenosquamous histology; 95% had M1 disease and 5% had recurrent disease; and 35% had one and 65% had two or more prior lines of therapy in the recurrent or metastatic setting.
No responses were observed in patients whose tumors did not have PD-L1 expression (CPS <1). Efficacy results are summarized in Table 88 for patients with PD-L1 expression (CPS ≥1).
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=77 Median follow-up time of 11.7 months (range 0.6 to 22.7 months)
|
|---|---|
| + Denotes ongoing response NR = not reached |
|
| Objective Response Rate | |
| ORR (95% CI) | 14.3% (7.4, 24.1) |
| Complete response rate | 2.6% |
| Partial response rate | 11.7% |
| Duration of Response | |
| Median in months (range) | NR (4.1, 18.6+) Based on patients (n=11) with a response by independent review
|
| % with duration ≥6 months | 91% |
2.1 Patient Selection
See information on FDA-authorized tests for intravenous pembrolizumab. Information on FDA-authorized tests for patient selection is available at: http://www.fda.gov/CompanionDiagnostics.
Patient Selection for Single-Agent Treatment
Select patients for treatment with KEYTRUDA QLEX as a single agent based on the presence of positive PD-L1 expression in:
- Stage III NSCLC who are not candidates for surgical resection or definitive chemoradiation [see Clinical Studies (14.3)].
- metastatic NSCLC [see Clinical Studies (14.3)].
- first-line treatment of metastatic or unresectable, recurrent HNSCC [see Clinical Studies (14.5)].
- previously treated recurrent locally advanced or metastatic esophageal cancer [see Clinical Studies (14.10)].
- recurrent or metastatic cervical cancer with disease progression on or after chemotherapy [see Clinical Studies (14.11)].
For the MSI-H/dMMR indications, select patients for treatment with KEYTRUDA QLEX as a single agent based on MSI-H/dMMR status in tumor specimens [see Clinical Studies (14.7, 14.8)].
For the TMB-H indication, select patients for treatment with KEYTRUDA QLEX as a single agent based on TMB-H status in tumor specimens [see Clinical Studies (14.17)].
Because subclonal dMMR mutations and microsatellite instability may arise in high-grade gliomas during temozolomide therapy, it is recommended to test for TMB-H, MSI-H, and dMMR in the primary tumor specimens obtained prior to initiation of temozolomide chemotherapy in patients with high-grade gliomas.
Additional Patient Selection Information for MSI-H or dMMR in Patients with non-CRC Solid Tumors
Due to discordance between local tests and FDA-authorized tests, confirmation of MSI-H or dMMR status is recommended by an FDA-authorized test in patients with MSI-H or dMMR solid tumors, if feasible. If unable to perform confirmatory MSI-H/dMMR testing, the presence of TMB ≥10 mut/Mb, as determined by an FDA-authorized test, may be used to select patients for treatment [see Clinical Studies (14.7)].
Patient Selection for Combination Therapy
For use of KEYTRUDA QLEX as a single agent as neoadjuvant treatment, then in combination with radiotherapy (RT) with or without chemotherapy then continued as a single agent as adjuvant treatment, select patients based on presence of positive PD-L1 expression (CPS ≥1) in resectable locally advanced HNSCC [see Clinical Studies (14.5)].
For use of KEYTRUDA QLEX in combination with chemotherapy, select patients based on the presence of positive PD-L1 expression (CPS ≥1) in locally advanced unresectable or metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma, and esophageal or gastroesophageal junction (GEJ) carcinoma [see Clinical Studies (14.9), (14.10)].
- An FDA-authorized test for the detection of PD-L1 for the selection of patients with PD-L1 (CPS ≥1) expression in esophageal carcinoma in combination with platinum- and fluoropyrimidine-based chemotherapy is not available.
For use of KEYTRUDA QLEX in combination with chemotherapy, with or without bevacizumab, select patients based on the presence of positive PD-L1 expression in persistent, recurrent, or metastatic cervical cancer [see Clinical Studies (14.11)].
For the pMMR/not MSI-H advanced endometrial carcinoma indication, select patients for treatment with KEYTRUDA QLEX in combination with lenvatinib based on MMR or MSI status in tumor specimens [see Clinical Studies (14.16)].
For use of KEYTRUDA QLEX in combination with chemotherapy, select patients based on the presence of positive PD-L1 expression in locally recurrent unresectable or metastatic TNBC [see Clinical Studies (14.19)].
For use of KEYTRUDA QLEX in combination with paclitaxel, with or without bevacizumab, select patients based on the presence of positive PD-L1 expression (CPS ≥1) in platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma [see Clinical Studies (14.20)].
2.3 Recommended Dosage
The recommended dosages of KEYTRUDA QLEX are presented in Table 1.
- Every 3-week dosing (395 mg pembrolizumab and 4,800 units berahyaluronidase alfa): inject 2.4 mL subcutaneously over 1 minute.
- Every 6-week dosing (790 mg pembrolizumab and 9,600 units berahyaluronidase alfa): inject 4.8 mL subcutaneously over 2 minutes.
| Indication |
Recommended Dosage of KEYTRUDA QLEX |
Duration/Timing of Treatment |
|---|---|---|
| Monotherapy | ||
| Adult patients with unresectable or metastatic melanoma |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease progression or unacceptable toxicity |
| Adjuvant treatment of adult patients with melanoma, NSCLC, or RCC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease recurrence, unacceptable toxicity, or up to 12 months |
| Adult patients with NSCLC, HNSCC, locally advanced or metastatic Urothelial Carcinoma, MSI-H or dMMR Cancer, MSI-H or dMMR CRC, MSI-H or dMMR Endometrial Carcinoma, Esophageal Cancer, Cervical Cancer, HCC, MCC, TMB-H Cancer, or cSCC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with high-risk BCG- unresponsive NMIBC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until persistent or recurrent high-risk NMIBC, disease progression, unacceptable toxicity, or up to 24 months |
| Pediatric patients The recommended dosage for melanoma, MSI-H or dMMR cancer, MCC and TMB-H cancer has not been established in pediatric patients 12 years and older who weigh 40 kg or less [see Use in Specific Populations (8.4)]. (12 years and olderwho weigh greater than 40 kg) with MSI-H or dMMR Cancer, MCC, or TMB- H Cancer |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Pediatric patients (12 years and older who weigh greater than 40 kg) for adjuvant treatment of melanoma |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease recurrence, unacceptable toxicity, or up to 12 months |
|
Combination Therapy
Refer to the Prescribing Information for the agents administered in combination with KEYTRUDA QLEX for recommended dosing information, as appropriate.
|
||
| Adult patients with resectable NSCLC | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Neoadjuvant treatment in combination with chemotherapy for 12 weeks or until disease progression that precludes definitive surgery or unacceptable toxicity, followed by adjuvant treatment with KEYTRUDA QLEX as a single agent after surgery for 39 weeks or until disease recurrence or unacceptable toxicity |
| Adult patients with NSCLC, MPM, HNSCC, HER2-negative Gastric Cancer, Esophageal Cancer, or BTC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with locally advanced or metastatic urothelial cancer |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX after enfortumab vedotin when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with locally advanced HNSCC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to cisplatin when given on the same day. |
Neoadjuvant:
disease recurrence or unacceptable toxicity or up to one year |
| Adult patients with MIBC | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX after enfortumab vedotin when given on the same day. |
Neoadjuvant:
|
| Adult patients with HER2-positive Gastric Cancer |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to trastuzumab and chemotherapy when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with Cervical Cancer | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemoradiotherapy or prior to chemotherapy with or without bevacizumab when given on the same day. |
Until disease progression, unacceptable toxicity, or for KEYTRUDA QLEX, up to 24 months |
| Adult patients with RCC | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX in combination with axitinib 5 mg orally twice daily When axitinib is used in combination with KEYTRUDA QLEX, dose escalation of axitinib above the initial 5 mg dose may be considered at intervals of six weeks or longer.
or Administer KEYTRUDA QLEX in combination with lenvatinib 20 mg orally once daily. |
Until disease progression, unacceptable toxicity, or for KEYTRUDA QLEX, up to 24 months |
| Adult patients with Endometrial Carcinoma |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to carboplatin and paclitaxel when given on the same day. or Administer KEYTRUDA QLEX in combination with lenvatinib 20 mg orally once daily. |
Until disease progression, unacceptable toxicity, or for KEYTRUDA QLEX, up to 24 months |
| Adult patients with high-risk early-stage TNBC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Neoadjuvant treatment in combination with chemotherapy for 24 weeks (8 doses of 395 mg/4,800 units every 3 weeks or 4 doses of 790 mg/9,600 units every 6 weeks) or until disease progression or unacceptable toxicity, followed by adjuvant treatment with KEYTRUDA QLEX as a single agent for up to 27 weeks (9 doses of 395 mg/4,800 units every 3 weeks or 5 doses of 790 mg/9,600 units every 6 weeks) or until disease recurrence or unacceptable toxicity Patients who experience disease progression or unacceptable toxicity related to KEYTRUDA QLEX with neoadjuvant treatment in combination with chemotherapy should not receive adjuvant single agent KEYTRUDA QLEX.
|
| Adult patients with locally recurrent unresectable or metastatic TNBC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with Ovarian Cancer | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to paclitaxel with or without bevacizumab when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
1 Indications and Usage
KEYTRUDA QLEX is a combination of pembrolizumab, a programmed death receptor-1 (PD-1)-blocking antibody, and berahyaluronidase alfa, an endoglycosidase, indicated:
Melanoma
- for the treatment of adult patients with unresectable or metastatic melanoma. (1.1)
- for the adjuvant treatment of adult and pediatric patients 12 years and older with Stage IIB, IIC, or III melanoma following complete resection. (1.1)
Non-Small Cell Lung Cancer (NSCLC)
- in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with metastatic nonsquamous NSCLC, with no EGFR or ALK genomic tumor aberrations. (1.2)
- in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, as first-line treatment of adult patients with metastatic squamous NSCLC. (1.2)
- as a single agent for the first-line treatment of adult patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-authorized test, with no EGFR or ALK genomic tumor aberrations, and is:
- as a single agent for the treatment of adult patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-authorized test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA QLEX. (1.2, 2.1)
- for the treatment of adult patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. (1.2)
- as a single agent, for adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC. (1.2)
Malignant Pleural Mesothelioma (MPM)
- in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with unresectable advanced or metastatic MPM. (1.3)
Head and Neck Squamous Cell Cancer (HNSCC)
- for the treatment of adult patients with resectable locally advanced HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test, as a single agent as neoadjuvant treatment, continued as adjuvant treatment in combination with radiotherapy (RT) with or without cisplatin and then as a single agent. (1.4)
- in combination with platinum and FU for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC. (1.4)
- as a single agent for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test. (1.4, 2.1)
- as a single agent for the treatment of adult patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy. (1.4)
Urothelial Cancer
- in combination with enfortumab vedotin, for the treatment of adult patients with locally advanced or metastatic urothelial cancer. (1.5)
- as a single agent for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma who:
- are not eligible for any platinum-containing chemotherapy, or
- who have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. (1.5)
- in combination with enfortumab vedotin, as neoadjuvant treatment and then continued after cystectomy as adjuvant treatment of adult patients with muscle invasive bladder cancer (MIBC) who are ineligible for cisplatin-containing chemotherapy. (1.5)
- as a single agent for the treatment of adult patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy. (1.5)
Microsatellite Instability-High or Mismatch Repair Deficient Cancer
- for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options. (1.6, 2.1)
Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer (CRC)
- for the treatment of adult patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-authorized test. (1.7, 2.1)
Gastric Cancer
- in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.8)
- in combination with fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.8)
Esophageal Cancer
- for the treatment of adult patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either:
- in combination with platinum- and fluoropyrimidine-based chemotherapy for patients whose tumors express PD-L1 (CPS ≥1), or
- as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. (1.9, 2.1)
Cervical Cancer
- in combination with chemoradiotherapy, for the treatment of adult patients with locally advanced cervical cancer involving the lower third of the vagina, with or without extension to pelvic sidewall, or hydronephrosis/non-functioning kidney, or spread to adjacent pelvic organs (FIGO 2014 Stage III-IVA). (1.10)
- in combination with chemotherapy, with or without bevacizumab, for the treatment of adult patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.10, 2.1)
- as a single agent for the treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.10, 2.1)
Hepatocellular Carcinoma (HCC)
- for the treatment of adult patients with HCC secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen. (1.11)
Biliary Tract Cancer (BTC)
- in combination with gemcitabine and cisplatin, for the treatment of adult patients with locally advanced unresectable or metastatic biliary tract cancer. (1.12)
Merkel Cell Carcinoma (MCC)
- for the treatment of adult and pediatric patients 12 years and older with recurrent locally advanced or metastatic Merkel cell carcinoma. (1.13)
Renal Cell Carcinoma (RCC)
- in combination with axitinib, for the first-line treatment of adult patients with advanced RCC. (1.14)
- in combination with lenvatinib, for the first-line treatment of adult patients with advanced RCC. (1.14)
- for the adjuvant treatment of adult patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions. (1.14)
Endometrial Carcinoma
- in combination with carboplatin and paclitaxel, followed by KEYTRUDA QLEX as a single agent, for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma. (1.15)
- in combination with lenvatinib, for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) or not MSI-H as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. (1.15, 2.1)
- as a single agent, for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. (1.15, 2.1)
Tumor Mutational Burden-High (TMB-H) Cancer
- for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.1 (1.16, 2.1)
- Limitations of Use: The safety and effectiveness of KEYTRUDA QLEX in pediatric patients 12 years and older with TMB-H central nervous system cancers have not been established.
Cutaneous Squamous Cell Carcinoma (cSCC)
- for the treatment of adult patients with recurrent or metastatic cSCC or locally advanced cSCC that is not curable by surgery or radiation. (1.17)
Triple-Negative Breast Cancer (TNBC)
- for the treatment of adult patients with high-risk early-stage TNBC in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. (1.18)
- in combination with chemotherapy, for the treatment of adult patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. (1.18, 2.1)
Ovarian Cancer
- in combination with paclitaxel, with or without bevacizumab, is indicated for the treatment of adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test, and who have received one or two prior systemic treatment regimens. (1.19, 2.1)
1 This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
12.1 Mechanism of Action
Binding of the PD-1 ligands, PD-L1 and PD-L2, to the PD-1 receptor found on T cells, inhibits T cell proliferation and cytokine production. Upregulation of PD-1 ligands occurs in some tumors and signaling through this pathway can contribute to inhibition of active T-cell immune surveillance of tumors. Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the anti-tumor immune response. In syngeneic mouse tumor models, blocking PD-1 activity resulted in decreased tumor growth.
In syngeneic mouse tumor models, combination treatment of a PD-1 blocking antibody and kinase inhibitor lenvatinib decreased tumor-associated macrophages, increased activated cytotoxic T cells, and reduced tumor growth compared to either treatment alone.
Berahyaluronidase alfa, an endoglycosidase, is a variant of human hyaluronidase PH20 that temporarily and locally breaks down hyaluronan. Hyaluronan is a polysaccharide found in the extracellular matrix of the subcutaneous tissue. Unlike the stable structural components of the interstitial matrix, hyaluronan has a half-life of approximately 0.5 days. Hyaluronidase increases permeability of the subcutaneous tissue by depolymerizing hyaluronan.
In the doses administered, the effects of berahyaluronidase alfa are reversible and permeability of the subcutaneous tissue is restored within 24 to 48 hours.
2.4 Dosage Modifications
No dose reduction for KEYTRUDA QLEX is recommended. In general, withhold KEYTRUDA QLEX for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue KEYTRUDA QLEX for Life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating steroids.
Dosage modifications for KEYTRUDA QLEX for adverse reactions that require management different from these general guidelines are summarized in Table 2.
| Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.0
|
Dosage Modification |
|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, DRESS = Drug Rash with Eosinophilia and Systemic Symptoms, SJS = Stevens Johnson Syndrome, TEN = toxic epidermal necrolysis, ULN = upper limit normal | ||
| Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)] | ||
| Pneumonitis | Grade 2 | Withhold Resume in patients with complete or partial resolution (Grades 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of initiating steroids or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating steroids.
|
| Grade 3 or 4 | Permanently discontinue | |
| Colitis | Grade 2 or 3 | Withhold |
| Grade 4 | Permanently discontinue | |
|
Hepatitis with no tumor involvement of the liver |
AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN |
Withhold |
| For liver enzyme elevations in patients treated with combination therapy with axitinib, see Table 3. |
AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue |
| Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue KEYTRUDA QLEX based on recommendations for hepatitis with no liver involvement.
|
Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN |
Withhold |
| ALT or AST increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Endocrinopathies | Grade 3 or 4 | Withhold until clinically stable or permanently discontinue depending on severity |
| Nephritis with Renal Dysfunction | Grade 2 or 3 increased blood creatinine | Withhold |
| Grade 4 increased blood creatinine | Permanently discontinue | |
| Exfoliative Dermatologic Conditions | Suspected SJS, TEN, or DRESS | Withhold |
| Confirmed SJS, TEN, or DRESS | Permanently discontinue | |
| Myocarditis | Grade 2, 3, or 4 | Permanently discontinue |
| Neurological Toxicities | Grade 2 | Withhold |
| Grade 3 or 4 | Permanently discontinue | |
| Other Adverse Reactions | ||
| Hypersensitivity and Administration-Related Systemic Reactions [see Warnings and Precautions (5.2)] |
Grade 1 or 2 | Interrupt injection (if not already fully administered). If symptoms resolve, resume injection |
| Grade 3 or 4 | Permanently discontinue |
The following table represents dosage modifications that are different from those described above for KEYTRUDA QLEX or in the Full Prescribing Information for the drug administered in combination.
| Treatment | Adverse Reaction | Severity | Dosage Modification |
|---|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal | |||
| KEYTRUDA QLEX in combination with axitinib |
Liver enzyme elevations Consider corticosteroid therapy
|
ALT or AST increases to at least 3 times but less than 10 times ULN without concurrent total bilirubin at least 2 times ULN | Withhold both KEYTRUDA QLEX and axitinib until resolution to Grades 0 or 1 Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. Consider rechallenge with a single drug or sequential rechallenge with both drugs after recovery. If rechallenging with axitinib, consider dose reduction as per the axitinib Prescribing Information.
|
| ALT or AST increases to more than 3 times ULN with concurrent total bilirubin at least 2 times ULN or ALT or AST ≥10 times ULN |
Permanently discontinue both KEYTRUDA QLEX and axitinib |
Recommended Dose Modifications for Adverse Reactions for KEYTRUDA QLEX in Combination with Lenvatinib
When administering KEYTRUDA QLEX in combination with lenvatinib, modify the dosage of one or both drugs. Withhold or discontinue KEYTRUDA QLEX as shown in Table 2. Refer to lenvatinib prescribing information for additional dose modification information.
1.12 Biliary Tract Cancer
KEYTRUDA QLEX, in combination with gemcitabine and cisplatin, is indicated for the treatment of adult patients with locally advanced unresectable or metastatic biliary tract cancer (BTC).
1.14 Renal Cell Carcinoma
KEYTRUDA QLEX, in combination with axitinib, is indicated for the first-line treatment of adult patients with advanced renal cell carcinoma (RCC).
KEYTRUDA QLEX, in combination with lenvatinib, is indicated for the first-line treatment of adult patients with advanced RCC.
KEYTRUDA QLEX is indicated for the adjuvant treatment of adult patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions [see Clinical Studies (14.15)].
5.5 Embryo Fetal Toxicity
Based on its mechanism of action, KEYTRUDA QLEX can cause fetal harm when administered to a pregnant woman. Animal studies have demonstrated that inhibition of the PD-L1/PD-1 pathway can lead to increased risk of immune-related rejection of the developing fetus resulting in fetal death. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with KEYTRUDA QLEX and for 4 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
1.13 Merkel Cell Carcinoma
KEYTRUDA QLEX is indicated for the treatment of adult and pediatric patients 12 years and older with recurrent locally advanced or metastatic Merkel cell carcinoma (MCC).
1.15 Endometrial Carcinoma
KEYTRUDA QLEX, in combination with carboplatin and paclitaxel, followed by KEYTRUDA QLEX as a single agent, is indicated for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma.
KEYTRUDA QLEX, in combination with lenvatinib, is indicated for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) or not MSI-H as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation [see Dosage and Administration (2.1)].
14.13 Biliary Tract Cancer
The efficacy of intravenous pembrolizumab in combination with gemcitabine and cisplatin chemotherapy was investigated in KEYNOTE-966 (NCT04003636), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 1069 patients with locally advanced unresectable or metastatic BTC, who had not received prior systemic therapy in the advanced disease setting. Patients with autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by region (Asia vs. non-Asia), locally advanced versus metastatic, and site of origin (gallbladder, intrahepatic or extrahepatic cholangiocarcinoma).
Patients were randomized (1:1) to intravenous pembrolizumab 200 mg on Day 1 plus gemcitabine 1000 mg/m2 and cisplatin 25 mg/m2 on Day 1 and Day 8 every 3 weeks, or placebo on Day 1 plus gemcitabine 1000 mg/m2 and cisplatin 25 mg/m2 on Day 1 and Day 8 every 3 weeks. Study medications were administered via intravenous infusion. Treatment continued until unacceptable toxicity or disease progression. For pembrolizumab, treatment continued for a maximum of 35 cycles, or approximately 24 months. For gemcitabine, treatment could be continued beyond 8 cycles while for cisplatin, treatment could be administered for a maximum of 8 cycles.
Administration of intravenous pembrolizumab with chemotherapy was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered by the investigator to be deriving clinical benefit. Assessment of tumor status was performed at baseline and then every 6 weeks through 54 weeks, followed by every 12 weeks thereafter.
Study population characteristics were median age of 64 years (range: 23 to 85), 47% age 65 or older; 52% male; 49% White, 46% Asian, 1.3% Black or African American; 10% Hispanic or Latino; 46% ECOG PS of 0 and 54% ECOG PS of 1; 31% of patients had a history of hepatitis B infection, and 3% had a history of hepatitis C infection.
The major efficacy outcome measure was OS. Additional efficacy outcome measures were PFS, ORR and DoR as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Table 90 and Figure 32 summarize the efficacy results for KEYNOTE-966.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks with gemcitabine/cisplatin n=533 |
Placebo with gemcitabine/cisplatin n=536 |
|---|---|---|
| NS = not significant | ||
|
OS Results at the pre-specified final OS analysis
|
||
| Number of patients with event (%) | 414 (78%) | 443 (83%) |
| Median in months (95% CI) | 12.7 (11.5, 13.6) | 10.9 (9.9, 11.6) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.83 (0.72, 0.95) | |
| p-Value One-sided p-Value based on a stratified log-rank test
|
0.0034 | |
|
PFS
Results at pre-specified final analysis of PFS and ORR
|
||
| Number (%) of patients with event | 361 (68%) | 391 (73%) |
| Median in months (95% CI) | 6.5 (5.7, 6.9) | 5.6 (5.1, 6.6) |
| Hazard ratio (95% CI) | 0.86 (0.75, 1.00) | |
| p-Value | NS | |
| Objective Response Rate | ||
| ORR Confirmed complete response or partial response (95% CI) |
29% (25, 33) | 29% (25, 33) |
| Number (%) of complete responses | 11 (2.1%) | 7 (1.3%) |
| Number (%) of partial responses | 142 (27%) | 146 (27%) |
| p-Value One-sided p-Value based on the stratified Miettinen and Nurminen analysis
|
NS | |
| Duration of Response | n=156 | n=152 |
| Median in months Based on Kaplan-Meier estimate (95% CI)
|
8.3 (6.9, 10.2) | 6.8 (5.7, 7.1) |
| Figure 32: Kaplan-Meier Curve for Overall Survival in KEYNOTE-966 |
|
|
14.15 Renal Cell Carcinoma
First-line treatment with axitinib
KEYNOTE-426
The efficacy of intravenous pembrolizumab in combination with axitinib was investigated in KEYNOTE-426 (NCT02853331), a randomized, multicenter, open-label trial conducted in 861 patients who had not received systemic therapy for advanced RCC. Patients were enrolled regardless of PD-L1 tumor expression status. Patients with active autoimmune disease requiring systemic immunosuppression within the last 2 years were ineligible. Randomization was stratified by International Metastatic RCC Database Consortium (IMDC) risk categories (favorable versus intermediate versus poor) and geographic region (North America versus Western Europe versus “Rest of the World”).
Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg intravenously every 3 weeks up to 24 months in combination with axitinib 5 mg orally, twice daily. Patients who tolerated axitinib 5 mg twice daily for 2 consecutive cycles (6 weeks) could increase to 7 mg and then subsequently to 10 mg twice daily. Axitinib could be interrupted or reduced to 3 mg twice daily and subsequently to 2 mg twice daily to manage toxicity.
- Sunitinib 50 mg orally, once daily for 4 weeks and then off treatment for 2 weeks.
Treatment with intravenous pembrolizumab and axitinib continued until RECIST v1.1-defined progression of disease or unacceptable toxicity. Administration of intravenous pembrolizumab and axitinib was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed at baseline, after randomization at Week 12, then every 6 weeks thereafter until Week 54, and then every 12 weeks thereafter.
The study population characteristics were: median age of 62 years (range: 26 to 90), 38% age 65 or older; 73% male; 79% White and 16% Asian; 20% and 80% of patients had a baseline KPS of 70 to 80 and 90 to 100, respectively; and patient distribution by IMDC risk categories was 31% favorable, 56% intermediate, and 13% poor.
The main efficacy outcome measures were OS and PFS as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ. Additional efficacy outcome measures included ORR, as assessed by BICR. A statistically significant improvement in OS was demonstrated at the first pre-specified interim analysis in patients randomized to intravenous pembrolizumab in combination with axitinib compared with sunitinib. The trial also demonstrated statistically significant improvements in PFS and ORR. An updated OS analysis was conducted when 418 deaths were observed based on the planned number of deaths for the pre-specified final analysis. Table 92 and Figure 33 summarize the efficacy results for KEYNOTE-426.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and Axitinib n=432 |
Sunitinib n=429 |
|---|---|---|
| NR = not reached | ||
| OS | ||
| Number of patients with event (%) | 59 (14%) | 97 (23%) |
| Median in months (95% CI) | NR (NR, NR) | NR (NR, NR) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.53 (0.38, 0.74) | |
| p-Value Based on stratified log-rank test
|
<0.0001 p-Value (one-sided) is compared with the allocated alpha of 0.0001 for this interim analysis (with 39% of the planned number of events for final analysis).
|
|
| Updated OS | ||
| Number of patients with event (%) | 193 (45%) | 225 (52%) |
| Median in months (95% CI) | 45.7 (43.6, NR) | 40.1 (34.3, 44.2) |
| Hazard ratio (95% CI) | 0.73 (0.60, 0.88) | |
| PFS | ||
| Number of patients with event (%) | 183 (42%) | 213 (50%) |
| Median in months (95% CI) | 15.1 (12.6, 17.7) | 11.0 (8.7, 12.5) |
| Hazard ratio (95% CI) | 0.69 (0.56, 0.84) | |
| p-Value | 0.0001 p-Value (one-sided) is compared with the allocated alpha of 0.0013 for this interim analysis (with 81% of the planned number of events for final analysis).
|
|
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
59% (54, 64) | 36% (31, 40) |
| Complete response rate | 6% | 2% |
| Partial response rate | 53% | 34% |
| p-Value Based on Miettinen and Nurminen method stratified by IMDC risk group and geographic region
|
<0.0001 |
| Figure 33: Kaplan-Meier Curve for Updated Overall Survival in KEYNOTE-426 |
|
|
In an exploratory analysis, the updated analysis of OS in patients with IMDC favorable, intermediate, intermediate/poor, and poor risk demonstrated a HR of 1.17 (95% CI: 0.76, 1.80), 0.67 (95% CI: 0.52, 0.86), 0.64 (95% CI: 0.52, 0.80), and 0.51 (95% CI: 0.32, 0.81), respectively.
First-line treatment with lenvatinib
KEYNOTE-581
The efficacy of intravenous pembrolizumab in combination with lenvatinib was investigated in KEYNOTE-581 (NCT02811861), a multicenter, open-label, randomized trial conducted in 1069 patients with advanced RCC in the first-line setting. Patients were enrolled regardless of PD-L1 tumor expression status. Patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible. Randomization was stratified by geographic region (North America versus Western Europe versus “Rest of the World”) and Memorial Sloan Kettering Cancer Center (MSKCC) prognostic groups (favorable versus intermediate versus poor risk).
Patients were randomized (1:1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg intravenously every 3 weeks up to 24 months in combination with lenvatinib 20 mg orally once daily.
- Lenvatinib 18 mg orally once daily in combination with everolimus 5 mg orally once daily.
- Sunitinib 50 mg orally once daily for 4 weeks then off treatment for 2 weeks.
Treatment continued until unacceptable toxicity or disease progression. Administration of intravenous pembrolizumab with lenvatinib was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered by the investigator to be deriving clinical benefit. intravenous pembrolizumab was continued for a maximum of 24 months; however, treatment with lenvatinib could be continued beyond 24 months. Assessment of tumor status was performed at baseline and then every 8 weeks.
The study population characteristics were: median age of 62 years (range: 29 to 88 years), 42% age 65 or older; 75% male; 74% White, 21% Asian, 1% Black, and 2% other races; 18% and 82% of patients had a baseline KPS of 70 to 80 and 90 to 100, respectively; patient distribution by MSKCC risk categories was 27% favorable, 64% intermediate, and 9% poor. Common sites of metastases in patients were lung (68%), lymph node (45%), and bone (25%).
The major efficacy outcome measures were PFS, as assessed by independent radiologic review (IRC) according to RECIST v1.1, and OS. Additional efficacy outcome measures included confirmed ORR as assessed by IRC. intravenous pembrolizumab in combination with lenvatinib demonstrated statistically significant improvements in PFS, OS, and ORR compared with sunitinib. An updated OS analysis was conducted when 304 deaths were observed based on the planned number of deaths for the pre-specified final analysis. Table 93 and Figures 34 and 35 summarize the efficacy results for KEYNOTE-581.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and Lenvatinib n=355 |
Sunitinib n=357 |
|---|---|---|
| Tumor assessments were based on RECIST 1.1; only confirmed responses are included for ORR. Data cutoff date = 28 Aug 2020, Updated OS cutoff date = 31 July 2022 CI = confidence interval; NR= Not reached |
||
| Progression-Free Survival (PFS) | ||
| Number of events, n (%) | 160 (45%) | 205 (57%) |
| Progressive disease | 145 (41%) | 196 (55%) |
| Death | 15 (4%) | 9 (3%) |
| Median PFS in months (95% CI) | 23.9 (20.8, 27.7) | 9.2 (6.0, 11.0) |
| Hazard ratio Hazard ratio is based on a Cox Proportional Hazards Model. Stratified by geographic region and MSKCC prognostic groups. (95% CI) |
0.39 (0.32, 0.49) | |
| p-Value Two-sided p-Value based on stratified log-rank test.
|
<0.0001 | |
| Overall Survival (OS) | ||
| Number of deaths, n (%) | 80 (23%) | 101 (28%) |
| Median OS in months (95% CI) | NR (33.6, NR) | NR (NR, NR) |
| Hazard ratio (95% CI) | 0.66 (0.49, 0.88) | |
| p-Value | 0.0049 | |
| Updated OS | ||
| Number of deaths, n (%) | 149 (42%) | 159 (45%) |
| Median OS in months (95% CI) | 53.7 (48.7, NR) | 54.3 (40.9, NR) |
| Hazard ratio (95% CI) | 0.79 (0.63, 0.99) | |
| Objective Response Rate (Confirmed) | ||
| ORR, n (%) | 252 (71%) | 129 (36%) |
| (95% CI) | (66, 76) | (31, 41) |
| Complete response rate | 16% | 4% |
| Partial response rate | 55% | 32% |
| p-Value Two-sided p-Value based upon CMH test.
|
<0.0001 |
| Figure 34: Kaplan-Meier Curve for PFS in KEYNOTE-581 |
|
|
| Figure 35: Kaplan-Meier Curve for Updated Overall Survival in KEYNOTE-581 |
|
|
KEYNOTE-B61
The efficacy of intravenous pembrolizumab in combination with lenvatinib was investigated in KEYNOTE-B61 (NCT04704219), a multicenter, single-arm trial that enrolled 160 patients with advanced or metastatic non-clear cell RCC in the first-line setting. Patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible.
Patients received intravenous pembrolizumab 400 mg every 6 weeks in combination with lenvatinib 20 mg orally once daily. Intravenous pembrolizumab was continued for a maximum of 24 months; however, lenvatinib could be continued beyond 24 months. Treatment continued until unacceptable toxicity or disease progression. Administration of intravenous pembrolizumab with lenvatinib was permitted beyond RECIST-defined disease progression if the patient was considered by the investigator to be deriving clinical benefit.
Among the 158 treated patients, the baseline characteristics were: median age of 60 years (range: 24 to 87 years); 71% male; 86% White, 8% Asian, and 3% Black; <1% Hispanic or Latino; 22% and 78% of patients had a baseline KPS of 70 to 80 and 90 to 100, respectively; histologic subtypes were 59% papillary, 18% chromophobe, 4% translocation, <1% medullary, 13% unclassified, and 6% other; patient distribution by IMDC risk categories was 35% favorable, 54% intermediate, and 10% poor. Common sites of metastases in patients were lymph node (65%), lung (35%), bone (30%), and liver (21%).
The major efficacy outcome measure was ORR as assessed by BICR using RECIST 1.1. Additional efficacy outcome measures included DOR as assessed by BICR using RECIST 1.1. Efficacy results are summarized in Table 94.
| Endpoint | Intravenous Pembrolizumab 400 mg every 6 weeks and Lenvatinib n=158 |
|---|---|
| CI = confidence interval + Denotes ongoing response |
|
| Objective Response Rate (Confirmed) | |
| ORR (95% CI) | 51% (43, 59) |
| Complete response | 8% |
| Partial response | 42% |
|
Duration of Response Based on Kaplan-Meier estimates
|
|
| Median in months (range) | 19.5 (1.5+, 23.5+) |
Adjuvant Treatment of RCC (KEYNOTE-564)
The efficacy of intravenous pembrolizumab was investigated as adjuvant therapy for RCC in KEYNOTE-564 (NCT03142334), a multicenter, randomized (1:1), double-blind, placebo-controlled trial in 994 patients with intermediate-high or high risk of recurrence of RCC, or M1 no evidence of disease (NED). The intermediate-high risk category included: pT2 with Grade 4 or sarcomatoid features; pT3, any Grade without nodal involvement (N0) or distant metastases (M0). The high risk category included: pT4, any Grade N0 and M0; any pT, any Grade with nodal involvement and M0. The M1 NED category included patients with metastatic disease who had undergone complete resection of primary and metastatic lesions. Patients must have undergone a partial nephroprotective or radical complete nephrectomy (and complete resection of solid, isolated, soft tissue metastatic lesion(s) in M1 NED participants) with negative surgical margins ≥4 weeks prior to the time of screening. Patients were excluded from the trial if they had received prior systemic therapy for advanced RCC. Patients with active autoimmune disease or a medical condition that required immunosuppression were also ineligible. Patients were randomized to intravenous pembrolizumab 200 mg administered intravenously every 3 weeks or placebo for up to 1 year until disease recurrence or unacceptable toxicity. Randomization was stratified by metastasis status (M0, M1 NED); M0 group was further stratified by ECOG PS (0,1) and geographic region (US, non-US).
The study population characteristics were: median age of 60 years (range: 25 to 84), 33% age 65 or older; 71% male; 75% White, 14% Asian, 9% Unknown, 1% Black or African American, 1% American Indian or Alaska Native, 1% Multiracial; 13% Hispanic or Latino, 78% Not Hispanic or Latino, 8% Unknown; and 85% ECOG PS of 0 and 15% ECOG PS of 1. Ninety-four percent of patients enrolled had N0 disease; 11% had sarcomatoid features; 86% were intermediate-high risk; 8% were high risk; and 6% were M1 NED. Ninety-two percent of patients had a radical nephrectomy, and 8% had a partial nephrectomy.
The major efficacy outcome measure was investigator-assessed disease-free survival (DFS), defined as time to recurrence, metastasis, or death. An additional outcome measure was OS. Statistically significant improvements in DFS and OS were demonstrated at pre-specified interim analyses in patients randomized to the intravenous pembrolizumab arm compared with placebo. Efficacy results are summarized in Table 95 and Figures 36 and 37.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=496 |
Placebo n=498 |
|---|---|---|
| NR = not reached | ||
| DFS | ||
| Number (%) of patients with event | 109 (22%) | 151 (30%) |
| Median in months (95% CI) | NR | NR |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.68 (0.53, 0.87) | |
| p-Value Based on stratified log-rank test
|
0.0010 p-Value (one-sided) is compared with a boundary of 0.0114.
|
|
| 24-month DFS rate (95% CI) | 77% (73, 81) | 68% (64, 72) |
| OS | ||
| Number (%) of patients with event | 55 (11%) | 86 (17%) |
| Median in months (95% CI) | NR (NR, NR) | NR (NR, NR) |
| Hazard ratio (95% CI) | 0.62 (0.44, 0.87) | |
| p-Value | 0.0024 p-Value (one-sided) is compared with a boundary of 0.0072.
|
|
| 48-month OS rate (95% CI) | 91% (88, 93) | 86% (83, 89) |
| Figure 36: Kaplan-Meier Curve for Disease-Free Survival in KEYNOTE-564 |
|
|
| Figure 37: Kaplan-Meier Curve for Overall Survival in KEYNOTE-564 |
|
|
5 Warnings and Precautions
- Immune-Mediated Adverse Reactions (5.1)
- Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and solid organ transplant rejection.
- Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment.
- Withhold or permanently discontinue based on severity and type of reaction.
- Hypersensitivity and Administration-Related Reactions: Interrupt injection and resume upon symptom resolution, or permanently discontinue KEYTRUDA QLEX based on the severity of reaction. (5.2)
- Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. (5.3)
- Treatment of patients with multiple myeloma with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials. (5.4)
- Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective method of contraception. (5.5, 8.1, 8.3)
14.14 Merkel Cell Carcinoma
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-017 (NCT02267603) and KEYNOTE-913 (NCT03783078), two multicenter, non-randomized, open-label trials that enrolled 105 patients with recurrent locally advanced or metastatic MCC who had not received prior systemic therapy for their advanced disease. Patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible.
Patients received intravenous pembrolizumab 2 mg/kg (KEYNOTE-017) or 200 mg (KEYNOTE-913) every 3 weeks until unacceptable toxicity or disease progression that was symptomatic, rapidly progressive, required urgent intervention, occurred with a decline in performance status, or was confirmed at least 4 weeks later with repeat imaging. Patients without disease progression were treated for up to 24 months.
The major efficacy outcome measures were ORR and DoR as assessed by BICR per RECIST v1.1.
Among the 105 patients enrolled, the median age was 73 years (range: 38 to 91), 79% were age 65 or older; 62% were male; 80% were White, race in 19% was unknown or missing, and 1% were Asian; 53% had ECOG PS of 0, and 47% had ECOG PS of 1. Thirteen percent had stage IIIB disease and 84% had stage IV. Seventy-six percent of patients had prior surgery and 51% had prior radiation therapy.
Efficacy results are summarized in Table 91.
| Endpoint | KEYNOTE-017 Intravenous Pembrolizumab 2 mg/kg every 3 weeks n=50 |
KEYNOTE-913 Intravenous Pembrolizumab 200 mg or 2 mg/kg every 3 weeks n=55 |
|---|---|---|
| + Denotes ongoing response | ||
| NR = not reached | ||
| Objective Response Rate | ||
| ORR (95% CI) | 56% (41, 70) | 49% (35, 63) |
| Complete responses, n (%) | 12 (24%) | 9 (16%) |
| Partial responses, n (%) | 16 (32%) | 18 (33%) |
| Duration of Response | n=28 | n=27 |
| Median DoR in months (range) | NR (5.9, 34.5+) | NR (4.8, 25.4+) |
| Patients with duration ≥6 months, n (%) | 27 (96%) | 25 (93%) |
| Patients with duration ≥12 months, n (%) | 15 (54%) | 19 (70%) |
14.16 Endometrial Carcinoma
In Combination with Paclitaxel and Carboplatin for the Treatment of Primary Advanced or Recurrent Endometrial Carcinoma
The efficacy of intravenous pembrolizumab in combination with paclitaxel and carboplatin was investigated in KEYNOTE-868/NRG-GY018 (NCT03914612), a multicenter, randomized, double-blind, placebo-controlled trial in 810 patients with advanced or recurrent endometrial carcinoma. The study design included two separate cohorts based on MMR status; 222 (27%) patients were in dMMR cohort, 588 (73%) patients were in pMMR cohort. The trial enrolled measurable Stage III, measurable Stage IVA, Stage IVB or recurrent endometrial cancer (with or without measurable disease). Patients who had not received prior systemic therapy or had received prior chemotherapy in the adjuvant setting were eligible. Patients who had received prior adjuvant chemotherapy were only eligible if their chemotherapy-free interval was at least 12 months. Patients with endometrial sarcoma, including carcinosarcoma, or patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible. Randomization was stratified according to MMR status, ECOG PS (0 or 1 vs. 2), and prior adjuvant chemotherapy.
Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg every 3 weeks, paclitaxel 175 mg/m2 and carboplatin AUC 5 mg/mL/min for 6 cycles, followed by intravenous pembrolizumab 400 mg every 6 weeks for up to 14 cycles.
- Placebo every 3 weeks, paclitaxel 175 mg/m2 and carboplatin AUC 5 mg/mL/min for 6 cycles, followed by placebo every 6 weeks for up to 14 cycles.
All study medications were administered as an intravenous infusion on Day 1 of each treatment cycle. Treatment continued until disease progression, unacceptable toxicity, or a maximum of 20 cycles (up to approximately 24 months). Patients with measurable disease who had RECIST-defined stable disease or partial response at the completion of cycle 6 were permitted to continue receiving paclitaxel and carboplatin with intravenous pembrolizumab or placebo for up to 10 cycles as determined by the investigator. Assessment of tumor status was performed every 9 weeks for the first 9 months and then every 12 weeks thereafter. The major efficacy outcome measure was PFS as assessed by the investigator according to RECIST 1.1. An additional efficacy outcome measure was OS.
The dMMR population characteristics were: median age of 66 years (range: 37 to 86), 55% age 65 or older; 79% White, 9% Black, and 3% Asian; 5% Hispanic or Latino; 64% ECOG PS of 0, 33% ECOG PS of 1, and 3% ECOG PS of 2; 61% had recurrent disease and 39% had primary or persistent disease; 5% received prior adjuvant chemotherapy and 43% received prior radiotherapy. The histologic subtypes were endometrioid carcinoma (81%), adenocarcinoma NOS (11%), serous carcinoma (2%), and other (6%).
The pMMR population characteristics were: median age of 66 years (range: 29 to 94), 54% age 65 or older; 72% White, 16% Black, and 5% Asian; 6% Hispanic or Latino; 67% ECOG PS of 0, 30% ECOG PS of 1, and 3% ECOG PS of 2; 56% had recurrent disease and 44% had primary or persistent disease; 26% received prior adjuvant chemotherapy and 41% received prior radiotherapy. The histologic subtypes were endometrioid carcinoma (52%), serous carcinoma (26%), adenocarcinoma NOS (10%), clear cell carcinoma (7%), and other (5%).
The trial demonstrated statistically significant improvements in PFS for patients randomized to intravenous pembrolizumab in combination with paclitaxel and carboplatin compared to placebo in combination with paclitaxel and carboplatin in both the dMMR and pMMR populations. Table 96 and Figures 38 and 39 summarize the efficacy results for KEYNOTE-868 by MMR status. At the time of the PFS analysis, OS data were not mature with 12% deaths in the dMMR population and 17% deaths in the pMMR population.
| Endpoint | dMMR Population | pMMR Population | ||
|---|---|---|---|---|
| Intravenous Pembrolizumab with paclitaxel and carboplatin n=110 |
Placebo with paclitaxel and carboplatin n=112 |
Intravenous Pembrolizumab with paclitaxel and carboplatin n=294 |
Placebo with paclitaxel and carboplatin n=294 |
|
| NR = not reached | ||||
|
PFS Based on interim PFS analysis; the information fractions for interim analyses were 49% for dMMR and 55% for pMMR.
|
||||
| Number (%) of patients with event | 26 (24%) | 57 (51%) | 91 (31%) | 124 (42%) |
| Median in months (95% CI) | NR (30.7, NR) | 6.5 (6.4, 8.7) | 11.1 (8.7, 13.5) | 8.5 (7.2, 8.8) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.30 (0.19, 0.48) | 0.60 (0.46, 0.78) | ||
| p-Value Based on the stratified log-rank test
|
<0.0001 | <0.0001 |
| Figure 38: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-868 (dMMR Population) |
|
|
| Figure 39: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-868 (pMMR Population) |
|
|
Lack of Effectiveness for Adjuvant Treatment of Patients with Endometrial Carcinoma
The efficacy of intravenous pembrolizumab in combination with carboplatin and paclitaxel, with or without radiation, was investigated in KEYNOTE‑B21 (NCT04634877), a randomized, multicenter, double-blind, placebo-controlled trial in 1,095 patients with newly-diagnosed, high‑risk endometrial cancer with no evidence of disease on imaging following curative intent surgery. High-risk disease was defined as any of the following (staging per FIGO 2009): Stage I/II with myometrial invasion and either non-endometrioid histology or aberrant p53 expression or p53 mutation, or Stage III/IVA. The trial did not meet the prespecified primary endpoint for investigator-assessed DFS, with a HR of 1.02 (95% CI: 0.79, 1.32).
In Combination with Lenvatinib for the Treatment of Advanced Endometrial Carcinoma That Is pMMR or Not MSI-H
The efficacy of intravenous pembrolizumab in combination with lenvatinib was investigated in KEYNOTE-775 (NCT03517449), a multicenter, open-label, randomized, active-controlled trial that enrolled 827 patients with advanced endometrial carcinoma who had been previously treated with at least one prior platinum-based chemotherapy regimen in any setting, including in the neoadjuvant and adjuvant settings. Patients with endometrial sarcoma, including carcinosarcoma, or patients who had active autoimmune disease or a medical condition that required immunosuppression were ineligible. Patients with endometrial carcinoma that were pMMR (using the VENTANA MMR RxDx Panel test) or not MSI-H were stratified by ECOG performance status, geographic region, and history of pelvic radiation. Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg intravenously every 3 weeks in combination with lenvatinib 20 mg orally once daily.
- Investigator’s choice, consisting of either doxorubicin 60 mg/m2 every 3 weeks or paclitaxel 80 mg/m2 given weekly, 3 weeks on/1 week off.
Treatment with intravenous pembrolizumab and lenvatinib continued until RECIST v1.1-defined progression of disease as verified by BICR, unacceptable toxicity, or for intravenous pembrolizumab, a maximum of 24 months. Treatment was permitted beyond RECIST v1.1-defined disease progression if the treating investigator considered the patient to be deriving clinical benefit, and the treatment was tolerated. Assessment of tumor status was performed every 8 weeks. The major efficacy outcome measures were OS and PFS as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ. Additional efficacy outcome measures included ORR and DoR, as assessed by BICR.
Among the 697 pMMR patients, 346 patients were randomized to intravenous pembrolizumab in combination with lenvatinib, and 351 patients were randomized to investigator’s choice of doxorubicin (n=254) or paclitaxel (n=97). The pMMR population characteristics were: median age of 65 years (range: 30 to 86), 52% age 65 or older; 62% White, 22% Asian, and 3% Black; 60% ECOG PS of 0 and 40% ECOG PS of 1. The histologic subtypes were endometrioid carcinoma (55%), serous (30%), clear cell carcinoma (7%), mixed (4%), and other (3%). All 697 of these patients received prior systemic therapy for endometrial carcinoma: 67% had one, 30% had two, and 3% had three or more prior systemic therapies. Thirty-seven percent of patients received only prior neoadjuvant or adjuvant therapy.
Efficacy results for the pMMR or not MSI-H patients are summarized in Table 97 and Figures 40 and 41.
| Endometrial Carcinoma (pMMR or not MSI-H) | ||
|---|---|---|
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and Lenvatinib n=346 |
Doxorubicin or Paclitaxel n=351 |
| OS | ||
| Number (%) of patients with event | 165 (48%) | 203 (58%) |
| Median in months (95% CI) | 17.4 (14.2, 19.9) | 12.0 (10.8, 13.3) |
| Hazard ratio Based on the stratified Cox regression model (95% CI) |
0.68 (0.56, 0.84) | |
| p-Value Based on stratified log-rank test
|
0.0001 | |
| PFS | ||
| Number (%) of patients with event | 247 (71%) | 238 (68%) |
| Median in months (95% CI) | 6.6 (5.6, 7.4) | 3.8 (3.6, 5.0) |
| Hazard ratio (95% CI) | 0.60 (0.50, 0.72) | |
| p-Value | <0.0001 | |
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
30% (26, 36) | 15% (12, 19) |
| Complete response rate | 5% | 3% |
| Partial response rate | 25% | 13% |
| p-Value Based on Miettinen and Nurminen method stratified by ECOG performance status, geographic region, and history of pelvic radiation
|
<0.0001 | |
| Duration of Response | n=105 | n=53 |
| Median in months (range) | 9.2 (1.6+, 23.7+) | 5.7 (0.0+, 24.2+) |
| Figure 40: Kaplan-Meier Curve for Overall Survival in KEYNOTE-775 (pMMR or Not MSI-H) |
|
|
| Figure 41: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-775 (pMMR or Not MSI-H) |
|
|
As a Single Agent for the Treatment of Advanced MSI-H or dMMR Endometrial Carcinoma
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-158 (NCT02628067), a multicenter, non-randomized, open-label, multi-cohort trial. The trial enrolled 90 patients with unresectable or metastatic MSI-H or dMMR endometrial carcinoma in Cohorts D and K. MSI or MMR tumor status was determined using polymerase chain reaction (PCR) or immunohistochemistry (IHC), respectively. Patients with autoimmune disease or a medical condition that required immunosuppression were ineligible. Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression. Patients treated with intravenous pembrolizumab without disease progression could be treated for up to 24 months. Assessment of tumor status was performed every 9 weeks for the first 12 months, and every 12 weeks thereafter. The major efficacy outcome measures were ORR and DoR as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among the 90 patients evaluated, the baseline characteristics were: median age of 64 years (range: 42 to 86); 83% White, 8% Asian, and 3% Black; 12% Hispanic or Latino; 39% ECOG PS of 0 and 61% ECOG PS of 1; 96% had M1 disease and 4% had M0 disease at study entry; and 51% had one and 48% had two or more prior lines of therapy. Nine patients received only adjuvant therapy and one patient received only neoadjuvant and adjuvant therapy before participating in the study.
Efficacy results are summarized in Table 98.
| Endpoint | Intravenous Pembrolizumab n=90 Median follow-up time of 16.0 months (range 0.5 to 62.1 months)
|
|---|---|
| + Denotes ongoing response NR = not reached |
|
| Objective Response Rate | |
| ORR (95% CI) | 46% (35, 56) |
| Complete response rate | 12% |
| Partial response rate | 33% |
| Duration of Response | n=41 |
| Median in months (range) | NR (2.9, 55.7+) |
| % with duration ≥12 months | 68% |
| % with duration ≥24 months | 44% |
2 Dosage and Administration
KEYTRUDA QLEX has different recommended dosage and administration than intravenous pembrolizumab. (2.2)
- KEYTRUDA QLEX is for subcutaneous use in the thigh or abdomen only. (2.2)
- Do not administer KEYTRUDA QLEX intravenously. (2.2)
- KEYTRUDA QLEX must be administered by a healthcare provider. (2.2)
The recommended dose for adults and pediatric patients 12 years and older who weigh greater than 40 kg is:
- Every 3-week dosing (395 mg/4,800 units): Inject 2.4 mL subcutaneously in the abdomen or thigh over 1 minute. (2.3)
- Every 6-week dosing (790 mg/9,600 units): Inject 4.8 mL subcutaneously in the abdomen or thigh over 2 minutes. (2.3)
- For RCC, administer KEYTRUDA QLEX as a single agent in the adjuvant setting, or in the advanced setting with either:
- axitinib 5 mg orally twice daily or
- lenvatinib 20 mg orally once daily. (2.3)
- For Endometrial Carcinoma, administer KEYTRUDA QLEX:
- in combination with carboplatin and paclitaxel regardless of MMR or MSI status, or
- in combination with lenvatinib 20 mg orally once daily for pMMR or not MSI-H tumors, or
- as a single agent for MSI-H or dMMR tumors. (2.3)
3 Dosage Forms and Strengths
KEYTRUDA QLEX is a clear to slightly opalescent, colorless to slightly yellow solution provided as:
- Injection: 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/2,000 units per mL) in a single-dose vial
- Injection: 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/2,000 units per mL) in a single-dose vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of intravenous pembrolizumab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal: Exocrine pancreatic insufficiency
Hepatobiliary: sclerosing cholangitis
1.11 Hepatocellular Carcinoma
KEYTRUDA QLEX is indicated for the treatment of adult patients with hepatocellular carcinoma (HCC) secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen.
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
1.2 Non Small Cell Lung Cancer
KEYTRUDA QLEX, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of adult patients with metastatic nonsquamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations.
KEYTRUDA QLEX, in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, is indicated for the first-line treatment of adult patients with metastatic squamous NSCLC.
KEYTRUDA QLEX, as a single agent, is indicated for the first-line treatment of adult patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-authorized test [see Dosage and Administration (2.1)], with no EGFR or ALK genomic tumor aberrations, and is:
- Stage III where patients are not candidates for surgical resection or definitive chemoradiation, or
- metastatic.
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-authorized test [see Dosage and Administration (2.1)], with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA QLEX.
KEYTRUDA QLEX is indicated for the treatment of adult patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA QLEX, as a single agent, is indicated as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC.
14.12 Hepatocellular Carcinoma
Previously Treated HCC
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-394 (NCT03062358), a multicenter, randomized, placebo-controlled, double-blind trial conducted in Asia in patients with Barcelona Clinic Liver Cancer (BCLC) Stage B or C HCC, who were previously treated with sorafenib or oxaliplatin-based chemotherapy and who were not amenable to or were refractory to local-regional therapy. Patients were also required to have Child-Pugh A liver function.
Patients with hepatitis B had treated controlled disease (HBV viral load <2000 IU/mL or <104 copies/mL). Patients with an autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Patients with hepatic encephalopathy, main branch portal venous invasion, clinically apparent ascites, or esophageal or gastric variceal bleeding within the last 6 months were also ineligible.
Randomization was stratified by prior treatment: sorafenib vs. oxaliplatin-based chemotherapy, macrovascular invasion, and etiology (active HBV vs. others (active HCV, non-infected)). Patients were randomized (2:1) to receive pembrolizumab 200 mg intravenously every 3 weeks or placebo.
Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by BICR, unacceptable toxicity, or a maximum of 24 months. Assessment of tumor status was performed every 6 weeks. The main efficacy outcome measure was OS. Additional efficacy outcome measures were PFS, ORR, and DoR, as assessed by BICR using RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
The study enrolled 453 patients, and 360 (79%) had active hepatitis B. The population characteristics in patients with active hepatitis B were: median age of 52 years (range: 23 to 82), 16% age 65 or older; 86% male; 100% Asian; 42% ECOG PS of 0 and 58% ECOG PS of 1; 90% received prior sorafenib and 10% received prior oxaliplatin-based chemotherapy. Patient characteristics also included extrahepatic disease (77%), macrovascular invasion (10%), BCLC stage C (93%) and B (7%), and baseline AFP ≥200 ng/mL (57%).
KEYNOTE-394 demonstrated improved OS in patients with HCC secondary to hepatitis B randomized to intravenous pembrolizumab compared with placebo. Efficacy results are summarized in Table 89 and Figure 31.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=236 |
Placebo n=124 |
|---|---|---|
| + Denotes ongoing response | ||
|
OS
Results at the pre-specified final OS analysis
|
||
| Number (%) of patients with events | 172 (73) | 105 (85) |
| Median in months (95% CI) | 13.9 (12.5, 17.9) | 13.0 (10.1, 15.6) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.78 (0.61, 0.99) | |
|
PFS
Results at pre-specified interim OS analysis
|
||
| Number (%) of patients with events | 189 (80) | 108 (87) |
| Median in months (95% CI) | 2 (1.4, 2.7) | 2.3 (1.4, 2.8) |
| Hazard ratio (95% CI) | 0.78 (0.61, 1.00) | |
| Objective Response Rate | ||
| ORR Confirmed complete response or partial response (95% CI) |
11% (7, 16) | 1.6% (0.2, 5.7) |
| Number (%) of complete responses | 2 (0.9%) | 1 (0.8%) |
| Number (%) of partial responses | 24 (10%) | 1 (0.8%) |
| Duration of Response | n=28 | n=2 |
| Median in months Based on Kaplan-Meier estimate (range)
|
23.9 (2.6+, 44.4+) | 5.6 (3.0+, 5.6) |
| Figure 31: Kaplan-Meier Curve for Overall Survival in KEYNOTE-394 |
|
|
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described in the WARNINGS AND PRECAUTIONS reflect exposure to intravenous pembrolizumab as a single agent in 2799 patients in three randomized, open-label, active-controlled trials (KEYNOTE-002, KEYNOTE-006, and KEYNOTE-010), which enrolled 912 patients with melanoma and 682 patients with NSCLC, and one single-arm trial (KEYNOTE-001), which enrolled 655 patients with melanoma and 550 patients with NSCLC. In addition to the 2799 patients, certain subsections in the WARNINGS AND PRECAUTIONS describe adverse reactions observed with exposure to KEYTRUDA QLEX in combination with platinum doublet chemotherapy in a randomized, open-label, active-controlled trial (Study MK-3475A-D77), which enrolled 251 patients with NSCLC; intravenous pembrolizumab as a single agent in a randomized, placebo-controlled trial (KEYNOTE-091), which enrolled 580 patients with resected NSCLC; a non-randomized, open-label, multi-cohort trial (KEYNOTE-012), a non-randomized, open-label, single-cohort trial (KEYNOTE-055), and two randomized, open-label, active-controlled trials (KEYNOTE-040 and KEYNOTE-048 single agent arms), which enrolled 909 patients with HNSCC; in a randomized, open-label, active-controlled trial (KEYNOTE-048 combination arm), which enrolled 276 patients with HNSCC; in combination with axitinib in a randomized, active-controlled trial (KEYNOTE-426), which enrolled 429 patients with RCC; and in post-marketing use. Across all trials, patients were administered either KEYTRUDA QLEX 790 mg/9,600 units every 6 weeks or intravenous pembrolizumab at doses of 2 mg/kg every 3 weeks, 10 mg/kg every 2 weeks, 10 mg/kg every 3 weeks, or 200 mg every 3 weeks. Among the 2799 patients who received intravenous pembrolizumab, 41% were exposed for 6 months or more and 21% were exposed for 12 months or more.
The most common adverse reactions (≥20%) in patients who received KEYTRUDA QLEX in combination with chemotherapy were nausea (25%), fatigue (25%), and musculoskeletal pain (21%).
The safety of KEYTRUDA QLEX for the approved indications is also based on the safety of intravenous pembrolizumab given as a single agent or in combination with other antitumor medicines.
The most common adverse reactions (≥20%) in patients who received intravenous pembrolizumab were:
- as a single agent: fatigue, musculoskeletal pain, rash, diarrhea, pyrexia, cough, decreased appetite, pruritus, dyspnea, constipation, pain, abdominal pain, nausea, and hypothyroidism.
- in combination with chemotherapy or chemoradiotherapy: fatigue/asthenia, nausea, constipation, diarrhea, decreased appetite, rash, vomiting, cough, dyspnea, pyrexia, alopecia, peripheral neuropathy, mucosal inflammation, stomatitis, headache, weight loss, abdominal pain, arthralgia, myalgia, insomnia, palmar-plantar erythrodysesthesia, urinary tract infection, hypothyroidism, radiation skin injury, dysphagia, dry mouth, and musculoskeletal pain.
- in combination with chemotherapy and bevacizumab: peripheral neuropathy, alopecia, anemia, fatigue/asthenia, nausea, neutropenia, diarrhea, hypertension, thrombocytopenia, constipation, arthralgia, vomiting, urinary tract infection, rash, leukopenia, hypothyroidism, decreased appetite, pyrexia, epistaxis, decreased white blood cell count, and stomatitis.
- in combination with axitinib: diarrhea, fatigue/asthenia, hypertension, hepatotoxicity, hypothyroidism, decreased appetite, palmar-plantar erythrodysesthesia, nausea, stomatitis/mucosal inflammation, dysphonia, rash, cough, and constipation.
- in combination with lenvatinib: hypothyroidism, hypertension, fatigue, diarrhea, musculoskeletal disorders, nausea, decreased appetite, vomiting, stomatitis, weight loss, abdominal pain, urinary tract infection, proteinuria, constipation, headache, hemorrhagic events, palmar-plantar erythrodysesthesia, dysphonia, rash, hepatotoxicity, and acute kidney injury.
- in combination with enfortumab vedotin: rash, peripheral neuropathy, fatigue, pruritus, diarrhea, alopecia, weight loss, decreased appetite, dry eye, nausea, constipation, dysgeusia, and urinary tract infection.
Adverse Reactions in Patients with NSCLC Treated with KEYTRUDA QLEX
The safety of KEYTRUDA QLEX compared to intravenous pembrolizumab in patients with previously untreated, metastatic NSCLC with no EGFR, ALK or ROS1 genomic tumor aberrations was evaluated in Study MK-3475A-D77 [see Clinical Studies (14.1)]. A total of 377 patients received either KEYTRUDA QLEX 790 mg/9,600 units every 6 weeks in combination with platinum doublet chemotherapy (n=251) or intravenous pembrolizumab 400 mg every 6 weeks in combination with platinum doublet chemotherapy (n=126).
Among patients who received KEYTRUDA QLEX, 58% were exposed for 6 months or longer and 3.2% were exposed for greater than one year.
The median age of patients who received KEYTRUDA QLEX was 65 years (range: 39 to 87); 73% male, 63% White; 29% Asian, 4.8% multiple races, 2% Black or African American, 0.8% Alaska Native or American Indian; and 29% were of Hispanic or Latino ethnicity.
Serious adverse reactions occurred in 39% of patients who received KEYTRUDA QLEX in combination with chemotherapy. Serious adverse reactions in ≥1% of patients who received KEYTRUDA QLEX were pneumonia (10%), thrombocytopenia (4%), febrile neutropenia (4%), neutropenia (2.8%), musculoskeletal pain (2%), pneumonitis (2%), diarrhea (1.6%), rash (1.2%), respiratory failure (1.2%), and anemia (1.2%). Fatal adverse reactions occurred in 10% of patients who received KEYTRUDA QLEX in combination with chemotherapy including pneumonia (3.2%), neutropenic sepsis (2%), death not otherwise specified (1.6%), respiratory failure (1.2%), parotitis (0.4%), pneumonitis (0.4%), pneumothorax (0.4%), pulmonary embolism (0.4%), neutropenic colitis (0.4%), and seizure (0.4%).
Permanent discontinuation of KEYTRUDA QLEX due to an adverse reaction occurred in 16% of patients. Adverse reactions which resulted in permanent discontinuation of KEYTRUDA QLEX in ≥2% of patients included pneumonia and pneumonitis.
Dosage interruptions of KEYTRUDA QLEX due to an adverse reaction occurred in 45% of patients. Adverse reactions which required dosage interruption in ≥2% of patients included neutropenia, anemia, thrombocytopenia, pneumonia, rash, and increased aspartate aminotransferase.
Tables 4 and 5 summarize the adverse reactions and laboratory abnormalities, respectively, in patients on KEYTRUDA QLEX in Study MK-3475A-D77.
| Adverse Reaction | KEYTRUDA QLEX and Platinum Doublet Chemotherapy |
Intravenous Pembrolizumab and Platinum Doublet Chemotherapy |
||
|---|---|---|---|---|
| (n=251) | (n=126) | |||
| All Grades Graded per NCI CTCAE V5.0
(%) |
Grades 3-4 (%) |
All Grades
(%) |
Grades 3-4 (%) |
|
| Gastrointestinal | ||||
| Nausea | 25 | 1.2 | 25 | 0.8 |
| Diarrhea Includes diarrhea, colitis, and enterocolitis.
|
16 | 2 | 14 | 0.8 |
| Constipation | 14 | 0 | 18 | 1.6 |
| General | ||||
| Fatigue Includes fatigue, asthenia.
|
25 | 3.6 | 26 | 3.2 |
| Musculoskeletal and Connective Tissue | ||||
| Musculoskeletal pain Includes musculoskeletal pain, arthralgia, arthritis, back pain, bone pain, musculoskeletal chest pain, musculoskeletal stiffness, myalgia, non-cardiac chest pain, and pain in extremity.
|
21 | 2.4 | 30 | 2.4 |
| Skin and Subcutaneous Tissue | ||||
| Rash Includes rash, dermatitis, dermatitis acneiform, dermatitis bullous, dermatitis exfoliative, eczema, erythema multiforme, immune-mediated dermatitis, rash erythematous, rash follicular, rash generalized, rash macular, rash maculo-papular, rash papular, rash pruritic, and skin exfoliation.
|
18 | 2 | 19 | 0.8 |
| Pruritus | 12 | 0 | 13 | 0.8 |
| Endocrine | ||||
| Hypothyroidism | 14 | 0 | 12 | 0 |
| Infections | ||||
| Pneumonia Includes pneumonia, COVID-19 pneumonia, lower respiratory tract infection, lung abscess, pneumocystis jirovecii pneumonia, pneumonia bacterial, and pneumonia mycoplasmal.
|
17 | 10 | 16 | 7 |
| Nervous System | ||||
| Peripheral neuropathy Includes neuropathy peripheral, hypoaesthesia, neuralgia, paraesthesia, and peripheral sensory neuropathy.
|
11 | 0.4 | 14 | 0 |
| Metabolism and Nutrition | ||||
| Decreased appetite | 11 | 0.8 | 21 | 2.4 |
| Hyperglycemia | 11 | 0.8 | 11 | 0.8 |
| Respiratory, Thoracic and Mediastinal | ||||
| Cough Includes cough, productive cough, and upper-airway cough syndrome.
|
10 | 0 | 11 | 0.8 |
Clinically relevant adverse reactions in <10% of patients who received KEYTRUDA QLEX included local injection site reactions (2.4%).
| Laboratory Test Each test incidence is based on the number of patients who had both baseline and at least one on-study laboratory measurement available: KEYTRUDA QLEX plus platinum doublet chemotherapy (range: 240 to 246 patients) and intravenous pembrolizumab plus platinum doublet chemotherapy (range: 124 to 125 patients).
|
KEYTRUDA QLEX and Platinum Doublet Chemotherapy |
Intravenous Pembrolizumab and Platinum Doublet Chemotherapy |
||
|---|---|---|---|---|
| All Grades Graded per NCI CTCAE V5.0
(%) |
Grades 3-4 (%) |
All Grades
(%) |
Grades 3-4 (%) |
|
| Hematology | ||||
| Anemia | 80 | 22 | 86 | 26 |
| Leukopenia | 61 | 13 | 52 | 10 |
| Neutropenia | 58 | 28 | 49 | 19 |
| Lymphopenia | 55 | 22 | 54 | 18 |
| Thrombocytopenia | 43 | 11 | 41 | 6 |
| Chemistry | ||||
| Increased AST | 43 | 2.5 | 38 | 3.2 |
| Hypoalbuminemia | 38 | 0.4 | 39 | 0 |
| Increased ALT | 37 | 2.1 | 36 | 0.8 |
| Hyponatremia | 35 | 4.1 | 42 | 7 |
| Increased creatinine | 33 | 4.5 | 38 | 6 |
| Hypocalcemia | 31 | 2.1 | 31 | 2.4 |
| Increased alkaline phosphatase | 29 | 0.4 | 34 | 0 |
| Hypokalemia | 21 | 5 | 24 | 6 |
Adverse Reactions in Adult and Pediatric Patients Treated with Intravenous Pembrolizumab
The safety of KEYTRUDA QLEX for its approved indications [see Indications and Usage (1)] has been established in adequate and well-controlled studies of KEYTRUDA QLEX in combination with platinum doublet chemotherapy (Study MK-3475A-D77) and intravenous pembrolizumab, as a single agent or in combination therapy, across tumor types.
Below is a description of adverse reactions of intravenous pembrolizumab in these adequate and well-controlled studies.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
1.18 Triple Negative Breast Cancer
KEYTRUDA QLEX is indicated for the treatment of adult patients with high-risk early-stage triple-negative breast cancer (TNBC) in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA QLEX, in combination with chemotherapy, is indicated for the treatment of adult patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
1.3 Malignant Pleural Mesothelioma
KEYTRUDA QLEX, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of adult patients with unresectable advanced or metastatic malignant pleural mesothelioma (MPM).
14.19 Triple Negative Breast Cancer
Neoadjuvant and Adjuvant Treatment of High-Risk Early-Stage TNBC
The efficacy of intravenous pembrolizumab in combination with neoadjuvant chemotherapy followed by surgery and continued adjuvant treatment with intravenous pembrolizumab as a single agent was investigated in KEYNOTE-522 (NCT03036488), a randomized (2:1), multicenter, double-blind, placebo-controlled trial conducted in 1174 patients with newly diagnosed previously untreated high-risk early-stage TNBC (tumor size >1 cm but ≤2 cm in diameter with nodal involvement or tumor size >2 cm in diameter regardless of nodal involvement). Patients were enrolled regardless of tumor PD-L1 expression. Patients with active autoimmune disease that required systemic therapy within two years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by nodal status (positive vs. negative), tumor size (T1/T2 vs. T3/T4), and choice of carboplatin (dosed every 3 weeks vs. weekly).
Patients were randomized (2:1) to one of the following two treatment arms; all study medications were administered intravenously:
-
Arm 1:
- Four cycles of preoperative intravenous pembrolizumab 200 mg every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
- Carboplatin
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
-or- - AUC 1.5 mg/mL/min every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
-and-
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
- Paclitaxel 80 mg/m2 every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
- Carboplatin
- Followed by four additional cycles of preoperative intravenous pembrolizumab 200 mg every 3 weeks on Day 1 of cycles 5-8 of treatment regimen in combination with:
- Doxorubicin 60 mg/m2 -or- epirubicin 90 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen -and-
- Cyclophosphamide 600 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen
- Following surgery, nine cycles of intravenous pembrolizumab 200 mg every 3 weeks were administered.
- Four cycles of preoperative intravenous pembrolizumab 200 mg every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
-
Arm 2:
- Four cycles of preoperative placebo every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
- Carboplatin
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
-or- - AUC 1.5 mg/mL/min every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
- and-
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
- Paclitaxel 80 mg/m2 every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
- Carboplatin
- Followed by four cycles of preoperative placebo every 3 weeks on Day 1 of cycles 5-8 of treatment regimen in combination with:
- Doxorubicin 60 mg/m2 -or- epirubicin 90 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen -and-
- Cyclophosphamide 600 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen
- Following surgery, nine cycles of placebo every 3 weeks were administered.
- Four cycles of preoperative placebo every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
The trial was not designed to isolate the effect of intravenous pembrolizumab in each phase (neoadjuvant or adjuvant) of treatment.
The main efficacy outcomes were pCR rate and EFS. pCR was defined as absence of invasive cancer in the breast and lymph nodes (ypT0/Tis ypN0) and was assessed by the blinded local pathologist at the time of definitive surgery. EFS was defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause. An additional efficacy outcome was overall survival (OS).
The study population characteristics were: median age of 49 years (range: 22 to 80), 11% age 65 or older; 99.9% female; 64% White, 20% Asian, 4.5% Black, and 1.8% American Indian or Alaska Native; 87% ECOG PS of 0 and 13% ECOG PS of 1; 56% were pre-menopausal status and 44% were post-menopausal status; 7% were primary Tumor 1 (T1), 68% T2, 19% T3, and 7% T4; 49% were nodal involvement 0 (N0), 40% N1, 11% N2, and 0.2% N3; 75% of patients were overall Stage II and 25% were Stage III.
Statistically significant improvements in pCR, EFS, and OS were demonstrated at pre-specified interim analyses for patients randomized to intravenous pembrolizumab in combination with chemotherapy followed by intravenous pembrolizumab as a single agent compared with patients randomized to placebo in combination with chemotherapy followed by placebo alone.
Table 102 and Figures 42 and 43 summarize the efficacy results for KEYNOTE-522.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks with chemotherapy/Intravenous Pembrolizumab n=784 |
Placebo with chemotherapy/Placebo n=390 |
|---|---|---|
|
pCR (ypT0/Tis ypN0)
Based on the entire intention-to-treat population n=1174 patients
|
||
| Number of patients with pCR | 494 | 217 |
| pCR rate (%), (95% CI) | 63.0 (59.5, 66.4) | 55.6 (50.6, 60.6) |
| Treatment difference (%) estimate (95% CI) Based on a pre-specified pCR interim analysis in n=602 patients, the pCR rate difference was statistically significant (p=0.00055 compared to a significance level of 0.003).
,
Based on Miettinen and Nurminen method stratified by nodal status, tumor size, and choice of carboplatin
|
7.5 (1.6, 13.4) | |
| EFS | ||
| Number of patients with event (%) | 123 (16%) | 93 (24%) |
| Hazard ratio (95% CI) Based on stratified Cox regression model
|
0.63 (0.48, 0.82) | |
| p-Value Based on a pre-specified EFS interim analysis (compared to a significance level of 0.0052)
,
Based on log-rank test stratified by nodal status, tumor size, and choice of carboplatin
|
0.00031 | |
| OS | ||
| Number of patients with event (%) | 115 (15%) | 85 (22%) |
| Hazard ratio (95% CI) | 0.66 (0.50, 0.87) | |
| p-Value
,
Based on a pre-specified OS interim analysis (compared to a significance level of 0.0050)
|
0.00150 |
| Figure 42: Kaplan-Meier Curve for Event-Free Survival in KEYNOTE-522 |
|
|
| Figure 43: Kaplan-Meier Curve for Overall Survival in KEYNOTE-522 |
|
|
Locally Recurrent Unresectable or Metastatic TNBC for Tumors Expressing PD-L1 (CPS≥ 10)
The efficacy of intravenous pembrolizumab in combination with paclitaxel, paclitaxel protein-bound, or gemcitabine and carboplatin was investigated in KEYNOTE-355 (NCT02819518), a multicenter, double-blind, randomized, placebo-controlled trial conducted in 847 patients with locally recurrent unresectable or metastatic TNBC, regardless of tumor PD-L1 expression, who had not been previously treated with chemotherapy in the metastatic setting. Patients with active autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by chemotherapy treatment (paclitaxel or paclitaxel protein-bound vs. gemcitabine and carboplatin), tumor PD-L1 expression (CPS ≥1 vs. CPS <1) according to the PD-L1 IHC 22C3 pharmDx assay, and prior treatment with the same class of chemotherapy in the neoadjuvant setting (yes vs. no).
