These Highlights Do Not Include All The Information Needed To Use Bylvay Safely And Effectively. See Full Prescribing Information For Bylvay.
0935cd31-3251-4a0a-ac23-25ca4be12be9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Contraindications ( 4 ) 03/2025 Warnings and Precautions ( 5.1 ) 03/2025 Warnings and Precautions ( 5.3 ) 12/2025
Indications and Usage
BYLVAY is an ileal bile acid transporter (IBAT) inhibitor indicated for: Progressive Familial Intrahepatic Cholestasis (PFIC) the treatment of pruritus in patients 3 months of age and older with progressive familial intrahepatic cholestasis (PFIC). ( 1.1 ) Limitation of Use : BYLVAY is not recommended in a subgroup of PFIC type 2 patients with specific ABCB11 variants resulting in non-functional or complete absence of the bile salt export pump protein. ( 12.5 , 14.1 ) Alagille Syndrome (ALGS) the treatment of cholestatic pruritus in patients 12 months of age and older with Alagille syndrome (ALGS). ( 1.2 )
Dosage and Administration
Recommended Dosage PFIC: Patients 3 months and older: 40 mcg/kg taken orally once daily. ( 2.1 ) If there is no improvement in pruritus after 3 months, the dosage may be increased in 40 mcg/kg increments up to 120 mcg/kg once daily, not to exceed a daily dosage of 6 mg/day. ( 2.1 ) ALGS: Patients 12 months and older: 120 mcg/kg taken orally once daily. ( 2.2 ) Preparation and Administration Instructions Administer BYLVAY in the morning with a meal. ( 2.4 ) Do not crush or chew capsules. ( 2.4 ) See full prescribing information for preparation and administration instructions. ( 2.4 )
Warnings and Precautions
Hepatoxicity : Obtain baseline liver tests and monitor patients frequently for the first 6 to 8 months after starting therapy, and as clinically indicated thereafter during treatment. If liver test abnormalities or signs of clinical hepatitis occur, consider dose reduction or treatment interruption. For persistent or recurrent liver test abnormalities relative to baseline, discontinue BYLVAY. Monitor patients with compensated cirrhosis or portal hypertension more frequently. Permanently discontinue BYLVAY if hepatic decompensation occurs. ( 2.3 , 5.1 ) Diarrhea : Treat dehydration. Treatment interruption or discontinuation may be required for persistent diarrhea. ( 5.2 ) Fat-Soluble Vitamin (FSV) Deficiency : Obtain baseline levels and monitor during treatment. Supplement with FSV if deficiency is observed. If FSV deficiency persists or worsens despite FSV supplementation, consider discontinuing BYLVAY treatment. Fracture: Consider interrupting BYLVAY treatment. Supplement with FSV if indicated. Bleeding: Interrupt treatment with BYLVAY. Optimize treatment of FSV deficiency and consider restarting BYLVAY once the patient is clinically stable. ( 5.3 )
Contraindications
IBAT inhibitors, including BYLVAY, are contraindicated in patients with prior or active hepatic decompensation events (e.g., variceal hemorrhage, ascites, hepatic encephalopathy) [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label: Hepatotoxicity [ see Warnings and Precautions (5.1) ] Diarrhea [see Warnings and Precautions (5.2) ] Fat-Soluble Vitamin Deficiency [see Warnings and Precautions (5.3) ]
Medication Information
Warnings and Precautions
Hepatoxicity : Obtain baseline liver tests and monitor patients frequently for the first 6 to 8 months after starting therapy, and as clinically indicated thereafter during treatment. If liver test abnormalities or signs of clinical hepatitis occur, consider dose reduction or treatment interruption. For persistent or recurrent liver test abnormalities relative to baseline, discontinue BYLVAY. Monitor patients with compensated cirrhosis or portal hypertension more frequently. Permanently discontinue BYLVAY if hepatic decompensation occurs. ( 2.3 , 5.1 ) Diarrhea : Treat dehydration. Treatment interruption or discontinuation may be required for persistent diarrhea. ( 5.2 ) Fat-Soluble Vitamin (FSV) Deficiency : Obtain baseline levels and monitor during treatment. Supplement with FSV if deficiency is observed. If FSV deficiency persists or worsens despite FSV supplementation, consider discontinuing BYLVAY treatment. Fracture: Consider interrupting BYLVAY treatment. Supplement with FSV if indicated. Bleeding: Interrupt treatment with BYLVAY. Optimize treatment of FSV deficiency and consider restarting BYLVAY once the patient is clinically stable. ( 5.3 )
Indications and Usage
BYLVAY is an ileal bile acid transporter (IBAT) inhibitor indicated for: Progressive Familial Intrahepatic Cholestasis (PFIC) the treatment of pruritus in patients 3 months of age and older with progressive familial intrahepatic cholestasis (PFIC). ( 1.1 ) Limitation of Use : BYLVAY is not recommended in a subgroup of PFIC type 2 patients with specific ABCB11 variants resulting in non-functional or complete absence of the bile salt export pump protein. ( 12.5 , 14.1 ) Alagille Syndrome (ALGS) the treatment of cholestatic pruritus in patients 12 months of age and older with Alagille syndrome (ALGS). ( 1.2 )
Dosage and Administration
Recommended Dosage PFIC: Patients 3 months and older: 40 mcg/kg taken orally once daily. ( 2.1 ) If there is no improvement in pruritus after 3 months, the dosage may be increased in 40 mcg/kg increments up to 120 mcg/kg once daily, not to exceed a daily dosage of 6 mg/day. ( 2.1 ) ALGS: Patients 12 months and older: 120 mcg/kg taken orally once daily. ( 2.2 ) Preparation and Administration Instructions Administer BYLVAY in the morning with a meal. ( 2.4 ) Do not crush or chew capsules. ( 2.4 ) See full prescribing information for preparation and administration instructions. ( 2.4 )
Contraindications
IBAT inhibitors, including BYLVAY, are contraindicated in patients with prior or active hepatic decompensation events (e.g., variceal hemorrhage, ascites, hepatic encephalopathy) [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label: Hepatotoxicity [ see Warnings and Precautions (5.1) ] Diarrhea [see Warnings and Precautions (5.2) ] Fat-Soluble Vitamin Deficiency [see Warnings and Precautions (5.3) ]
Description
Contraindications ( 4 ) 03/2025 Warnings and Precautions ( 5.1 ) 03/2025 Warnings and Precautions ( 5.3 ) 12/2025
Section 42229-5
Limitations of Use
- BYLVAY is not recommended in a subgroup of PFIC type 2 patients with specific ABCB11 variants resulting in non-functional or complete absence of the bile salt export pump (BSEP) protein [see Clinical Pharmacology (12.5) and Clinical Studies (14.1)].
Section 43683-2
Section 44425-7
Storage and Handling:
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (between 59°F and 86°F) [See USP Controlled Room Temperature].
14.1 Pfic
The efficacy of BYLVAY was evaluated in Trial 1 (NCT03566238), a 24-week, randomized, double-blind, placebo-controlled trial. Trial 1 was conducted in 62 pediatric patients, aged 6 months to 17 years, with a confirmed molecular diagnosis of PFIC type 1 or type 2, and presence of pruritus at baseline. Patients with variants in the ABCB11 gene that predict non-function or complete absence of the bile salt export pump (BSEP) protein, who had experienced prior hepatic decompensation events, who had other concomitant liver disease, whose INR was greater than 1.4, whose ALT or total bilirubin was greater than 10-times the upper limit of normal (ULN), or who had received a liver transplant were excluded in Trial 1.
Patients were randomized to placebo (n=20), 40 mcg/kg (n=23), or 120 mcg/kg (n=19). Study drug was administered once daily with a meal in the morning. In patients weighing less than 19.5 kg or patients who could not swallow the whole capsule, study drug was sprinkled on soft food and then administered orally.
Median age (range) of the patients in Trial 1 was 3.2 (0.5 to 15.9) years; 3 patients were older than 12 years of age. Of the 62 patients, 50% were male and 84% were white; 27% had PFIC type 1, and 73% had PFIC type 2. The mean (standard error [SE]) scratching score in the 2 weeks prior to baseline was 2.9 (0.08). Baseline mean (SE) eGFR was 164 (30.6) mL/min/1.73 m2. Baseline median (range) ALT, AST, and total bilirubin were 65 (16-798) U/L, 83.5 (32-405) U/L, and 2.2 (0.2-18.6) mg/dL, respectively.
In Trial 1, a total of 13 patients discontinued from trial prematurely either due to no improvement in pruritus (n=11) or due to adverse reactions (n=2); 5/20 (25%) patients discontinued from the placebo arm and 8/42 (19%) patients discontinued from the BYLVAY arms. A total of 11 of the 13 patients rolled over to Trial 2 to receive BYLVAY 120 mcg/kg/day. One patient treated with BYLVAY 120 mcg/kg/day withdrew from the trial due to an adverse reaction of diarrhea [see Adverse Reactions (6)].
Given the patients' young age, a single-item observer-reported outcome (ObsRO) was used to measure patients' scratching severity as observed by their caregiver twice daily (once in the morning and once in the evening). Scratching severity was assessed on a 5-point ordinal response scale, with scores ranging from 0 (no scratching) to 4 (worst possible scratching). Patients were included in Trial 1 if their average scratching score was greater than or equal to 2 (medium scratching) in the 2 weeks prior to baseline.
Table 5 presents the results of the comparison between BYLVAY and placebo on the mean of patients' percentage of ObsRO assessments over the 24-week treatment period that were scored as 0 (no scratching) or 1 (a little scratching). Patients treated with BYLVAY demonstrated greater improvement in pruritus compared with placebo. Figure 1 displays the mean of patients' worst weekly average scratching scores in each treatment group for each month, where the weekly average utilized the worst score from each day (morning or evening score).
| Placebo (n=20) |
BYLVAY | ||
|---|---|---|---|
| 40 mcg/kg/day (n=23) |
120 mcg/kg/day (n=19) |
||
|
Mean Based on least squares means from analysis of covariance model with daytime and nighttime baseline pruritus scores as covariates and treatment group and stratification factors (i.e., PFIC type and age category) as fixed effects. Percentage of Assessments Over the Treatment Period Scored as 0 (No Scratching) or 1 (A Little Scratching) (%)
|
|||
| Mean (SE) | 13.2 (8.7) | 35.4 (8.1) | 30.1 (9.0) |
| Mean Difference vs Placebo (95% CI) | 22.2 (4.7, 39.6) | 16.9 (-2.0, 35.7) |
|
Figure 1: Mean Figure 1 presents least squares means of the Worst Weekly Average Scratching Scores for Each Month in Trial 1
Based on a mixed model repeated measure (MMRM) analysis accounting for baseline score, treatment group, time (in months), treatment-by-baseline interaction, treatment-by-time interaction, and stratification factors (i.e., PFIC type and age category). Missing data were accounted for using placebo-reference multiple imputation. |
|
|
14.2 Algs
The efficacy of BYLVAY was evaluated in Trial 3 (NCT04674761), a 24-week, randomized, double-blind, placebo-controlled trial. Trial 3 was conducted in 52 pediatric patients, aged 6 months to 15 years, with a confirmed diagnosis of ALGS and presence of pruritus at baseline. Patients who had decompensated liver disease, who had other concomitant liver disease, whose INR was greater than 1.4, whose ALT was greater than 10-times the upper limit of normal (ULN) at screening, whose total bilirubin was greater than 15-times the ULN at screening, or who had received a liver transplant were excluded from Trial 3.
Patients were randomized to placebo (n=17) or 120 mcg/kg (n=35). Study drug was administered once daily with a meal in the morning. In patients weighing less than 19.5 kg or patients who could not swallow the whole capsule, study drug was sprinkled on soft food and then administered orally.
Median age (range) of the patients in Trial 3 was 6.1 (1.7 to 15.5) years in the BYLVAY group and 4.2 (0.5 to 14.3) years in the placebo group; 5 patients were older than 12 years of age. Of the 52 patients, 52% were male and 83% were white; 92% of patients had the JAG1 mutation and 8% had the NOTCH2 mutation. The mean (standard deviation [SD]) scratching score in the 2 weeks prior to baseline was 2.9 (0.6). Baseline mean (SD) eGFR was 159 (51.4) mL/min/1.73 m2. Baseline median (range) ALT, AST, and total bilirubin were 152 (39-403) U/L, 135 (57-427) U/L, and 2.0 (0.4-11.4) mg/dL, respectively.
Given the patients' young ages, a single-item observer-reported outcome (ObsRO) was used to measure patients' scratching severity as observed by their caregiver twice daily (once in the morning and once in the evening). Scratching severity was assessed on a 5-point ordinal response scale, with scores ranging from 0 (no scratching) to 4 (worst possible scratching). Patients were included in Trial 3 if the average scratching score was greater than or equal to 2 (medium scratching) in the 14 days prior to baseline.
Table 6 presents the results of the comparison between BYLVAY and placebo on the change from baseline in average scratching score based on ObsRO assessments to Month 6 (Weeks 21 to 24). The average scratching score for each patient for each month post-baseline was calculated by: (Step 1) averaging the morning scores and averaging the evening scores within a week; (Step 2) averaging the morning and evening weekly scores to yield a single weekly score; and finally (Step 3) averaging the 4 weekly scores within the month. The baseline average scratching score for each patient was calculated by averaging the weekly scores obtained in Step 2 across the 2 weeks prior to randomization and initiation of blinded treatment. Patients treated with BYLVAY demonstrated greater improvement in pruritus compared with placebo. Figure 2 displays the means (95% confidence interval) of patients' average scratching scores in each treatment group for each month.
| Placebo (n=17) |
BYLVAY 120 mcg/kg/day (n=35) |
|
|---|---|---|
| Baseline Average Scratching Score | ||
| Mean (SD) | 3.0 (0.6) | 2.8 (0.5) |
|
Change from Baseline in Average Scratching Score to Month 6 (Weeks 21 to 24) Based on least square means from a mixed-effect model for repeated measures (MMRM) for change from baseline to each month accounting for baseline average scratching score, baseline age stratification (<10, ≥10 years), baseline direct bilirubin, treatment group, time (in months), and treatment-by-time interaction.
|
||
| Mean (SE) | -0.8 (0.2) | -1.7 (0.2) |
| Mean Difference vs Placebo (95% CI) p-value |
-0.9 (-1.4, -0.3) 0.002 |
|
Figure 2: Mean Figure 2 presents means for baseline and least squares means for Month 1 to 6 of the Average Scratching Scores for Each Month in Trial 3
Least squares means are based on a mixed model repeated measure (MMRM) analysis accounting for baseline average scratching score, baseline age stratification (<10, ≥10 years), baseline direct bilirubin, treatment group, time (in months), and treatment-by-time interaction. |
5.2 Diarrhea
In Trial 1, diarrhea in PFIC patients was reported in 2 (10%) placebo-treated patients, 9 (39%) BYLVAY-treated 40 mcg/kg/day patients and 4 (21%) BYLVAY-treated 120 mcg/kg/day patients. Treatment interruption due to diarrhea occurred in 2 patients with 3 events during treatment with BYLVAY 120 mcg/kg/day. Treatment interruption due to diarrhea ranged between 3 to 7 days [see Adverse Reactions (6.1)]. One patient treated with BYLVAY 120 mcg/kg/day withdrew from Trial 1 due to persistent diarrhea.