Patients were randomized (2:1) to one of the following treatment arms; all study medications were administered via intravenous infusion:
- Intravenous pembrolizumab 200 mg on Day 1 every 3 weeks in combination with paclitaxel protein-bound 100 mg/m2 on Days 1, 8 and 15 every 28 days, paclitaxel 90 mg/m2 on Days 1, 8, and 15 every 28 days, or gemcitabine 1000 mg/m2 and carboplatin AUC 2 mg/mL/min on Days 1 and 8 every 21 days.
- Placebo on Day 1 every 3 weeks in combination with paclitaxel protein-bound 100 mg/m2 on Days 1, 8 and 15 every 28 days, paclitaxel 90 mg/m2 on Days 1, 8, and 15 every 28 days, or gemcitabine 1000 mg/m2 and carboplatin AUC 2 mg/mL/min on Days 1 and 8 every 21 days.
Assessment of tumor status was performed at Weeks 8, 16, and 24, then every 9 weeks for the first year, and every 12 weeks thereafter. The main efficacy outcome measures were OS and PFS as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, tested in the subgroup of patients with CPS ≥10. Additional efficacy outcome measures were ORR and DoR as assessed by BICR.
The study population characteristics for patients were: median age of 53 years (range: 22 to 85), 21% age 65 or older; 100% female; 68% White, 21% Asian, and 4% Black; 60% ECOG PS of 0 and 40% ECOG PS of 1; and 68% were post-menopausal status. Seventy-five percent of patients had tumor PD-L1 expression CPS ≥1 and 38% had tumor PD-L1 expression CPS ≥10.
Table 103 and Figures 44 and 45 summarize the efficacy results for KEYNOTE-355.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks with chemotherapy n=220 |
Placebo every 3 weeks with chemotherapy n=103 |
|---|---|---|
|
OS Based on the pre-specified final analysis
|
||
| Number of patients with event (%) | 155 (70%) | 84 (82%) |
| Median in months (95% CI) | 23 (19.0, 26.3) | 16.1 (12.6, 18.8) |
| Hazard ratio Based on stratified Cox regression model (95% CI) |
0.73 (0.55, 0.95) | |
| p-Value One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.0113)
|
0.0093 | |
|
PFS Based on a pre-specified interim analysis
|
||
| Number of patients with event (%) |
136 (62%) | 79 (77%) |
| Median in months (95% CI) | 9.7 (7.6, 11.3) | 5.6 (5.3, 7.5) |
| Hazard ratio (95% CI) | 0.65 (0.49, 0.86) | |
| p-Value One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.00411)
|
0.0012 | |
| Objective Response Rate (Confirmed) | ||
| ORR (95% CI) | 53% (46, 59) | 41% (31, 51) |
| Complete response rate | 17% | 14% |
| Partial response rate | 35% | 27% |
| Duration of Response | n=116 | n=42 |
| Median in months (95% CI) | 12.8 (9.9, 25.9) | 7.3 (5.5, 15.4) |
| Figure 44: Kaplan-Meier Curve for Overall Survival in KEYNOTE-355 (CPS ≥10) |
|
|
| Figure 45: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-355 (CPS ≥10) |
|
|
14.4 Malignant Pleural Mesothelioma
First-line treatment of unresectable advanced or metastatic malignant pleural mesothelioma (MPM) with pemetrexed and platinum chemotherapy
The efficacy of intravenous pembrolizumab in combination with pemetrexed and platinum chemotherapy was investigated in KEYNOTE-483 (NCT02784171), a multicenter, randomized, open-label, active-controlled trial that enrolled 440 patients with unresectable advanced or metastatic MPM and no prior systemic therapy for advanced/metastatic disease. Patients were enrolled regardless of tumor PD-L1 expression. Patients with autoimmune disease that required systemic therapy within 3 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by histological subtype (epithelioid vs. non-epithelioid). Patients were randomized (1:1) to one of the following treatment arms; all study medications were administered via intravenous infusion:
- Intravenous pembrolizumab 200 mg with pemetrexed 500 mg/m2 and cisplatin 75 mg/m2 or carboplatin AUC 5-6 mg/mL/min on Day 1 of each 21-day cycle for up to 6 cycles, followed by intravenous pembrolizumab 200 mg every 3 weeks.
- Pemetrexed 500 mg/m2 and cisplatin 75 mg/m2 or carboplatin AUC 5-6 mg/mL/min on Day 1 of each 21-day cycle for up to 6 cycles.
Treatment with intravenous pembrolizumab continued until disease progression as determined by the investigator according to modified RECIST 1.1 for mesothelioma (mRECIST), unacceptable toxicity, or a maximum of 24 months. Assessment of tumor status was performed every 6 weeks for 18 weeks, followed by every 12 weeks thereafter. The main efficacy outcome measure was OS. Additional efficacy outcome measures were PFS, ORR, and DoR, as assessed by BICR according to mRECIST.
The study population characteristics were: median age of 70 years (77% age 65 or older); 76% male; 79% White, 21% race not reported or unknown; 2% Hispanic or Latino; and 53% ECOG performance status of 1. Seventy-eight percent had epithelioid and 22% had non-epithelioid histology; 60% had tumors with PD-L1 CPS ≥1 and 30% had tumors with PD-L1 CPS <1.
The trial demonstrated a statistically significant improvement in OS, PFS, and ORR in patients randomized to intravenous pembrolizumab in combination with chemotherapy compared with patients randomized to chemotherapy alone. Table 69 and Figure 12 summarize the efficacy results for KEYNOTE-483.
| Endpoint | Intravenous pembrolizumab 200 mg every 3 weeks Pemetrexed Platinum Chemotherapy |
Pemetrexed Platinum Chemotherapy |
|---|---|---|
| (n=222) | (n=218) | |
| OS | ||
| Number (%) of patients with event | 167 (75%) | 175 (80%) |
| Median in months (95% CI) | 17.3 (14.4, 21.3) | 16.1 (13.1, 18.2) |
| Hazard ratio Based on stratified Cox proportional hazard model (95% CI) |
0.79 (0.64, 0.98) | |
| p-Value Based on stratified log-rank test
|
0.0162 | |
| PFS | ||
| Number (%) of patients with event | 190 (86%) | 166 (76%) |
| Median in months (95% CI) | 7.1 (6.9, 8.1) | 7.1 (6.8, 7.7) |
| Hazard ratio (95% CI) | 0.80 (0.65, 0.99) | |
| p-Value | 0.0194 | |
| Objective Response Rate | ||
| ORR % (95% CI) | 52% (45.5, 59.0) | 29% (23.0, 35.4) |
| Complete responses | 1 (0.5%) | 0 (0%) |
| Partial responses | 115 (52%) | 63 (29%) |
| p-Value Based on Miettinen and Nurminen method stratified by histological subtype at randomization (epithelioid vs. non-epithelioid)
|
<0.00001 | |
|
Duration of Response
Based on patients with a best overall response as confirmed complete or partial response; n=116 for patients in the intravenous pembrolizumab combination arm; n=63 for patients in the chemotherapy arm
|
||
| Median in months (95% CI) | 6.9 (5.8, 8.3) | 6.8 (5.5, 8.5) |
In a pre-specified exploratory analysis based on histology, in the subgroup of patients with epithelioid histology (n=345), the hazard ratio (HR) for OS was 0.89 (95% CI: 0.70, 1.13), with median OS of 19.8 months in intravenous pembrolizumab in combination with chemotherapy and 18.2 months in chemotherapy alone. In the subgroup of patients with non-epithelioid histology (n=95), the HR for OS was 0.57 (95% CI: 0.36, 0.89), with median OS of 12.3 months in intravenous pembrolizumab in combination with chemotherapy and 8.2 months in chemotherapy alone.
16 How Supplied/storage and Handling
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration. Each carton contains one single-dose vial either as:
- 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/ 2,000 units per mL), NDC 0006-3083-01
- 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/ 2,000 units per mL), NDC 0006-5083-01
1.17 Cutaneous Squamous Cell Carcinoma
KEYTRUDA QLEX is indicated for the treatment of adult patients with recurrent or metastatic cutaneous squamous cell carcinoma (cSCC) or locally advanced cSCC that is not curable by surgery or radiation.
1.4 Head and Neck Squamous Cell Cancer
KEYTRUDA QLEX is indicated for the treatment of adult patients with resectable locally advanced HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test [see Dosage and Administration (2.1)], as a single agent as neoadjuvant treatment, continued as adjuvant treatment in combination with radiotherapy (RT) with or without cisplatin and then as a single agent.
KEYTRUDA QLEX, in combination with platinum and fluorouracil (FU), is indicated for the first-line treatment of adult patients with metastatic or with unresectable, recurrent head and neck squamous cell carcinoma (HNSCC).
KEYTRUDA QLEX, as a single agent, is indicated for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy.
14.18 Cutaneous Squamous Cell Carcinoma
The efficacy of intravenous pembrolizumab was investigated in patients with recurrent or metastatic cSCC or locally advanced cSCC enrolled in KEYNOTE-629 (NCT03284424), a multicenter, multi-cohort, non-randomized, open-label trial. The trial excluded patients with autoimmune disease or a medical condition that required immunosuppression.
Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until documented disease progression, unacceptable toxicity, or a maximum of 24 months. Patients with initial radiographic disease progression could receive additional doses of intravenous pembrolizumab during confirmation of progression unless disease progression was symptomatic, rapidly progressive, required urgent intervention, or occurred with a decline in performance status.
Assessment of tumor status was performed every 6 weeks during the first year, and every 9 weeks during the second year. The major efficacy outcome measures were ORR and DoR as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among the 105 patients with recurrent or metastatic cSCC treated, the study population characteristics were: median age of 72 years (range: 29 to 95), 71% age 65 or older; 76% male; 70% White, 25% race unknown; 34% ECOG PS of 0 and 66% ECOG PS of 1. Forty-five percent of patients had locally recurrent only cSCC, 24% had metastatic only cSCC, and 31% had both locally recurrent and metastatic cSCC. Eighty-seven percent received one or more prior lines of therapy; 73% received prior radiation therapy.
Among the 54 patients with locally advanced cSCC treated, the study population characteristics were: median age of 76 years (range: 35 to 95), 80% age 65 or older; 72% male; 83% White, 13% race unknown; 41% ECOG PS of 0 and 59% ECOG PS of 1. Twenty-two percent received one or more prior lines of therapy; 63% received prior radiation therapy.
Efficacy results are summarized in Table 101.
| Endpoint | Intravenous Pembrolizumab Recurrent or Metastatic cSCC n=105 |
Intravenous Pembrolizumab Locally Advanced cSCC n=54 |
|---|---|---|
| + Denotes ongoing response | ||
| Objective Response Rate | ||
| ORR (95% CI) | 35% (26, 45) | 52% (38, 66) |
| Complete response rate | 12% | 22% |
| Partial response rate | 23% | 30% |
|
Duration of Response Median follow-up time: recurrent or metastatic cSCC: 23.8 months; locally advanced cSCC: 48.0 months
|
n=37 | n=28 |
| Median in months (range) | NR (2.7, 64.2+) | 47.2 (1.0+, 49.9+) |
| % with duration ≥6 months | 76% | 89% |
| % with duration ≥12 months | 68% | 75% |
1.16 Tumor Mutational Burden High Cancer
KEYTRUDA QLEX is indicated for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-authorized test [see Dosage and Administration (2.1)], that have progressed following prior treatment and who have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on tumor response rate and durability of response [see Clinical Studies (14.17)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Limitations of Use: The safety and effectiveness of KEYTRUDA QLEX in pediatric patients 12 years and older with TMB-H central nervous system cancers have not been established.
14.17 Tumor Mutational Burden High Cancer
The efficacy of intravenous pembrolizumab was investigated in a prospectively-planned retrospective analysis of 10 cohorts (A through J) of patients with various previously treated unresectable or metastatic solid tumors with high tumor mutation burden (TMB-H) who were enrolled in a multicenter, non-randomized, open-label trial, KEYNOTE-158 (NCT02628067). The trial excluded patients who previously received an anti-PD-1 or other immune-modulating monoclonal antibody, or who had an autoimmune disease, or a medical condition that required immunosuppression. Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression. Assessment of tumor status was performed every 9 weeks for the first 12 months and every 12 weeks thereafter.
The statistical analysis plan pre-specified ≥10 and ≥13 mutations per megabase using the FoundationOne CDx assay as cutpoints to assess TMB. Testing of TMB was blinded with respect to clinical outcomes. The major efficacy outcome measures were ORR and DoR in patients who received at least one dose of intravenous pembrolizumab as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
In KEYNOTE-158, 1050 patients were included in the efficacy analysis population. TMB was analyzed in the subset of 790 patients with sufficient tissue for testing based on protocol-specified testing requirements. Of the 790 patients, 102 (13%) had tumors identified as TMB-H, defined as TMB ≥10 mutations per megabase. Among the 102 patients with TMB-H advanced solid tumors, the study population characteristics were: median age of 61 years (range: 27 to 80), 34% age 65 or older; 34% male; 81% White; and 41% ECOG PS of 0 and 58% ECOG PS of 1. Fifty-six percent of patients had at least two prior lines of therapy.
Efficacy results are summarized in Tables 99 and 100.
|
Endpoint |
Intravenous Pembrolizumab 200 mg every 3 weeks |
|
|---|---|---|
| TMB ≥10 mut/Mb n=102 Median follow-up time of 11.1 months
|
TMB ≥13 mut/Mb n=70 |
|
| + Denotes ongoing response NR = not reached |
||
| Objective Response Rate | ||
| ORR (95% CI) | 29% (21, 39) | 37% (26, 50) |
| Complete response rate | 4% | 3% |
| Partial response rate | 25% | 34% |
| Duration of Response | n=30 | n=26 |
| Median in months (range) From product-limit (Kaplan-Meier) method for censored data
|
NR (2.2+, 34.8+) | NR (2.2+, 34.8+) |
| % with duration ≥12 months | 57% | 58% |
| % with duration ≥24 months | 50% | 50% |
| Objective Response Rate | Duration of Response range | |||
|---|---|---|---|---|
| N | n (%) | 95% CI | (months) | |
| CR = complete response PR = partial response SD = stable disease PD = progressive disease |
||||
|
Overall No TMB-H patients were identified in the cholangiocarcinoma cohort
|
102 | 30 (29%) | (21%, 39%) | (2.2+, 34.8+) |
| Small cell lung cancer | 34 | 10 (29%) | (15%, 47%) | (4.1, 32.5+) |
| Cervical cancer | 16 | 5 (31%) | (11%, 59%) | (3.7+, 34.8+) |
| Endometrial cancer | 15 | 7 (47%) | (21%, 73%) | (8.4+, 33.9+) |
| Anal cancer | 14 | 1 (7%) | (0.2%, 34%) | 18.8+ |
| Vulvar cancer | 12 | 2 (17%) | (2%, 48%) | (8.8, 11.0) |
| Neuroendocrine cancer | 5 | 2 (40%) | (5%, 85%) | (2.2+, 32.6+) |
| Salivary cancer | 3 | PR, SD, PD | 31.3+ | |
| Thyroid cancer | 2 | CR, CR | (8.2, 33.2+) | |
| Mesothelioma cancer | 1 | PD |
In an exploratory analysis in 32 patients enrolled in KEYNOTE-158 whose cancer had TMB ≥10 mut/Mb and <13 mut/Mb, the ORR was 13% (95% CI: 4%, 29%), including two complete responses and two partial responses.
13.2 Animal Toxicology And/or Pharmacology
In animal models, inhibition of PD-1/PD-L1 signaling increased the severity of some infections and enhanced inflammatory responses. Mycobacterium tuberculosis-infected PD-1 knockout mice exhibit markedly decreased survival compared with wild-type controls, which correlated with increased bacterial proliferation and inflammatory responses in these animals. PD-1 blockade using a primate anti-PD-1 antibody was also shown to exacerbate M. tuberculosis infection in rhesus macaques. PD-1 and PD-L1 knockout mice and mice receiving PD-L1-blocking antibody have also shown decreased survival following infection with lymphocytic choriomeningitis virus. Administration of pembrolizumab in chimpanzees with naturally occurring chronic hepatitis B infection resulted in two out of four animals with significantly increased levels of serum ALT, AST, and GGT, which persisted for at least 1 month after discontinuation of pembrolizumab.
2.2 Important Dosage and Administration Information
- KEYTRUDA QLEX has different recommended dosage and administration instructions than intravenous pembrolizumab.
- To reduce the risk of medication errors, check the vial labels to ensure that the drug being prepared and administered is KEYTRUDA QLEX for subcutaneous use and not intravenous pembrolizumab.
- Do not substitute KEYTRUDA QLEX with intravenous pembrolizumab because they have different recommended dosages and routes of administration.
- Patients receiving intravenous pembrolizumab can switch to subcutaneous KEYTRUDA QLEX at their next scheduled dose.
- Patients receiving subcutaneous KEYTRUDA QLEX can switch to intravenous pembrolizumab at their next scheduled dose.
- Administer KEYTRUDA QLEX as a subcutaneous injection into the thigh or abdomen, avoiding the 5 cm area around the navel.
- Every 3-week dosing (395 mg/4,800 units): inject 2.4 mL subcutaneously over 1 minute. Treatment duration is provided in Recommended Dosage (Table 1).
- Every 6-week dosing (790 mg/9,600 units): inject 4.8 mL subcutaneously over 2 minutes. Treatment duration is provided in Recommended Dosage (Table 1).
- Inject into healthy skin and never into areas where the skin is red, bruised, tender, or hard.
- Ensure the injection site is at least 2.5 cm from the previous injection site.
- During treatment with KEYTRUDA QLEX, do not administer other medications for subcutaneous use at the same site as KEYTRUDA QLEX.
- Do not administer KEYTRUDA QLEX intravenously.
- KEYTRUDA QLEX must be administered by a healthcare provider.
5.1 Severe and Fatal Immune Mediated Adverse Reactions
KEYTRUDA QLEX is a monoclonal antibody that belongs to a class of drugs that bind to either the programmed death-receptor 1 (PD-1) or the PD-ligand 1 (PD-L1), blocking the PD-1/PD-L1 pathway, thereby removing inhibition of the immune response, potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Important immune-mediated adverse reactions listed under WARNINGS AND PRECAUTIONS may not include all possible severe and fatal immune-mediated adverse reactions.
Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue and can affect more than one body system simultaneously. Immune-mediated adverse reactions can occur at any time after starting treatment with a PD-1/PD-L1 blocking antibody. While immune-mediated adverse reactions usually manifest during treatment with PD-1/PD-L1 blocking antibodies, immune-mediated adverse reactions can also manifest after discontinuation of PD-1/PD-L1 blocking antibodies.
Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of PD-1/PD-L1 blocking antibodies. Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. For patients with TNBC treated with KEYTRUDA QLEX in the neoadjuvant setting, monitor blood cortisol at baseline, prior to surgery, and as clinically indicated. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)]. In general, if KEYTRUDA QLEX requires interruption or discontinuation, administer systemic corticosteroid therapy (1 to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reactions are not controlled with corticosteroid therapy.
Toxicity management guidelines for adverse reactions that do not necessarily require systemic steroids (e.g., endocrinopathies and dermatologic reactions) are discussed below.
Immune-Mediated Pneumonitis
KEYTRUDA QLEX can cause immune-mediated pneumonitis. The incidence of pneumonitis is higher in patients who have received prior thoracic radiation. Immune-mediated pneumonitis occurred in 5% (13/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including fatal (0.4%), Grade 3 (2%), and Grade 2 (1.2%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated pneumonitis occurred in 3.4% (94/2799) of patients receiving intravenous pembrolizumab, including fatal (0.1%), Grade 4 (0.3%), Grade 3 (0.9%), and Grade 2 (1.3%) adverse reactions. Systemic corticosteroids were required in 67% (63/94) of patients with pneumonitis. Pneumonitis led to permanent discontinuation of intravenous pembrolizumab in 1.3% (36) of patients and withholding of intravenous pembrolizumab in 0.9% (26) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, 23% had recurrence of pneumonitis. Pneumonitis resolved in 59% of the 94 patients.
In a clinical study enrolling 580 adult patients with resected NSCLC (KEYNOTE-091) who received intravenous pembrolizumab as a single agent for adjuvant treatment, pneumonitis occurred in 41 (7%) patients, including fatal (0.2%), Grade 4 (0.3%), and Grade 3 (1%) adverse reactions. Patients received high-dose corticosteroids for a median duration of 10 days (range: 1 day to 2.3 months). Pneumonitis led to discontinuation of intravenous pembrolizumab in 26 (4.5%) of patients. Of the patients who developed pneumonitis, 54% interrupted intravenous pembrolizumab, 63% discontinued intravenous pembrolizumab, and 71% had resolution.
Immune-Mediated Colitis
KEYTRUDA QLEX can cause immune-mediated colitis, which may present with diarrhea. Cytomegalovirus (CMV) infection/reactivation has been reported in patients with corticosteroid-refractory immune-mediated colitis. In cases of corticosteroid-refractory colitis, consider repeating infectious workup to exclude alternative etiologies. Immune-mediated colitis occurred in 1.2% (3/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 3 (0.8%), and Grade 2 (0.4%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated colitis occurred in 1.7% (48/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (1.1%), and Grade 2 (0.4%) adverse reactions. Systemic corticosteroids were required in 69% (33/48) of patients with colitis. Additional immunosuppressant therapy was required in 4.2% of patients. Colitis led to permanent discontinuation of intravenous pembrolizumab in 0.5% (15) of patients and withholding of intravenous pembrolizumab in 0.5% (13) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, 23% had recurrence of colitis. Colitis resolved in 85% of the 48 patients.
Hepatotoxicity and Immune-Mediated Hepatitis
KEYTRUDA QLEX can cause immune-mediated hepatitis. Immune-mediated hepatitis occurred in 0.4% (1/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (0.4%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated hepatitis occurred in 0.7% (19/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.4%), and Grade 2 (0.1%) adverse reactions. Systemic corticosteroids were required in 68% (13/19) of patients with hepatitis. Eleven percent of these patients required additional immunosuppressant therapy. Hepatitis led to permanent discontinuation of intravenous pembrolizumab in 0.2% (6) of patients and withholding of intravenous pembrolizumab in 0.3% (9) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, none had recurrence of hepatitis. Hepatitis resolved in 79% of the 19 patients.
In Combination with Axitinib
KEYTRUDA QLEX in combination with axitinib can cause hepatic toxicity with higher than expected frequencies of Grades 3 and 4 ALT and AST elevations compared to KEYTRUDA QLEX alone. Monitor liver enzymes before initiation of and periodically throughout treatment. Consider more frequent monitoring of liver enzymes as compared to when the drugs are administered as single agents. For elevated liver enzymes, interrupt KEYTRUDA QLEX and axitinib, and consider administering corticosteroids as needed [see Dosage and Administration (2.4)].
Intravenous Pembrolizumab in Combination with Axitinib
With the combination of intravenous pembrolizumab and axitinib, Grades 3 and 4 increased ALT (20%) and increased AST (13%) were seen. Fifty-nine percent of the patients with increased ALT received systemic corticosteroids. In patients with ALT ≥3 times ULN (Grades 2-4, n=116), ALT resolved to Grades 0-1 in 94%. Among the 92 patients who were rechallenged with either intravenous pembrolizumab (n=3) or axitinib (n=34) administered as a single agent or with both (n=55), recurrence of ALT ≥3 times ULN was observed in 1 patient receiving intravenous pembrolizumab, 16 patients receiving axitinib, and 24 patients receiving both intravenous pembrolizumab and axitinib. All patients with a recurrence of ALT ≥3 ULN subsequently recovered from the event.
Immune-Mediated Endocrinopathies
Adrenal Insufficiency
KEYTRUDA QLEX can cause primary or secondary adrenal insufficiency. For Grade 2 or higher adrenal insufficiency, initiate symptomatic treatment, including hormone replacement as clinically indicated. Withhold KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Adrenal insufficiency occurred in 2% (5/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 3 (0.4%), and Grade 2 (0.8%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Adrenal insufficiency occurred in 0.8% (22/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.3%), and Grade 2 (0.3%) adverse reactions. Systemic corticosteroids were required in 77% (17/22) of patients with adrenal insufficiency; of these, the majority remained on systemic corticosteroids. Adrenal insufficiency led to permanent discontinuation of intravenous pembrolizumab in <0.1% (1) of patients and withholding of intravenous pembrolizumab in 0.3% (8) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement.
Hypophysitis
KEYTRUDA QLEX can cause immune-mediated hypophysitis. Hypophysitis can present with acute symptoms associated with mass effect such as headache, photophobia, or visual field defects. Hypophysitis can cause hypopituitarism. Initiate hormone replacement as indicated. Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Intravenous Pembrolizumab as a Single Agent
Hypophysitis occurred in 0.6% (17/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.3%), and Grade 2 (0.2%) adverse reactions. Systemic corticosteroids were required in 94% (16/17) of patients with hypophysitis; of these, the majority remained on systemic corticosteroids. Hypophysitis led to permanent discontinuation of intravenous pembrolizumab in 0.1% (4) of patients and withholding of intravenous pembrolizumab in 0.3% (7) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement.
Thyroid Disorders
KEYTRUDA QLEX can cause immune-mediated thyroid disorders. Thyroiditis can present with or without endocrinopathy. Hypothyroidism can follow hyperthyroidism. Initiate hormone replacement for hypothyroidism or institute medical management of hyperthyroidism as clinically indicated. Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Thyroiditis occurred in 0.4% (1/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (0.4%). Hyperthyroidism occurred in 8% (20/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (3.2%). Hypothyroidism occurred in 14% (35/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (11%).
Intravenous Pembrolizumab as a Single Agent
Thyroiditis occurred in 0.6% (16/2799) of patients receiving intravenous pembrolizumab, including Grade 2 (0.3%). No patients discontinued intravenous pembrolizumab due to thyroiditis. Intravenous pembrolizumab was withheld in <0.1% (1) of patients.
Hyperthyroidism occurred in 3.4% (96/2799) of patients receiving intravenous pembrolizumab, including Grade 3 (0.1%) and Grade 2 (0.8%). Hyperthyroidism led to permanent discontinuation of intravenous pembrolizumab in <0.1% (2) of patients and withholding of intravenous pembrolizumab in 0.3% (7) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement.
The incidence of new or worsening hyperthyroidism was higher in 580 patients with resected NSCLC, occurring in 11% of patients receiving intravenous pembrolizumab as a single agent as adjuvant treatment (KEYNOTE-091), including Grade 3 (0.2%) hyperthyroidism.
Hypothyroidism occurred in 8% (237/2799) of patients receiving intravenous pembrolizumab, including Grade 3 (0.1%) and Grade 2 (6.2%). Hypothyroidism led to permanent discontinuation of intravenous pembrolizumab in <0.1% (1) of patients and withholding of intravenous pembrolizumab in 0.5% (14) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement. The majority of patients with hypothyroidism required long-term thyroid hormone replacement.
The incidence of new or worsening hypothyroidism was higher in 1185 patients with HNSCC, occurring in 16% of patients receiving intravenous pembrolizumab as a single agent or in combination with platinum and FU, including Grade 3 (0.3%) hypothyroidism.
The incidence of new or worsening hypothyroidism was higher in 580 patients with resected NSCLC, occurring in 22% of patients receiving intravenous pembrolizumab as a single agent as adjuvant treatment (KEYNOTE-091), including Grade 3 (0.3%) hypothyroidism.
Type 1 Diabetes Mellitus, which can present with Diabetic Ketoacidosis
Monitor patients for hyperglycemia or other signs and symptoms of diabetes. Initiate treatment with insulin as clinically indicated. Withhold KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Type 1 diabetes mellitus occurred in 0.4% (1/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy.
Intravenous Pembrolizumab as a Single Agent
Type 1 diabetes mellitus occurred in 0.2% (6/2799) of patients receiving intravenous pembrolizumab. Type 1 diabetes mellitus led to permanent discontinuation in <0.1% (1) of patients and withholding of intravenous pembrolizumab in <0.1% (1) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement. All patients with Type 1 diabetes mellitus required long-term insulin therapy.
Immune-Mediated Nephritis with Renal Dysfunction
KEYTRUDA QLEX can cause immune-mediated nephritis.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated nephritis occurred in 0.3% (9/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.1%), and Grade 2 (0.1%) adverse reactions. Systemic corticosteroids were required in 89% (8/9) of patients with nephritis. Nephritis led to permanent discontinuation of intravenous pembrolizumab in 0.1% (3) of patients and withholding of intravenous pembrolizumab in 0.1% (3) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, none had recurrence of nephritis. Nephritis resolved in 56% of the 9 patients.
Immune-Mediated Dermatologic Adverse Reactions
KEYTRUDA QLEX can cause immune-mediated rash or dermatitis. Exfoliative dermatitis, including Stevens Johnson Syndrome, DRESS, and toxic epidermal necrolysis (TEN), has occurred with PD-1/PD-L1 blocking antibodies. Topical emollients and/or topical corticosteroids may be adequate to treat mild to moderate non-exfoliative rashes. Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Immune-mediated dermatologic adverse reactions occurred in 1.6% (4/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 4 (0.8%), and Grade 3 (0.8%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated dermatologic adverse reactions occurred in 1.4% (38/2799) of patients receiving intravenous pembrolizumab, including Grade 3 (1%) and Grade 2 (0.1%) adverse reactions. Systemic corticosteroids were required in 40% (15/38) of patients with immune-mediated dermatologic adverse reactions. Immune-mediated dermatologic adverse reactions led to permanent discontinuation of intravenous pembrolizumab in 0.1% (2) of patients and withholding of intravenous pembrolizumab in 0.6% (16) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, 6% had recurrence of immune-mediated dermatologic adverse reactions. Immune-mediated dermatologic adverse reactions resolved in 79% of the 38 patients.
Other Immune-Mediated Adverse Reactions
The following clinically significant immune-mediated adverse reactions occurred at an incidence of <1% (unless otherwise noted) in patients who received KEYTRUDA QLEX, intravenous pembrolizumab, or were reported with the use of other PD-1/PD-L1 blocking antibodies. Severe or fatal cases have been reported for some of these adverse reactions.
Cardiac/Vascular: Myocarditis, pericarditis, vasculitis
Nervous System: Meningitis, encephalitis, myelitis and demyelination, myasthenic syndrome/myasthenia gravis (including exacerbation), Guillain-Barré syndrome, nerve paresis, autoimmune neuropathy
Ocular: Uveitis, iritis and other ocular inflammatory toxicities can occur. Some cases can be associated with retinal detachment. Various grades of visual impairment, including blindness, can occur. If uveitis occurs in combination with other immune-mediated adverse reactions, consider a Vogt-Koyanagi-Harada-like syndrome, as this may require treatment with systemic steroids to reduce the risk of permanent vision loss.
Gastrointestinal: Pancreatitis, to include increases in serum amylase and lipase levels, gastritis (2.8%), duodenitis
Musculoskeletal and Connective Tissue: Myositis/polymyositis, rhabdomyolysis (and associated sequelae, including renal failure), arthritis (1.5%), polymyalgia rheumatica
Endocrine: Hypoparathyroidism
Hematologic/Immune: Hemolytic anemia, aplastic anemia, hemophagocytic lymphohistiocytosis, systemic inflammatory response syndrome, histiocytic necrotizing lymphadenitis (Kikuchi lymphadenitis), sarcoidosis, immune thrombocytopenic purpura, solid organ transplant rejection, other transplant (including corneal graft) rejection
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
KEYTRUDA QLEX contains pembrolizumab and berahyaluronidase alfa.
Pembrolizumab
No studies have been performed to test the potential of pembrolizumab for carcinogenicity or genotoxicity.
Fertility studies have not been conducted with pembrolizumab. In 1-month and 6-month repeat-dose toxicology studies in monkeys, there were no notable effects in the male and female reproductive organs; however, most animals in these studies were not sexually mature.
Berahyaluronidase alfa
Hyaluronidases are found in most tissues of the body. Long-term animal studies have not been performed to assess the carcinogenic or mutagenic potential of berahyaluronidase alfa.
In a fertility and early embryonic development study, male and female rats were administered daily subcutaneous injections of 280,000, 840,000, or 2,520,000 U/kg berahyaluronidase alfa. Males were dosed for 9 weeks prior to mating and throughout mating to termination. Females were dosed for 2 weeks prior to mating, throughout mating, and up to gestation day 7. Although treatment with ≥840,000 U/kg berahyaluronidase alfa (>5,200 times higher than the human dose) resulted in an increased incidence of abnormal sperm morphology, there were no adverse effects on mating, fertility or embryogenesis observed at doses up to 2,520,000 U/kg (>15,000 times higher than the human dose).
5.2 Hypersensitivity and Administration Related Reactions
KEYTRUDA QLEX can cause severe or life-threatening administration-related reactions, including hypersensitivity and anaphylaxis. In Study MK-3475A-D77, hypersensitivity and administration-related systemic reactions occurred in 3.2% (8/251) of patients receiving KEYTRUDA QLEX, including Grade 2 (2.8%). Monitor patients for signs and symptoms of administration-related systemic reactions including rigors, chills, wheezing, pruritus, flushing, rash, hypotension, hypoxemia, and fever. Interrupt injection (if not already fully administered) and resume if symptoms resolve for mild or moderate hypersensitivity and administration-related systemic reactions. For severe or life-threatening hypersensitivity and administration-related systemic reactions, stop injection and permanently discontinue KEYTRUDA QLEX [see Dosage and Administration (2.4)].
Principal Display Panel 395 Mg and 4,800 Units/2.4 Ml Vial Carton
NDC 0006-3083-01
Keytruda Qlex ™
(pembrolizumab and
berahyaluronidase alfa-pmph) Injection
395 mg and 4,800 units/2.4 mL
(165 mg and 2,000 units/mL)
For Subcutaneous Use Only
Administer subcutaneous injection
over 1 minute
Dispense the enclosed Medication Guide to each patient.
One Single-dose vial. Discard unused portion.
Rx only
Principal Display Panel 790 Mg and 9,600 Units/4.8 Ml Vial Carton
NDC 0006-5083-01
Keytruda Qlex ™
(pembrolizumab and
berahyaluronidase alfa-pmph) Injection
790 mg and 9,600 units/4.8 mL
(165 mg and 2,000 units/mL)
For Subcutaneous Use Only
Administer subcutaneous injection
over 2 minutes
Dispense the enclosed Medication Guide to each patient.
One Single-dose vial. Discard unused portion.
Rx only
1.6 Microsatellite Instability High Or Mismatch Repair Deficient Cancer
KEYTRUDA QLEX is indicated for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options [see Dosage and Administration (2.1)].
14.7 Microsatellite Instability High Or Mismatch Repair Deficient Cancer
The efficacy of intravenous pembrolizumab was investigated in 504 patients with MSI-H or dMMR cancers enrolled in three multicenter, non-randomized, open-label, multi-cohort trials: KEYNOTE-164 (NCT02460198), KEYNOTE-158 (NCT02628067), and KEYNOTE-051 (NCT02332668). All trials excluded patients with autoimmune disease or a medical condition that required immunosuppression. Regardless of histology, MSI or MMR tumor status was determined using polymerase chain reaction (PCR; local or central) or immunohistochemistry (IHC; local or central), respectively.
- KEYNOTE-164 enrolled 124 patients with advanced MSI-H or dMMR colorectal cancer (CRC) that progressed following treatment with fluoropyrimidine and either oxaliplatin or irinotecan +/- anti-VEGF/EGFR mAb-based therapy.
- KEYNOTE-158 enrolled 373 patients with advanced MSI-H or dMMR non-colorectal cancers (non-CRC) who had disease progression following prior therapy. Patients were either prospectively enrolled with MSI-H/dMMR tumors (Cohort K) or retrospectively identified in one of 10 solid tumor cohorts (Cohorts A-J).
- KEYNOTE-051 enrolled 7 pediatric patients with MSI-H or dMMR cancers.
Adult patients received intravenous pembrolizumab 200 mg every 3 weeks (pediatric patients received 2 mg/kg every 3 weeks) until unacceptable toxicity, disease progression, or a maximum of 24 months. In KEYNOTE-164 and KEYNOTE-158, assessment of tumor status was performed every 9 weeks through the first year, then every 12 weeks thereafter. In KEYNOTE-051, assessment of tumor status was performed every 8 weeks for 24 weeks, and then every 12 weeks thereafter. The major efficacy outcome measures were ORR and DoR as assessed by BICR according to RECIST v1.1 (modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ in KEYNOTE-158) and as assessed by the investigator according to RECIST v1.1 in KEYNOTE-051.