In Trial 3, diarrhea in ALGS patients was reported in 1 placebo-treated patient (6%) and in 10 (29%) BYLVAY-treated patients [see Adverse Reactions (6.1)]. No patients interrupted or permanently discontinued BYLVAY due to diarrhea.
If diarrhea occurs, monitor for dehydration and treat promptly. Interrupt BYLVAY dosing if a patient experiences persistent diarrhea. Restart BYLVAY at 40 mcg/kg/day when diarrhea resolves, and increase the dose as tolerated if appropriate. If diarrhea persists and no alternate etiology is identified, stop BYLVAY treatment.
11 Description
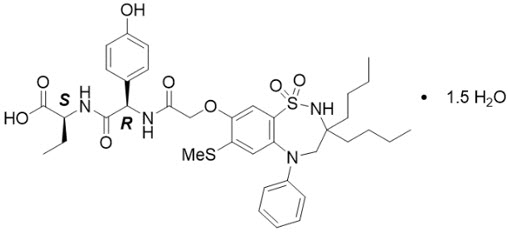
The active ingredient in BYLVAY (odevixibat) capsules and BYLVAY (odevixibat) oral pellets, an ileal bile acid transporter (IBAT) inhibitor, is (2S)-2-{[(2R)-2-(2-{[3,3-dibutyl-7-(methylsulfanyl)-1,1-dioxo-5-phenyl-2,3,4,5-tetrahydro-1H-1λ6,2,5-benzothiadiazepin-8yl]oxy}acetamido)-2-(4-hydroxyphenyl)acetly]amino}butanoic acid, which is formulated as the sesquihydrate having the following chemical structure:
The molecular formula is C37H48N4O8S2 × 1.5 H2O, with a molecular weight of 768.0 g/mol (anhydrous 740.9 g/mol). Odevixibat sesquihydrate is a white to off-white solid. Its solubility in aqueous solutions is pH-dependent and increases with increased pH.
BYLVAY is available for oral administration as oral pellets containing odevixibat sesquihydrate equivalent to 200 mcg or 600 mcg of odevixibat, and as capsules containing odevixibat sesquihydrate equivalent to 400 mcg or 1200 mcg of odevixibat, and the following excipients: hypromellose and microcrystalline cellulose.
The capsule shells for the oral pellets contain hypromellose, titanium dioxide and yellow iron oxide.
The capsule shells for the capsules contain hypromellose, red iron oxide, titanium dioxide and yellow iron oxide.
The imprinting ink contains ferrosoferric oxide/black iron oxide and shellac glaze.
5.1 Hepatoxicity
BYLVAY treatment is associated with a potential for drug-induced liver injury (DILI).
In the PFIC and ALGS trials, treatment-emergent elevations of liver tests or worsening of liver tests occurred. Of the six patients who experienced DILI, two underwent liver transplant.
Obtain baseline liver tests because some ALGS and PFIC patients have abnormal liver tests at baseline. Monitor patients frequently for the first 6 to 8 months after starting therapy and as clinically indicated thereafter during treatment with BYLVAY. Monitor for elevation in liver tests, for the development of liver-related adverse reactions, and for physical signs of hepatic decompensation. If liver test abnormalities or signs of clinical hepatitis occur in the absence of other causes, consider dose reduction or treatment interruption.
Permanently discontinue BYLVAY if a patient experiences the following:
- persistent or recurrent liver test abnormalities, or
- upon rechallenge, signs and symptoms consistent with clinical hepatitis, or
- a hepatic decompensation event.
The safety and effectiveness of BYLVAY have not been established in patients with decompensated cirrhosis. Monitor patients with compensated cirrhosis or portal hypertension more frequently and discontinue BYLVAY if hepatic decompensation occurs. IBAT inhibitors, including BYLVAY, are contraindicated in patients with prior or active hepatic decompensation events [see Contraindications (4)].
8.4 Pediatric Use
The safety and effectiveness of BYLVAY have been established in pediatric patients 3 months to 17 years of age for the treatment of pruritus in PFIC. Use of BYLVAY in this age group is supported by evidence from one randomized, double-blind, placebo-controlled trial conducted in 62 patients with a confirmed diagnosis of PFIC type 1 or type 2 (Trial 1), and an open-label extension trial in PFIC patients (Trial 2) [see Adverse Reactions (6.1) and Clinical Studies (14.1)].
The safety and effectiveness of BYLVAY for the treatment of pruritus in PFIC in pediatric patients less than 3 months of age have not been established.
The safety and effectiveness of BYLVAY have been established in pediatric patients 12 months to 17 years of age for the treatment of pruritus in ALGS. Use of BYLVAY in this age group is supported by evidence from one randomized, double-blind, placebo-controlled trial conducted in 52 patients with a confirmed diagnosis of ALGS (Trial 3) and one open-label extension trial in ALGS patients (Trial 4) [see Adverse Reactions (6.1) and Clinical Studies (14.2)].
The safety and effectiveness of BYLVAY for the treatment of pruritus in ALGS in pediatric patients less than 12 months of age have not been established.
8.5 Geriatric Use
PFIC and ALGS are largely diseases of pediatric and young adult patients. Clinical studies in BYLVAY did not include patients 65 years of age and older.
4 Contraindications
IBAT inhibitors, including BYLVAY, are contraindicated in patients with prior or active hepatic decompensation events (e.g., variceal hemorrhage, ascites, hepatic encephalopathy) [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following adverse reactions are discussed in greater detail in other sections of the label:
- Hepatotoxicity [see Warnings and Precautions (5.1) ]
- Diarrhea [see Warnings and Precautions (5.2)]
- Fat-Soluble Vitamin Deficiency [see Warnings and Precautions (5.3)]
Instructions for Use
This Instructions for Use contains information on how to give BYLVAY Capsules and Oral Pellets. This information does not take the place of talking to your healthcare provider about your child's medical condition or their treatment.
Important information you need to know before giving or taking BYLVAY
- Give BYLVAY along with the morning meal.
- Mix BYLVAY in a small amount of soft food (up to 2 tablespoons [30 mL]), such as apple sauce, oatmeal, banana or carrot puree, chocolate or rice pudding, in a bowl. You may also mix BYLVAY with an age-appropriate liquid and give through an oral syringe.
- If your child is taking bile acid binding resins (for example, cholestyramine, colestipol), give them BYLVAY at least 4 hours before or 4 hours after they take the bile acid binding resin.
Preparing to Give BYLVAY
You will be provided with the number of BYLVAY Capsules or Oral Pellets prescribed by your child's healthcare provider in a child-resistant closure.
Giving BYLVAY Oral Pellets with soft food:
- The shell containing Oral Pellets are to be opened and sprinkled. Do not let your child swallow the shell containing the Oral Pellets. Dispose of (throw away) the emptied shell.
- Mix the contents of the Oral Pellets with soft food as shown in Steps 1 through 9 below.
| Step 1. | Give BYLVAY with the first morning meal. Place a small amount of soft food (up to 2 tablespoons [30 mL], such as apple sauce, oatmeal, banana or carrot puree, chocolate or rice pudding) in a bowl. Keep the soft food at, or cooler than, room temperature. Note: This small amount of soft food should be less than what your child would normally eat. |
| Step 2. | Hold the shell containing Oral Pellets horizontally on both ends, twist in opposite directions and pull apart (see Figure A ). |
|
Figure A |
|
| Step 3. | Empty the Oral Pellets into the bowl of soft food (see Figure B ). |
|
Figure B |
|
| Step 4. | Gently tap the shell containing Oral Pellets to make sure that all pellets come out (see Figure C ). |
|
Figure C |
|
| Step 5. | If the dose requires more than 1 capsule shell, repeat Step 2 and Step 3. |
| Step 6. | Gently mix the Oral Pellets with a spoon into the soft food. Note that the Oral Pellets will not dissolve (see Figure D ). |
|
Figure D |
|
| Step 7. | Give the entire dose right away after mixing. Do not store the BYLVAY mixture for later use. |
| Step 8. | Give water or an age-appropriate liquid, such as breast milk or infant formula, after the dose is taken. |
| Step 9. | Dispose of (throw away) the empty Oral Pellet shells in the trash. |
Giving BYLVAY Oral Pellets with liquids (Using an oral dosing syringe):
- The shell containing the Oral Pellets are to be opened.