In KEYNOTE-164 and KEYNOTE-158, the study population characteristics were median age of 60 years, 36% age 65 or older; 44% male; 78% White, 14% Asian, 4% American Indian or Alaska Native, and 3% Black; and 45% ECOG PS of 0 and 55% ECOG PS of 1. Ninety-two percent of patients had metastatic disease and 4% had locally advanced, unresectable disease. Thirty-seven percent of patients received one prior line of therapy and 61% received two or more prior lines of therapy.
In KEYNOTE-051, the study population characteristics were median age of 11 years (range: 3 to 16); 71% female; 86% White and 14% Asian; and 57% had a Lansky/Karnofsky Score of 100. Seventy-one percent of patients had Stage IV and 14% had Stage III disease. Fifty-seven percent of patients received one prior line of therapy and 29% received two prior lines of therapy.
Discordant results were observed between local MSI-H or dMMR tests and central testing among patients enrolled in Cohort K of KEYNOTE-158. Among 104 tumor samples that were MSI-H or dMMR by local testing and also tested using the FoundationOne®CDx (F1CDx) test, 59 (56.7%) were MSI-H and 45 (43.3%) were not MSI-H. Among 169 tumor samples that were MSI-H or dMMR by local testing and also tested using the VENTANA MMR RxDx Panel, 105 (62.1%) were dMMR and 64 (37.9%) were pMMR.
Efficacy results are summarized in Tables 79 and 80.
| Endpoint | Intravenous Pembrolizumab n=504 Median follow-up time of 20.1 months (range 0.1 to 71.4 months)
|
|---|---|
| + Denotes ongoing response | |
| Objective Response Rate | |
| ORR (95% CI) Of the 7 pediatric patients from KEYNOTE-051, 1 patient had a radiographic complete response after initial growth of their tumor but is not reflected in the results.
|
33.3% (29.2, 37.6) |
| Complete response rate | 10.3% |
| Partial response rate | 23.0% |
| Duration of Response | n=168 |
| Median in months (range) | 63.2 (1.9+, 63.9+) |
| % with duration ≥12 months | 77% |
| % with duration ≥36 months | 39% |
| Objective Response Rate | Duration of Response range |
|||
|---|---|---|---|---|
| N | n (%) | 95% CI | (months) | |
| + Denotes ongoing response | ||||
| CRC | 124 | 42 (34%) | (26%, 43%) | (4.4, 58.5+) |
|
Non-CRC Results include patients in Cohort K of KEYNOTE-158 that were later determined to be pMMR or not MSI-H by central testing
|
380 | 126 (33%) | (28%, 38%) | (1.9+, 63.9+) |
| Endometrial cancer | 94 | 47 (50%) | (40%, 61%) | (2.9, 63.2) |
| Gastric or GE junction cancer | 51 | 20 (39%) | (26%, 54%) | (1.9+, 63.0+) |
| Small intestinal cancer | 27 | 16 (59%) | (39%, 78%) | (3.7+, 57.3+) |
| Brain cancer | 27 Includes 6 pediatric patients with brain cancer
|
1 (4%) In addition to the 1 adult responder, 1 pediatric patient had a radiographic complete response after initial growth of their tumor.
|
(0%, 19%) | 18.9 |
| Ovarian cancer | 25 | 8 (32%) | (15%, 54%) | (4.2, 56.6+) |
| Biliary cancer | 22 | 9 (41%) | (21%, 64%) | (6.2, 49.0+) |
| Pancreatic cancer | 22 | 4 (18%) | (5%, 40%) | (8.1, 24.3+) |
| Sarcoma | 14 | 3 (21%) | (5%, 51%) | (35.4+, 57.2+) |
| Breast cancer | 13 | 1 (8%) | (0%,36%) | 24.3+ |
| Other Includes tumor type (n): anal (3), HNSCC (1), nasopharyngeal (1), retroperitoneal (1), testicular (1), vaginal (1), vulvar (1), appendiceal adenocarcinoma, NOS (1), hepatocellular carcinoma (1), and carcinoma of unknown origin (1). Includes 1 pediatric patient with abdominal adenocarcinoma.
|
13 | 4 (31%) | (9%, 61%) | (6.2+, 32.3+) |
| Cervical cancer | 11 | 1 (9%) | (0%, 41%) | 63.9+ |
| Neuroendocrine cancer | 11 | 1 (9%) | (0%, 41%) | 13.3 |
| Prostate cancer | 8 | 1 (13%) | (0%, 53%) | 24.5+ |
| Adrenocortical cancer | 7 | 1 (14%) | (0%, 58%) | 4.2 |
| Mesothelioma | 7 | 0 (0%) | (0%, 41%) | |
| Thyroid cancer | 7 | 1 (14%) | (0%, 58%) | 8.2 |
| Small cell lung cancer | 6 | 2 (33%) | (4%, 78%) | (20.0, 47.5) |
| Bladder cancer | 6 | 3 (50%) | (12%, 88%) | (35.6+, 57.5+) |
| Salivary cancer | 5 | 2 (40%) | (5%, 85%) | (42.6+, 57.8+) |
| Renal cell cancer | 4 | 1 (25%) | (0%, 81%) | 22.0 |
Exploratory analysis by TMB
In an exploratory analysis performed in 138 patients (Cohort K of KEYNOTE-158) who were tested retrospectively for tumor mutation burden (TMB) using an FDA-authorized test, 45 (33%) had tumors with TMB score of <10 mut/Mb; ORR in these 45 patients was 6.7% (95% CI: 1.4, 18.3). Among the 45 patients with TMB score of <10 mut/Mb, 39 of the patients were pMMR/not MSI-H when tested using an FDA-authorized test.
1.7 Microsatellite Instability High Or Mismatch Repair Deficient Colorectal Cancer
KEYTRUDA QLEX is indicated for the treatment of adult patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
14.8 Microsatellite Instability High Or Mismatch Repair Deficient Colorectal Cancer
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-177 (NCT02563002), a multicenter, randomized, open-label, active-controlled trial that enrolled 307 patients with previously untreated unresectable or metastatic MSI-H or dMMR CRC. MSI or MMR tumor status was determined locally using polymerase chain reaction (PCR) or immunohistochemistry (IHC), respectively. Patients with autoimmune disease or a medical condition that required immunosuppression were ineligible.
Patients were randomized (1:1) to receive intravenous pembrolizumab 200 mg intravenously every 3 weeks or investigator’s choice of the following chemotherapy regimens given intravenously every 2 weeks:
- mFOLFOX6 (oxaliplatin, leucovorin, and FU) or mFOLFOX6 in combination with either bevacizumab or cetuximab: Oxaliplatin 85 mg/m2, leucovorin 400 mg/m2 (or levoleucovorin 200 mg/m2), and FU 400 mg/m2 bolus on Day 1, then FU 2400 mg/m2 over 46-48 hours. Bevacizumab 5 mg/kg on Day 1 or cetuximab 400 mg/m2 on first infusion, then 250 mg/m2 weekly.
- FOLFIRI (irinotecan, leucovorin, and FU) or FOLFIRI in combination with either bevacizumab or cetuximab: Irinotecan 180 mg/m2, leucovorin 400 mg/m2 (or levoleucovorin 200 mg/m2), and FU 400 mg/m2 bolus on Day 1, then FU 2400 mg/m2 over 46-48 hours. Bevacizumab 5 mg/kg on Day 1 or cetuximab 400 mg/m2 on first infusion, then 250 mg/m2 weekly.
Treatment with intravenous pembrolizumab or chemotherapy continued until RECIST v1.1-defined progression of disease as determined by the investigator or unacceptable toxicity. Patients treated with intravenous pembrolizumab without disease progression could be treated for up to 24 months. Assessment of tumor status was performed every 9 weeks. Patients randomized to chemotherapy were offered intravenous pembrolizumab at the time of disease progression. The main efficacy outcome measures were PFS (as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ) and OS. Additional efficacy outcome measures were ORR and DoR.
A total of 307 patients were enrolled and randomized to intravenous pembrolizumab (n=153) or chemotherapy (n=154). The baseline characteristics of these 307 patients were: median age of 63 years (range: 24 to 93), 47% age 65 or older; 50% male; 75% White and 16% Asian; 52% had an ECOG PS of 0 and 48% had an ECOG PS of 1; and 27% received prior adjuvant or neoadjuvant chemotherapy. Among 154 patients randomized to receive chemotherapy,143 received chemotherapy per the protocol. Of the 143 patients, 56% received mFOLFOX6, 44% received FOLFIRI, 70% received bevacizumab plus mFOLFOX6 or FOLFIRI, and 11% received cetuximab plus mFOLFOX6 or FOLFIRI.
The trial demonstrated a statistically significant improvement in PFS for patients randomized to intravenous pembrolizumab compared with chemotherapy. There was no statistically significant difference between intravenous pembrolizumab and chemotherapy in the final OS analysis. Sixty percent of the patients who had been randomized to receive chemotherapy had crossed over to receive subsequent anti-PD-1/PD-L1 therapies including intravenous pembrolizumab. The median follow-up time at the final analysis was 38.1 months (range: 0.2 to 58.7 months). Table 81 and Figure 21 summarize the key efficacy measures for KEYNOTE-177.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=153 |
Chemotherapy n=154 |
|---|---|---|
| + Denotes ongoing response NR = not reached |
||
| PFS | ||
| Number (%) of patients with event | 82 (54%) | 113 (73%) |
| Median in months (95% CI) | 16.5 (5.4, 32.4) | 8.2 (6.1, 10.2) |
| Hazard ratio
Based on Cox regression model
(95% CI) |
0.60 (0.45, 0.80) | |
| p-Value Two-sided p-Value based on log-rank test (compared to a significance level of 0.0234)
|
0.0004 | |
|
OS Final OS analysis
|
||
| Number (%) of patients with event | 62 (41%) | 78 (51%) |
| Median in months (95% CI) | NR (49.2, NR) | 36.7 (27.6, NR) |
| Hazard ratio (95% CI) | 0.74 (0.53, 1.03) | |
| p-Value Two-sided p-Value based on log-rank test (compared to a significance level of 0.0492)
|
0.0718 | |
|
Objective Response Rate
Based on confirmed response by BICR review
|
||
| ORR (95% CI) | 44% (35.8, 52.0) | 33% (25.8, 41.1) |
| Complete response rate | 11% | 4% |
| Partial response rate | 33% | 29% |
|
Duration of Response
,
Based on n=67 patients with a response in the intravenous pembrolizumab arm and n=51 patients with a response in the chemotherapy arm
|
||
| Median in months (range) | NR (2.3+, 41.4+) | 10.6 (2.8, 37.5+) |
| % with duration ≥12 months Based on observed duration of response
|
75% | 37% |
| % with duration ≥24 months | 43% | 18% |
|
|
5.4 Increased Mortality in Patients With Multiple Myeloma When Pembrolizumab Is Added to A Thalidomide Analogue and Dexamethasone
In two randomized trials in patients with multiple myeloma, the addition of intravenous pembrolizumab to a thalidomide analogue plus dexamethasone, a use for which no PD-1 or PD-L1 blocking antibody is indicated, resulted in increased mortality. Treatment of patients with multiple myeloma with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled trials.
Structured Label Content
Section 42229-5 (42229-5)
KEYTRUDA QLEX is a ready-to-use solution. Do not dilute KEYTRUDA QLEX.
Do not shake.
Preparation of the Syringe
- Remove KEYTRUDA QLEX vial from refrigerated storage [2°C to 8°C (36°F to 46°F)] and allow it to equilibrate to room temperature [20°C to 25°C (68°F to 77°F)] for at least 30 minutes.
- Prior to preparation for administration, if needed, the unpunctured vial may be stored at room temperature for up to 24 hours.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. The solution is clear to slightly opalescent, colorless to slightly yellow. Discard the vial if visible particles are observed.
- Use a sterile, polypropylene or polycarbonate syringe and a stainless steel transfer needle (18 to 21 gauge) to withdraw KEYTRUDA QLEX from the vial.
- Every 3-week dosing (395 mg pembrolizumab/4,800 units berahyaluronidase alfa): withdraw 2.4 mL into the syringe.
- Every 6-week dosing (790 mg pembrolizumab/9,600 units berahyaluronidase alfa): withdraw 4.8 mL into the syringe.
- To avoid needle clogging, change the needle to a 25 to 30 gauge, ½-inch, stainless steel hypodermic injection needle immediately prior to subcutaneous injection.
- Discard any unused portion left in the vial.
Storage of Prepared Syringe
The product does not contain a preservative and should be used immediately after withdrawing from the vial. If not used immediately, store the syringe containing KEYTRUDA QLEX with the transfer needle and cap in place:
- At room temperature 20°C to 25°C (68°F to 77°F) for up to 8 hours, or
- In the refrigerator at 2°C to 8°C (36°F to 46°F) for up to 24 hours. The 24-hour period may include up to 8 hours at room temperature.
Discard if storage time exceeds these limits.
If refrigerated, allow the filled syringe to come to room temperature for at least 30 minutes prior to administration.
Do not freeze.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Issued: 02/2026 | ||
|
MEDICATION GUIDE KEYTRUDA QLEX™ (key-true-duh Q-lex) (pembrolizumab and berahyaluronidase alfa-pmph) injection, for subcutaneous use |
|||
|
What is the most important information I should know about KEYTRUDA QLEX? |
|||
| KEYTRUDA QLEX is a medicine that may treat certain cancers by working with your immune system. KEYTRUDA QLEX can cause your immune system to attack normal organs and tissues in any area of your body and can affect the way they work. These problems can sometimes become severe or life-threatening and can lead to death. You can have more than one of these problems at the same time. These problems may happen anytime during treatment or even after your treatment has ended. | |||
| Call or see your healthcare provider right away if you develop any new or worsening signs or symptoms, including: | |||
| Lung problems | |||
|
|
|
|
| Intestinal problems | |||
|
|||
| Liver problems | |||
|
|
||
| Hormone gland problems | |||
|
|
||
| Kidney problems | |||
|
|
||
| Skin problems | |||
|
|
||
| Problems can also happen in other organs and tissues. These are not all of the signs and symptoms of immune system problems that can happen with KEYTRUDA QLEX. Call or see your healthcare provider right away for any new or worsening signs or symptoms, which may include: | |||
|
|||
| Allergic and injection-related reactions that can sometimes be severe or life-threatening, can happen during treatment with KEYTRUDA QLEX. Tell your healthcare provider right away if you get any signs or symptoms, including: | |||
|
|
||
| Rejection of a transplanted organ or tissue. Your healthcare provider should tell you what signs and symptoms you should report and monitor you depending on the type of organ or tissue transplant that you have had. | |||
| Complications, including graft-versus-host-disease (GVHD), in people who have received a bone marrow (stem cell) transplant that uses donor stem cells (allogeneic). These complications can be serious and can lead to death. These complications may happen if you underwent transplantation either before or after being treated with KEYTRUDA QLEX. Your healthcare provider will monitor you for these complications. | |||
|
Getting medical treatment right away may help keep these problems from becoming more serious. Your healthcare provider will check you for these problems during treatment with KEYTRUDA QLEX. Your healthcare provider may treat you with corticosteroid or hormone replacement medicines. Your healthcare provider may also need to delay or completely stop treatment with KEYTRUDA QLEX if you have severe side effects. |
|||
| What is KEYTRUDA QLEX? | |||
| KEYTRUDA QLEX is a prescription medicine used to treat: | |||
|
|||
|
Who should not receive KEYTRUDA QLEX?
Do not receive KEYTRUDA QLEX if you are allergic to berahyaluronidase, hyaluronidase or any of the inactive ingredients in KEYTRUDA QLEX. See the end of this Medication Guide for a complete list of ingredients in KEYTRUDA QLEX. |
|||
Before receiving KEYTRUDA QLEX, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
| How will I receive KEYTRUDA QLEX? | |||
|
|||
| What are the possible side effects of KEYTRUDA QLEX? | |||
| KEYTRUDA QLEX can cause serious side effects. See “What is the most important information I should know about KEYTRUDA QLEX?” | |||
| The most common side effects of KEYTRUDA QLEX when given with certain chemotherapy medicines include: nausea, tiredness, and muscle, bone, and joint pain. | |||
| The most common side effects seen with pembrolizumab given into the vein (intravenous pembrolizumab), which may happen with KEYTRUDA QLEX, are shown below: | |||
|
|||
| These are not all of the possible side effects of KEYTRUDA QLEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
| General information about the safe and effective use of KEYTRUDA QLEX | |||
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about KEYTRUDA QLEX that is written for health professionals. | |||
| What are the ingredients in KEYTRUDA QLEX? | |||
| Active ingredients: pembrolizumab and berahyaluronidase alfa-pmph | |||
| Inactive ingredients: histidine, histidine hydrochloride monohydrate, methionine, polysorbate 80, sucrose, and Water for Injection. | |||
| Manufactured by: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA U.S. License No. 0002 |
For patent information: www.msd.com/research/patent
Copyright © 2026 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved. usmg-mk3475a-i-2602r003 For more information, go to www.keytruda.com/qlex . |
Section 44425-7 (44425-7)
Store vials refrigerated at 2°C to 8°C (36°F to 46°F) in original carton to protect from light. Do not freeze. Do not shake.
1.1 Melanoma
KEYTRUDA QLEX™ is indicated for the treatment of adult patients with unresectable or metastatic melanoma.
KEYTRUDA QLEX is indicated for the adjuvant treatment of adult and pediatric patients 12 years and older with Stage IIB, IIC, or III melanoma following complete resection.
11 Description (11 DESCRIPTION)
KEYTRUDA QLEX is a fixed-combination drug product containing pembrolizumab and berahyaluronidase alfa.
Pembrolizumab is a programmed death receptor-1 (PD 1)-blocking antibody. Pembrolizumab is a humanized monoclonal IgG4 kappa antibody with an approximate molecular weight of 149 kDa. Pembrolizumab is produced in recombinant Chinese hamster ovary (CHO) cells.
Berahyaluronidase alfa is an endoglycosidase used to enhance dispersion and permeation, which facilitates delivery of increased volume of pembrolizumab that is co-administered subcutaneously. It is produced by mammalian CHO (Chinese Hamster Ovary) cells containing a DNA plasmid encoding a variant of human hyaluronidase PH20. It is a glycosylated protein with an approximate molecular weight of 49 kDa under nonreducing, deglycosylated conditions.
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration.
KEYTRUDA QLEX is supplied as two different configurations:
- Each KEYTRUDA QLEX 2.4 mL single-dose vial contains 395 mg of pembrolizumab and 4,800 units of berahyaluronidase alfa, and histidine (0.7 mg), histidine hydrochloride monohydrate (4.1 mg), methionine (3.6 mg), polysorbate 80 (0.5 mg), sucrose (168 mg), and Water for Injection, USP. The pH is 5.3-5.9.
- Each KEYTRUDA QLEX 4.8 mL single-dose vial contains 790 mg of pembrolizumab and 9,600 units of berahyaluronidase alfa, and histidine (1.4 mg), histidine hydrochloride monohydrate (8.2 mg), methionine (7.2 mg), polysorbate 80 (1 mg), sucrose (336 mg), and Water for Injection, USP. The pH is 5.3-5.9.
8.4 Pediatric Use
The safety and effectiveness of KEYTRUDA QLEX for the treatment of pediatric patients 12 years and older who weigh greater than 40 kg have been established for:
- Stage IIB, IIC, or III melanoma following complete resection [see Indications and Usage (1.1)]
- Unresectable or metastatic microsatellite instability high (MSI-H) or mismatch repair deficient (dMMR) solid tumors [see Indications and Usage (1.6)]
- Recurrent locally advanced or metastatic Merkel cell carcinoma [see Indications and Usage (1.13)]
- Unresectable or metastatic tumor mutational burden high solid tumors (TMB-H) [see Indications and Usage (1.16)]
Use of KEYTRUDA QLEX in pediatric patients for these indications is supported by evidence from adequate and well-controlled studies of intravenous pembrolizumab in adults and additional pharmacokinetic and safety data for intravenous pembrolizumab in pediatric patients 12 years and older [see Adverse Reactions (6.1), Clinical Studies (14)]. Pembrolizumab exposures in pediatric patients 12 years and older who weigh greater than 40 kg are predicted to be within range of those observed in adults at the same dosage [see Clinical Pharmacology (12.3)].
The safety and effectiveness of KEYTRUDA QLEX have not been established in pediatric patients younger than 12 years of age for the treatment of melanoma, MCC, MSI-H or dMMR cancer, and TMB-H cancer.
The safety and effectiveness of KEYTRUDA QLEX have not been established in pediatric patients for other approved indications [see Indications and Usage (1)].
Intravenous pembrolizumab
In KEYNOTE-051, 173 pediatric patients (including 108 pediatric patients aged 12 to 17 years) with advanced melanoma, lymphoma, or PD-L1 positive or MSI-H solid tumors received intravenous pembrolizumab 2 mg/kg every 3 weeks. The median duration of exposure was 2.1 months (range: 1 day to 25 months). Adverse reactions that occurred at a ≥10% higher rate in pediatric patients when compared to adults included pyrexia (33%), vomiting (29%), headache (25%), abdominal pain (23%), decreased lymphocyte count (13%), and decreased white blood cell count (11%). Laboratory abnormalities that occurred at a ≥10% higher rate in pediatric patients when compared to adults were leukopenia (30%), neutropenia (28%), thrombocytopenia (22%), and Grade 3 anemia (17%).
8.5 Geriatric Use
Of the 251 patients treated with KEYTRUDA QLEX in combination with platinum doublet chemotherapy in Study MK-3745A-D77, 53% were 65 years and older and 16% were 75 years and older. No overall differences in safety or effectiveness of KEYTRUDA QLEX have been observed between patients aged 65 years or older and younger adult patients.
The safety of KEYTRUDA QLEX as monotherapy or in combination with other antineoplastic drugs for its approved indications [see Indications and Usage (1)] has been established in adequate and well-controlled studies of intravenous pembrolizumab as a single agent and in combination with other antineoplastic drugs. Below is a description of geriatric use information from the intravenous pembrolizumab studies.
- Of 3781 patients with melanoma, NSCLC, HNSCC, or urothelial carcinoma who were treated with intravenous pembrolizumab in clinical studies, 48% were 65 years and over and 17% were 75 years and over.
- Of 506 adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC following complete resection and platinum-based chemotherapy who were treated with intravenous pembrolizumab in KEYNOTE-091, 242 (48%) were 65 years and over.
- Of 596 adult patients with TNBC who were treated with intravenous pembrolizumab in combination with paclitaxel, paclitaxel protein-bound, or gemcitabine and carboplatin in KEYNOTE-355, 137 (23%) were 65 years and over.
- Of 406 adult patients with endometrial carcinoma who were treated with intravenous pembrolizumab in combination with lenvatinib in KEYNOTE-775, 201 (50%) were 65 years and over.
- Of the 564 patients with locally advanced or metastatic urothelial cancer treated with intravenous pembrolizumab in combination with enfortumab vedotin, 44% (n=247) were 65-74 years and 26% (n=144) were 75 years or older. No overall differences in effectiveness were observed between patients 65 years of age or older and younger patients. Patients 75 years of age or older treated with intravenous pembrolizumab in combination with enfortumab vedotin experienced a higher incidence of fatal adverse reactions than younger patients. The incidence of fatal adverse reactions was 4% in patients younger than 75 and 7% in patients 75 years or older.
- Of the 167 patients with MIBC treated with intravenous pembrolizumab in combination with enfortumab vedotin, 37% (n=61) were 65-74 years and 46% (n=77) were 75 years or older. Patients 75 years of age or older treated with intravenous pembrolizumab in combination with enfortumab vedotin experienced a higher incidence of fatal adverse reactions than younger patients. The incidence of fatal adverse reactions was 4% in patients younger than 75 and 12% in patients 75 years or older.
- Of the 432 patients randomized to intravenous pembrolizumab in combination with axitinib in the KEYNOTE-426 trial, 40% were 65 years or older.
- Of 294 adult patients with FIGO 2014 Stage III-IVA cervical cancer who were treated with intravenous pembrolizumab in combination with CRT in KEYNOTE-A18, 42 (14%) were 65 years and over.
- Of 643 adult patients with ovarian cancer who were treated with intravenous pembrolizumab in combination with paclitaxel with or without bevacizumab in KEYNOTE-B96, 236 (37%) were 65 years and over and 58 (9%) were 75 years and over.
No overall differences in safety or effectiveness were observed between intravenous pembrolizumab-treated patients aged 65 years or older and younger adult patients.
1.8 Gastric Cancer
KEYTRUDA QLEX, in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, in combination with fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥ 1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
14.1 Keytruda Qlex (14.1 KEYTRUDA QLEX)
NSCLC (Study MK-3475A-D77)
KEYTRUDA QLEX was evaluated in Study MK-3475A-D77 (NCT05722015), a randomized, multicenter, open-label, active-controlled trial conducted in patients with treatment-naïve metastatic NSCLC, in whom there were no EGFR, ALK, or ROS1 genomic tumor aberrations. Patients were excluded if they had a history of autoimmune disease that required systemic therapy within 2 years of treatment; had a medical condition that required immunosuppression; or had received more than 30 Gy of thoracic radiation within the prior 26 weeks.
A total of 377 patients were randomized (2:1) to receive either KEYTRUDA QLEX (containing 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa) administered subcutaneously every 6 weeks with platinum doublet chemotherapy (n=251) or pembrolizumab 400 mg intravenously every 6 weeks with platinum doublet chemotherapy (n=126).
The chemotherapy regimens were as follows:
- Non-squamous NSCLC: pemetrexed 500 mg/m2 and a platinum chemotherapy (cisplatin 75 mg/m2 or carboplatin AUC 5 mg/mL/min) intravenously every 3 weeks for 4 cycles, followed by pemetrexed 500 mg/m2 intravenously every 3 weeks.
- Squamous NSCLC: carboplatin AUC 6 mg/mL/min and a taxane (paclitaxel 200 mg/m2 on Day 1 of each 21-day cycle or paclitaxel protein-bound 100 mg/m2 on Days 1, 8 and 15 of each 21-day cycle) intravenously every 3 weeks for 4 cycles.
Randomization was stratified by ECOG performance status (0 vs. 1), histology (squamous vs. non-squamous), PD-L1 TPS (<50% vs. ≥50%), and geographic region (East Asia vs. North America/Western Europe/Australia/New Zealand vs. Rest of the World).
Treatment with KEYTRUDA QLEX or intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by the investigator, unacceptable toxicity, or a maximum of 18 cycles (approximately 24 months). Administration of KEYTRUDA QLEX or intravenous pembrolizumab was permitted beyond RECIST-defined disease progression by BICR or beyond discontinuation of pemetrexed if the patient was clinically stable and deriving clinical benefit as determined by the investigator.
The primary outcome measure was pembrolizumab exposure [Cycle 1 AUC0-6 weeks and Cycle 3 (i.e., Steady State) Ctrough] of subcutaneous KEYTRUDA QLEX as compared to intravenous pembrolizumab [see Clinical Pharmacology (12.3)]. Additional descriptive efficacy outcome measures were overall response rate (ORR) by blinded independent central review (BICR), progression-free survival (PFS) by BICR, and overall survival (OS).
The study population characteristics were: median age 65 years (range: 37 to 87); 71% male; 63% White, 29% Asian, 4% multiracial, 3% Black or African American, 2% American Indian or Alaska Native; 31% of Hispanic or Latino ethnicity; 35% ECOG performance status (PS) of 0 and 65% ECOG PS of 1. Nineteen percent had PD-L1 TPS ≥50%; 34% had tumors with squamous histology and 66% had tumors with non-squamous histology; and 9% had brain metastases at baseline.
At the primary analysis, the confirmed ORR was 45% (95% CI: 39, 52) in the subcutaneous KEYTRUDA QLEX arm and 42% (95% CI: 33, 51) in the intravenous pembrolizumab arm. There were no notable differences in PFS or OS observed in patients who received KEYTRUDA QLEX compared to patients who received intravenous pembrolizumab.
Intravenous Pembrolizumab
The effectiveness of KEYTRUDA QLEX for its approved indications [see Indications and Usage (1)] has been established based upon the evidence from the adequate and well-controlled studies conducted with intravenous pembrolizumab [see Clinical Studies (14.2-14.20)] and additional data that demonstrated comparable pharmacokinetic, efficacy, and safety profiles between KEYTRUDA QLEX and intravenous pembrolizumab in Study MK-3475A-D77 [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.1)].
Below is a description of the efficacy results of these adequate and well-controlled studies of intravenous pembrolizumab in these patient populations.
1.19 Ovarian Cancer
KEYTRUDA QLEX, in combination with paclitaxel, with or without bevacizumab, is indicated for the treatment of adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)], and who have received one or two prior systemic treatment regimens.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described in this section with the incidence of ADA in other studies, including those of KEYTRUDA QLEX or of other pembrolizumab products or berahyaluronidase alfa products.
With a median (min, max) duration of treatment on KEYTRUDA QLEX of 6.9 months (1 day, 1 year) in Study MK-3475A-D77, 1.4% (3/211) of patients developed anti-pembrolizumab antibodies, and one ADA-positive patient developed neutralizing antibodies (NAb) against pembrolizumab. The incidence of anti-berahyaluronidase alfa antibodies was 1.5% (3/194). No analysis of neutralizing antibodies was performed for berahyaluronidase alfa ADA-positive samples. Because of the low occurrence of anti-pembrolizumab or anti-berahyaluronidase antibodies, the effect of these antibodies on the pharmacokinetics, safety and effectiveness of KEYTRUDA QLEX is unknown.
14.9 Gastric Cancer
First-line Treatment of Locally Advanced Unresectable or Metastatic HER2-Positive Gastric or Gastroesophageal Junction Adenocarcinoma for Tumors Expressing PD-L1 (CPS ≥1)
The efficacy of intravenous pembrolizumab in combination with trastuzumab plus fluoropyrimidine and platinum chemotherapy was investigated in KEYNOTE-811 (NCT03615326), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 698 patients with HER2-positive advanced gastric or gastroesophageal junction (GEJ) adenocarcinoma who had not previously received systemic therapy for metastatic disease. PD-L1 status was determined using the PD-L1 IHC 22C3 pharmDx assay. Patients with an autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by PD-L1 expression (CPS ≥1 or CPS <1), chemotherapy regimen (5-FU plus cisplatin [FP] or capecitabine plus oxaliplatin [CAPOX]), and geographic region (Europe/Israel/North America/Australia, Asia, or Rest of the World). Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg, trastuzumab 8 mg/kg on first infusion and 6 mg/kg in subsequent cycles, followed by investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 for up to 6 cycles and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 up to 6-8 cycles and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
- Placebo, trastuzumab 8 mg/kg on first infusion and 6 mg/kg in subsequent cycles, followed by investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 for up to 6 cycles and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 up to 6-8 cycles and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
All study medications, except oral capecitabine, were administered as an intravenous infusion every 3-week cycle. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by BICR, unacceptable toxicity, or a maximum of 24 months. The major outcome measures assessed were PFS by BICR using RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, and OS.
Additional outcome measures included ORR and DoR, based on BICR using RECIST 1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among the 698 patients randomized, 594 (85%) had tumors that expressed PD-L1 with a CPS ≥1. PD-L1 status was determined using the PD-L1 IHC 22C3 pharmDx assay. The population characteristics of these 594 patients were: median age of 63 years (range: 19 to 85), 43% age 65 or older; 80% male; 63% White, 33% Asian, and 0.7% Black; 42% ECOG PS of 0 and 58% ECOG PS of 1. Ninety-eight percent of patients had metastatic disease (Stage IV) and 2% had locally advanced unresectable disease. Ninety-five percent (n=562) had tumors that were not MSI-H, 1% (n=8) had tumors that were MSI-H, and in 4% (n=24) the status was not known. Eighty-five percent of patients received CAPOX.
A statistically significant improvement in OS and PFS was demonstrated in patients randomized to intravenous pembrolizumab in combination with trastuzumab and chemotherapy compared with placebo in combination with trastuzumab and chemotherapy; however, an exploratory analysis of OS in the PD-L1 CPS <1 population showed a HR of 1.10 (95% CI: 0.72, 1.68), indicating that the improvement in the ITT population was primarily attributed to the results observed in the subgroup of patients with PD-L1 CPS ≥1.
Efficacy results at the final analysis for the subgroup of patients whose tumors expressed PD-L1 with a CPS ≥1 are summarized in Table 82 and Figure 22.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks Trastuzumab Fluoropyrimidine and Platinum Chemotherapy n=298 |
Placebo Trastuzumab Fluoropyrimidine and Platinum Chemotherapy n=296 |
|---|---|---|
| + Denotes ongoing response | ||
| OS | ||
| Number (%) of patients with event | 226 (76%) | 244 (82%) |
| Median in months Based on Kaplan-Meier estimation (95% CI) |
20.1 (17.9, 22.9) | 15.7 (13.5, 18.5) |
| Hazard ratio Based on the unstratified Cox proportional hazard model (95% CI) |
0.79 (0.66, 0.95) | |
| PFS | ||
| Number (%) of patients with event | 221 (74%) | 226 (76%) |
| Median in months (95% CI) | 10.9 (8.5, 12.5) | 7.3 (6.8, 8.4) |
| Hazard ratio (95% CI) | 0.72 (0.60, 0.87) | |
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
73% (68, 78) | 58% (53, 64) |
| Complete response rate | 17% | 10% |
| Partial response rate | 56% | 48% |
| Duration of Response | n=218 | n=173 |
| Median in months (95% CI) Range in months |
11.3 (9.9, 13.7) 1.1+, 60.8+ |
9.6 (7.1, 11.2) 1.4+, 60.5+ |
|
|
First-line Treatment of Locally Unresectable or Metastatic HER2-Negative Gastric or Gastroesophageal Junction Adenocarcinoma for Tumors Expressing PD-L1 (CPS ≥1)
The efficacy of intravenous pembrolizumab in combination with fluoropyrimidine- and platinum-containing chemotherapy was investigated in KEYNOTE-859 (NCT03675737), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 1579 patients with HER2-negative advanced gastric or GEJ adenocarcinoma who had not previously received systemic therapy for metastatic disease. Patients with an autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by PD-L1 expression (CPS ≥1 or CPS <1), chemotherapy regimen (FP or CAPOX), and geographic region (Europe/Israel/North America/Australia, Asia, or Rest of the World). Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg, investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
- Placebo, investigator’s choice of combination chemotherapy of cisplatin 80 mg/m2 and 5-FU 800 mg/m2/day for 5 days (FP) or oxaliplatin 130 mg/m2 and capecitabine 1000 mg/m2 bid for 14 days (CAPOX).
All study medications, except oral capecitabine, were administered as an intravenous infusion every 3-week cycle. Platinum agents could be administered for 6 or more cycles following local guidelines. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by BICR, unacceptable toxicity, or a maximum of 24 months. The major efficacy outcome measure was OS. Additional secondary efficacy outcome measures included PFS, ORR, and DoR as assessed by BICR using RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among 1,579 patients, 1,235 (78%) had tumors expressing PD-L1 CPS ≥ 1. The population characteristics in patients with PD-L1 CPS ≥ 1 expressing tumors were: median age of 62 years (range: 24 to 86), 40% age 65 or older; 70% male and 30% female; 55% White, 33% Asian, 4.6% Multiple, 4.3% American Indian or Alaskan Native, 1.3% Black, and 0.2% Native Hawaiian or other Pacific Islander; 76% Not Hispanic or Latino and 21% Hispanic or Latino; 37% ECOG PS of 0 and 63% ECOG PS of 1. Ninety-six percent of patients had metastatic disease (Stage IV) and 3% had locally advanced unresectable disease. Five percent (n=66) had tumors that were MSI-H. Eighty-six percent of patients received CAPOX.
A statistically significant improvement in OS, PFS, and ORR was demonstrated in patients randomized to intravenous pembrolizumab in combination with chemotherapy compared with placebo in combination with chemotherapy at the time of a pre-specified interim analysis of OS; however, an exploratory analysis of OS in the PD-L1 CPS <1 population showed a HR of 0.92 (95% CI 0.73, 1.17) indicating that the improvement in the ITT population was primarily attributed to the results observed in the subgroup of patients with PD-L1 CPS ≥1. Efficacy results for patients whose tumors expressed PD-L1 CPS ≥1 and CPS ≥10 are summarized in Table 83 and Figures 23 and 24.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and FP or CAPOX n=618 |
Placebo and FP or CAPOX n=617 |
Intravenous Pembrolizumab 200 mg every 3 weeks and FP or CAPOX n=279 |
Placebo and FP or CAPOX n=272 |
|---|---|---|---|---|
| CPS ≥1 | CPS ≥10 | |||
| + Denotes ongoing response | ||||
| OS | ||||
| Number (%) of patients with event | 464 (75) | 526 (85) | 188 (67) | 226 (83) |
| Median in months (95% CI) | 13.0 (11.6, 14.2) | 11.4 (10.5, 12.0) | 15.7 (13.8, 19.3) | 11.8 (10.3, 12.7) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.74 (0.65, 0.84) | 0.65 (0.53, 0.79) | ||
| p-Value (stratified log-rank) One-sided p-Value based on stratified log-rank test
|
<0.0001 | <0.0001 | ||
| PFS | ||||
| Number (%) of patients with event | 443 (72%) | 483 (78%) | 190 (68) | 210 (77) |
| Median in months (95% CI) | 6.9 (6.0, 7.2) | 5.6 (5.4, 5.7) | 8.1 (6.8, 8.5) | 5.6 (5.4, 6.7) |
| Hazard ratio (95% CI) | 0.72 (0.63, 0.82) | 0.62 (0.51, 0.76) | ||
| p-Value (stratified log-rank) | <0.0001 | <0.0001 | ||
| Objective Response Rate | ||||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
52% (48, 56) | 43% (39, 47) | 61% (55, 66) | 43% (37, 49) |
| Complete response rate | 10% | 6% | 13% | 5% |
| Partial response rate | 42% | 37% | 48% | 38% |
| p-Value One-sided p-Value based on stratified Miettinen & Nurminen method
|
0.0004 | <0.0001 | ||
| Duration of Response | n=322 | n=263 | n=169 | n=117 |
| Median in months Based on Kaplan-Meier estimates (95% CI) |
8.3 (7.0, 10.9) | 5.6 (5.4, 6.9) | 10.9 (8.0, 13.8) | 5.8 (5.3, 7.0) |
| Range in months | 1.2+, 41.5+ | 1.3+, 34.2+ | 1.2+, 41.5+ | 1.4+, 31.2+ |
|
|
|
|
An exploratory analysis of OS in the 74 patients with MSI-H tumors irrespective of PD-L1 status showed a HR of 0.34 (95% CI: 0.18, 0.66).