- Mix the Oral Pellets with liquid as shown in Steps 1 through 14 below.
- Dispose of (throw away) the emptied shells. Do not let your child swallow the unopened shells containing the Oral Pellets.
| Step 1. | Give BYLVAY with the first morning meal. |
| Step 2. | Hold the shell containing the Oral Pellets horizontally on both ends, twist in opposite directions and pull apart (see Figure A ). |
| Step 3. | Empty the Oral Pellets into a small mixing cup. Gently tap the Oral Pellet shell to ensure that all contents have been emptied into the mixing cup (see Figure E ). |
|
Figure E |
|
| Step 4. | If the dose requires more than 1 capsule shell, repeat Step 2 and Step 3. |
| Step 5. | Add 1 teaspoon (5 mL) of an age-appropriate liquid (for example, breast milk, infant formula, or water). |
| Step 6. | Let the pellets sit in the liquid for about 5 minutes to allow complete wetting. REMINDER: The pellets will not dissolve in the liquid. |
| Step 7. | After 5 minutes, place the tip of the oral syringe completely into the mixing cup. Pull the plunger of the syringe up slowly to withdraw the liquid and pellet mixture into the syringe. Gently push the plunger down again to expel the liquid and pellet mixture back into the mixing cup. Do this 2 to 3 times to ensure complete mixing of the pellets into the liquid. |
| Step 8. | Withdraw the entire contents into the syringe by pulling the plunger on the end of the syringe (see Figure F). |
|
Figure F |
|
| Step 9. | Place the tip of the syringe into the front of the child's mouth between the tongue and the side of the mouth, and then gently push the plunger down to squirt the liquid and pellet mixture between the tongue and the side of the mouth. Do not squirt the liquid and pellet mixture in the back of the throat because this could cause gagging or choking (see Figure G). |
|
Figure G |
|
| Step 10. | Do not give using a bottle or "sippy cup" because the Oral Pellets will not pass through the opening. The oral pellets will not dissolve in liquid. |
| Step 11. | Give water or an age-appropriate liquid such as breast milk or infant formula after the dose is taken. |
| Step 12. | Repeat Steps 8 and 9 until the entire dose has been given. |
| Step 13. | Check to make sure all of the liquid and pellet mixture has been swallowed. |
| Step 14. | Dispose of (throw away) the empty Oral Pellet shells in the trash. |
Taking BYLVAY Capsules
- Take BYLVAY Capsules along with your morning meal. Swallow BYLVAY Capsules whole with a glass of water. Do not chew or crush the Capsules.
- For children unable to swallow BYLVAY Capsules whole, follow instructions under Preparing to Give BYLVAY above.
How should I store BYLVAY Capsules or Oral Pellets?
Store BYLVAY at room temperature between 68°F to 77°F (20°C to 25°C).
Disposing (throwing away) of BYLVAY Capsules or Oral Pellets shells.
Dispose of (throw away) the empty BYLVAY Capsule or Oral Pellets shells in the household trash.
What are the ingredients in BYLVAY?
Active ingredient: odevixibat.
Inactive ingredients: hypromellose and microcrystalline cellulose.
Manufactured for:
Ipsen Biopharmaceuticals, Inc.
One Main Street,
Cambridge, MA 02142
© 2024 Ipsen Biopharmaceuticals, Inc. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 01/2024
12.2 Pharmacodynamics
Odevixibat reduces serum bile acids in patients with PFIC and ALGS.
In Trial 1, a 24-week, randomized, double-blind, placebo-controlled trial conducted in 62 patients with a confirmed diagnosis of PFIC type 1 or type 2, the majority of patients (88.7%) had elevated serum bile acids above 100 mcmol/L at baseline [see Clinical Studies (14.1)]. Serum bile acids concentrations were reduced from baseline within 4-8 weeks of odevixibat treatment compared to placebo treatment. The decreased concentrations of serum bile acids fluctuated over time but generally were maintained during the treatment over 24 weeks. The extent of decrease in serum bile acids was similar between 40 and 120 mcg/kg.
Trial 3 is a 24-week, randomized, double-blind, placebo-controlled trial conducted in 52 patients with a confirmed diagnosis of ALGS who were administered treatment with BYLVAY 120 mcg/kg once daily [see Clinical Studies (14.2)]. At baseline, serum bile acids were variable ranging from 93 to 510 mcmol/L. Serum bile acid concentrations were reduced from baseline as early as Week 4 of odevixibat treatment and the reduction was generally maintained during treatment over 24 weeks.
12.3 Pharmacokinetics
In pediatric patients with PFIC, 6 months to 17 years of age who received BYLVAY 40 mcg/kg or 120 mcg/kg once daily with food in the morning, the measurable odevixibat concentrations ranged from 0.06 to 0.72 ng/mL, and odevixibat concentrations were below the limit of quantification (0.05 ng/mL) in the majority of plasma samples.
In pediatric patients with ALGS who received BYLVAY 120 mcg/kg once daily with food in the morning, the measurable odevixibat concentrations ranged from 0.05 to 3.4 ng/mL.
Following single and repeated oral administration of odevixibat from 0.1 to 3 mg in healthy adults, plasma concentrations of odevixibat were mostly below the limit of quantification (0.05 ng/mL); therefore, AUC and peak plasma concentration (Cmax) could not be calculated.
Following a single administration of odevixibat 7.2 mg in healthy adults, the mean (%CV) Cmax and AUC0-24h were 0.47 ng/mL (34.8) and 2.19 ng∙h/mL (36.2), respectively. No accumulation of odevixibat was observed following once-daily dosing.
12.5 Pharmacogenomics
PFIC is a heterogenous disease caused by homozygous or compound heterozygous variants, with different PFIC subtypes occurring in the general population. PFIC1 is caused by variants in the ATP8B1 gene, which encodes FIC1. PFIC2 is caused by variants in the ABCB11 gene, which encodes BSEP. PFIC2 is further categorized into BSEP subgroups based on specific variants. The BSEP-1 subgroup includes patients with at least one p.D482G (c.1445A>G) or p.E297G (c.890A>G) variant, BSEP-2 includes patients with at least one missense variant other than p.D482G or p.E297G (non BSEP-1), and BSEP-3 includes patients with variants that are predicted to encode a non-functional protein. PFIC3 is caused by variants in the ABCB4 gene, which encodes MDR3. PFIC4 is caused by variants in the TJP2 gene, which encodes TJP2. PFIC6 is caused by variants in the MYO5B gene, which encodes MYO5B. Patients can be clinically diagnosed with PFIC without a known pathogenic variant.
PFIC2 is the most common subtype accounting for 37-90% of PFIC patients. The prevalence of BSEP-1, BSEP-2, and BSEP-3 subgroups are approximately 27%, 52%, and 21%, respectively, based on data from a global consortium characterizing the natural history of severe BSEP deficiency.
8.6 Hepatic Impairment
Patients with PFIC and ALGS may have impaired hepatic function. The efficacy and safety of BYLVAY in PFIC and ALGS patients with clinically significant portal hypertension, and in patients with decompensated cirrhosis have not been established [see Dosage and Administration (2.3), Contraindications (4), Warning and Precautions (5.1), and Clinical Studies (14)].
1 Indications and Usage
BYLVAY is an ileal bile acid transporter (IBAT) inhibitor indicated for:
Progressive Familial Intrahepatic Cholestasis (PFIC)
- the treatment of pruritus in patients 3 months of age and older with progressive familial intrahepatic cholestasis (PFIC). (1.1)
Limitation of Use:
BYLVAY is not recommended in a subgroup of PFIC type 2 patients with specific ABCB11 variants resulting in non-functional or complete absence of the bile salt export pump protein. (12.5, 14.1)
Alagille Syndrome (ALGS)
- the treatment of cholestatic pruritus in patients 12 months of age and older with Alagille syndrome (ALGS). (1.2)
12.1 Mechanism of Action
Odevixibat is a reversible inhibitor of the ileal bile acid transporter (IBAT). It decreases the reabsorption of bile acids (primarily the salt forms) from the terminal ileum.
Pruritus is a common symptom in patients with PFIC and ALGS; the pathophysiology of pruritus in patients with PFIC is not completely understood. Although the complete mechanism by which odevixibat improves pruritus in both PFIC and ALGS patients is unknown, it may involve inhibition of the IBAT, which results in decreased reuptake of bile salts, as observed by a decrease in serum bile acids [see Clinical Pharmacology (12.2)].
5 Warnings and Precautions
- Hepatoxicity: Obtain baseline liver tests and monitor patients frequently for the first 6 to 8 months after starting therapy, and as clinically indicated thereafter during treatment. If liver test abnormalities or signs of clinical hepatitis occur, consider dose reduction or treatment interruption. For persistent or recurrent liver test abnormalities relative to baseline, discontinue BYLVAY. Monitor patients with compensated cirrhosis or portal hypertension more frequently. Permanently discontinue BYLVAY if hepatic decompensation occurs. (2.3, 5.1)
- Diarrhea: Treat dehydration. Treatment interruption or discontinuation may be required for persistent diarrhea. (5.2)
-
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement with FSV if deficiency is observed. If FSV deficiency persists or worsens despite FSV supplementation, consider discontinuing BYLVAY treatment.
- Fracture: Consider interrupting BYLVAY treatment. Supplement with FSV if indicated.
- Bleeding: Interrupt treatment with BYLVAY. Optimize treatment of FSV deficiency and consider restarting BYLVAY once the patient is clinically stable. (5.3)
2 Dosage and Administration
Recommended Dosage
PFIC:
- Patients 3 months and older: 40 mcg/kg taken orally once daily. (2.1)
- If there is no improvement in pruritus after 3 months, the dosage may be increased in 40 mcg/kg increments up to 120 mcg/kg once daily, not to exceed a daily dosage of 6 mg/day. (2.1)
ALGS:
- Patients 12 months and older: 120 mcg/kg taken orally once daily. (2.2)
- Preparation and Administration Instructions
1.2 Alagille Syndrome (algs)
BYLVAY is indicated for the treatment of cholestatic pruritus in patients 12 months of age and older with ALGS.
3 Dosage Forms and Strengths
- Oral Pellets:
- 200 mcg: capsule with ivory opaque cap and white opaque body; imprinted "A200" (black ink).
- 600 mcg: capsule with ivory opaque cap and body; imprinted "A600" (black ink).
- Capsules:
- 400 mcg: capsule with medium orange opaque cap and white opaque body; imprinted "A400" (black ink).
- 1200 mcg: capsule with medium orange opaque cap and body; imprinted "A1200" (black ink).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of BYLVAY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders: gastrointestinal hemorrhage, gingival hemorrhage, liver transplant
Investigations: gamma-glutamyltransferase increased, hemoglobin decreased
Nervous system disorders: extra-axial hemorrhage (subdural hemorrhage)
Respiratory, thoracic, and mediastinal disorders: epistaxis
7.1 Bile Acid Binding Resins
Administer bile acid binding resins (e.g., cholestyramine, colesevelam, or colestipol) at least 4 hours before or 4 hours after administration of BYLVAY [see Dosage and Administration (2.3)]. Bile acid binding resins may bind odevixibat in the gut, which may reduce BYLVAY efficacy.
8 Use in Specific Populations
Pregnancy: Based on animal data, may cause cardiac malformations (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
5.3 Fat Soluble Vitamin Deficiency
BYLVAY may adversely affect absorption of fat-soluble vitamins (FSV). FSV include vitamin A, D, E, and K (measured using INR levels). PFIC and ALGS patients can have FSV deficiency at baseline and are frequently supplemented with FSV.
In Trial 1 in PFIC patients, new onset or worsening of existing FSV deficiency was reported in 1 (5%) placebo-treated patient, and 3 (16%) BYLVAY-treated 120 mcg/kg/day patients; none of the patients treated with BYLVAY dosage 40 mcg/kg/day had new onset or worsening of existing FSV deficiency.
In Trial 3 in ALGS patients, new or worsening of existing FSV deficiency was reported in 3 (17.6%) placebo- treated patients and 3 (8.6%) BYLVAY-treated patients [see Adverse Reactions (6.1)].
Obtain serum FSV levels at baseline and monitor during treatment, along with any clinical manifestations of FSV deficiency. If FSV deficiency is diagnosed, supplement with FSV. If FSV deficiency persists or worsens despite adequate FSV supplementation, consider permanent discontinuation of BYLVAY depending on the benefit and risk balance.
If complications of FSV deficiency occur, consider interrupting BYLVAY treatment and reassess to ensure adequate supplementation with FSV. Consider restarting BYLVAY once the patient is clinically stable.
2.4 Preparation and Administration Instructions
- For patients taking bile acid binding resins, take BYLVAY at least 4 hours before or 4 hours after taking a bile acid binding resin [see Drug Interactions (7.1)].
- Do not crush or chew capsules.
Oral Pellets:
- Mix the contents of the shell containing Oral Pellets into soft food or a liquid (as described below).
- Discard the emptied shells. Do not let your child swallow the unopened shells containing the Oral Pellets.
Administration Instructions for patients capable of swallowing soft food:
- Administer BYLVAY with the first morning meal.
- Place a small amount (up to 2 tablespoons [30 mL]) of soft food (apple sauce, oatmeal, banana or carrot puree, chocolate or rice pudding) in a bowl. Keep the soft food at, or cooler than, room temperature.
- Open the shell containing Oral Pellets and empty the contents into the bowl of soft food. Gently tap the Oral Pellet shell to ensure that all contents have been dispersed.
- If the dose requires more than one shell of Oral Pellets, repeat Step 3.
- Gently mix until well dispersed and administer the entire dose immediately.
- Follow the dose with breast milk, infant formula, or other age-appropriate liquid.
- Do not store the mixture for future use.
- For patients unable to swallow soft food, see the instructions below.
Administration Instructions with liquids (Using an oral dosing syringe):
- Administer BYLVAY with the first morning meal.
- Open the shell containing Oral Pellets and empty the contents into a small mixing cup. Gently tap the shell containing Oral Pellets to ensure that all contents have been emptied into the mixing cup.
- If the dose requires more than one shell of Oral Pellets, repeat Step 2.
- Add 1 teaspoon (5 mL) of an age-appropriate liquid (for example, breast milk, infant formula, or water).
- Let the pellets sit in the liquid for approximately 5 minutes to allow complete wetting REMINDER: The Oral Pellets will not dissolve in the liquid.