4 Contraindications (4 CONTRAINDICATIONS)
KEYTRUDA QLEX is contraindicated in patients with known hypersensitivity to berahyaluronidase alfa, hyaluronidase or to any of its excipients.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling.
- Severe and fatal immune-mediated adverse reactions [see Warnings and Precautions (5.1)].
- Hypersensitivity and Administration-Related Reactions [see Warnings and Precautions (5.2)].
1.10 Cervical Cancer
KEYTRUDA QLEX, in combination with chemoradiotherapy (CRT), is indicated for the treatment of adult patients with locally advanced cervical cancer involving the lower third of the vagina, with or without extension to pelvic sidewall, or hydronephrosis/non-functioning kidney, or spread to adjacent pelvic organs (FIGO 2014 Stage III-IVA).
KEYTRUDA QLEX, in combination with chemotherapy, with or without bevacizumab, is indicated for the treatment of adult patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
14.20 Ovarian Cancer
The efficacy of intravenous pembrolizumab in combination with paclitaxel, with or without bevacizumab, was evaluated in KEYNOTE-B96 (NCT05116189), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 643 patients with platinum-resistant, epithelial ovarian, fallopian tube, or primary peritoneal carcinoma who received one or two prior lines of systemic therapy for ovarian carcinoma. Patients must have received at least one line of platinum-based chemotherapy for ovarian cancer with radiographic evidence of disease progression within 6 months after the last dose. Prior therapy with an anti-PD-1/PD-L1 inhibitor, PARP inhibitor, or bevacizumab was permitted. Patients were enrolled regardless of PD-L1 tumor expression status. Patients with autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by investigator decision to use bevacizumab, geographic region (U.S. or European Union or Rest of World), and PD-L1 status according to the PD-L1 IHC 22C3 pharmDx assay (CPS <1 or CPS 1 to <10 or CPS ≥10). Patients were randomized (1:1) to one of the two treatment groups:
- Intravenous pembrolizumab 400 mg every 6 weeks plus paclitaxel 80 mg/m2 with or without bevacizumab 10 mg/kg
- Placebo every 6 weeks plus paclitaxel 80 mg/m2 with or without bevacizumab 10 mg/kg
All study medications were administered as an intravenous infusion. Intravenous pembrolizumab 400 mg or placebo were administered on Day 1 of each 6-week treatment cycle and paclitaxel 80 mg/m2 was administered on Days 1, 8, and 15 of each 3-week treatment cycle. The option to use bevacizumab was by investigator choice prior to randomization. Bevacizumab 10 mg/kg was administered on Day 1 of a 2-week treatment cycle. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease, unacceptable toxicity, or a maximum of 24 months. Administration of intravenous pembrolizumab was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed every 9 weeks for the first year, followed by every 12 weeks thereafter. The major efficacy outcome measure was PFS as assessed by investigator according to RECIST v1.1. An additional efficacy outcome measure was OS.
Among the 643 patients randomized, 466 patients (72%) had tumors expressing PD-L1 with a CPS ≥1. The population characteristics of these 466 patients were: median age of 62 years (range: 37 to 85), 38% age 65 or older; 67% White, 20% Asian, 8% Missing, 3% Multiple, 2% Black, 0.4% Native Hawaiian or other Pacific Islander; 13% Hispanic or Latino; 55% and 44% ECOG performance status of 0 or 1, respectively; 73% received bevacizumab as study treatment; 36% of patients had received one prior line and 64% had received two prior lines of therapy; prior systemic therapy included: 46% with bevacizumab; 39% with a PARP inhibitor, or 3% with an anti-PD-1/PD-L1 inhibitor. The platinum-free interval following the most recent line of therapy was <3 months in 47% of patients, and 3 to 6 months in 53% of patients.
The study demonstrated statistically significant improvement in PFS and OS for patients randomized to intravenous pembrolizumab in combination with paclitaxel with or without bevacizumab compared to placebo in combination with paclitaxel with or without bevacizumab in patients whose tumors expressed PD-L1 CPS ≥1. Efficacy results are summarized in Table 104 and Figures 46 and 47.
| Endpoint | Intravenous pembrolizumab 400 mg every 6 weeks plus paclitaxel with or without bevacizumab n=234 |
Placebo plus paclitaxel with or without bevacizumab n=232 |
|---|---|---|
| PFS | ||
| Number of patients with event (%) | 162 (69) | 180 (78) |
| Median in months (95% CI) | 8.3 (7.0, 9.4) | 7.2 (6.2, 8.1) |
| Hazard ratio (95% CI) | 0.72 (0.58, 0.89) | |
| p-Value | 0.0014 Based on stratified log-rank test (p-Value [one-sided] is compared to an alpha boundary of 0.0116)
|
|
| OS | ||
| Number of patients with event (%) | 157 (67) | 175 (75) |
| Median in months (95% CI) | 18.2 (15.3, 21.0) | 14.0 (12.5, 16.1) |
| Hazard ratio (95% CI) | 0.76 (0.61, 0.94) | |
| p-Value | 0.0053 Based on stratified log-rank test (p-Value [one-sided] is compared to an alpha boundary of 0.0083)
|
| Figure 46: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-B96 (CPS ≥1) |
|
|
| Figure 47: Kaplan-Meier Curve for Overall Survival in KEYNOTE-B96 (CPS ≥1) |
|
|
1.5 Urothelial Cancer
KEYTRUDA QLEX, in combination with enfortumab vedotin, is indicated for the treatment of adult patients with locally advanced or metastatic urothelial cancer.
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma:
- who are not eligible for any platinum-containing chemotherapy, or
- who have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy.
KEYTRUDA QLEX, in combination with enfortumab vedotin, as neoadjuvant treatment and then continued after cystectomy as adjuvant treatment, is indicated for the treatment of adult patients with muscle invasive bladder cancer (MIBC) who are ineligible for cisplatin-containing chemotherapy.
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy.
1.9 Esophageal Cancer
KEYTRUDA QLEX is indicated for the treatment of adult patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either:
- in combination with platinum- and fluoropyrimidine-based chemotherapy for patients with tumors that express PD-L1 (CPS ≥ 1) [see Dosage and Administration (2.1)], or
- as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
12.2 Pharmacodynamics
There are no clinically significant exposure-response relationships for efficacy or safety for intravenous pembrolizumab across the approved dosing regimens, regardless of cancer type. The exposures from subcutaneous KEYTRUDA QLEX doses of 395 mg/4,800 units every 3 weeks or 790 mg/9,600 units every 6 weeks are within the range of exposures from intravenous pembrolizumab doses.
12.3 Pharmacokinetics
Pembrolizumab pharmacokinetics were characterized at Cycle 1 and at steady state in patients with advanced solid tumors at the approved recommended dosages and are presented as mean (CV%) unless otherwise specified.
When comparing pembrolizumab exposure following subcutaneous administration every 6 weeks to that of intravenous administration every 6 weeks in Study MK-3475A-D77 [see Clinical Studies (14.1)], the geometric mean ratio (GMR) for Cycle 1 AUC0-6wks was 1.14 (96% CI: 1.06, 1.22) and Cycle 3 Ctrough (i.e., steady state) was 1.67 (94% CI: 1.52, 1.84).
Pembrolizumab steady state was reached by 16 weeks. At steady state following subcutaneous administration, the mean pembrolizumab AUC0-6wks was 2,798 mcg•day/mL for the every 6 week dosing and pembrolizumab AUC0-3wks was 1,343 mcg•day/mL for the every 3 week dosing. Pembrolizumab Ctrough was 39 mcg/mL for the every 6 week dosing and 49 mcg/mL for the every 3 week dosing.
The systemic accumulation ratio was 1.6-fold following administration of KEYTRUDA QLEX 790 mg/9,600 units every 6 weeks and 2.5-fold following administration of KEYTRUDA QLEX 395 mg/4,800 units every 3 weeks.
Absorption
Pembrolizumab bioavailability (CV%) is approximately 60% (14%). Peak concentrations occurred by approximately 4 days.
Distribution
The volume of distribution is 6 L.
Elimination
Pembrolizumab clearance decreases over time, resulting in a steady state clearance (CV%) of - 195 mL/day (40%); this decrease in clearance with time is not considered clinically significant. The terminal half-life is 22 days.
Specific Populations
No clinically significant differences in the pharmacokinetics of pembrolizumab were observed based on age (37 to 87 years), race (63% White, 28% Asian, 3% Black), sex, body weight (37 to 144 kg), tumor type, injection site (thigh or abdomen), estimated glomerular filtration rate (eGFR) ≥15 mL/min/1.73 m2, and mild to moderate hepatic impairment (total bilirubin ≤3 times ULN and any AST). The effect of severe hepatic impairment (total bilirubin >3 times ULN and any AST) on pembrolizumab pharmacokinetics is unknown.
Pediatric Patients
Pembrolizumab exposures in pediatric patients 12 years and older who weigh greater than 40 kg are predicted to be within range of those observed in adult patients at the same dosage.
14.11 Cervical Cancer
FIGO 2014 Stage III-IVA Cervical Cancer with Chemoradiotherapy
The efficacy of intravenous pembrolizumab in combination with CRT (cisplatin and external beam radiation therapy [EBRT] followed by brachytherapy [BT]) was investigated in KEYNOTE-A18 (NCT04221945), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 1060 patients with cervical cancer who had not previously received any definitive surgery, radiation, or systemic therapy for cervical cancer. There were 599 patients with FIGO 2014 Stage III-IVA disease (tumor involves the lower third of the vagina or the pelvic sidewall, or there is hydronephrosis/non-functioning kidney or spread to adjacent pelvic organs, all without spread to distant organs), and 459 patients with FIGO 2014 Stage IB2-IIB disease (clinical lesion >4 cm confined to the cervix, or clinical lesion of any size with extension beyond the uterus, but which has not extended to the pelvic wall or lower third of the vagina) with positive nodes. Two patients had FIGO 2014 Stage IVB disease. Randomization was stratified by planned type of EBRT (Intensity-modulated radiation therapy [IMRT] or volumetric modulated arc therapy [VMAT] vs. non-IMRT and non-VMAT), stage at screening of cervical cancer (FIGO 2014 Stage IB2-IIB vs. FIGO 2014 Stage III-IVA), and planned total radiotherapy dose (EBRT + brachytherapy dose of <70 Gy vs. ≥70 Gy as per equivalent dose [EQD2]).
Patients were randomized (1:1) to one of two treatment arms:
- Intravenous pembrolizumab 200 mg IV every 3 weeks (5 cycles) concurrent with cisplatin 40 mg/m2 IV weekly (5 cycles, an optional sixth infusion could be administered per local practice), and radiotherapy (EBRT followed by BT), followed by intravenous pembrolizumab 400 mg IV every 6 weeks (15 cycles)
- Placebo IV every 3 weeks (5 cycles) concurrent with cisplatin 40 mg/m2 IV weekly (5 cycles, an optional sixth infusion could be administered per local practice), and radiotherapy (EBRT followed by BT), followed by placebo IV every 6 weeks (15 cycles)
Treatment continued until RECIST v1.1-defined progression of disease as determined by investigator or unacceptable toxicity.
Assessment of tumor status was performed every 12 weeks from completion of CRT for the first two years, followed by every 24 weeks in year 3, and then annually. The major efficacy outcome measures were PFS as assessed by investigator according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, or histopathologic confirmation, and OS.
Among the 599 patients with FIGO 2014 Stage III-IVA disease, the baseline characteristics were: median age of 52 years (range: 22 to 87), 17% age 65 or older; 36% White, 34% Asian, 2% Black; 38% Hispanic or Latino; 68% ECOG PS 0 and 32% ECOG PS 1; 93% with CPS ≥1; 71% had positive pelvic and/or positive para-aortic lymph node(s) and 29% had neither positive pelvic nor para-aortic lymph node(s); 83% had squamous cell carcinoma and 17% had non-squamous histology. Regarding radiation, 86% of patients received IMRT or VMAT EBRT, and the median EQD2 dose was 87 Gy (range: 7 to 114).
The trial demonstrated statistically significant improvements in PFS and OS in the ITT population. Exploratory analyses of PFS and OS by the stratification factor of FIGO 2014 stage showed that the improvement in the ITT population was primarily attributed to the results seen in the patients with FIGO 2014 Stage III-IVA disease. Table 86 and Figures 28 and 29 summarize the results in exploratory subgroup analyses of 599 patients with FIGO 2014 Stage III-IVA disease.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and 400 mg every 6 weeks with CRT n=295 |
Placebo with CRT n=304 |
|---|---|---|
| CRT = Chemoradiotherapy NR = not reached |
||
|
OS Results at the time of pre-specified final analysis for OS
|
||
| Number of patients with event (%) | 61 (21) | 90 (30) |
| Hazard ratio Based on the unstratified Cox proportional hazard model (95% CI) |
0.65 (0.47, 0.90) | |
|
PFS by Investigator
Results at the time of first pre-specified interim analysis for PFS
|
||
| Number of patients with event (%) | 61 (21) | 94 (31) |
| Median in months (95% CI) | NR (NR, NR) | NR (18.8, NR) |
| 12-month PFS rate (95% CI) | 81% (75, 85) | 70% (64, 76) |
| Hazard ratio (95% CI) | 0.59 (0.43, 0.81) |
|
|
|
|
Persistent, Recurrent, or Metastatic Cervical Cancer for Tumors Expressing PD-L1 (CPS ≥1)
The efficacy of intravenous pembrolizumab in combination with paclitaxel and cisplatin or paclitaxel and carboplatin, with or without bevacizumab, was investigated in KEYNOTE-826 (NCT03635567), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 617 patients with persistent, recurrent, or first-line metastatic cervical cancer who had not been treated with chemotherapy except when used concurrently as a radio-sensitizing agent. Patients were enrolled regardless of tumor PD-L1 expression status. Patients with autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by metastatic status at initial diagnosis, investigator decision to use bevacizumab, and PD-L1 status (CPS <1 vs. CPS 1 to <10 vs. CPS ≥10). Patients were randomized (1:1) to one of the two treatment groups:
- Treatment Group 1: Intravenous pembrolizumab 200 mg plus chemotherapy with or without bevacizumab
- Treatment Group 2: Placebo plus chemotherapy with or without bevacizumab
The investigator selected one of the following four treatment regimens prior to randomization:
- Paclitaxel 175 mg/m2 + cisplatin 50 mg/m2
- Paclitaxel 175 mg/m2 + cisplatin 50 mg/m2 + bevacizumab 15 mg/kg
- Paclitaxel 175 mg/m2 + carboplatin AUC 5 mg/mL/min
- Paclitaxel 175 mg/m2 + carboplatin AUC 5 mg/mL/min + bevacizumab 15 mg/kg
All study medications were administered as an intravenous infusion. All study treatments were administered on Day 1 of each 3-week treatment cycle. Cisplatin could be administered on Day 2 of each 3-week treatment cycle. Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease, unacceptable toxicity, or a maximum of 24 months. Administration of intravenous pembrolizumab was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed every 9 weeks for the first year, followed by every 12 weeks thereafter. The main efficacy outcome measures were OS and PFS as assessed by investigator according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ. Additional efficacy outcome measures were ORR and DoR, according to RECIST v1.1, as assessed by investigator.
Of the 617 enrolled patients, 548 patients (89%) had tumors expressing PD-L1 with a CPS ≥1. Among these 548 enrolled patients with tumors expressing PD-L1, 273 patients were randomized to intravenous pembrolizumab in combination with chemotherapy with or without bevacizumab, and 275 patients were randomized to placebo in combination with chemotherapy with or without bevacizumab. Sixty-three percent of the 548 patients received bevacizumab as part of study treatment. The baseline characteristics of the 548 patients were: median age of 51 years (range: 22 to 82), 16% age 65 or older; 59% White, 18% Asian, 6% American Indian or Alaska Native, and 1% Black; 37% Hispanic or Latino; 56% ECOG performance status 0 and 43% ECOG performance status 1. Seventy-five percent had squamous cell carcinoma, 21% adenocarcinoma, and 5% adenosquamous histology, and 32% of patients had metastatic disease at diagnosis. At study entry, 21% of patients had metastatic disease only and 79% had persistent or recurrent disease with or without distant metastases, of whom 39% had received prior chemoradiation only and 17% had received prior chemoradiation plus surgery.
A statistically significant improvement in OS and PFS was demonstrated in patients randomized to receive intravenous pembrolizumab compared with patients randomized to receive placebo. An updated OS analysis was conducted at the time of final analysis when 354 deaths in the CPS ≥1 population were observed. Table 87 and Figure 30 summarize the key efficacy measures for KEYNOTE-826 for patients with tumors expressing PD-L1 (CPS ≥1).
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and chemotherapy Chemotherapy (paclitaxel and cisplatin or paclitaxel and carboplatin) with or without bevacizumabn=273 |
Placebo and chemotherapy with or without bevacizumab n=275 |
|---|---|---|
| + Denotes ongoing response NR = not reached |
||
| OS | ||
| Number of patients with event (%) | 118 (43.2) | 154 (56.0) |
| Median in months (95% CI) | NR (19.8, NR) | 16.3 (14.5, 19.4) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.64 (0.50, 0.81) | |
| p-Value p-Value (one-sided) is compared with the allocated alpha of 0.0055 for this interim analysis (with 72% of the planned number of events for final analysis)
|
0.0001 | |
| Updated OS | ||
| Number of patients with event (%) | 153 (56.0%) | 201 (73.1%) |
| Median in months (95% CI) | 28.6 (22.1, 38.0) | 16.5 (14.5, 20.0) |
| Hazard ratio (95% CI) | 0.60 (0.49, 0.74) | |
| PFS | ||
| Number of patients with event (%) | 157 (57.5) | 198 (72.0) |
| Median in months (95% CI) | 10.4 (9.7, 12.3) | 8.2 (6.3, 8.5) |
| Hazard ratio (95% CI) | 0.62 (0.50, 0.77) | |
| p-Value p-Value (one-sided) is compared with the allocated alpha of 0.0014 for this interim analysis (with 82% of the planned number of events for final analysis)
|
< 0.0001 | |
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
68% (62, 74) | 50% (44, 56) |
| Complete response rate | 23% | 13% |
| Partial response rate | 45% | 37% |
| Duration of Response | ||
| Median in months (range) | 18.0 (1.3+, 24.2+) | 10.4 (1.5+, 22.0+) |
|
|
Previously Treated Recurrent or Metastatic Cervical Cancer for Tumors Expressing PD-L1 (CPS≥ 1)
The efficacy of intravenous pembrolizumab was investigated in 98 patients with recurrent or metastatic cervical cancer enrolled in a single cohort (Cohort E) in KEYNOTE-158 (NCT02628067), a multicenter, non-randomized, open-label, multi-cohort trial. The trial excluded patients with autoimmune disease or a medical condition that required immunosuppression. Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression. Patients with initial radiographic disease progression could receive additional doses of treatment during confirmation of progression unless disease progression was symptomatic, was rapidly progressive, required urgent intervention, or occurred with a decline in performance status. Patients without disease progression could be treated for up to 24 months. Assessment of tumor status was performed every 9 weeks for the first 12 months, and every 12 weeks thereafter. The major efficacy outcome measures were ORR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, as assessed by BICR, and DoR.
Among the 98 patients in Cohort E, 77 (79%) had tumors that expressed PD-L1 with a CPS ≥ 1 and received at least one line of chemotherapy in the metastatic setting. PD-L1 status was determined using the IHC 22C3 pharmDx assay. The baseline characteristics of these 77 patients were: median age of 45 years (range: 27 to 75); 81% White, 14% Asian, and 3% Black; 32% ECOG PS of 0 and 68% ECOG PS of 1; 92% had squamous cell carcinoma, 6% adenocarcinoma, and 1% adenosquamous histology; 95% had M1 disease and 5% had recurrent disease; and 35% had one and 65% had two or more prior lines of therapy in the recurrent or metastatic setting.
No responses were observed in patients whose tumors did not have PD-L1 expression (CPS <1). Efficacy results are summarized in Table 88 for patients with PD-L1 expression (CPS ≥1).
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=77 Median follow-up time of 11.7 months (range 0.6 to 22.7 months)
|
|---|---|
| + Denotes ongoing response NR = not reached |
|
| Objective Response Rate | |
| ORR (95% CI) | 14.3% (7.4, 24.1) |
| Complete response rate | 2.6% |
| Partial response rate | 11.7% |
| Duration of Response | |
| Median in months (range) | NR (4.1, 18.6+) Based on patients (n=11) with a response by independent review
|
| % with duration ≥6 months | 91% |
2.1 Patient Selection
See information on FDA-authorized tests for intravenous pembrolizumab. Information on FDA-authorized tests for patient selection is available at: http://www.fda.gov/CompanionDiagnostics.
Patient Selection for Single-Agent Treatment
Select patients for treatment with KEYTRUDA QLEX as a single agent based on the presence of positive PD-L1 expression in:
- Stage III NSCLC who are not candidates for surgical resection or definitive chemoradiation [see Clinical Studies (14.3)].
- metastatic NSCLC [see Clinical Studies (14.3)].
- first-line treatment of metastatic or unresectable, recurrent HNSCC [see Clinical Studies (14.5)].
- previously treated recurrent locally advanced or metastatic esophageal cancer [see Clinical Studies (14.10)].
- recurrent or metastatic cervical cancer with disease progression on or after chemotherapy [see Clinical Studies (14.11)].
For the MSI-H/dMMR indications, select patients for treatment with KEYTRUDA QLEX as a single agent based on MSI-H/dMMR status in tumor specimens [see Clinical Studies (14.7, 14.8)].
For the TMB-H indication, select patients for treatment with KEYTRUDA QLEX as a single agent based on TMB-H status in tumor specimens [see Clinical Studies (14.17)].
Because subclonal dMMR mutations and microsatellite instability may arise in high-grade gliomas during temozolomide therapy, it is recommended to test for TMB-H, MSI-H, and dMMR in the primary tumor specimens obtained prior to initiation of temozolomide chemotherapy in patients with high-grade gliomas.
Additional Patient Selection Information for MSI-H or dMMR in Patients with non-CRC Solid Tumors
Due to discordance between local tests and FDA-authorized tests, confirmation of MSI-H or dMMR status is recommended by an FDA-authorized test in patients with MSI-H or dMMR solid tumors, if feasible. If unable to perform confirmatory MSI-H/dMMR testing, the presence of TMB ≥10 mut/Mb, as determined by an FDA-authorized test, may be used to select patients for treatment [see Clinical Studies (14.7)].
Patient Selection for Combination Therapy
For use of KEYTRUDA QLEX as a single agent as neoadjuvant treatment, then in combination with radiotherapy (RT) with or without chemotherapy then continued as a single agent as adjuvant treatment, select patients based on presence of positive PD-L1 expression (CPS ≥1) in resectable locally advanced HNSCC [see Clinical Studies (14.5)].
For use of KEYTRUDA QLEX in combination with chemotherapy, select patients based on the presence of positive PD-L1 expression (CPS ≥1) in locally advanced unresectable or metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma, and esophageal or gastroesophageal junction (GEJ) carcinoma [see Clinical Studies (14.9), (14.10)].
- An FDA-authorized test for the detection of PD-L1 for the selection of patients with PD-L1 (CPS ≥1) expression in esophageal carcinoma in combination with platinum- and fluoropyrimidine-based chemotherapy is not available.
For use of KEYTRUDA QLEX in combination with chemotherapy, with or without bevacizumab, select patients based on the presence of positive PD-L1 expression in persistent, recurrent, or metastatic cervical cancer [see Clinical Studies (14.11)].
For the pMMR/not MSI-H advanced endometrial carcinoma indication, select patients for treatment with KEYTRUDA QLEX in combination with lenvatinib based on MMR or MSI status in tumor specimens [see Clinical Studies (14.16)].
For use of KEYTRUDA QLEX in combination with chemotherapy, select patients based on the presence of positive PD-L1 expression in locally recurrent unresectable or metastatic TNBC [see Clinical Studies (14.19)].
For use of KEYTRUDA QLEX in combination with paclitaxel, with or without bevacizumab, select patients based on the presence of positive PD-L1 expression (CPS ≥1) in platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma [see Clinical Studies (14.20)].
2.3 Recommended Dosage
The recommended dosages of KEYTRUDA QLEX are presented in Table 1.
- Every 3-week dosing (395 mg pembrolizumab and 4,800 units berahyaluronidase alfa): inject 2.4 mL subcutaneously over 1 minute.
- Every 6-week dosing (790 mg pembrolizumab and 9,600 units berahyaluronidase alfa): inject 4.8 mL subcutaneously over 2 minutes.
| Indication |
Recommended Dosage of KEYTRUDA QLEX |
Duration/Timing of Treatment |
|---|---|---|
| Monotherapy | ||
| Adult patients with unresectable or metastatic melanoma |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease progression or unacceptable toxicity |
| Adjuvant treatment of adult patients with melanoma, NSCLC, or RCC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease recurrence, unacceptable toxicity, or up to 12 months |
| Adult patients with NSCLC, HNSCC, locally advanced or metastatic Urothelial Carcinoma, MSI-H or dMMR Cancer, MSI-H or dMMR CRC, MSI-H or dMMR Endometrial Carcinoma, Esophageal Cancer, Cervical Cancer, HCC, MCC, TMB-H Cancer, or cSCC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with high-risk BCG- unresponsive NMIBC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until persistent or recurrent high-risk NMIBC, disease progression, unacceptable toxicity, or up to 24 months |
| Pediatric patients The recommended dosage for melanoma, MSI-H or dMMR cancer, MCC and TMB-H cancer has not been established in pediatric patients 12 years and older who weigh 40 kg or less [see Use in Specific Populations (8.4)]. (12 years and olderwho weigh greater than 40 kg) with MSI-H or dMMR Cancer, MCC, or TMB- H Cancer |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Pediatric patients (12 years and older who weigh greater than 40 kg) for adjuvant treatment of melanoma |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks |
Until disease recurrence, unacceptable toxicity, or up to 12 months |
|
Combination Therapy
Refer to the Prescribing Information for the agents administered in combination with KEYTRUDA QLEX for recommended dosing information, as appropriate.
|
||
| Adult patients with resectable NSCLC | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Neoadjuvant treatment in combination with chemotherapy for 12 weeks or until disease progression that precludes definitive surgery or unacceptable toxicity, followed by adjuvant treatment with KEYTRUDA QLEX as a single agent after surgery for 39 weeks or until disease recurrence or unacceptable toxicity |
| Adult patients with NSCLC, MPM, HNSCC, HER2-negative Gastric Cancer, Esophageal Cancer, or BTC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with locally advanced or metastatic urothelial cancer |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX after enfortumab vedotin when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with locally advanced HNSCC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to cisplatin when given on the same day. |
Neoadjuvant:
disease recurrence or unacceptable toxicity or up to one year |
| Adult patients with MIBC | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX after enfortumab vedotin when given on the same day. |
Neoadjuvant:
|
| Adult patients with HER2-positive Gastric Cancer |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to trastuzumab and chemotherapy when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with Cervical Cancer | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemoradiotherapy or prior to chemotherapy with or without bevacizumab when given on the same day. |
Until disease progression, unacceptable toxicity, or for KEYTRUDA QLEX, up to 24 months |
| Adult patients with RCC | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX in combination with axitinib 5 mg orally twice daily When axitinib is used in combination with KEYTRUDA QLEX, dose escalation of axitinib above the initial 5 mg dose may be considered at intervals of six weeks or longer.
or Administer KEYTRUDA QLEX in combination with lenvatinib 20 mg orally once daily. |
Until disease progression, unacceptable toxicity, or for KEYTRUDA QLEX, up to 24 months |
| Adult patients with Endometrial Carcinoma |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to carboplatin and paclitaxel when given on the same day. or Administer KEYTRUDA QLEX in combination with lenvatinib 20 mg orally once daily. |
Until disease progression, unacceptable toxicity, or for KEYTRUDA QLEX, up to 24 months |
| Adult patients with high-risk early-stage TNBC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Neoadjuvant treatment in combination with chemotherapy for 24 weeks (8 doses of 395 mg/4,800 units every 3 weeks or 4 doses of 790 mg/9,600 units every 6 weeks) or until disease progression or unacceptable toxicity, followed by adjuvant treatment with KEYTRUDA QLEX as a single agent for up to 27 weeks (9 doses of 395 mg/4,800 units every 3 weeks or 5 doses of 790 mg/9,600 units every 6 weeks) or until disease recurrence or unacceptable toxicity Patients who experience disease progression or unacceptable toxicity related to KEYTRUDA QLEX with neoadjuvant treatment in combination with chemotherapy should not receive adjuvant single agent KEYTRUDA QLEX.
|
| Adult patients with locally recurrent unresectable or metastatic TNBC |
395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to chemotherapy when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
| Adult patients with Ovarian Cancer | 395 mg/4,800 units every 3 weeks
or 790 mg/9,600 units every 6 weeks Administer KEYTRUDA QLEX prior to paclitaxel with or without bevacizumab when given on the same day. |
Until disease progression, unacceptable toxicity, or up to 24 months |
1 Indications and Usage (1 INDICATIONS AND USAGE)
KEYTRUDA QLEX is a combination of pembrolizumab, a programmed death receptor-1 (PD-1)-blocking antibody, and berahyaluronidase alfa, an endoglycosidase, indicated:
Melanoma
- for the treatment of adult patients with unresectable or metastatic melanoma. (1.1)
- for the adjuvant treatment of adult and pediatric patients 12 years and older with Stage IIB, IIC, or III melanoma following complete resection. (1.1)
Non-Small Cell Lung Cancer (NSCLC)
- in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with metastatic nonsquamous NSCLC, with no EGFR or ALK genomic tumor aberrations. (1.2)
- in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, as first-line treatment of adult patients with metastatic squamous NSCLC. (1.2)
- as a single agent for the first-line treatment of adult patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-authorized test, with no EGFR or ALK genomic tumor aberrations, and is:
- as a single agent for the treatment of adult patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-authorized test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA QLEX. (1.2, 2.1)
- for the treatment of adult patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. (1.2)
- as a single agent, for adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC. (1.2)
Malignant Pleural Mesothelioma (MPM)
- in combination with pemetrexed and platinum chemotherapy, as first-line treatment of adult patients with unresectable advanced or metastatic MPM. (1.3)
Head and Neck Squamous Cell Cancer (HNSCC)
- for the treatment of adult patients with resectable locally advanced HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test, as a single agent as neoadjuvant treatment, continued as adjuvant treatment in combination with radiotherapy (RT) with or without cisplatin and then as a single agent. (1.4)
- in combination with platinum and FU for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC. (1.4)
- as a single agent for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test. (1.4, 2.1)
- as a single agent for the treatment of adult patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy. (1.4)
Urothelial Cancer
- in combination with enfortumab vedotin, for the treatment of adult patients with locally advanced or metastatic urothelial cancer. (1.5)
- as a single agent for the treatment of adult patients with locally advanced or metastatic urothelial carcinoma who:
- are not eligible for any platinum-containing chemotherapy, or
- who have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. (1.5)
- in combination with enfortumab vedotin, as neoadjuvant treatment and then continued after cystectomy as adjuvant treatment of adult patients with muscle invasive bladder cancer (MIBC) who are ineligible for cisplatin-containing chemotherapy. (1.5)
- as a single agent for the treatment of adult patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy. (1.5)
Microsatellite Instability-High or Mismatch Repair Deficient Cancer
- for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options. (1.6, 2.1)
Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer (CRC)
- for the treatment of adult patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-authorized test. (1.7, 2.1)
Gastric Cancer
- in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.8)
- in combination with fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.8)
Esophageal Cancer
- for the treatment of adult patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either:
- in combination with platinum- and fluoropyrimidine-based chemotherapy for patients whose tumors express PD-L1 (CPS ≥1), or
- as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. (1.9, 2.1)
Cervical Cancer
- in combination with chemoradiotherapy, for the treatment of adult patients with locally advanced cervical cancer involving the lower third of the vagina, with or without extension to pelvic sidewall, or hydronephrosis/non-functioning kidney, or spread to adjacent pelvic organs (FIGO 2014 Stage III-IVA). (1.10)
- in combination with chemotherapy, with or without bevacizumab, for the treatment of adult patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.10, 2.1)
- as a single agent for the treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test. (1.10, 2.1)
Hepatocellular Carcinoma (HCC)
- for the treatment of adult patients with HCC secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen. (1.11)
Biliary Tract Cancer (BTC)
- in combination with gemcitabine and cisplatin, for the treatment of adult patients with locally advanced unresectable or metastatic biliary tract cancer. (1.12)
Merkel Cell Carcinoma (MCC)
- for the treatment of adult and pediatric patients 12 years and older with recurrent locally advanced or metastatic Merkel cell carcinoma. (1.13)
Renal Cell Carcinoma (RCC)
- in combination with axitinib, for the first-line treatment of adult patients with advanced RCC. (1.14)
- in combination with lenvatinib, for the first-line treatment of adult patients with advanced RCC. (1.14)
- for the adjuvant treatment of adult patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions. (1.14)
Endometrial Carcinoma
- in combination with carboplatin and paclitaxel, followed by KEYTRUDA QLEX as a single agent, for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma. (1.15)
- in combination with lenvatinib, for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) or not MSI-H as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. (1.15, 2.1)
- as a single agent, for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation. (1.15, 2.1)
Tumor Mutational Burden-High (TMB-H) Cancer
- for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.1 (1.16, 2.1)
- Limitations of Use: The safety and effectiveness of KEYTRUDA QLEX in pediatric patients 12 years and older with TMB-H central nervous system cancers have not been established.
Cutaneous Squamous Cell Carcinoma (cSCC)
- for the treatment of adult patients with recurrent or metastatic cSCC or locally advanced cSCC that is not curable by surgery or radiation. (1.17)
Triple-Negative Breast Cancer (TNBC)
- for the treatment of adult patients with high-risk early-stage TNBC in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. (1.18)
- in combination with chemotherapy, for the treatment of adult patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-authorized test. (1.18, 2.1)
Ovarian Cancer
- in combination with paclitaxel, with or without bevacizumab, is indicated for the treatment of adult patients with platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal carcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-authorized test, and who have received one or two prior systemic treatment regimens. (1.19, 2.1)
1 This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
12.1 Mechanism of Action
Binding of the PD-1 ligands, PD-L1 and PD-L2, to the PD-1 receptor found on T cells, inhibits T cell proliferation and cytokine production. Upregulation of PD-1 ligands occurs in some tumors and signaling through this pathway can contribute to inhibition of active T-cell immune surveillance of tumors. Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the anti-tumor immune response. In syngeneic mouse tumor models, blocking PD-1 activity resulted in decreased tumor growth.
In syngeneic mouse tumor models, combination treatment of a PD-1 blocking antibody and kinase inhibitor lenvatinib decreased tumor-associated macrophages, increased activated cytotoxic T cells, and reduced tumor growth compared to either treatment alone.
Berahyaluronidase alfa, an endoglycosidase, is a variant of human hyaluronidase PH20 that temporarily and locally breaks down hyaluronan. Hyaluronan is a polysaccharide found in the extracellular matrix of the subcutaneous tissue. Unlike the stable structural components of the interstitial matrix, hyaluronan has a half-life of approximately 0.5 days. Hyaluronidase increases permeability of the subcutaneous tissue by depolymerizing hyaluronan.
In the doses administered, the effects of berahyaluronidase alfa are reversible and permeability of the subcutaneous tissue is restored within 24 to 48 hours.
2.4 Dosage Modifications
No dose reduction for KEYTRUDA QLEX is recommended. In general, withhold KEYTRUDA QLEX for severe (Grade 3) immune-mediated adverse reactions. Permanently discontinue KEYTRUDA QLEX for Life-threatening (Grade 4) immune-mediated adverse reactions, recurrent severe (Grade 3) immune-mediated reactions that require systemic immunosuppressive treatment, or an inability to reduce corticosteroid dose to 10 mg or less of prednisone or equivalent per day within 12 weeks of initiating steroids.