- After 5 minutes, place the tip of the oral syringe completely into the mixing cup. Pull the plunger of the syringe up slowly to withdraw the liquid/pellet mixture into the syringe. Gently push the plunger down again to expel the liquid/pellet mixture back into the mixing cup. Do this 2 to 3 times to ensure complete mixing of the pellets into the liquid.
- Withdraw the entire contents into the oral syringe by pulling the plunger on the end of the syringe.
- Place the tip of the syringe into the front of the patient's mouth between the tongue and the side of the mouth, and then gently push the plunger down to squirt the liquid/pellet mixture between your child's tongue and the side of the mouth. Do not squirt liquid/pellet mixture in the back of the child's throat because this could cause gagging or choking.
- Do not administer via a bottle or "sippy cup" because the Oral Pellets will not pass through the opening. The oral pellets will not dissolve in liquid.
- Follow the dose with breast milk, infant formula, or other age-appropriate liquid.
- If any pellet/liquid mixture remains in the mixing cup, repeat Step 7 and Step 8 until the entire dose has been administered.
- Check the child's mouth to make sure all of the liquid/pellet mixture has been swallowed.
Capsules:
Administration Instructions:
- Take in the morning with a meal.
- Swallow the capsule whole with a glass of water.
- Alternatively, for patients unable to swallow the capsules whole, BYLVAY capsules may be opened, and sprinkled and mixed with a small amount of soft food, or mixed with an age-appropriate liquid. Follow directions above for oral pellets to prepare and administer such a mixture.
Principal Display Panel 200 Mcg Capsule Bottle Carton
NDC 15054-3301-1
Bylvay®
(odevixibat)
ORAL PELLETS
200 mcg
RX ONLY
30 Capsules
Principal Display Panel 400 Mcg Capsule Bottle Carton
NDC 15054-3302-1
Bylvay®
(odevixibat)
CAPSULES
400 mcg
RX ONLY
30 Capsules
Principal Display Panel 600 Mcg Capsule Bottle Carton
NDC 15054-3303-1
Bylvay®
(odevixibat)
ORAL PELLETS
600 mcg
RX ONLY
30 Capsules
1.1 Progressive Familial Intrahepatic Cholestasis (pfic)
BYLVAY is indicated for the treatment of pruritus in patients 3 months of age and older with PFIC.
Principal Display Panel 1,200 Mcg Capsule Bottle Carton
NDC 15054-3304-1
Bylvay®
(odevixibat)
CAPSULES
1,200 mcg
RX ONLY
30 Capsules
2.2 Recommended Dosage for Alagille Syndrome (algs) in Patients Aged 12 Months and Older
- The recommended dosage of BYLVAY is 120 mcg/kg taken orally once daily in the morning with a meal. Table 2 below shows the recommended once daily dosage by body weight.
- BYLVAY oral pellets are intended for use by patients weighing less than 19.5 kilograms.
- BYLVAY capsules are intended for use by patients weighing 19.5 kilograms or above.
| Body Weight (kg) | Once Daily Dosage (mcg) |
|---|---|
| 7.4 and below | 600 |
| 7.5 to 12.4 | 1,200 |
| 12.5 to 17.4 | 1,800 |
| 17.5 to 25.4 | 2,400 |
| 25.5 to 35.4 | 3,600 |
| 35.5 to 45.4 | 4,800 |
| 45.5 to 55.4 | 6,000 |
| 55.5 and above | 7,200 |
2.1 Recommended Dosage for Progressive Familial Intrahepatic Cholestasis (pfic) in Patients Aged 3 Months and Older
- The recommended dosage of BYLVAY is 40 mcg/kg taken orally once daily in the morning with a meal. Table 1 below shows the recommended once daily dosage by body weight.
- If there is no improvement in pruritus after 3 months, the dosage may be increased in 40 mcg/kg increments up to 120 mcg/kg once daily not to exceed a daily dosage of 6 mg/day.
- BYLVAY oral pellets are intended for use by patients weighing less than 19.5 kilograms.
- BYLVAY capsules are intended for use by patients weighing 19.5 kilograms or above.
| Body Weight (kg) | Once Daily Dosage (mcg) |
|---|---|
| 7.4 and below | 200 |
| 7.5 to 12.4 | 400 |
| 12.5 to 17.4 | 600 |
| 17.5 to 25.4 | 800 |
| 25.5 to 35.4 | 1,200 |
| 35.5 to 45.4 | 1,600 |
| 45.5 to 55.4 | 2,000 |
| 55.5 and above | 2,400 |
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
- BYLVAY is not recommended in a subgroup of PFIC type 2 patients with specific ABCB11 variants resulting in non-functional or complete absence of the bile salt export pump (BSEP) protein [see Clinical Pharmacology (12.5) and Clinical Studies (14.1)].
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling:
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (between 59°F and 86°F) [See USP Controlled Room Temperature].
14.1 Pfic (14.1 PFIC)
The efficacy of BYLVAY was evaluated in Trial 1 (NCT03566238), a 24-week, randomized, double-blind, placebo-controlled trial. Trial 1 was conducted in 62 pediatric patients, aged 6 months to 17 years, with a confirmed molecular diagnosis of PFIC type 1 or type 2, and presence of pruritus at baseline. Patients with variants in the ABCB11 gene that predict non-function or complete absence of the bile salt export pump (BSEP) protein, who had experienced prior hepatic decompensation events, who had other concomitant liver disease, whose INR was greater than 1.4, whose ALT or total bilirubin was greater than 10-times the upper limit of normal (ULN), or who had received a liver transplant were excluded in Trial 1.
Patients were randomized to placebo (n=20), 40 mcg/kg (n=23), or 120 mcg/kg (n=19). Study drug was administered once daily with a meal in the morning. In patients weighing less than 19.5 kg or patients who could not swallow the whole capsule, study drug was sprinkled on soft food and then administered orally.
Median age (range) of the patients in Trial 1 was 3.2 (0.5 to 15.9) years; 3 patients were older than 12 years of age. Of the 62 patients, 50% were male and 84% were white; 27% had PFIC type 1, and 73% had PFIC type 2. The mean (standard error [SE]) scratching score in the 2 weeks prior to baseline was 2.9 (0.08). Baseline mean (SE) eGFR was 164 (30.6) mL/min/1.73 m2. Baseline median (range) ALT, AST, and total bilirubin were 65 (16-798) U/L, 83.5 (32-405) U/L, and 2.2 (0.2-18.6) mg/dL, respectively.
In Trial 1, a total of 13 patients discontinued from trial prematurely either due to no improvement in pruritus (n=11) or due to adverse reactions (n=2); 5/20 (25%) patients discontinued from the placebo arm and 8/42 (19%) patients discontinued from the BYLVAY arms. A total of 11 of the 13 patients rolled over to Trial 2 to receive BYLVAY 120 mcg/kg/day. One patient treated with BYLVAY 120 mcg/kg/day withdrew from the trial due to an adverse reaction of diarrhea [see Adverse Reactions (6)].
Given the patients' young age, a single-item observer-reported outcome (ObsRO) was used to measure patients' scratching severity as observed by their caregiver twice daily (once in the morning and once in the evening). Scratching severity was assessed on a 5-point ordinal response scale, with scores ranging from 0 (no scratching) to 4 (worst possible scratching). Patients were included in Trial 1 if their average scratching score was greater than or equal to 2 (medium scratching) in the 2 weeks prior to baseline.
Table 5 presents the results of the comparison between BYLVAY and placebo on the mean of patients' percentage of ObsRO assessments over the 24-week treatment period that were scored as 0 (no scratching) or 1 (a little scratching). Patients treated with BYLVAY demonstrated greater improvement in pruritus compared with placebo. Figure 1 displays the mean of patients' worst weekly average scratching scores in each treatment group for each month, where the weekly average utilized the worst score from each day (morning or evening score).
| Placebo (n=20) |
BYLVAY | ||
|---|---|---|---|
| 40 mcg/kg/day (n=23) |
120 mcg/kg/day (n=19) |
||
|
Mean Based on least squares means from analysis of covariance model with daytime and nighttime baseline pruritus scores as covariates and treatment group and stratification factors (i.e., PFIC type and age category) as fixed effects. Percentage of Assessments Over the Treatment Period Scored as 0 (No Scratching) or 1 (A Little Scratching) (%)
|
|||
| Mean (SE) | 13.2 (8.7) | 35.4 (8.1) | 30.1 (9.0) |
| Mean Difference vs Placebo (95% CI) | 22.2 (4.7, 39.6) | 16.9 (-2.0, 35.7) |
|
Figure 1: Mean Figure 1 presents least squares means of the Worst Weekly Average Scratching Scores for Each Month in Trial 1
Based on a mixed model repeated measure (MMRM) analysis accounting for baseline score, treatment group, time (in months), treatment-by-baseline interaction, treatment-by-time interaction, and stratification factors (i.e., PFIC type and age category). Missing data were accounted for using placebo-reference multiple imputation. |
|
|
14.2 Algs (14.2 ALGS)
The efficacy of BYLVAY was evaluated in Trial 3 (NCT04674761), a 24-week, randomized, double-blind, placebo-controlled trial. Trial 3 was conducted in 52 pediatric patients, aged 6 months to 15 years, with a confirmed diagnosis of ALGS and presence of pruritus at baseline. Patients who had decompensated liver disease, who had other concomitant liver disease, whose INR was greater than 1.4, whose ALT was greater than 10-times the upper limit of normal (ULN) at screening, whose total bilirubin was greater than 15-times the ULN at screening, or who had received a liver transplant were excluded from Trial 3.
Patients were randomized to placebo (n=17) or 120 mcg/kg (n=35). Study drug was administered once daily with a meal in the morning. In patients weighing less than 19.5 kg or patients who could not swallow the whole capsule, study drug was sprinkled on soft food and then administered orally.
Median age (range) of the patients in Trial 3 was 6.1 (1.7 to 15.5) years in the BYLVAY group and 4.2 (0.5 to 14.3) years in the placebo group; 5 patients were older than 12 years of age. Of the 52 patients, 52% were male and 83% were white; 92% of patients had the JAG1 mutation and 8% had the NOTCH2 mutation. The mean (standard deviation [SD]) scratching score in the 2 weeks prior to baseline was 2.9 (0.6). Baseline mean (SD) eGFR was 159 (51.4) mL/min/1.73 m2. Baseline median (range) ALT, AST, and total bilirubin were 152 (39-403) U/L, 135 (57-427) U/L, and 2.0 (0.4-11.4) mg/dL, respectively.
Given the patients' young ages, a single-item observer-reported outcome (ObsRO) was used to measure patients' scratching severity as observed by their caregiver twice daily (once in the morning and once in the evening). Scratching severity was assessed on a 5-point ordinal response scale, with scores ranging from 0 (no scratching) to 4 (worst possible scratching). Patients were included in Trial 3 if the average scratching score was greater than or equal to 2 (medium scratching) in the 14 days prior to baseline.
Table 6 presents the results of the comparison between BYLVAY and placebo on the change from baseline in average scratching score based on ObsRO assessments to Month 6 (Weeks 21 to 24). The average scratching score for each patient for each month post-baseline was calculated by: (Step 1) averaging the morning scores and averaging the evening scores within a week; (Step 2) averaging the morning and evening weekly scores to yield a single weekly score; and finally (Step 3) averaging the 4 weekly scores within the month. The baseline average scratching score for each patient was calculated by averaging the weekly scores obtained in Step 2 across the 2 weeks prior to randomization and initiation of blinded treatment. Patients treated with BYLVAY demonstrated greater improvement in pruritus compared with placebo. Figure 2 displays the means (95% confidence interval) of patients' average scratching scores in each treatment group for each month.
| Placebo (n=17) |
BYLVAY 120 mcg/kg/day (n=35) |
|
|---|---|---|
| Baseline Average Scratching Score | ||
| Mean (SD) | 3.0 (0.6) | 2.8 (0.5) |
|
Change from Baseline in Average Scratching Score to Month 6 (Weeks 21 to 24) Based on least square means from a mixed-effect model for repeated measures (MMRM) for change from baseline to each month accounting for baseline average scratching score, baseline age stratification (<10, ≥10 years), baseline direct bilirubin, treatment group, time (in months), and treatment-by-time interaction.
|
||
| Mean (SE) | -0.8 (0.2) | -1.7 (0.2) |
| Mean Difference vs Placebo (95% CI) p-value |
-0.9 (-1.4, -0.3) 0.002 |
|
Figure 2: Mean Figure 2 presents means for baseline and least squares means for Month 1 to 6 of the Average Scratching Scores for Each Month in Trial 3
Least squares means are based on a mixed model repeated measure (MMRM) analysis accounting for baseline average scratching score, baseline age stratification (<10, ≥10 years), baseline direct bilirubin, treatment group, time (in months), and treatment-by-time interaction. |
5.2 Diarrhea
In Trial 1, diarrhea in PFIC patients was reported in 2 (10%) placebo-treated patients, 9 (39%) BYLVAY-treated 40 mcg/kg/day patients and 4 (21%) BYLVAY-treated 120 mcg/kg/day patients. Treatment interruption due to diarrhea occurred in 2 patients with 3 events during treatment with BYLVAY 120 mcg/kg/day. Treatment interruption due to diarrhea ranged between 3 to 7 days [see Adverse Reactions (6.1)]. One patient treated with BYLVAY 120 mcg/kg/day withdrew from Trial 1 due to persistent diarrhea.
In Trial 3, diarrhea in ALGS patients was reported in 1 placebo-treated patient (6%) and in 10 (29%) BYLVAY-treated patients [see Adverse Reactions (6.1)]. No patients interrupted or permanently discontinued BYLVAY due to diarrhea.
If diarrhea occurs, monitor for dehydration and treat promptly. Interrupt BYLVAY dosing if a patient experiences persistent diarrhea. Restart BYLVAY at 40 mcg/kg/day when diarrhea resolves, and increase the dose as tolerated if appropriate. If diarrhea persists and no alternate etiology is identified, stop BYLVAY treatment.
11 Description (11 DESCRIPTION)
The active ingredient in BYLVAY (odevixibat) capsules and BYLVAY (odevixibat) oral pellets, an ileal bile acid transporter (IBAT) inhibitor, is (2S)-2-{[(2R)-2-(2-{[3,3-dibutyl-7-(methylsulfanyl)-1,1-dioxo-5-phenyl-2,3,4,5-tetrahydro-1H-1λ6,2,5-benzothiadiazepin-8yl]oxy}acetamido)-2-(4-hydroxyphenyl)acetly]amino}butanoic acid, which is formulated as the sesquihydrate having the following chemical structure:
The molecular formula is C37H48N4O8S2 × 1.5 H2O, with a molecular weight of 768.0 g/mol (anhydrous 740.9 g/mol). Odevixibat sesquihydrate is a white to off-white solid. Its solubility in aqueous solutions is pH-dependent and increases with increased pH.
BYLVAY is available for oral administration as oral pellets containing odevixibat sesquihydrate equivalent to 200 mcg or 600 mcg of odevixibat, and as capsules containing odevixibat sesquihydrate equivalent to 400 mcg or 1200 mcg of odevixibat, and the following excipients: hypromellose and microcrystalline cellulose.
The capsule shells for the oral pellets contain hypromellose, titanium dioxide and yellow iron oxide.
The capsule shells for the capsules contain hypromellose, red iron oxide, titanium dioxide and yellow iron oxide.