Dosage modifications for KEYTRUDA QLEX for adverse reactions that require management different from these general guidelines are summarized in Table 2.
| Adverse Reaction | Severity Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.0
|
Dosage Modification |
|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, DRESS = Drug Rash with Eosinophilia and Systemic Symptoms, SJS = Stevens Johnson Syndrome, TEN = toxic epidermal necrolysis, ULN = upper limit normal | ||
| Immune-Mediated Adverse Reactions [see Warnings and Precautions (5.1)] | ||
| Pneumonitis | Grade 2 | Withhold Resume in patients with complete or partial resolution (Grades 0 to 1) after corticosteroid taper. Permanently discontinue if no complete or partial resolution within 12 weeks of initiating steroids or inability to reduce prednisone to 10 mg per day or less (or equivalent) within 12 weeks of initiating steroids.
|
| Grade 3 or 4 | Permanently discontinue | |
| Colitis | Grade 2 or 3 | Withhold |
| Grade 4 | Permanently discontinue | |
|
Hepatitis with no tumor involvement of the liver |
AST or ALT increases to more than 3 and up to 8 times ULN or Total bilirubin increases to more than 1.5 and up to 3 times ULN |
Withhold |
| For liver enzyme elevations in patients treated with combination therapy with axitinib, see Table 3. |
AST or ALT increases to more than 8 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue |
| Hepatitis with tumor involvement of the liver If AST and ALT are less than or equal to ULN at baseline, withhold or permanently discontinue KEYTRUDA QLEX based on recommendations for hepatitis with no liver involvement.
|
Baseline AST or ALT is more than 1 and up to 3 times ULN and increases to more than 5 and up to 10 times ULN or Baseline AST or ALT is more than 3 and up to 5 times ULN and increases to more than 8 and up to 10 times ULN |
Withhold |
| ALT or AST increases to more than 10 times ULN or Total bilirubin increases to more than 3 times ULN |
Permanently discontinue | |
| Endocrinopathies | Grade 3 or 4 | Withhold until clinically stable or permanently discontinue depending on severity |
| Nephritis with Renal Dysfunction | Grade 2 or 3 increased blood creatinine | Withhold |
| Grade 4 increased blood creatinine | Permanently discontinue | |
| Exfoliative Dermatologic Conditions | Suspected SJS, TEN, or DRESS | Withhold |
| Confirmed SJS, TEN, or DRESS | Permanently discontinue | |
| Myocarditis | Grade 2, 3, or 4 | Permanently discontinue |
| Neurological Toxicities | Grade 2 | Withhold |
| Grade 3 or 4 | Permanently discontinue | |
| Other Adverse Reactions | ||
| Hypersensitivity and Administration-Related Systemic Reactions [see Warnings and Precautions (5.2)] |
Grade 1 or 2 | Interrupt injection (if not already fully administered). If symptoms resolve, resume injection |
| Grade 3 or 4 | Permanently discontinue |
The following table represents dosage modifications that are different from those described above for KEYTRUDA QLEX or in the Full Prescribing Information for the drug administered in combination.
| Treatment | Adverse Reaction | Severity | Dosage Modification |
|---|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal | |||
| KEYTRUDA QLEX in combination with axitinib |
Liver enzyme elevations Consider corticosteroid therapy
|
ALT or AST increases to at least 3 times but less than 10 times ULN without concurrent total bilirubin at least 2 times ULN | Withhold both KEYTRUDA QLEX and axitinib until resolution to Grades 0 or 1 Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. Consider rechallenge with a single drug or sequential rechallenge with both drugs after recovery. If rechallenging with axitinib, consider dose reduction as per the axitinib Prescribing Information.
|
| ALT or AST increases to more than 3 times ULN with concurrent total bilirubin at least 2 times ULN or ALT or AST ≥10 times ULN |
Permanently discontinue both KEYTRUDA QLEX and axitinib |
Recommended Dose Modifications for Adverse Reactions for KEYTRUDA QLEX in Combination with Lenvatinib
When administering KEYTRUDA QLEX in combination with lenvatinib, modify the dosage of one or both drugs. Withhold or discontinue KEYTRUDA QLEX as shown in Table 2. Refer to lenvatinib prescribing information for additional dose modification information.
1.12 Biliary Tract Cancer
KEYTRUDA QLEX, in combination with gemcitabine and cisplatin, is indicated for the treatment of adult patients with locally advanced unresectable or metastatic biliary tract cancer (BTC).
1.14 Renal Cell Carcinoma
KEYTRUDA QLEX, in combination with axitinib, is indicated for the first-line treatment of adult patients with advanced renal cell carcinoma (RCC).
KEYTRUDA QLEX, in combination with lenvatinib, is indicated for the first-line treatment of adult patients with advanced RCC.
KEYTRUDA QLEX is indicated for the adjuvant treatment of adult patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions [see Clinical Studies (14.15)].
5.5 Embryo Fetal Toxicity (5.5 Embryo-Fetal Toxicity)
Based on its mechanism of action, KEYTRUDA QLEX can cause fetal harm when administered to a pregnant woman. Animal studies have demonstrated that inhibition of the PD-L1/PD-1 pathway can lead to increased risk of immune-related rejection of the developing fetus resulting in fetal death. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with KEYTRUDA QLEX and for 4 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
1.13 Merkel Cell Carcinoma
KEYTRUDA QLEX is indicated for the treatment of adult and pediatric patients 12 years and older with recurrent locally advanced or metastatic Merkel cell carcinoma (MCC).
1.15 Endometrial Carcinoma
KEYTRUDA QLEX, in combination with carboplatin and paclitaxel, followed by KEYTRUDA QLEX as a single agent, is indicated for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma.
KEYTRUDA QLEX, in combination with lenvatinib, is indicated for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) or not MSI-H as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-authorized test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation [see Dosage and Administration (2.1)].
14.13 Biliary Tract Cancer
The efficacy of intravenous pembrolizumab in combination with gemcitabine and cisplatin chemotherapy was investigated in KEYNOTE-966 (NCT04003636), a multicenter, randomized, double-blind, placebo-controlled trial that enrolled 1069 patients with locally advanced unresectable or metastatic BTC, who had not received prior systemic therapy in the advanced disease setting. Patients with autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by region (Asia vs. non-Asia), locally advanced versus metastatic, and site of origin (gallbladder, intrahepatic or extrahepatic cholangiocarcinoma).
Patients were randomized (1:1) to intravenous pembrolizumab 200 mg on Day 1 plus gemcitabine 1000 mg/m2 and cisplatin 25 mg/m2 on Day 1 and Day 8 every 3 weeks, or placebo on Day 1 plus gemcitabine 1000 mg/m2 and cisplatin 25 mg/m2 on Day 1 and Day 8 every 3 weeks. Study medications were administered via intravenous infusion. Treatment continued until unacceptable toxicity or disease progression. For pembrolizumab, treatment continued for a maximum of 35 cycles, or approximately 24 months. For gemcitabine, treatment could be continued beyond 8 cycles while for cisplatin, treatment could be administered for a maximum of 8 cycles.
Administration of intravenous pembrolizumab with chemotherapy was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered by the investigator to be deriving clinical benefit. Assessment of tumor status was performed at baseline and then every 6 weeks through 54 weeks, followed by every 12 weeks thereafter.
Study population characteristics were median age of 64 years (range: 23 to 85), 47% age 65 or older; 52% male; 49% White, 46% Asian, 1.3% Black or African American; 10% Hispanic or Latino; 46% ECOG PS of 0 and 54% ECOG PS of 1; 31% of patients had a history of hepatitis B infection, and 3% had a history of hepatitis C infection.
The major efficacy outcome measure was OS. Additional efficacy outcome measures were PFS, ORR and DoR as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Table 90 and Figure 32 summarize the efficacy results for KEYNOTE-966.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks with gemcitabine/cisplatin n=533 |
Placebo with gemcitabine/cisplatin n=536 |
|---|---|---|
| NS = not significant | ||
|
OS Results at the pre-specified final OS analysis
|
||
| Number of patients with event (%) | 414 (78%) | 443 (83%) |
| Median in months (95% CI) | 12.7 (11.5, 13.6) | 10.9 (9.9, 11.6) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.83 (0.72, 0.95) | |
| p-Value One-sided p-Value based on a stratified log-rank test
|
0.0034 | |
|
PFS
Results at pre-specified final analysis of PFS and ORR
|
||
| Number (%) of patients with event | 361 (68%) | 391 (73%) |
| Median in months (95% CI) | 6.5 (5.7, 6.9) | 5.6 (5.1, 6.6) |
| Hazard ratio (95% CI) | 0.86 (0.75, 1.00) | |
| p-Value | NS | |
| Objective Response Rate | ||
| ORR Confirmed complete response or partial response (95% CI) |
29% (25, 33) | 29% (25, 33) |
| Number (%) of complete responses | 11 (2.1%) | 7 (1.3%) |
| Number (%) of partial responses | 142 (27%) | 146 (27%) |
| p-Value One-sided p-Value based on the stratified Miettinen and Nurminen analysis
|
NS | |
| Duration of Response | n=156 | n=152 |
| Median in months Based on Kaplan-Meier estimate (95% CI)
|
8.3 (6.9, 10.2) | 6.8 (5.7, 7.1) |
| Figure 32: Kaplan-Meier Curve for Overall Survival in KEYNOTE-966 |
|
|
14.15 Renal Cell Carcinoma
First-line treatment with axitinib
KEYNOTE-426
The efficacy of intravenous pembrolizumab in combination with axitinib was investigated in KEYNOTE-426 (NCT02853331), a randomized, multicenter, open-label trial conducted in 861 patients who had not received systemic therapy for advanced RCC. Patients were enrolled regardless of PD-L1 tumor expression status. Patients with active autoimmune disease requiring systemic immunosuppression within the last 2 years were ineligible. Randomization was stratified by International Metastatic RCC Database Consortium (IMDC) risk categories (favorable versus intermediate versus poor) and geographic region (North America versus Western Europe versus “Rest of the World”).
Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg intravenously every 3 weeks up to 24 months in combination with axitinib 5 mg orally, twice daily. Patients who tolerated axitinib 5 mg twice daily for 2 consecutive cycles (6 weeks) could increase to 7 mg and then subsequently to 10 mg twice daily. Axitinib could be interrupted or reduced to 3 mg twice daily and subsequently to 2 mg twice daily to manage toxicity.
- Sunitinib 50 mg orally, once daily for 4 weeks and then off treatment for 2 weeks.
Treatment with intravenous pembrolizumab and axitinib continued until RECIST v1.1-defined progression of disease or unacceptable toxicity. Administration of intravenous pembrolizumab and axitinib was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered to be deriving clinical benefit by the investigator. Assessment of tumor status was performed at baseline, after randomization at Week 12, then every 6 weeks thereafter until Week 54, and then every 12 weeks thereafter.
The study population characteristics were: median age of 62 years (range: 26 to 90), 38% age 65 or older; 73% male; 79% White and 16% Asian; 20% and 80% of patients had a baseline KPS of 70 to 80 and 90 to 100, respectively; and patient distribution by IMDC risk categories was 31% favorable, 56% intermediate, and 13% poor.
The main efficacy outcome measures were OS and PFS as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ. Additional efficacy outcome measures included ORR, as assessed by BICR. A statistically significant improvement in OS was demonstrated at the first pre-specified interim analysis in patients randomized to intravenous pembrolizumab in combination with axitinib compared with sunitinib. The trial also demonstrated statistically significant improvements in PFS and ORR. An updated OS analysis was conducted when 418 deaths were observed based on the planned number of deaths for the pre-specified final analysis. Table 92 and Figure 33 summarize the efficacy results for KEYNOTE-426.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and Axitinib n=432 |
Sunitinib n=429 |
|---|---|---|
| NR = not reached | ||
| OS | ||
| Number of patients with event (%) | 59 (14%) | 97 (23%) |
| Median in months (95% CI) | NR (NR, NR) | NR (NR, NR) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.53 (0.38, 0.74) | |
| p-Value Based on stratified log-rank test
|
<0.0001 p-Value (one-sided) is compared with the allocated alpha of 0.0001 for this interim analysis (with 39% of the planned number of events for final analysis).
|
|
| Updated OS | ||
| Number of patients with event (%) | 193 (45%) | 225 (52%) |
| Median in months (95% CI) | 45.7 (43.6, NR) | 40.1 (34.3, 44.2) |
| Hazard ratio (95% CI) | 0.73 (0.60, 0.88) | |
| PFS | ||
| Number of patients with event (%) | 183 (42%) | 213 (50%) |
| Median in months (95% CI) | 15.1 (12.6, 17.7) | 11.0 (8.7, 12.5) |
| Hazard ratio (95% CI) | 0.69 (0.56, 0.84) | |
| p-Value | 0.0001 p-Value (one-sided) is compared with the allocated alpha of 0.0013 for this interim analysis (with 81% of the planned number of events for final analysis).
|
|
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
59% (54, 64) | 36% (31, 40) |
| Complete response rate | 6% | 2% |
| Partial response rate | 53% | 34% |
| p-Value Based on Miettinen and Nurminen method stratified by IMDC risk group and geographic region
|
<0.0001 |
| Figure 33: Kaplan-Meier Curve for Updated Overall Survival in KEYNOTE-426 |
|
|
In an exploratory analysis, the updated analysis of OS in patients with IMDC favorable, intermediate, intermediate/poor, and poor risk demonstrated a HR of 1.17 (95% CI: 0.76, 1.80), 0.67 (95% CI: 0.52, 0.86), 0.64 (95% CI: 0.52, 0.80), and 0.51 (95% CI: 0.32, 0.81), respectively.
First-line treatment with lenvatinib
KEYNOTE-581
The efficacy of intravenous pembrolizumab in combination with lenvatinib was investigated in KEYNOTE-581 (NCT02811861), a multicenter, open-label, randomized trial conducted in 1069 patients with advanced RCC in the first-line setting. Patients were enrolled regardless of PD-L1 tumor expression status. Patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible. Randomization was stratified by geographic region (North America versus Western Europe versus “Rest of the World”) and Memorial Sloan Kettering Cancer Center (MSKCC) prognostic groups (favorable versus intermediate versus poor risk).
Patients were randomized (1:1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg intravenously every 3 weeks up to 24 months in combination with lenvatinib 20 mg orally once daily.
- Lenvatinib 18 mg orally once daily in combination with everolimus 5 mg orally once daily.
- Sunitinib 50 mg orally once daily for 4 weeks then off treatment for 2 weeks.
Treatment continued until unacceptable toxicity or disease progression. Administration of intravenous pembrolizumab with lenvatinib was permitted beyond RECIST-defined disease progression if the patient was clinically stable and considered by the investigator to be deriving clinical benefit. intravenous pembrolizumab was continued for a maximum of 24 months; however, treatment with lenvatinib could be continued beyond 24 months. Assessment of tumor status was performed at baseline and then every 8 weeks.
The study population characteristics were: median age of 62 years (range: 29 to 88 years), 42% age 65 or older; 75% male; 74% White, 21% Asian, 1% Black, and 2% other races; 18% and 82% of patients had a baseline KPS of 70 to 80 and 90 to 100, respectively; patient distribution by MSKCC risk categories was 27% favorable, 64% intermediate, and 9% poor. Common sites of metastases in patients were lung (68%), lymph node (45%), and bone (25%).
The major efficacy outcome measures were PFS, as assessed by independent radiologic review (IRC) according to RECIST v1.1, and OS. Additional efficacy outcome measures included confirmed ORR as assessed by IRC. intravenous pembrolizumab in combination with lenvatinib demonstrated statistically significant improvements in PFS, OS, and ORR compared with sunitinib. An updated OS analysis was conducted when 304 deaths were observed based on the planned number of deaths for the pre-specified final analysis. Table 93 and Figures 34 and 35 summarize the efficacy results for KEYNOTE-581.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and Lenvatinib n=355 |
Sunitinib n=357 |
|---|---|---|
| Tumor assessments were based on RECIST 1.1; only confirmed responses are included for ORR. Data cutoff date = 28 Aug 2020, Updated OS cutoff date = 31 July 2022 CI = confidence interval; NR= Not reached |
||
| Progression-Free Survival (PFS) | ||
| Number of events, n (%) | 160 (45%) | 205 (57%) |
| Progressive disease | 145 (41%) | 196 (55%) |
| Death | 15 (4%) | 9 (3%) |
| Median PFS in months (95% CI) | 23.9 (20.8, 27.7) | 9.2 (6.0, 11.0) |
| Hazard ratio Hazard ratio is based on a Cox Proportional Hazards Model. Stratified by geographic region and MSKCC prognostic groups. (95% CI) |
0.39 (0.32, 0.49) | |
| p-Value Two-sided p-Value based on stratified log-rank test.
|
<0.0001 | |
| Overall Survival (OS) | ||
| Number of deaths, n (%) | 80 (23%) | 101 (28%) |
| Median OS in months (95% CI) | NR (33.6, NR) | NR (NR, NR) |
| Hazard ratio (95% CI) | 0.66 (0.49, 0.88) | |
| p-Value | 0.0049 | |
| Updated OS | ||
| Number of deaths, n (%) | 149 (42%) | 159 (45%) |
| Median OS in months (95% CI) | 53.7 (48.7, NR) | 54.3 (40.9, NR) |
| Hazard ratio (95% CI) | 0.79 (0.63, 0.99) | |
| Objective Response Rate (Confirmed) | ||
| ORR, n (%) | 252 (71%) | 129 (36%) |
| (95% CI) | (66, 76) | (31, 41) |
| Complete response rate | 16% | 4% |
| Partial response rate | 55% | 32% |
| p-Value Two-sided p-Value based upon CMH test.
|
<0.0001 |
| Figure 34: Kaplan-Meier Curve for PFS in KEYNOTE-581 |
|
|
| Figure 35: Kaplan-Meier Curve for Updated Overall Survival in KEYNOTE-581 |
|
|
KEYNOTE-B61
The efficacy of intravenous pembrolizumab in combination with lenvatinib was investigated in KEYNOTE-B61 (NCT04704219), a multicenter, single-arm trial that enrolled 160 patients with advanced or metastatic non-clear cell RCC in the first-line setting. Patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible.
Patients received intravenous pembrolizumab 400 mg every 6 weeks in combination with lenvatinib 20 mg orally once daily. Intravenous pembrolizumab was continued for a maximum of 24 months; however, lenvatinib could be continued beyond 24 months. Treatment continued until unacceptable toxicity or disease progression. Administration of intravenous pembrolizumab with lenvatinib was permitted beyond RECIST-defined disease progression if the patient was considered by the investigator to be deriving clinical benefit.
Among the 158 treated patients, the baseline characteristics were: median age of 60 years (range: 24 to 87 years); 71% male; 86% White, 8% Asian, and 3% Black; <1% Hispanic or Latino; 22% and 78% of patients had a baseline KPS of 70 to 80 and 90 to 100, respectively; histologic subtypes were 59% papillary, 18% chromophobe, 4% translocation, <1% medullary, 13% unclassified, and 6% other; patient distribution by IMDC risk categories was 35% favorable, 54% intermediate, and 10% poor. Common sites of metastases in patients were lymph node (65%), lung (35%), bone (30%), and liver (21%).
The major efficacy outcome measure was ORR as assessed by BICR using RECIST 1.1. Additional efficacy outcome measures included DOR as assessed by BICR using RECIST 1.1. Efficacy results are summarized in Table 94.
| Endpoint | Intravenous Pembrolizumab 400 mg every 6 weeks and Lenvatinib n=158 |
|---|---|
| CI = confidence interval + Denotes ongoing response |
|
| Objective Response Rate (Confirmed) | |
| ORR (95% CI) | 51% (43, 59) |
| Complete response | 8% |
| Partial response | 42% |
|
Duration of Response Based on Kaplan-Meier estimates
|
|
| Median in months (range) | 19.5 (1.5+, 23.5+) |
Adjuvant Treatment of RCC (KEYNOTE-564)
The efficacy of intravenous pembrolizumab was investigated as adjuvant therapy for RCC in KEYNOTE-564 (NCT03142334), a multicenter, randomized (1:1), double-blind, placebo-controlled trial in 994 patients with intermediate-high or high risk of recurrence of RCC, or M1 no evidence of disease (NED). The intermediate-high risk category included: pT2 with Grade 4 or sarcomatoid features; pT3, any Grade without nodal involvement (N0) or distant metastases (M0). The high risk category included: pT4, any Grade N0 and M0; any pT, any Grade with nodal involvement and M0. The M1 NED category included patients with metastatic disease who had undergone complete resection of primary and metastatic lesions. Patients must have undergone a partial nephroprotective or radical complete nephrectomy (and complete resection of solid, isolated, soft tissue metastatic lesion(s) in M1 NED participants) with negative surgical margins ≥4 weeks prior to the time of screening. Patients were excluded from the trial if they had received prior systemic therapy for advanced RCC. Patients with active autoimmune disease or a medical condition that required immunosuppression were also ineligible. Patients were randomized to intravenous pembrolizumab 200 mg administered intravenously every 3 weeks or placebo for up to 1 year until disease recurrence or unacceptable toxicity. Randomization was stratified by metastasis status (M0, M1 NED); M0 group was further stratified by ECOG PS (0,1) and geographic region (US, non-US).
The study population characteristics were: median age of 60 years (range: 25 to 84), 33% age 65 or older; 71% male; 75% White, 14% Asian, 9% Unknown, 1% Black or African American, 1% American Indian or Alaska Native, 1% Multiracial; 13% Hispanic or Latino, 78% Not Hispanic or Latino, 8% Unknown; and 85% ECOG PS of 0 and 15% ECOG PS of 1. Ninety-four percent of patients enrolled had N0 disease; 11% had sarcomatoid features; 86% were intermediate-high risk; 8% were high risk; and 6% were M1 NED. Ninety-two percent of patients had a radical nephrectomy, and 8% had a partial nephrectomy.
The major efficacy outcome measure was investigator-assessed disease-free survival (DFS), defined as time to recurrence, metastasis, or death. An additional outcome measure was OS. Statistically significant improvements in DFS and OS were demonstrated at pre-specified interim analyses in patients randomized to the intravenous pembrolizumab arm compared with placebo. Efficacy results are summarized in Table 95 and Figures 36 and 37.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=496 |
Placebo n=498 |
|---|---|---|
| NR = not reached | ||
| DFS | ||
| Number (%) of patients with event | 109 (22%) | 151 (30%) |
| Median in months (95% CI) | NR | NR |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.68 (0.53, 0.87) | |
| p-Value Based on stratified log-rank test
|
0.0010 p-Value (one-sided) is compared with a boundary of 0.0114.
|
|
| 24-month DFS rate (95% CI) | 77% (73, 81) | 68% (64, 72) |
| OS | ||
| Number (%) of patients with event | 55 (11%) | 86 (17%) |
| Median in months (95% CI) | NR (NR, NR) | NR (NR, NR) |
| Hazard ratio (95% CI) | 0.62 (0.44, 0.87) | |
| p-Value | 0.0024 p-Value (one-sided) is compared with a boundary of 0.0072.
|
|
| 48-month OS rate (95% CI) | 91% (88, 93) | 86% (83, 89) |
| Figure 36: Kaplan-Meier Curve for Disease-Free Survival in KEYNOTE-564 |
|
|
| Figure 37: Kaplan-Meier Curve for Overall Survival in KEYNOTE-564 |
|
|
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Immune-Mediated Adverse Reactions (5.1)
- Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, including the following: immune-mediated pneumonitis, immune-mediated colitis, immune-mediated hepatitis, immune-mediated endocrinopathies, immune-mediated nephritis with renal dysfunction, immune-mediated dermatologic adverse reactions, and solid organ transplant rejection.
- Monitor for early identification and management. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment.
- Withhold or permanently discontinue based on severity and type of reaction.
- Hypersensitivity and Administration-Related Reactions: Interrupt injection and resume upon symptom resolution, or permanently discontinue KEYTRUDA QLEX based on the severity of reaction. (5.2)
- Complications of Allogeneic HSCT: Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after being treated with a PD-1/PD-L1 blocking antibody. (5.3)
- Treatment of patients with multiple myeloma with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled clinical trials. (5.4)
- Embryo-Fetal toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective method of contraception. (5.5, 8.1, 8.3)
14.14 Merkel Cell Carcinoma
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-017 (NCT02267603) and KEYNOTE-913 (NCT03783078), two multicenter, non-randomized, open-label trials that enrolled 105 patients with recurrent locally advanced or metastatic MCC who had not received prior systemic therapy for their advanced disease. Patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible.
Patients received intravenous pembrolizumab 2 mg/kg (KEYNOTE-017) or 200 mg (KEYNOTE-913) every 3 weeks until unacceptable toxicity or disease progression that was symptomatic, rapidly progressive, required urgent intervention, occurred with a decline in performance status, or was confirmed at least 4 weeks later with repeat imaging. Patients without disease progression were treated for up to 24 months.
The major efficacy outcome measures were ORR and DoR as assessed by BICR per RECIST v1.1.
Among the 105 patients enrolled, the median age was 73 years (range: 38 to 91), 79% were age 65 or older; 62% were male; 80% were White, race in 19% was unknown or missing, and 1% were Asian; 53% had ECOG PS of 0, and 47% had ECOG PS of 1. Thirteen percent had stage IIIB disease and 84% had stage IV. Seventy-six percent of patients had prior surgery and 51% had prior radiation therapy.
Efficacy results are summarized in Table 91.
| Endpoint | KEYNOTE-017 Intravenous Pembrolizumab 2 mg/kg every 3 weeks n=50 |
KEYNOTE-913 Intravenous Pembrolizumab 200 mg or 2 mg/kg every 3 weeks n=55 |
|---|---|---|
| + Denotes ongoing response | ||
| NR = not reached | ||
| Objective Response Rate | ||
| ORR (95% CI) | 56% (41, 70) | 49% (35, 63) |
| Complete responses, n (%) | 12 (24%) | 9 (16%) |
| Partial responses, n (%) | 16 (32%) | 18 (33%) |
| Duration of Response | n=28 | n=27 |
| Median DoR in months (range) | NR (5.9, 34.5+) | NR (4.8, 25.4+) |
| Patients with duration ≥6 months, n (%) | 27 (96%) | 25 (93%) |
| Patients with duration ≥12 months, n (%) | 15 (54%) | 19 (70%) |
14.16 Endometrial Carcinoma
In Combination with Paclitaxel and Carboplatin for the Treatment of Primary Advanced or Recurrent Endometrial Carcinoma
The efficacy of intravenous pembrolizumab in combination with paclitaxel and carboplatin was investigated in KEYNOTE-868/NRG-GY018 (NCT03914612), a multicenter, randomized, double-blind, placebo-controlled trial in 810 patients with advanced or recurrent endometrial carcinoma. The study design included two separate cohorts based on MMR status; 222 (27%) patients were in dMMR cohort, 588 (73%) patients were in pMMR cohort. The trial enrolled measurable Stage III, measurable Stage IVA, Stage IVB or recurrent endometrial cancer (with or without measurable disease). Patients who had not received prior systemic therapy or had received prior chemotherapy in the adjuvant setting were eligible. Patients who had received prior adjuvant chemotherapy were only eligible if their chemotherapy-free interval was at least 12 months. Patients with endometrial sarcoma, including carcinosarcoma, or patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible. Randomization was stratified according to MMR status, ECOG PS (0 or 1 vs. 2), and prior adjuvant chemotherapy.
Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg every 3 weeks, paclitaxel 175 mg/m2 and carboplatin AUC 5 mg/mL/min for 6 cycles, followed by intravenous pembrolizumab 400 mg every 6 weeks for up to 14 cycles.
- Placebo every 3 weeks, paclitaxel 175 mg/m2 and carboplatin AUC 5 mg/mL/min for 6 cycles, followed by placebo every 6 weeks for up to 14 cycles.
All study medications were administered as an intravenous infusion on Day 1 of each treatment cycle. Treatment continued until disease progression, unacceptable toxicity, or a maximum of 20 cycles (up to approximately 24 months). Patients with measurable disease who had RECIST-defined stable disease or partial response at the completion of cycle 6 were permitted to continue receiving paclitaxel and carboplatin with intravenous pembrolizumab or placebo for up to 10 cycles as determined by the investigator. Assessment of tumor status was performed every 9 weeks for the first 9 months and then every 12 weeks thereafter. The major efficacy outcome measure was PFS as assessed by the investigator according to RECIST 1.1. An additional efficacy outcome measure was OS.
The dMMR population characteristics were: median age of 66 years (range: 37 to 86), 55% age 65 or older; 79% White, 9% Black, and 3% Asian; 5% Hispanic or Latino; 64% ECOG PS of 0, 33% ECOG PS of 1, and 3% ECOG PS of 2; 61% had recurrent disease and 39% had primary or persistent disease; 5% received prior adjuvant chemotherapy and 43% received prior radiotherapy. The histologic subtypes were endometrioid carcinoma (81%), adenocarcinoma NOS (11%), serous carcinoma (2%), and other (6%).
The pMMR population characteristics were: median age of 66 years (range: 29 to 94), 54% age 65 or older; 72% White, 16% Black, and 5% Asian; 6% Hispanic or Latino; 67% ECOG PS of 0, 30% ECOG PS of 1, and 3% ECOG PS of 2; 56% had recurrent disease and 44% had primary or persistent disease; 26% received prior adjuvant chemotherapy and 41% received prior radiotherapy. The histologic subtypes were endometrioid carcinoma (52%), serous carcinoma (26%), adenocarcinoma NOS (10%), clear cell carcinoma (7%), and other (5%).
The trial demonstrated statistically significant improvements in PFS for patients randomized to intravenous pembrolizumab in combination with paclitaxel and carboplatin compared to placebo in combination with paclitaxel and carboplatin in both the dMMR and pMMR populations. Table 96 and Figures 38 and 39 summarize the efficacy results for KEYNOTE-868 by MMR status. At the time of the PFS analysis, OS data were not mature with 12% deaths in the dMMR population and 17% deaths in the pMMR population.
| Endpoint | dMMR Population | pMMR Population | ||
|---|---|---|---|---|
| Intravenous Pembrolizumab with paclitaxel and carboplatin n=110 |
Placebo with paclitaxel and carboplatin n=112 |
Intravenous Pembrolizumab with paclitaxel and carboplatin n=294 |
Placebo with paclitaxel and carboplatin n=294 |
|
| NR = not reached | ||||
|
PFS Based on interim PFS analysis; the information fractions for interim analyses were 49% for dMMR and 55% for pMMR.
|
||||
| Number (%) of patients with event | 26 (24%) | 57 (51%) | 91 (31%) | 124 (42%) |
| Median in months (95% CI) | NR (30.7, NR) | 6.5 (6.4, 8.7) | 11.1 (8.7, 13.5) | 8.5 (7.2, 8.8) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.30 (0.19, 0.48) | 0.60 (0.46, 0.78) | ||
| p-Value Based on the stratified log-rank test
|
<0.0001 | <0.0001 |
| Figure 38: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-868 (dMMR Population) |
|
|
| Figure 39: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-868 (pMMR Population) |
|
|
Lack of Effectiveness for Adjuvant Treatment of Patients with Endometrial Carcinoma
The efficacy of intravenous pembrolizumab in combination with carboplatin and paclitaxel, with or without radiation, was investigated in KEYNOTE‑B21 (NCT04634877), a randomized, multicenter, double-blind, placebo-controlled trial in 1,095 patients with newly-diagnosed, high‑risk endometrial cancer with no evidence of disease on imaging following curative intent surgery. High-risk disease was defined as any of the following (staging per FIGO 2009): Stage I/II with myometrial invasion and either non-endometrioid histology or aberrant p53 expression or p53 mutation, or Stage III/IVA. The trial did not meet the prespecified primary endpoint for investigator-assessed DFS, with a HR of 1.02 (95% CI: 0.79, 1.32).
In Combination with Lenvatinib for the Treatment of Advanced Endometrial Carcinoma That Is pMMR or Not MSI-H
The efficacy of intravenous pembrolizumab in combination with lenvatinib was investigated in KEYNOTE-775 (NCT03517449), a multicenter, open-label, randomized, active-controlled trial that enrolled 827 patients with advanced endometrial carcinoma who had been previously treated with at least one prior platinum-based chemotherapy regimen in any setting, including in the neoadjuvant and adjuvant settings. Patients with endometrial sarcoma, including carcinosarcoma, or patients who had active autoimmune disease or a medical condition that required immunosuppression were ineligible. Patients with endometrial carcinoma that were pMMR (using the VENTANA MMR RxDx Panel test) or not MSI-H were stratified by ECOG performance status, geographic region, and history of pelvic radiation. Patients were randomized (1:1) to one of the following treatment arms:
- Intravenous pembrolizumab 200 mg intravenously every 3 weeks in combination with lenvatinib 20 mg orally once daily.
- Investigator’s choice, consisting of either doxorubicin 60 mg/m2 every 3 weeks or paclitaxel 80 mg/m2 given weekly, 3 weeks on/1 week off.
Treatment with intravenous pembrolizumab and lenvatinib continued until RECIST v1.1-defined progression of disease as verified by BICR, unacceptable toxicity, or for intravenous pembrolizumab, a maximum of 24 months. Treatment was permitted beyond RECIST v1.1-defined disease progression if the treating investigator considered the patient to be deriving clinical benefit, and the treatment was tolerated. Assessment of tumor status was performed every 8 weeks. The major efficacy outcome measures were OS and PFS as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ. Additional efficacy outcome measures included ORR and DoR, as assessed by BICR.
Among the 697 pMMR patients, 346 patients were randomized to intravenous pembrolizumab in combination with lenvatinib, and 351 patients were randomized to investigator’s choice of doxorubicin (n=254) or paclitaxel (n=97). The pMMR population characteristics were: median age of 65 years (range: 30 to 86), 52% age 65 or older; 62% White, 22% Asian, and 3% Black; 60% ECOG PS of 0 and 40% ECOG PS of 1. The histologic subtypes were endometrioid carcinoma (55%), serous (30%), clear cell carcinoma (7%), mixed (4%), and other (3%). All 697 of these patients received prior systemic therapy for endometrial carcinoma: 67% had one, 30% had two, and 3% had three or more prior systemic therapies. Thirty-seven percent of patients received only prior neoadjuvant or adjuvant therapy.
Efficacy results for the pMMR or not MSI-H patients are summarized in Table 97 and Figures 40 and 41.
| Endometrial Carcinoma (pMMR or not MSI-H) | ||
|---|---|---|
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks and Lenvatinib n=346 |
Doxorubicin or Paclitaxel n=351 |
| OS | ||
| Number (%) of patients with event | 165 (48%) | 203 (58%) |
| Median in months (95% CI) | 17.4 (14.2, 19.9) | 12.0 (10.8, 13.3) |
| Hazard ratio Based on the stratified Cox regression model (95% CI) |
0.68 (0.56, 0.84) | |
| p-Value Based on stratified log-rank test
|
0.0001 | |
| PFS | ||
| Number (%) of patients with event | 247 (71%) | 238 (68%) |
| Median in months (95% CI) | 6.6 (5.6, 7.4) | 3.8 (3.6, 5.0) |
| Hazard ratio (95% CI) | 0.60 (0.50, 0.72) | |
| p-Value | <0.0001 | |
| Objective Response Rate | ||
| ORR Response: Best objective response as confirmed complete response or partial response (95% CI) |
30% (26, 36) | 15% (12, 19) |
| Complete response rate | 5% | 3% |
| Partial response rate | 25% | 13% |
| p-Value Based on Miettinen and Nurminen method stratified by ECOG performance status, geographic region, and history of pelvic radiation
|
<0.0001 | |
| Duration of Response | n=105 | n=53 |
| Median in months (range) | 9.2 (1.6+, 23.7+) | 5.7 (0.0+, 24.2+) |
| Figure 40: Kaplan-Meier Curve for Overall Survival in KEYNOTE-775 (pMMR or Not MSI-H) |
|
|
| Figure 41: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-775 (pMMR or Not MSI-H) |
|
|
As a Single Agent for the Treatment of Advanced MSI-H or dMMR Endometrial Carcinoma
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-158 (NCT02628067), a multicenter, non-randomized, open-label, multi-cohort trial. The trial enrolled 90 patients with unresectable or metastatic MSI-H or dMMR endometrial carcinoma in Cohorts D and K. MSI or MMR tumor status was determined using polymerase chain reaction (PCR) or immunohistochemistry (IHC), respectively. Patients with autoimmune disease or a medical condition that required immunosuppression were ineligible. Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression. Patients treated with intravenous pembrolizumab without disease progression could be treated for up to 24 months. Assessment of tumor status was performed every 9 weeks for the first 12 months, and every 12 weeks thereafter. The major efficacy outcome measures were ORR and DoR as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among the 90 patients evaluated, the baseline characteristics were: median age of 64 years (range: 42 to 86); 83% White, 8% Asian, and 3% Black; 12% Hispanic or Latino; 39% ECOG PS of 0 and 61% ECOG PS of 1; 96% had M1 disease and 4% had M0 disease at study entry; and 51% had one and 48% had two or more prior lines of therapy. Nine patients received only adjuvant therapy and one patient received only neoadjuvant and adjuvant therapy before participating in the study.
Efficacy results are summarized in Table 98.
| Endpoint | Intravenous Pembrolizumab n=90 Median follow-up time of 16.0 months (range 0.5 to 62.1 months)
|
|---|---|
| + Denotes ongoing response NR = not reached |
|
| Objective Response Rate | |
| ORR (95% CI) | 46% (35, 56) |
| Complete response rate | 12% |
| Partial response rate | 33% |
| Duration of Response | n=41 |
| Median in months (range) | NR (2.9, 55.7+) |
| % with duration ≥12 months | 68% |
| % with duration ≥24 months | 44% |
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
KEYTRUDA QLEX has different recommended dosage and administration than intravenous pembrolizumab. (2.2)
- KEYTRUDA QLEX is for subcutaneous use in the thigh or abdomen only. (2.2)
- Do not administer KEYTRUDA QLEX intravenously. (2.2)
- KEYTRUDA QLEX must be administered by a healthcare provider. (2.2)
The recommended dose for adults and pediatric patients 12 years and older who weigh greater than 40 kg is:
- Every 3-week dosing (395 mg/4,800 units): Inject 2.4 mL subcutaneously in the abdomen or thigh over 1 minute. (2.3)
- Every 6-week dosing (790 mg/9,600 units): Inject 4.8 mL subcutaneously in the abdomen or thigh over 2 minutes. (2.3)
- For RCC, administer KEYTRUDA QLEX as a single agent in the adjuvant setting, or in the advanced setting with either:
- axitinib 5 mg orally twice daily or
- lenvatinib 20 mg orally once daily. (2.3)
- For Endometrial Carcinoma, administer KEYTRUDA QLEX:
- in combination with carboplatin and paclitaxel regardless of MMR or MSI status, or
- in combination with lenvatinib 20 mg orally once daily for pMMR or not MSI-H tumors, or
- as a single agent for MSI-H or dMMR tumors. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
KEYTRUDA QLEX is a clear to slightly opalescent, colorless to slightly yellow solution provided as:
- Injection: 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/2,000 units per mL) in a single-dose vial
- Injection: 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/2,000 units per mL) in a single-dose vial
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of intravenous pembrolizumab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal: Exocrine pancreatic insufficiency
Hepatobiliary: sclerosing cholangitis
1.11 Hepatocellular Carcinoma
KEYTRUDA QLEX is indicated for the treatment of adult patients with hepatocellular carcinoma (HCC) secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
1.2 Non Small Cell Lung Cancer (1.2 Non-Small Cell Lung Cancer)
KEYTRUDA QLEX, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of adult patients with metastatic nonsquamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations.
KEYTRUDA QLEX, in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, is indicated for the first-line treatment of adult patients with metastatic squamous NSCLC.
KEYTRUDA QLEX, as a single agent, is indicated for the first-line treatment of adult patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-authorized test [see Dosage and Administration (2.1)], with no EGFR or ALK genomic tumor aberrations, and is:
- Stage III where patients are not candidates for surgical resection or definitive chemoradiation, or
- metastatic.
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-authorized test [see Dosage and Administration (2.1)], with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA QLEX.
KEYTRUDA QLEX is indicated for the treatment of adult patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA QLEX, as a single agent, is indicated as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC.
14.12 Hepatocellular Carcinoma
Previously Treated HCC
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-394 (NCT03062358), a multicenter, randomized, placebo-controlled, double-blind trial conducted in Asia in patients with Barcelona Clinic Liver Cancer (BCLC) Stage B or C HCC, who were previously treated with sorafenib or oxaliplatin-based chemotherapy and who were not amenable to or were refractory to local-regional therapy. Patients were also required to have Child-Pugh A liver function.
Patients with hepatitis B had treated controlled disease (HBV viral load <2000 IU/mL or <104 copies/mL). Patients with an autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Patients with hepatic encephalopathy, main branch portal venous invasion, clinically apparent ascites, or esophageal or gastric variceal bleeding within the last 6 months were also ineligible.
Randomization was stratified by prior treatment: sorafenib vs. oxaliplatin-based chemotherapy, macrovascular invasion, and etiology (active HBV vs. others (active HCV, non-infected)). Patients were randomized (2:1) to receive pembrolizumab 200 mg intravenously every 3 weeks or placebo.
Treatment with intravenous pembrolizumab continued until RECIST v1.1-defined progression of disease as determined by BICR, unacceptable toxicity, or a maximum of 24 months. Assessment of tumor status was performed every 6 weeks. The main efficacy outcome measure was OS. Additional efficacy outcome measures were PFS, ORR, and DoR, as assessed by BICR using RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
The study enrolled 453 patients, and 360 (79%) had active hepatitis B. The population characteristics in patients with active hepatitis B were: median age of 52 years (range: 23 to 82), 16% age 65 or older; 86% male; 100% Asian; 42% ECOG PS of 0 and 58% ECOG PS of 1; 90% received prior sorafenib and 10% received prior oxaliplatin-based chemotherapy. Patient characteristics also included extrahepatic disease (77%), macrovascular invasion (10%), BCLC stage C (93%) and B (7%), and baseline AFP ≥200 ng/mL (57%).
KEYNOTE-394 demonstrated improved OS in patients with HCC secondary to hepatitis B randomized to intravenous pembrolizumab compared with placebo. Efficacy results are summarized in Table 89 and Figure 31.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=236 |
Placebo n=124 |
|---|---|---|
| + Denotes ongoing response | ||
|
OS
Results at the pre-specified final OS analysis
|
||
| Number (%) of patients with events | 172 (73) | 105 (85) |
| Median in months (95% CI) | 13.9 (12.5, 17.9) | 13.0 (10.1, 15.6) |
| Hazard ratio Based on the stratified Cox proportional hazard model (95% CI) |
0.78 (0.61, 0.99) | |
|
PFS
Results at pre-specified interim OS analysis
|
||
| Number (%) of patients with events | 189 (80) | 108 (87) |
| Median in months (95% CI) | 2 (1.4, 2.7) | 2.3 (1.4, 2.8) |
| Hazard ratio (95% CI) | 0.78 (0.61, 1.00) | |
| Objective Response Rate | ||
| ORR Confirmed complete response or partial response (95% CI) |
11% (7, 16) | 1.6% (0.2, 5.7) |
| Number (%) of complete responses | 2 (0.9%) | 1 (0.8%) |
| Number (%) of partial responses | 24 (10%) | 1 (0.8%) |
| Duration of Response | n=28 | n=2 |
| Median in months Based on Kaplan-Meier estimate (range)
|
23.9 (2.6+, 44.4+) | 5.6 (3.0+, 5.6) |
| Figure 31: Kaplan-Meier Curve for Overall Survival in KEYNOTE-394 |
|
|
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described in the WARNINGS AND PRECAUTIONS reflect exposure to intravenous pembrolizumab as a single agent in 2799 patients in three randomized, open-label, active-controlled trials (KEYNOTE-002, KEYNOTE-006, and KEYNOTE-010), which enrolled 912 patients with melanoma and 682 patients with NSCLC, and one single-arm trial (KEYNOTE-001), which enrolled 655 patients with melanoma and 550 patients with NSCLC. In addition to the 2799 patients, certain subsections in the WARNINGS AND PRECAUTIONS describe adverse reactions observed with exposure to KEYTRUDA QLEX in combination with platinum doublet chemotherapy in a randomized, open-label, active-controlled trial (Study MK-3475A-D77), which enrolled 251 patients with NSCLC; intravenous pembrolizumab as a single agent in a randomized, placebo-controlled trial (KEYNOTE-091), which enrolled 580 patients with resected NSCLC; a non-randomized, open-label, multi-cohort trial (KEYNOTE-012), a non-randomized, open-label, single-cohort trial (KEYNOTE-055), and two randomized, open-label, active-controlled trials (KEYNOTE-040 and KEYNOTE-048 single agent arms), which enrolled 909 patients with HNSCC; in a randomized, open-label, active-controlled trial (KEYNOTE-048 combination arm), which enrolled 276 patients with HNSCC; in combination with axitinib in a randomized, active-controlled trial (KEYNOTE-426), which enrolled 429 patients with RCC; and in post-marketing use. Across all trials, patients were administered either KEYTRUDA QLEX 790 mg/9,600 units every 6 weeks or intravenous pembrolizumab at doses of 2 mg/kg every 3 weeks, 10 mg/kg every 2 weeks, 10 mg/kg every 3 weeks, or 200 mg every 3 weeks. Among the 2799 patients who received intravenous pembrolizumab, 41% were exposed for 6 months or more and 21% were exposed for 12 months or more.
The most common adverse reactions (≥20%) in patients who received KEYTRUDA QLEX in combination with chemotherapy were nausea (25%), fatigue (25%), and musculoskeletal pain (21%).
The safety of KEYTRUDA QLEX for the approved indications is also based on the safety of intravenous pembrolizumab given as a single agent or in combination with other antitumor medicines.
The most common adverse reactions (≥20%) in patients who received intravenous pembrolizumab were:
- as a single agent: fatigue, musculoskeletal pain, rash, diarrhea, pyrexia, cough, decreased appetite, pruritus, dyspnea, constipation, pain, abdominal pain, nausea, and hypothyroidism.
- in combination with chemotherapy or chemoradiotherapy: fatigue/asthenia, nausea, constipation, diarrhea, decreased appetite, rash, vomiting, cough, dyspnea, pyrexia, alopecia, peripheral neuropathy, mucosal inflammation, stomatitis, headache, weight loss, abdominal pain, arthralgia, myalgia, insomnia, palmar-plantar erythrodysesthesia, urinary tract infection, hypothyroidism, radiation skin injury, dysphagia, dry mouth, and musculoskeletal pain.
- in combination with chemotherapy and bevacizumab: peripheral neuropathy, alopecia, anemia, fatigue/asthenia, nausea, neutropenia, diarrhea, hypertension, thrombocytopenia, constipation, arthralgia, vomiting, urinary tract infection, rash, leukopenia, hypothyroidism, decreased appetite, pyrexia, epistaxis, decreased white blood cell count, and stomatitis.
- in combination with axitinib: diarrhea, fatigue/asthenia, hypertension, hepatotoxicity, hypothyroidism, decreased appetite, palmar-plantar erythrodysesthesia, nausea, stomatitis/mucosal inflammation, dysphonia, rash, cough, and constipation.
- in combination with lenvatinib: hypothyroidism, hypertension, fatigue, diarrhea, musculoskeletal disorders, nausea, decreased appetite, vomiting, stomatitis, weight loss, abdominal pain, urinary tract infection, proteinuria, constipation, headache, hemorrhagic events, palmar-plantar erythrodysesthesia, dysphonia, rash, hepatotoxicity, and acute kidney injury.
- in combination with enfortumab vedotin: rash, peripheral neuropathy, fatigue, pruritus, diarrhea, alopecia, weight loss, decreased appetite, dry eye, nausea, constipation, dysgeusia, and urinary tract infection.
Adverse Reactions in Patients with NSCLC Treated with KEYTRUDA QLEX
The safety of KEYTRUDA QLEX compared to intravenous pembrolizumab in patients with previously untreated, metastatic NSCLC with no EGFR, ALK or ROS1 genomic tumor aberrations was evaluated in Study MK-3475A-D77 [see Clinical Studies (14.1)]. A total of 377 patients received either KEYTRUDA QLEX 790 mg/9,600 units every 6 weeks in combination with platinum doublet chemotherapy (n=251) or intravenous pembrolizumab 400 mg every 6 weeks in combination with platinum doublet chemotherapy (n=126).
Among patients who received KEYTRUDA QLEX, 58% were exposed for 6 months or longer and 3.2% were exposed for greater than one year.
The median age of patients who received KEYTRUDA QLEX was 65 years (range: 39 to 87); 73% male, 63% White; 29% Asian, 4.8% multiple races, 2% Black or African American, 0.8% Alaska Native or American Indian; and 29% were of Hispanic or Latino ethnicity.
Serious adverse reactions occurred in 39% of patients who received KEYTRUDA QLEX in combination with chemotherapy. Serious adverse reactions in ≥1% of patients who received KEYTRUDA QLEX were pneumonia (10%), thrombocytopenia (4%), febrile neutropenia (4%), neutropenia (2.8%), musculoskeletal pain (2%), pneumonitis (2%), diarrhea (1.6%), rash (1.2%), respiratory failure (1.2%), and anemia (1.2%). Fatal adverse reactions occurred in 10% of patients who received KEYTRUDA QLEX in combination with chemotherapy including pneumonia (3.2%), neutropenic sepsis (2%), death not otherwise specified (1.6%), respiratory failure (1.2%), parotitis (0.4%), pneumonitis (0.4%), pneumothorax (0.4%), pulmonary embolism (0.4%), neutropenic colitis (0.4%), and seizure (0.4%).
Permanent discontinuation of KEYTRUDA QLEX due to an adverse reaction occurred in 16% of patients. Adverse reactions which resulted in permanent discontinuation of KEYTRUDA QLEX in ≥2% of patients included pneumonia and pneumonitis.
Dosage interruptions of KEYTRUDA QLEX due to an adverse reaction occurred in 45% of patients. Adverse reactions which required dosage interruption in ≥2% of patients included neutropenia, anemia, thrombocytopenia, pneumonia, rash, and increased aspartate aminotransferase.
Tables 4 and 5 summarize the adverse reactions and laboratory abnormalities, respectively, in patients on KEYTRUDA QLEX in Study MK-3475A-D77.
| Adverse Reaction | KEYTRUDA QLEX and Platinum Doublet Chemotherapy |
Intravenous Pembrolizumab and Platinum Doublet Chemotherapy |
||
|---|---|---|---|---|
| (n=251) | (n=126) | |||
| All Grades Graded per NCI CTCAE V5.0
(%) |
Grades 3-4 (%) |
All Grades
(%) |
Grades 3-4 (%) |
|
| Gastrointestinal | ||||
| Nausea | 25 | 1.2 | 25 | 0.8 |
| Diarrhea Includes diarrhea, colitis, and enterocolitis.
|
16 | 2 | 14 | 0.8 |
| Constipation | 14 | 0 | 18 | 1.6 |
| General | ||||
| Fatigue Includes fatigue, asthenia.
|
25 | 3.6 | 26 | 3.2 |
| Musculoskeletal and Connective Tissue | ||||
| Musculoskeletal pain Includes musculoskeletal pain, arthralgia, arthritis, back pain, bone pain, musculoskeletal chest pain, musculoskeletal stiffness, myalgia, non-cardiac chest pain, and pain in extremity.
|
21 | 2.4 | 30 | 2.4 |
| Skin and Subcutaneous Tissue | ||||
| Rash Includes rash, dermatitis, dermatitis acneiform, dermatitis bullous, dermatitis exfoliative, eczema, erythema multiforme, immune-mediated dermatitis, rash erythematous, rash follicular, rash generalized, rash macular, rash maculo-papular, rash papular, rash pruritic, and skin exfoliation.
|
18 | 2 | 19 | 0.8 |
| Pruritus | 12 | 0 | 13 | 0.8 |
| Endocrine | ||||
| Hypothyroidism | 14 | 0 | 12 | 0 |
| Infections | ||||
| Pneumonia Includes pneumonia, COVID-19 pneumonia, lower respiratory tract infection, lung abscess, pneumocystis jirovecii pneumonia, pneumonia bacterial, and pneumonia mycoplasmal.
|
17 | 10 | 16 | 7 |
| Nervous System | ||||
| Peripheral neuropathy Includes neuropathy peripheral, hypoaesthesia, neuralgia, paraesthesia, and peripheral sensory neuropathy.
|
11 | 0.4 | 14 | 0 |
| Metabolism and Nutrition | ||||
| Decreased appetite | 11 | 0.8 | 21 | 2.4 |
| Hyperglycemia | 11 | 0.8 | 11 | 0.8 |
| Respiratory, Thoracic and Mediastinal | ||||
| Cough Includes cough, productive cough, and upper-airway cough syndrome.
|
10 | 0 | 11 | 0.8 |
Clinically relevant adverse reactions in <10% of patients who received KEYTRUDA QLEX included local injection site reactions (2.4%).
| Laboratory Test Each test incidence is based on the number of patients who had both baseline and at least one on-study laboratory measurement available: KEYTRUDA QLEX plus platinum doublet chemotherapy (range: 240 to 246 patients) and intravenous pembrolizumab plus platinum doublet chemotherapy (range: 124 to 125 patients).
|
KEYTRUDA QLEX and Platinum Doublet Chemotherapy |
Intravenous Pembrolizumab and Platinum Doublet Chemotherapy |
||
|---|---|---|---|---|
| All Grades Graded per NCI CTCAE V5.0
(%) |
Grades 3-4 (%) |
All Grades
(%) |
Grades 3-4 (%) |
|
| Hematology | ||||
| Anemia | 80 | 22 | 86 | 26 |
| Leukopenia | 61 | 13 | 52 | 10 |
| Neutropenia | 58 | 28 | 49 | 19 |
| Lymphopenia | 55 | 22 | 54 | 18 |
| Thrombocytopenia | 43 | 11 | 41 | 6 |
| Chemistry | ||||
| Increased AST | 43 | 2.5 | 38 | 3.2 |
| Hypoalbuminemia | 38 | 0.4 | 39 | 0 |
| Increased ALT | 37 | 2.1 | 36 | 0.8 |
| Hyponatremia | 35 | 4.1 | 42 | 7 |
| Increased creatinine | 33 | 4.5 | 38 | 6 |
| Hypocalcemia | 31 | 2.1 | 31 | 2.4 |
| Increased alkaline phosphatase | 29 | 0.4 | 34 | 0 |
| Hypokalemia | 21 | 5 | 24 | 6 |
Adverse Reactions in Adult and Pediatric Patients Treated with Intravenous Pembrolizumab
The safety of KEYTRUDA QLEX for its approved indications [see Indications and Usage (1)] has been established in adequate and well-controlled studies of KEYTRUDA QLEX in combination with platinum doublet chemotherapy (Study MK-3475A-D77) and intravenous pembrolizumab, as a single agent or in combination therapy, across tumor types.
Below is a description of adverse reactions of intravenous pembrolizumab in these adequate and well-controlled studies.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
1.18 Triple Negative Breast Cancer (1.18 Triple-Negative Breast Cancer)
KEYTRUDA QLEX is indicated for the treatment of adult patients with high-risk early-stage triple-negative breast cancer (TNBC) in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA QLEX, in combination with chemotherapy, is indicated for the treatment of adult patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
1.3 Malignant Pleural Mesothelioma
KEYTRUDA QLEX, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of adult patients with unresectable advanced or metastatic malignant pleural mesothelioma (MPM).
14.19 Triple Negative Breast Cancer (14.19 Triple-Negative Breast Cancer)
Neoadjuvant and Adjuvant Treatment of High-Risk Early-Stage TNBC
The efficacy of intravenous pembrolizumab in combination with neoadjuvant chemotherapy followed by surgery and continued adjuvant treatment with intravenous pembrolizumab as a single agent was investigated in KEYNOTE-522 (NCT03036488), a randomized (2:1), multicenter, double-blind, placebo-controlled trial conducted in 1174 patients with newly diagnosed previously untreated high-risk early-stage TNBC (tumor size >1 cm but ≤2 cm in diameter with nodal involvement or tumor size >2 cm in diameter regardless of nodal involvement). Patients were enrolled regardless of tumor PD-L1 expression. Patients with active autoimmune disease that required systemic therapy within two years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by nodal status (positive vs. negative), tumor size (T1/T2 vs. T3/T4), and choice of carboplatin (dosed every 3 weeks vs. weekly).
Patients were randomized (2:1) to one of the following two treatment arms; all study medications were administered intravenously:
-
Arm 1:
- Four cycles of preoperative intravenous pembrolizumab 200 mg every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
- Carboplatin
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
-or- - AUC 1.5 mg/mL/min every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
-and-
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
- Paclitaxel 80 mg/m2 every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
- Carboplatin
- Followed by four additional cycles of preoperative intravenous pembrolizumab 200 mg every 3 weeks on Day 1 of cycles 5-8 of treatment regimen in combination with:
- Doxorubicin 60 mg/m2 -or- epirubicin 90 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen -and-
- Cyclophosphamide 600 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen
- Following surgery, nine cycles of intravenous pembrolizumab 200 mg every 3 weeks were administered.
- Four cycles of preoperative intravenous pembrolizumab 200 mg every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
-
Arm 2:
- Four cycles of preoperative placebo every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
- Carboplatin
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
-or- - AUC 1.5 mg/mL/min every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
- and-
- AUC 5 mg/mL/min every 3 weeks on Day 1 of cycles 1-4 of treatment regimen
- Paclitaxel 80 mg/m2 every week on Days 1, 8, and 15 of cycles 1-4 of treatment regimen
- Carboplatin
- Followed by four cycles of preoperative placebo every 3 weeks on Day 1 of cycles 5-8 of treatment regimen in combination with:
- Doxorubicin 60 mg/m2 -or- epirubicin 90 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen -and-
- Cyclophosphamide 600 mg/m2 every 3 weeks on Day 1 of cycles 5-8 of treatment regimen
- Following surgery, nine cycles of placebo every 3 weeks were administered.
- Four cycles of preoperative placebo every 3 weeks on Day 1 of cycles 1-4 of treatment regimen in combination with:
The trial was not designed to isolate the effect of intravenous pembrolizumab in each phase (neoadjuvant or adjuvant) of treatment.
The main efficacy outcomes were pCR rate and EFS. pCR was defined as absence of invasive cancer in the breast and lymph nodes (ypT0/Tis ypN0) and was assessed by the blinded local pathologist at the time of definitive surgery. EFS was defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause. An additional efficacy outcome was overall survival (OS).
The study population characteristics were: median age of 49 years (range: 22 to 80), 11% age 65 or older; 99.9% female; 64% White, 20% Asian, 4.5% Black, and 1.8% American Indian or Alaska Native; 87% ECOG PS of 0 and 13% ECOG PS of 1; 56% were pre-menopausal status and 44% were post-menopausal status; 7% were primary Tumor 1 (T1), 68% T2, 19% T3, and 7% T4; 49% were nodal involvement 0 (N0), 40% N1, 11% N2, and 0.2% N3; 75% of patients were overall Stage II and 25% were Stage III.
Statistically significant improvements in pCR, EFS, and OS were demonstrated at pre-specified interim analyses for patients randomized to intravenous pembrolizumab in combination with chemotherapy followed by intravenous pembrolizumab as a single agent compared with patients randomized to placebo in combination with chemotherapy followed by placebo alone.
Table 102 and Figures 42 and 43 summarize the efficacy results for KEYNOTE-522.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks with chemotherapy/Intravenous Pembrolizumab n=784 |
Placebo with chemotherapy/Placebo n=390 |
|---|---|---|
|
pCR (ypT0/Tis ypN0)
Based on the entire intention-to-treat population n=1174 patients
|
||
| Number of patients with pCR | 494 | 217 |
| pCR rate (%), (95% CI) | 63.0 (59.5, 66.4) | 55.6 (50.6, 60.6) |
| Treatment difference (%) estimate (95% CI) Based on a pre-specified pCR interim analysis in n=602 patients, the pCR rate difference was statistically significant (p=0.00055 compared to a significance level of 0.003).
,
Based on Miettinen and Nurminen method stratified by nodal status, tumor size, and choice of carboplatin
|
7.5 (1.6, 13.4) | |
| EFS | ||
| Number of patients with event (%) | 123 (16%) | 93 (24%) |
| Hazard ratio (95% CI) Based on stratified Cox regression model
|
0.63 (0.48, 0.82) | |
| p-Value Based on a pre-specified EFS interim analysis (compared to a significance level of 0.0052)
,
Based on log-rank test stratified by nodal status, tumor size, and choice of carboplatin
|
0.00031 | |
| OS | ||
| Number of patients with event (%) | 115 (15%) | 85 (22%) |
| Hazard ratio (95% CI) | 0.66 (0.50, 0.87) | |
| p-Value
,
Based on a pre-specified OS interim analysis (compared to a significance level of 0.0050)
|
0.00150 |
| Figure 42: Kaplan-Meier Curve for Event-Free Survival in KEYNOTE-522 |
|
|
| Figure 43: Kaplan-Meier Curve for Overall Survival in KEYNOTE-522 |
|
|
Locally Recurrent Unresectable or Metastatic TNBC for Tumors Expressing PD-L1 (CPS≥ 10)
The efficacy of intravenous pembrolizumab in combination with paclitaxel, paclitaxel protein-bound, or gemcitabine and carboplatin was investigated in KEYNOTE-355 (NCT02819518), a multicenter, double-blind, randomized, placebo-controlled trial conducted in 847 patients with locally recurrent unresectable or metastatic TNBC, regardless of tumor PD-L1 expression, who had not been previously treated with chemotherapy in the metastatic setting. Patients with active autoimmune disease that required systemic therapy within 2 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by chemotherapy treatment (paclitaxel or paclitaxel protein-bound vs. gemcitabine and carboplatin), tumor PD-L1 expression (CPS ≥1 vs. CPS <1) according to the PD-L1 IHC 22C3 pharmDx assay, and prior treatment with the same class of chemotherapy in the neoadjuvant setting (yes vs. no).
Patients were randomized (2:1) to one of the following treatment arms; all study medications were administered via intravenous infusion:
- Intravenous pembrolizumab 200 mg on Day 1 every 3 weeks in combination with paclitaxel protein-bound 100 mg/m2 on Days 1, 8 and 15 every 28 days, paclitaxel 90 mg/m2 on Days 1, 8, and 15 every 28 days, or gemcitabine 1000 mg/m2 and carboplatin AUC 2 mg/mL/min on Days 1 and 8 every 21 days.
- Placebo on Day 1 every 3 weeks in combination with paclitaxel protein-bound 100 mg/m2 on Days 1, 8 and 15 every 28 days, paclitaxel 90 mg/m2 on Days 1, 8, and 15 every 28 days, or gemcitabine 1000 mg/m2 and carboplatin AUC 2 mg/mL/min on Days 1 and 8 every 21 days.
Assessment of tumor status was performed at Weeks 8, 16, and 24, then every 9 weeks for the first year, and every 12 weeks thereafter. The main efficacy outcome measures were OS and PFS as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ, tested in the subgroup of patients with CPS ≥10. Additional efficacy outcome measures were ORR and DoR as assessed by BICR.
The study population characteristics for patients were: median age of 53 years (range: 22 to 85), 21% age 65 or older; 100% female; 68% White, 21% Asian, and 4% Black; 60% ECOG PS of 0 and 40% ECOG PS of 1; and 68% were post-menopausal status. Seventy-five percent of patients had tumor PD-L1 expression CPS ≥1 and 38% had tumor PD-L1 expression CPS ≥10.
Table 103 and Figures 44 and 45 summarize the efficacy results for KEYNOTE-355.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks with chemotherapy n=220 |
Placebo every 3 weeks with chemotherapy n=103 |
|---|---|---|
|
OS Based on the pre-specified final analysis
|
||
| Number of patients with event (%) | 155 (70%) | 84 (82%) |
| Median in months (95% CI) | 23 (19.0, 26.3) | 16.1 (12.6, 18.8) |
| Hazard ratio Based on stratified Cox regression model (95% CI) |
0.73 (0.55, 0.95) | |
| p-Value One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.0113)
|
0.0093 | |
|
PFS Based on a pre-specified interim analysis
|
||
| Number of patients with event (%) |
136 (62%) | 79 (77%) |
| Median in months (95% CI) | 9.7 (7.6, 11.3) | 5.6 (5.3, 7.5) |
| Hazard ratio (95% CI) | 0.65 (0.49, 0.86) | |
| p-Value One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.00411)
|
0.0012 | |
| Objective Response Rate (Confirmed) | ||
| ORR (95% CI) | 53% (46, 59) | 41% (31, 51) |
| Complete response rate | 17% | 14% |
| Partial response rate | 35% | 27% |
| Duration of Response | n=116 | n=42 |
| Median in months (95% CI) | 12.8 (9.9, 25.9) | 7.3 (5.5, 15.4) |
| Figure 44: Kaplan-Meier Curve for Overall Survival in KEYNOTE-355 (CPS ≥10) |
|
|
| Figure 45: Kaplan-Meier Curve for Progression-Free Survival in KEYNOTE-355 (CPS ≥10) |
|
|
14.4 Malignant Pleural Mesothelioma
First-line treatment of unresectable advanced or metastatic malignant pleural mesothelioma (MPM) with pemetrexed and platinum chemotherapy
The efficacy of intravenous pembrolizumab in combination with pemetrexed and platinum chemotherapy was investigated in KEYNOTE-483 (NCT02784171), a multicenter, randomized, open-label, active-controlled trial that enrolled 440 patients with unresectable advanced or metastatic MPM and no prior systemic therapy for advanced/metastatic disease. Patients were enrolled regardless of tumor PD-L1 expression. Patients with autoimmune disease that required systemic therapy within 3 years of treatment or a medical condition that required immunosuppression were ineligible. Randomization was stratified by histological subtype (epithelioid vs. non-epithelioid). Patients were randomized (1:1) to one of the following treatment arms; all study medications were administered via intravenous infusion:
- Intravenous pembrolizumab 200 mg with pemetrexed 500 mg/m2 and cisplatin 75 mg/m2 or carboplatin AUC 5-6 mg/mL/min on Day 1 of each 21-day cycle for up to 6 cycles, followed by intravenous pembrolizumab 200 mg every 3 weeks.
- Pemetrexed 500 mg/m2 and cisplatin 75 mg/m2 or carboplatin AUC 5-6 mg/mL/min on Day 1 of each 21-day cycle for up to 6 cycles.
Treatment with intravenous pembrolizumab continued until disease progression as determined by the investigator according to modified RECIST 1.1 for mesothelioma (mRECIST), unacceptable toxicity, or a maximum of 24 months. Assessment of tumor status was performed every 6 weeks for 18 weeks, followed by every 12 weeks thereafter. The main efficacy outcome measure was OS. Additional efficacy outcome measures were PFS, ORR, and DoR, as assessed by BICR according to mRECIST.
The study population characteristics were: median age of 70 years (77% age 65 or older); 76% male; 79% White, 21% race not reported or unknown; 2% Hispanic or Latino; and 53% ECOG performance status of 1. Seventy-eight percent had epithelioid and 22% had non-epithelioid histology; 60% had tumors with PD-L1 CPS ≥1 and 30% had tumors with PD-L1 CPS <1.
The trial demonstrated a statistically significant improvement in OS, PFS, and ORR in patients randomized to intravenous pembrolizumab in combination with chemotherapy compared with patients randomized to chemotherapy alone. Table 69 and Figure 12 summarize the efficacy results for KEYNOTE-483.
| Endpoint | Intravenous pembrolizumab 200 mg every 3 weeks Pemetrexed Platinum Chemotherapy |
Pemetrexed Platinum Chemotherapy |
|---|---|---|
| (n=222) | (n=218) | |
| OS | ||
| Number (%) of patients with event | 167 (75%) | 175 (80%) |
| Median in months (95% CI) | 17.3 (14.4, 21.3) | 16.1 (13.1, 18.2) |
| Hazard ratio Based on stratified Cox proportional hazard model (95% CI) |
0.79 (0.64, 0.98) | |
| p-Value Based on stratified log-rank test
|
0.0162 | |
| PFS | ||
| Number (%) of patients with event | 190 (86%) | 166 (76%) |
| Median in months (95% CI) | 7.1 (6.9, 8.1) | 7.1 (6.8, 7.7) |
| Hazard ratio (95% CI) | 0.80 (0.65, 0.99) | |
| p-Value | 0.0194 | |
| Objective Response Rate | ||
| ORR % (95% CI) | 52% (45.5, 59.0) | 29% (23.0, 35.4) |
| Complete responses | 1 (0.5%) | 0 (0%) |
| Partial responses | 115 (52%) | 63 (29%) |
| p-Value Based on Miettinen and Nurminen method stratified by histological subtype at randomization (epithelioid vs. non-epithelioid)
|
<0.00001 | |
|
Duration of Response
Based on patients with a best overall response as confirmed complete or partial response; n=116 for patients in the intravenous pembrolizumab combination arm; n=63 for patients in the chemotherapy arm
|
||
| Median in months (95% CI) | 6.9 (5.8, 8.3) | 6.8 (5.5, 8.5) |
In a pre-specified exploratory analysis based on histology, in the subgroup of patients with epithelioid histology (n=345), the hazard ratio (HR) for OS was 0.89 (95% CI: 0.70, 1.13), with median OS of 19.8 months in intravenous pembrolizumab in combination with chemotherapy and 18.2 months in chemotherapy alone. In the subgroup of patients with non-epithelioid histology (n=95), the HR for OS was 0.57 (95% CI: 0.36, 0.89), with median OS of 12.3 months in intravenous pembrolizumab in combination with chemotherapy and 8.2 months in chemotherapy alone.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
KEYTRUDA QLEX (pembrolizumab and berahyaluronidase alfa-pmph) injection is a sterile, preservative-free, clear to slightly opalescent, colorless to slightly yellow solution supplied in single-dose vials for subcutaneous administration. Each carton contains one single-dose vial either as:
- 395 mg pembrolizumab and 4,800 units berahyaluronidase alfa per 2.4 mL (165 mg/ 2,000 units per mL), NDC 0006-3083-01
- 790 mg pembrolizumab and 9,600 units berahyaluronidase alfa per 4.8 mL (165 mg/ 2,000 units per mL), NDC 0006-5083-01
1.17 Cutaneous Squamous Cell Carcinoma
KEYTRUDA QLEX is indicated for the treatment of adult patients with recurrent or metastatic cutaneous squamous cell carcinoma (cSCC) or locally advanced cSCC that is not curable by surgery or radiation.
1.4 Head and Neck Squamous Cell Cancer
KEYTRUDA QLEX is indicated for the treatment of adult patients with resectable locally advanced HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test [see Dosage and Administration (2.1)], as a single agent as neoadjuvant treatment, continued as adjuvant treatment in combination with radiotherapy (RT) with or without cisplatin and then as a single agent.
KEYTRUDA QLEX, in combination with platinum and fluorouracil (FU), is indicated for the first-line treatment of adult patients with metastatic or with unresectable, recurrent head and neck squamous cell carcinoma (HNSCC).
KEYTRUDA QLEX, as a single agent, is indicated for the first-line treatment of adult patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
KEYTRUDA QLEX, as a single agent, is indicated for the treatment of adult patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy.
14.18 Cutaneous Squamous Cell Carcinoma
The efficacy of intravenous pembrolizumab was investigated in patients with recurrent or metastatic cSCC or locally advanced cSCC enrolled in KEYNOTE-629 (NCT03284424), a multicenter, multi-cohort, non-randomized, open-label trial. The trial excluded patients with autoimmune disease or a medical condition that required immunosuppression.
Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until documented disease progression, unacceptable toxicity, or a maximum of 24 months. Patients with initial radiographic disease progression could receive additional doses of intravenous pembrolizumab during confirmation of progression unless disease progression was symptomatic, rapidly progressive, required urgent intervention, or occurred with a decline in performance status.
Assessment of tumor status was performed every 6 weeks during the first year, and every 9 weeks during the second year. The major efficacy outcome measures were ORR and DoR as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
Among the 105 patients with recurrent or metastatic cSCC treated, the study population characteristics were: median age of 72 years (range: 29 to 95), 71% age 65 or older; 76% male; 70% White, 25% race unknown; 34% ECOG PS of 0 and 66% ECOG PS of 1. Forty-five percent of patients had locally recurrent only cSCC, 24% had metastatic only cSCC, and 31% had both locally recurrent and metastatic cSCC. Eighty-seven percent received one or more prior lines of therapy; 73% received prior radiation therapy.
Among the 54 patients with locally advanced cSCC treated, the study population characteristics were: median age of 76 years (range: 35 to 95), 80% age 65 or older; 72% male; 83% White, 13% race unknown; 41% ECOG PS of 0 and 59% ECOG PS of 1. Twenty-two percent received one or more prior lines of therapy; 63% received prior radiation therapy.
Efficacy results are summarized in Table 101.
| Endpoint | Intravenous Pembrolizumab Recurrent or Metastatic cSCC n=105 |
Intravenous Pembrolizumab Locally Advanced cSCC n=54 |
|---|---|---|
| + Denotes ongoing response | ||
| Objective Response Rate | ||
| ORR (95% CI) | 35% (26, 45) | 52% (38, 66) |
| Complete response rate | 12% | 22% |
| Partial response rate | 23% | 30% |
|
Duration of Response Median follow-up time: recurrent or metastatic cSCC: 23.8 months; locally advanced cSCC: 48.0 months
|
n=37 | n=28 |
| Median in months (range) | NR (2.7, 64.2+) | 47.2 (1.0+, 49.9+) |
| % with duration ≥6 months | 76% | 89% |
| % with duration ≥12 months | 68% | 75% |
1.16 Tumor Mutational Burden High Cancer (1.16 Tumor Mutational Burden-High Cancer)
KEYTRUDA QLEX is indicated for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-authorized test [see Dosage and Administration (2.1)], that have progressed following prior treatment and who have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on tumor response rate and durability of response [see Clinical Studies (14.17)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Limitations of Use: The safety and effectiveness of KEYTRUDA QLEX in pediatric patients 12 years and older with TMB-H central nervous system cancers have not been established.
14.17 Tumor Mutational Burden High Cancer (14.17 Tumor Mutational Burden-High Cancer)
The efficacy of intravenous pembrolizumab was investigated in a prospectively-planned retrospective analysis of 10 cohorts (A through J) of patients with various previously treated unresectable or metastatic solid tumors with high tumor mutation burden (TMB-H) who were enrolled in a multicenter, non-randomized, open-label trial, KEYNOTE-158 (NCT02628067). The trial excluded patients who previously received an anti-PD-1 or other immune-modulating monoclonal antibody, or who had an autoimmune disease, or a medical condition that required immunosuppression. Patients received intravenous pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression. Assessment of tumor status was performed every 9 weeks for the first 12 months and every 12 weeks thereafter.
The statistical analysis plan pre-specified ≥10 and ≥13 mutations per megabase using the FoundationOne CDx assay as cutpoints to assess TMB. Testing of TMB was blinded with respect to clinical outcomes. The major efficacy outcome measures were ORR and DoR in patients who received at least one dose of intravenous pembrolizumab as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ.
In KEYNOTE-158, 1050 patients were included in the efficacy analysis population. TMB was analyzed in the subset of 790 patients with sufficient tissue for testing based on protocol-specified testing requirements. Of the 790 patients, 102 (13%) had tumors identified as TMB-H, defined as TMB ≥10 mutations per megabase. Among the 102 patients with TMB-H advanced solid tumors, the study population characteristics were: median age of 61 years (range: 27 to 80), 34% age 65 or older; 34% male; 81% White; and 41% ECOG PS of 0 and 58% ECOG PS of 1. Fifty-six percent of patients had at least two prior lines of therapy.
Efficacy results are summarized in Tables 99 and 100.
|
Endpoint |
Intravenous Pembrolizumab 200 mg every 3 weeks |
|
|---|---|---|
| TMB ≥10 mut/Mb n=102 Median follow-up time of 11.1 months
|
TMB ≥13 mut/Mb n=70 |
|
| + Denotes ongoing response NR = not reached |
||
| Objective Response Rate | ||
| ORR (95% CI) | 29% (21, 39) | 37% (26, 50) |
| Complete response rate | 4% | 3% |
| Partial response rate | 25% | 34% |
| Duration of Response | n=30 | n=26 |
| Median in months (range) From product-limit (Kaplan-Meier) method for censored data
|
NR (2.2+, 34.8+) | NR (2.2+, 34.8+) |
| % with duration ≥12 months | 57% | 58% |
| % with duration ≥24 months | 50% | 50% |
| Objective Response Rate | Duration of Response range | |||
|---|---|---|---|---|
| N | n (%) | 95% CI | (months) | |
| CR = complete response PR = partial response SD = stable disease PD = progressive disease |
||||
|
Overall No TMB-H patients were identified in the cholangiocarcinoma cohort
|
102 | 30 (29%) | (21%, 39%) | (2.2+, 34.8+) |
| Small cell lung cancer | 34 | 10 (29%) | (15%, 47%) | (4.1, 32.5+) |
| Cervical cancer | 16 | 5 (31%) | (11%, 59%) | (3.7+, 34.8+) |
| Endometrial cancer | 15 | 7 (47%) | (21%, 73%) | (8.4+, 33.9+) |
| Anal cancer | 14 | 1 (7%) | (0.2%, 34%) | 18.8+ |
| Vulvar cancer | 12 | 2 (17%) | (2%, 48%) | (8.8, 11.0) |
| Neuroendocrine cancer | 5 | 2 (40%) | (5%, 85%) | (2.2+, 32.6+) |
| Salivary cancer | 3 | PR, SD, PD | 31.3+ | |
| Thyroid cancer | 2 | CR, CR | (8.2, 33.2+) | |
| Mesothelioma cancer | 1 | PD |
In an exploratory analysis in 32 patients enrolled in KEYNOTE-158 whose cancer had TMB ≥10 mut/Mb and <13 mut/Mb, the ORR was 13% (95% CI: 4%, 29%), including two complete responses and two partial responses.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In animal models, inhibition of PD-1/PD-L1 signaling increased the severity of some infections and enhanced inflammatory responses. Mycobacterium tuberculosis-infected PD-1 knockout mice exhibit markedly decreased survival compared with wild-type controls, which correlated with increased bacterial proliferation and inflammatory responses in these animals. PD-1 blockade using a primate anti-PD-1 antibody was also shown to exacerbate M. tuberculosis infection in rhesus macaques. PD-1 and PD-L1 knockout mice and mice receiving PD-L1-blocking antibody have also shown decreased survival following infection with lymphocytic choriomeningitis virus. Administration of pembrolizumab in chimpanzees with naturally occurring chronic hepatitis B infection resulted in two out of four animals with significantly increased levels of serum ALT, AST, and GGT, which persisted for at least 1 month after discontinuation of pembrolizumab.
2.2 Important Dosage and Administration Information
- KEYTRUDA QLEX has different recommended dosage and administration instructions than intravenous pembrolizumab.
- To reduce the risk of medication errors, check the vial labels to ensure that the drug being prepared and administered is KEYTRUDA QLEX for subcutaneous use and not intravenous pembrolizumab.
- Do not substitute KEYTRUDA QLEX with intravenous pembrolizumab because they have different recommended dosages and routes of administration.
- Patients receiving intravenous pembrolizumab can switch to subcutaneous KEYTRUDA QLEX at their next scheduled dose.
- Patients receiving subcutaneous KEYTRUDA QLEX can switch to intravenous pembrolizumab at their next scheduled dose.
- Administer KEYTRUDA QLEX as a subcutaneous injection into the thigh or abdomen, avoiding the 5 cm area around the navel.
- Every 3-week dosing (395 mg/4,800 units): inject 2.4 mL subcutaneously over 1 minute. Treatment duration is provided in Recommended Dosage (Table 1).
- Every 6-week dosing (790 mg/9,600 units): inject 4.8 mL subcutaneously over 2 minutes. Treatment duration is provided in Recommended Dosage (Table 1).
- Inject into healthy skin and never into areas where the skin is red, bruised, tender, or hard.
- Ensure the injection site is at least 2.5 cm from the previous injection site.
- During treatment with KEYTRUDA QLEX, do not administer other medications for subcutaneous use at the same site as KEYTRUDA QLEX.
- Do not administer KEYTRUDA QLEX intravenously.
- KEYTRUDA QLEX must be administered by a healthcare provider.
5.1 Severe and Fatal Immune Mediated Adverse Reactions (5.1 Severe and Fatal Immune-Mediated Adverse Reactions)
KEYTRUDA QLEX is a monoclonal antibody that belongs to a class of drugs that bind to either the programmed death-receptor 1 (PD-1) or the PD-ligand 1 (PD-L1), blocking the PD-1/PD-L1 pathway, thereby removing inhibition of the immune response, potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Important immune-mediated adverse reactions listed under WARNINGS AND PRECAUTIONS may not include all possible severe and fatal immune-mediated adverse reactions.
Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue and can affect more than one body system simultaneously. Immune-mediated adverse reactions can occur at any time after starting treatment with a PD-1/PD-L1 blocking antibody. While immune-mediated adverse reactions usually manifest during treatment with PD-1/PD-L1 blocking antibodies, immune-mediated adverse reactions can also manifest after discontinuation of PD-1/PD-L1 blocking antibodies.
Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of PD-1/PD-L1 blocking antibodies. Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. For patients with TNBC treated with KEYTRUDA QLEX in the neoadjuvant setting, monitor blood cortisol at baseline, prior to surgery, and as clinically indicated. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)]. In general, if KEYTRUDA QLEX requires interruption or discontinuation, administer systemic corticosteroid therapy (1 to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose immune-mediated adverse reactions are not controlled with corticosteroid therapy.
Toxicity management guidelines for adverse reactions that do not necessarily require systemic steroids (e.g., endocrinopathies and dermatologic reactions) are discussed below.
Immune-Mediated Pneumonitis
KEYTRUDA QLEX can cause immune-mediated pneumonitis. The incidence of pneumonitis is higher in patients who have received prior thoracic radiation. Immune-mediated pneumonitis occurred in 5% (13/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including fatal (0.4%), Grade 3 (2%), and Grade 2 (1.2%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated pneumonitis occurred in 3.4% (94/2799) of patients receiving intravenous pembrolizumab, including fatal (0.1%), Grade 4 (0.3%), Grade 3 (0.9%), and Grade 2 (1.3%) adverse reactions. Systemic corticosteroids were required in 67% (63/94) of patients with pneumonitis. Pneumonitis led to permanent discontinuation of intravenous pembrolizumab in 1.3% (36) of patients and withholding of intravenous pembrolizumab in 0.9% (26) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, 23% had recurrence of pneumonitis. Pneumonitis resolved in 59% of the 94 patients.
In a clinical study enrolling 580 adult patients with resected NSCLC (KEYNOTE-091) who received intravenous pembrolizumab as a single agent for adjuvant treatment, pneumonitis occurred in 41 (7%) patients, including fatal (0.2%), Grade 4 (0.3%), and Grade 3 (1%) adverse reactions. Patients received high-dose corticosteroids for a median duration of 10 days (range: 1 day to 2.3 months). Pneumonitis led to discontinuation of intravenous pembrolizumab in 26 (4.5%) of patients. Of the patients who developed pneumonitis, 54% interrupted intravenous pembrolizumab, 63% discontinued intravenous pembrolizumab, and 71% had resolution.
Immune-Mediated Colitis
KEYTRUDA QLEX can cause immune-mediated colitis, which may present with diarrhea. Cytomegalovirus (CMV) infection/reactivation has been reported in patients with corticosteroid-refractory immune-mediated colitis. In cases of corticosteroid-refractory colitis, consider repeating infectious workup to exclude alternative etiologies. Immune-mediated colitis occurred in 1.2% (3/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 3 (0.8%), and Grade 2 (0.4%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated colitis occurred in 1.7% (48/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (1.1%), and Grade 2 (0.4%) adverse reactions. Systemic corticosteroids were required in 69% (33/48) of patients with colitis. Additional immunosuppressant therapy was required in 4.2% of patients. Colitis led to permanent discontinuation of intravenous pembrolizumab in 0.5% (15) of patients and withholding of intravenous pembrolizumab in 0.5% (13) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, 23% had recurrence of colitis. Colitis resolved in 85% of the 48 patients.
Hepatotoxicity and Immune-Mediated Hepatitis
KEYTRUDA QLEX can cause immune-mediated hepatitis. Immune-mediated hepatitis occurred in 0.4% (1/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (0.4%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated hepatitis occurred in 0.7% (19/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.4%), and Grade 2 (0.1%) adverse reactions. Systemic corticosteroids were required in 68% (13/19) of patients with hepatitis. Eleven percent of these patients required additional immunosuppressant therapy. Hepatitis led to permanent discontinuation of intravenous pembrolizumab in 0.2% (6) of patients and withholding of intravenous pembrolizumab in 0.3% (9) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, none had recurrence of hepatitis. Hepatitis resolved in 79% of the 19 patients.
In Combination with Axitinib
KEYTRUDA QLEX in combination with axitinib can cause hepatic toxicity with higher than expected frequencies of Grades 3 and 4 ALT and AST elevations compared to KEYTRUDA QLEX alone. Monitor liver enzymes before initiation of and periodically throughout treatment. Consider more frequent monitoring of liver enzymes as compared to when the drugs are administered as single agents. For elevated liver enzymes, interrupt KEYTRUDA QLEX and axitinib, and consider administering corticosteroids as needed [see Dosage and Administration (2.4)].
Intravenous Pembrolizumab in Combination with Axitinib
With the combination of intravenous pembrolizumab and axitinib, Grades 3 and 4 increased ALT (20%) and increased AST (13%) were seen. Fifty-nine percent of the patients with increased ALT received systemic corticosteroids. In patients with ALT ≥3 times ULN (Grades 2-4, n=116), ALT resolved to Grades 0-1 in 94%. Among the 92 patients who were rechallenged with either intravenous pembrolizumab (n=3) or axitinib (n=34) administered as a single agent or with both (n=55), recurrence of ALT ≥3 times ULN was observed in 1 patient receiving intravenous pembrolizumab, 16 patients receiving axitinib, and 24 patients receiving both intravenous pembrolizumab and axitinib. All patients with a recurrence of ALT ≥3 ULN subsequently recovered from the event.
Immune-Mediated Endocrinopathies
Adrenal Insufficiency
KEYTRUDA QLEX can cause primary or secondary adrenal insufficiency. For Grade 2 or higher adrenal insufficiency, initiate symptomatic treatment, including hormone replacement as clinically indicated. Withhold KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Adrenal insufficiency occurred in 2% (5/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 3 (0.4%), and Grade 2 (0.8%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Adrenal insufficiency occurred in 0.8% (22/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.3%), and Grade 2 (0.3%) adverse reactions. Systemic corticosteroids were required in 77% (17/22) of patients with adrenal insufficiency; of these, the majority remained on systemic corticosteroids. Adrenal insufficiency led to permanent discontinuation of intravenous pembrolizumab in <0.1% (1) of patients and withholding of intravenous pembrolizumab in 0.3% (8) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement.
Hypophysitis
KEYTRUDA QLEX can cause immune-mediated hypophysitis. Hypophysitis can present with acute symptoms associated with mass effect such as headache, photophobia, or visual field defects. Hypophysitis can cause hypopituitarism. Initiate hormone replacement as indicated. Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Intravenous Pembrolizumab as a Single Agent
Hypophysitis occurred in 0.6% (17/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.3%), and Grade 2 (0.2%) adverse reactions. Systemic corticosteroids were required in 94% (16/17) of patients with hypophysitis; of these, the majority remained on systemic corticosteroids. Hypophysitis led to permanent discontinuation of intravenous pembrolizumab in 0.1% (4) of patients and withholding of intravenous pembrolizumab in 0.3% (7) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement.
Thyroid Disorders
KEYTRUDA QLEX can cause immune-mediated thyroid disorders. Thyroiditis can present with or without endocrinopathy. Hypothyroidism can follow hyperthyroidism. Initiate hormone replacement for hypothyroidism or institute medical management of hyperthyroidism as clinically indicated. Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Thyroiditis occurred in 0.4% (1/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (0.4%). Hyperthyroidism occurred in 8% (20/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (3.2%). Hypothyroidism occurred in 14% (35/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 2 (11%).
Intravenous Pembrolizumab as a Single Agent
Thyroiditis occurred in 0.6% (16/2799) of patients receiving intravenous pembrolizumab, including Grade 2 (0.3%). No patients discontinued intravenous pembrolizumab due to thyroiditis. Intravenous pembrolizumab was withheld in <0.1% (1) of patients.
Hyperthyroidism occurred in 3.4% (96/2799) of patients receiving intravenous pembrolizumab, including Grade 3 (0.1%) and Grade 2 (0.8%). Hyperthyroidism led to permanent discontinuation of intravenous pembrolizumab in <0.1% (2) of patients and withholding of intravenous pembrolizumab in 0.3% (7) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement.
The incidence of new or worsening hyperthyroidism was higher in 580 patients with resected NSCLC, occurring in 11% of patients receiving intravenous pembrolizumab as a single agent as adjuvant treatment (KEYNOTE-091), including Grade 3 (0.2%) hyperthyroidism.
Hypothyroidism occurred in 8% (237/2799) of patients receiving intravenous pembrolizumab, including Grade 3 (0.1%) and Grade 2 (6.2%). Hypothyroidism led to permanent discontinuation of intravenous pembrolizumab in <0.1% (1) of patients and withholding of intravenous pembrolizumab in 0.5% (14) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement. The majority of patients with hypothyroidism required long-term thyroid hormone replacement.
The incidence of new or worsening hypothyroidism was higher in 1185 patients with HNSCC, occurring in 16% of patients receiving intravenous pembrolizumab as a single agent or in combination with platinum and FU, including Grade 3 (0.3%) hypothyroidism.
The incidence of new or worsening hypothyroidism was higher in 580 patients with resected NSCLC, occurring in 22% of patients receiving intravenous pembrolizumab as a single agent as adjuvant treatment (KEYNOTE-091), including Grade 3 (0.3%) hypothyroidism.
Type 1 Diabetes Mellitus, which can present with Diabetic Ketoacidosis
Monitor patients for hyperglycemia or other signs and symptoms of diabetes. Initiate treatment with insulin as clinically indicated. Withhold KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Type 1 diabetes mellitus occurred in 0.4% (1/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy.
Intravenous Pembrolizumab as a Single Agent
Type 1 diabetes mellitus occurred in 0.2% (6/2799) of patients receiving intravenous pembrolizumab. Type 1 diabetes mellitus led to permanent discontinuation in <0.1% (1) of patients and withholding of intravenous pembrolizumab in <0.1% (1) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement. All patients with Type 1 diabetes mellitus required long-term insulin therapy.
Immune-Mediated Nephritis with Renal Dysfunction
KEYTRUDA QLEX can cause immune-mediated nephritis.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated nephritis occurred in 0.3% (9/2799) of patients receiving intravenous pembrolizumab, including Grade 4 (<0.1%), Grade 3 (0.1%), and Grade 2 (0.1%) adverse reactions. Systemic corticosteroids were required in 89% (8/9) of patients with nephritis. Nephritis led to permanent discontinuation of intravenous pembrolizumab in 0.1% (3) of patients and withholding of intravenous pembrolizumab in 0.1% (3) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, none had recurrence of nephritis. Nephritis resolved in 56% of the 9 patients.
Immune-Mediated Dermatologic Adverse Reactions
KEYTRUDA QLEX can cause immune-mediated rash or dermatitis. Exfoliative dermatitis, including Stevens Johnson Syndrome, DRESS, and toxic epidermal necrolysis (TEN), has occurred with PD-1/PD-L1 blocking antibodies. Topical emollients and/or topical corticosteroids may be adequate to treat mild to moderate non-exfoliative rashes. Withhold or permanently discontinue KEYTRUDA QLEX depending on severity [see Dosage and Administration (2.4)].
Immune-mediated dermatologic adverse reactions occurred in 1.6% (4/251) of patients receiving KEYTRUDA QLEX in combination with chemotherapy, including Grade 4 (0.8%), and Grade 3 (0.8%) adverse reactions.
Intravenous Pembrolizumab as a Single Agent
Immune-mediated dermatologic adverse reactions occurred in 1.4% (38/2799) of patients receiving intravenous pembrolizumab, including Grade 3 (1%) and Grade 2 (0.1%) adverse reactions. Systemic corticosteroids were required in 40% (15/38) of patients with immune-mediated dermatologic adverse reactions. Immune-mediated dermatologic adverse reactions led to permanent discontinuation of intravenous pembrolizumab in 0.1% (2) of patients and withholding of intravenous pembrolizumab in 0.6% (16) of patients. All patients who were withheld reinitiated intravenous pembrolizumab after symptom improvement; of these, 6% had recurrence of immune-mediated dermatologic adverse reactions. Immune-mediated dermatologic adverse reactions resolved in 79% of the 38 patients.
Other Immune-Mediated Adverse Reactions
The following clinically significant immune-mediated adverse reactions occurred at an incidence of <1% (unless otherwise noted) in patients who received KEYTRUDA QLEX, intravenous pembrolizumab, or were reported with the use of other PD-1/PD-L1 blocking antibodies. Severe or fatal cases have been reported for some of these adverse reactions.
Cardiac/Vascular: Myocarditis, pericarditis, vasculitis
Nervous System: Meningitis, encephalitis, myelitis and demyelination, myasthenic syndrome/myasthenia gravis (including exacerbation), Guillain-Barré syndrome, nerve paresis, autoimmune neuropathy
Ocular: Uveitis, iritis and other ocular inflammatory toxicities can occur. Some cases can be associated with retinal detachment. Various grades of visual impairment, including blindness, can occur. If uveitis occurs in combination with other immune-mediated adverse reactions, consider a Vogt-Koyanagi-Harada-like syndrome, as this may require treatment with systemic steroids to reduce the risk of permanent vision loss.
Gastrointestinal: Pancreatitis, to include increases in serum amylase and lipase levels, gastritis (2.8%), duodenitis
Musculoskeletal and Connective Tissue: Myositis/polymyositis, rhabdomyolysis (and associated sequelae, including renal failure), arthritis (1.5%), polymyalgia rheumatica
Endocrine: Hypoparathyroidism
Hematologic/Immune: Hemolytic anemia, aplastic anemia, hemophagocytic lymphohistiocytosis, systemic inflammatory response syndrome, histiocytic necrotizing lymphadenitis (Kikuchi lymphadenitis), sarcoidosis, immune thrombocytopenic purpura, solid organ transplant rejection, other transplant (including corneal graft) rejection
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
KEYTRUDA QLEX contains pembrolizumab and berahyaluronidase alfa.
Pembrolizumab
No studies have been performed to test the potential of pembrolizumab for carcinogenicity or genotoxicity.
Fertility studies have not been conducted with pembrolizumab. In 1-month and 6-month repeat-dose toxicology studies in monkeys, there were no notable effects in the male and female reproductive organs; however, most animals in these studies were not sexually mature.
Berahyaluronidase alfa
Hyaluronidases are found in most tissues of the body. Long-term animal studies have not been performed to assess the carcinogenic or mutagenic potential of berahyaluronidase alfa.
In a fertility and early embryonic development study, male and female rats were administered daily subcutaneous injections of 280,000, 840,000, or 2,520,000 U/kg berahyaluronidase alfa. Males were dosed for 9 weeks prior to mating and throughout mating to termination. Females were dosed for 2 weeks prior to mating, throughout mating, and up to gestation day 7. Although treatment with ≥840,000 U/kg berahyaluronidase alfa (>5,200 times higher than the human dose) resulted in an increased incidence of abnormal sperm morphology, there were no adverse effects on mating, fertility or embryogenesis observed at doses up to 2,520,000 U/kg (>15,000 times higher than the human dose).
5.2 Hypersensitivity and Administration Related Reactions (5.2 Hypersensitivity and Administration-Related Reactions)
KEYTRUDA QLEX can cause severe or life-threatening administration-related reactions, including hypersensitivity and anaphylaxis. In Study MK-3475A-D77, hypersensitivity and administration-related systemic reactions occurred in 3.2% (8/251) of patients receiving KEYTRUDA QLEX, including Grade 2 (2.8%). Monitor patients for signs and symptoms of administration-related systemic reactions including rigors, chills, wheezing, pruritus, flushing, rash, hypotension, hypoxemia, and fever. Interrupt injection (if not already fully administered) and resume if symptoms resolve for mild or moderate hypersensitivity and administration-related systemic reactions. For severe or life-threatening hypersensitivity and administration-related systemic reactions, stop injection and permanently discontinue KEYTRUDA QLEX [see Dosage and Administration (2.4)].
Principal Display Panel 395 Mg and 4,800 Units/2.4 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 395 mg and 4,800 units/2.4 mL Vial Carton)
NDC 0006-3083-01
Keytruda Qlex ™
(pembrolizumab and
berahyaluronidase alfa-pmph) Injection
395 mg and 4,800 units/2.4 mL
(165 mg and 2,000 units/mL)
For Subcutaneous Use Only
Administer subcutaneous injection
over 1 minute
Dispense the enclosed Medication Guide to each patient.
One Single-dose vial. Discard unused portion.
Rx only
Principal Display Panel 790 Mg and 9,600 Units/4.8 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 790 mg and 9,600 units/4.8 mL Vial Carton)
NDC 0006-5083-01
Keytruda Qlex ™
(pembrolizumab and
berahyaluronidase alfa-pmph) Injection
790 mg and 9,600 units/4.8 mL
(165 mg and 2,000 units/mL)
For Subcutaneous Use Only
Administer subcutaneous injection
over 2 minutes
Dispense the enclosed Medication Guide to each patient.
One Single-dose vial. Discard unused portion.
Rx only
1.6 Microsatellite Instability High Or Mismatch Repair Deficient Cancer (1.6 Microsatellite Instability-High or Mismatch Repair Deficient Cancer)
KEYTRUDA QLEX is indicated for the treatment of adult and pediatric patients 12 years and older with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-authorized test, that have progressed following prior treatment and who have no satisfactory alternative treatment options [see Dosage and Administration (2.1)].
14.7 Microsatellite Instability High Or Mismatch Repair Deficient Cancer (14.7 Microsatellite Instability-High or Mismatch Repair Deficient Cancer)
The efficacy of intravenous pembrolizumab was investigated in 504 patients with MSI-H or dMMR cancers enrolled in three multicenter, non-randomized, open-label, multi-cohort trials: KEYNOTE-164 (NCT02460198), KEYNOTE-158 (NCT02628067), and KEYNOTE-051 (NCT02332668). All trials excluded patients with autoimmune disease or a medical condition that required immunosuppression. Regardless of histology, MSI or MMR tumor status was determined using polymerase chain reaction (PCR; local or central) or immunohistochemistry (IHC; local or central), respectively.
- KEYNOTE-164 enrolled 124 patients with advanced MSI-H or dMMR colorectal cancer (CRC) that progressed following treatment with fluoropyrimidine and either oxaliplatin or irinotecan +/- anti-VEGF/EGFR mAb-based therapy.
- KEYNOTE-158 enrolled 373 patients with advanced MSI-H or dMMR non-colorectal cancers (non-CRC) who had disease progression following prior therapy. Patients were either prospectively enrolled with MSI-H/dMMR tumors (Cohort K) or retrospectively identified in one of 10 solid tumor cohorts (Cohorts A-J).
- KEYNOTE-051 enrolled 7 pediatric patients with MSI-H or dMMR cancers.
Adult patients received intravenous pembrolizumab 200 mg every 3 weeks (pediatric patients received 2 mg/kg every 3 weeks) until unacceptable toxicity, disease progression, or a maximum of 24 months. In KEYNOTE-164 and KEYNOTE-158, assessment of tumor status was performed every 9 weeks through the first year, then every 12 weeks thereafter. In KEYNOTE-051, assessment of tumor status was performed every 8 weeks for 24 weeks, and then every 12 weeks thereafter. The major efficacy outcome measures were ORR and DoR as assessed by BICR according to RECIST v1.1 (modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ in KEYNOTE-158) and as assessed by the investigator according to RECIST v1.1 in KEYNOTE-051.
In KEYNOTE-164 and KEYNOTE-158, the study population characteristics were median age of 60 years, 36% age 65 or older; 44% male; 78% White, 14% Asian, 4% American Indian or Alaska Native, and 3% Black; and 45% ECOG PS of 0 and 55% ECOG PS of 1. Ninety-two percent of patients had metastatic disease and 4% had locally advanced, unresectable disease. Thirty-seven percent of patients received one prior line of therapy and 61% received two or more prior lines of therapy.
In KEYNOTE-051, the study population characteristics were median age of 11 years (range: 3 to 16); 71% female; 86% White and 14% Asian; and 57% had a Lansky/Karnofsky Score of 100. Seventy-one percent of patients had Stage IV and 14% had Stage III disease. Fifty-seven percent of patients received one prior line of therapy and 29% received two prior lines of therapy.
Discordant results were observed between local MSI-H or dMMR tests and central testing among patients enrolled in Cohort K of KEYNOTE-158. Among 104 tumor samples that were MSI-H or dMMR by local testing and also tested using the FoundationOne®CDx (F1CDx) test, 59 (56.7%) were MSI-H and 45 (43.3%) were not MSI-H. Among 169 tumor samples that were MSI-H or dMMR by local testing and also tested using the VENTANA MMR RxDx Panel, 105 (62.1%) were dMMR and 64 (37.9%) were pMMR.
Efficacy results are summarized in Tables 79 and 80.
| Endpoint | Intravenous Pembrolizumab n=504 Median follow-up time of 20.1 months (range 0.1 to 71.4 months)
|
|---|---|
| + Denotes ongoing response | |
| Objective Response Rate | |
| ORR (95% CI) Of the 7 pediatric patients from KEYNOTE-051, 1 patient had a radiographic complete response after initial growth of their tumor but is not reflected in the results.
|
33.3% (29.2, 37.6) |
| Complete response rate | 10.3% |
| Partial response rate | 23.0% |
| Duration of Response | n=168 |
| Median in months (range) | 63.2 (1.9+, 63.9+) |
| % with duration ≥12 months | 77% |
| % with duration ≥36 months | 39% |
| Objective Response Rate | Duration of Response range |
|||
|---|---|---|---|---|
| N | n (%) | 95% CI | (months) | |
| + Denotes ongoing response | ||||
| CRC | 124 | 42 (34%) | (26%, 43%) | (4.4, 58.5+) |
|
Non-CRC Results include patients in Cohort K of KEYNOTE-158 that were later determined to be pMMR or not MSI-H by central testing
|
380 | 126 (33%) | (28%, 38%) | (1.9+, 63.9+) |
| Endometrial cancer | 94 | 47 (50%) | (40%, 61%) | (2.9, 63.2) |
| Gastric or GE junction cancer | 51 | 20 (39%) | (26%, 54%) | (1.9+, 63.0+) |
| Small intestinal cancer | 27 | 16 (59%) | (39%, 78%) | (3.7+, 57.3+) |
| Brain cancer | 27 Includes 6 pediatric patients with brain cancer
|
1 (4%) In addition to the 1 adult responder, 1 pediatric patient had a radiographic complete response after initial growth of their tumor.
|
(0%, 19%) | 18.9 |
| Ovarian cancer | 25 | 8 (32%) | (15%, 54%) | (4.2, 56.6+) |
| Biliary cancer | 22 | 9 (41%) | (21%, 64%) | (6.2, 49.0+) |
| Pancreatic cancer | 22 | 4 (18%) | (5%, 40%) | (8.1, 24.3+) |
| Sarcoma | 14 | 3 (21%) | (5%, 51%) | (35.4+, 57.2+) |
| Breast cancer | 13 | 1 (8%) | (0%,36%) | 24.3+ |
| Other Includes tumor type (n): anal (3), HNSCC (1), nasopharyngeal (1), retroperitoneal (1), testicular (1), vaginal (1), vulvar (1), appendiceal adenocarcinoma, NOS (1), hepatocellular carcinoma (1), and carcinoma of unknown origin (1). Includes 1 pediatric patient with abdominal adenocarcinoma.
|
13 | 4 (31%) | (9%, 61%) | (6.2+, 32.3+) |
| Cervical cancer | 11 | 1 (9%) | (0%, 41%) | 63.9+ |
| Neuroendocrine cancer | 11 | 1 (9%) | (0%, 41%) | 13.3 |
| Prostate cancer | 8 | 1 (13%) | (0%, 53%) | 24.5+ |
| Adrenocortical cancer | 7 | 1 (14%) | (0%, 58%) | 4.2 |
| Mesothelioma | 7 | 0 (0%) | (0%, 41%) | |
| Thyroid cancer | 7 | 1 (14%) | (0%, 58%) | 8.2 |
| Small cell lung cancer | 6 | 2 (33%) | (4%, 78%) | (20.0, 47.5) |
| Bladder cancer | 6 | 3 (50%) | (12%, 88%) | (35.6+, 57.5+) |
| Salivary cancer | 5 | 2 (40%) | (5%, 85%) | (42.6+, 57.8+) |
| Renal cell cancer | 4 | 1 (25%) | (0%, 81%) | 22.0 |
Exploratory analysis by TMB
In an exploratory analysis performed in 138 patients (Cohort K of KEYNOTE-158) who were tested retrospectively for tumor mutation burden (TMB) using an FDA-authorized test, 45 (33%) had tumors with TMB score of <10 mut/Mb; ORR in these 45 patients was 6.7% (95% CI: 1.4, 18.3). Among the 45 patients with TMB score of <10 mut/Mb, 39 of the patients were pMMR/not MSI-H when tested using an FDA-authorized test.
1.7 Microsatellite Instability High Or Mismatch Repair Deficient Colorectal Cancer (1.7 Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer)
KEYTRUDA QLEX is indicated for the treatment of adult patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-authorized test [see Dosage and Administration (2.1)].
14.8 Microsatellite Instability High Or Mismatch Repair Deficient Colorectal Cancer (14.8 Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer)
The efficacy of intravenous pembrolizumab was investigated in KEYNOTE-177 (NCT02563002), a multicenter, randomized, open-label, active-controlled trial that enrolled 307 patients with previously untreated unresectable or metastatic MSI-H or dMMR CRC. MSI or MMR tumor status was determined locally using polymerase chain reaction (PCR) or immunohistochemistry (IHC), respectively. Patients with autoimmune disease or a medical condition that required immunosuppression were ineligible.
Patients were randomized (1:1) to receive intravenous pembrolizumab 200 mg intravenously every 3 weeks or investigator’s choice of the following chemotherapy regimens given intravenously every 2 weeks:
- mFOLFOX6 (oxaliplatin, leucovorin, and FU) or mFOLFOX6 in combination with either bevacizumab or cetuximab: Oxaliplatin 85 mg/m2, leucovorin 400 mg/m2 (or levoleucovorin 200 mg/m2), and FU 400 mg/m2 bolus on Day 1, then FU 2400 mg/m2 over 46-48 hours. Bevacizumab 5 mg/kg on Day 1 or cetuximab 400 mg/m2 on first infusion, then 250 mg/m2 weekly.
- FOLFIRI (irinotecan, leucovorin, and FU) or FOLFIRI in combination with either bevacizumab or cetuximab: Irinotecan 180 mg/m2, leucovorin 400 mg/m2 (or levoleucovorin 200 mg/m2), and FU 400 mg/m2 bolus on Day 1, then FU 2400 mg/m2 over 46-48 hours. Bevacizumab 5 mg/kg on Day 1 or cetuximab 400 mg/m2 on first infusion, then 250 mg/m2 weekly.
Treatment with intravenous pembrolizumab or chemotherapy continued until RECIST v1.1-defined progression of disease as determined by the investigator or unacceptable toxicity. Patients treated with intravenous pembrolizumab without disease progression could be treated for up to 24 months. Assessment of tumor status was performed every 9 weeks. Patients randomized to chemotherapy were offered intravenous pembrolizumab at the time of disease progression. The main efficacy outcome measures were PFS (as assessed by BICR according to RECIST v1.1, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ) and OS. Additional efficacy outcome measures were ORR and DoR.
A total of 307 patients were enrolled and randomized to intravenous pembrolizumab (n=153) or chemotherapy (n=154). The baseline characteristics of these 307 patients were: median age of 63 years (range: 24 to 93), 47% age 65 or older; 50% male; 75% White and 16% Asian; 52% had an ECOG PS of 0 and 48% had an ECOG PS of 1; and 27% received prior adjuvant or neoadjuvant chemotherapy. Among 154 patients randomized to receive chemotherapy,143 received chemotherapy per the protocol. Of the 143 patients, 56% received mFOLFOX6, 44% received FOLFIRI, 70% received bevacizumab plus mFOLFOX6 or FOLFIRI, and 11% received cetuximab plus mFOLFOX6 or FOLFIRI.
The trial demonstrated a statistically significant improvement in PFS for patients randomized to intravenous pembrolizumab compared with chemotherapy. There was no statistically significant difference between intravenous pembrolizumab and chemotherapy in the final OS analysis. Sixty percent of the patients who had been randomized to receive chemotherapy had crossed over to receive subsequent anti-PD-1/PD-L1 therapies including intravenous pembrolizumab. The median follow-up time at the final analysis was 38.1 months (range: 0.2 to 58.7 months). Table 81 and Figure 21 summarize the key efficacy measures for KEYNOTE-177.
| Endpoint | Intravenous Pembrolizumab 200 mg every 3 weeks n=153 |
Chemotherapy n=154 |
|---|---|---|
| + Denotes ongoing response NR = not reached |
||
| PFS | ||
| Number (%) of patients with event | 82 (54%) | 113 (73%) |
| Median in months (95% CI) | 16.5 (5.4, 32.4) | 8.2 (6.1, 10.2) |
| Hazard ratio
Based on Cox regression model
(95% CI) |
0.60 (0.45, 0.80) | |
| p-Value Two-sided p-Value based on log-rank test (compared to a significance level of 0.0234)
|
0.0004 | |
|
OS Final OS analysis
|
||
| Number (%) of patients with event | 62 (41%) | 78 (51%) |
| Median in months (95% CI) | NR (49.2, NR) | 36.7 (27.6, NR) |
| Hazard ratio (95% CI) | 0.74 (0.53, 1.03) | |
| p-Value Two-sided p-Value based on log-rank test (compared to a significance level of 0.0492)
|
0.0718 | |
|
Objective Response Rate
Based on confirmed response by BICR review
|
||
| ORR (95% CI) | 44% (35.8, 52.0) | 33% (25.8, 41.1) |
| Complete response rate | 11% | 4% |
| Partial response rate | 33% | 29% |
|
Duration of Response
,
Based on n=67 patients with a response in the intravenous pembrolizumab arm and n=51 patients with a response in the chemotherapy arm
|
||
| Median in months (range) | NR (2.3+, 41.4+) | 10.6 (2.8, 37.5+) |
| % with duration ≥12 months Based on observed duration of response
|
75% | 37% |
| % with duration ≥24 months | 43% | 18% |
|
|
5.4 Increased Mortality in Patients With Multiple Myeloma When Pembrolizumab Is Added to A Thalidomide Analogue and Dexamethasone (5.4 Increased Mortality in Patients with Multiple Myeloma when Pembrolizumab is Added to a Thalidomide Analogue and Dexamethasone)
In two randomized trials in patients with multiple myeloma, the addition of intravenous pembrolizumab to a thalidomide analogue plus dexamethasone, a use for which no PD-1 or PD-L1 blocking antibody is indicated, resulted in increased mortality. Treatment of patients with multiple myeloma with a PD-1 or PD-L1 blocking antibody in combination with a thalidomide analogue plus dexamethasone is not recommended outside of controlled trials.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:44.317445 · Updated: 2026-03-14T22:51:58.103963