The imprinting ink contains ferrosoferric oxide/black iron oxide and shellac glaze.
5.1 Hepatoxicity
BYLVAY treatment is associated with a potential for drug-induced liver injury (DILI).
In the PFIC and ALGS trials, treatment-emergent elevations of liver tests or worsening of liver tests occurred. Of the six patients who experienced DILI, two underwent liver transplant.
Obtain baseline liver tests because some ALGS and PFIC patients have abnormal liver tests at baseline. Monitor patients frequently for the first 6 to 8 months after starting therapy and as clinically indicated thereafter during treatment with BYLVAY. Monitor for elevation in liver tests, for the development of liver-related adverse reactions, and for physical signs of hepatic decompensation. If liver test abnormalities or signs of clinical hepatitis occur in the absence of other causes, consider dose reduction or treatment interruption.
Permanently discontinue BYLVAY if a patient experiences the following:
- persistent or recurrent liver test abnormalities, or
- upon rechallenge, signs and symptoms consistent with clinical hepatitis, or
- a hepatic decompensation event.
The safety and effectiveness of BYLVAY have not been established in patients with decompensated cirrhosis. Monitor patients with compensated cirrhosis or portal hypertension more frequently and discontinue BYLVAY if hepatic decompensation occurs. IBAT inhibitors, including BYLVAY, are contraindicated in patients with prior or active hepatic decompensation events [see Contraindications (4)].
8.4 Pediatric Use
The safety and effectiveness of BYLVAY have been established in pediatric patients 3 months to 17 years of age for the treatment of pruritus in PFIC. Use of BYLVAY in this age group is supported by evidence from one randomized, double-blind, placebo-controlled trial conducted in 62 patients with a confirmed diagnosis of PFIC type 1 or type 2 (Trial 1), and an open-label extension trial in PFIC patients (Trial 2) [see Adverse Reactions (6.1) and Clinical Studies (14.1)].
The safety and effectiveness of BYLVAY for the treatment of pruritus in PFIC in pediatric patients less than 3 months of age have not been established.
The safety and effectiveness of BYLVAY have been established in pediatric patients 12 months to 17 years of age for the treatment of pruritus in ALGS. Use of BYLVAY in this age group is supported by evidence from one randomized, double-blind, placebo-controlled trial conducted in 52 patients with a confirmed diagnosis of ALGS (Trial 3) and one open-label extension trial in ALGS patients (Trial 4) [see Adverse Reactions (6.1) and Clinical Studies (14.2)].
The safety and effectiveness of BYLVAY for the treatment of pruritus in ALGS in pediatric patients less than 12 months of age have not been established.
8.5 Geriatric Use
PFIC and ALGS are largely diseases of pediatric and young adult patients. Clinical studies in BYLVAY did not include patients 65 years of age and older.
4 Contraindications (4 CONTRAINDICATIONS)
IBAT inhibitors, including BYLVAY, are contraindicated in patients with prior or active hepatic decompensation events (e.g., variceal hemorrhage, ascites, hepatic encephalopathy) [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in greater detail in other sections of the label:
- Hepatotoxicity [see Warnings and Precautions (5.1) ]
- Diarrhea [see Warnings and Precautions (5.2)]
- Fat-Soluble Vitamin Deficiency [see Warnings and Precautions (5.3)]
Instructions for Use (Instructions For Use)
This Instructions for Use contains information on how to give BYLVAY Capsules and Oral Pellets. This information does not take the place of talking to your healthcare provider about your child's medical condition or their treatment.
Important information you need to know before giving or taking BYLVAY
- Give BYLVAY along with the morning meal.
- Mix BYLVAY in a small amount of soft food (up to 2 tablespoons [30 mL]), such as apple sauce, oatmeal, banana or carrot puree, chocolate or rice pudding, in a bowl. You may also mix BYLVAY with an age-appropriate liquid and give through an oral syringe.
- If your child is taking bile acid binding resins (for example, cholestyramine, colestipol), give them BYLVAY at least 4 hours before or 4 hours after they take the bile acid binding resin.
Preparing to Give BYLVAY
You will be provided with the number of BYLVAY Capsules or Oral Pellets prescribed by your child's healthcare provider in a child-resistant closure.
Giving BYLVAY Oral Pellets with soft food:
- The shell containing Oral Pellets are to be opened and sprinkled. Do not let your child swallow the shell containing the Oral Pellets. Dispose of (throw away) the emptied shell.
- Mix the contents of the Oral Pellets with soft food as shown in Steps 1 through 9 below.
| Step 1. | Give BYLVAY with the first morning meal. Place a small amount of soft food (up to 2 tablespoons [30 mL], such as apple sauce, oatmeal, banana or carrot puree, chocolate or rice pudding) in a bowl. Keep the soft food at, or cooler than, room temperature. Note: This small amount of soft food should be less than what your child would normally eat. |
| Step 2. | Hold the shell containing Oral Pellets horizontally on both ends, twist in opposite directions and pull apart (see Figure A ). |
|
Figure A |
|
| Step 3. | Empty the Oral Pellets into the bowl of soft food (see Figure B ). |
|
Figure B |
|
| Step 4. | Gently tap the shell containing Oral Pellets to make sure that all pellets come out (see Figure C ). |
|
Figure C |
|
| Step 5. | If the dose requires more than 1 capsule shell, repeat Step 2 and Step 3. |
| Step 6. | Gently mix the Oral Pellets with a spoon into the soft food. Note that the Oral Pellets will not dissolve (see Figure D ). |
|
Figure D |
|
| Step 7. | Give the entire dose right away after mixing. Do not store the BYLVAY mixture for later use. |
| Step 8. | Give water or an age-appropriate liquid, such as breast milk or infant formula, after the dose is taken. |
| Step 9. | Dispose of (throw away) the empty Oral Pellet shells in the trash. |
Giving BYLVAY Oral Pellets with liquids (Using an oral dosing syringe):
- The shell containing the Oral Pellets are to be opened.
- Mix the Oral Pellets with liquid as shown in Steps 1 through 14 below.
- Dispose of (throw away) the emptied shells. Do not let your child swallow the unopened shells containing the Oral Pellets.
| Step 1. | Give BYLVAY with the first morning meal. |
| Step 2. | Hold the shell containing the Oral Pellets horizontally on both ends, twist in opposite directions and pull apart (see Figure A ). |
| Step 3. | Empty the Oral Pellets into a small mixing cup. Gently tap the Oral Pellet shell to ensure that all contents have been emptied into the mixing cup (see Figure E ). |
|
Figure E |
|
| Step 4. | If the dose requires more than 1 capsule shell, repeat Step 2 and Step 3. |
| Step 5. | Add 1 teaspoon (5 mL) of an age-appropriate liquid (for example, breast milk, infant formula, or water). |
| Step 6. | Let the pellets sit in the liquid for about 5 minutes to allow complete wetting. REMINDER: The pellets will not dissolve in the liquid. |
| Step 7. | After 5 minutes, place the tip of the oral syringe completely into the mixing cup. Pull the plunger of the syringe up slowly to withdraw the liquid and pellet mixture into the syringe. Gently push the plunger down again to expel the liquid and pellet mixture back into the mixing cup. Do this 2 to 3 times to ensure complete mixing of the pellets into the liquid. |
| Step 8. | Withdraw the entire contents into the syringe by pulling the plunger on the end of the syringe (see Figure F). |
|
Figure F |
|
| Step 9. | Place the tip of the syringe into the front of the child's mouth between the tongue and the side of the mouth, and then gently push the plunger down to squirt the liquid and pellet mixture between the tongue and the side of the mouth. Do not squirt the liquid and pellet mixture in the back of the throat because this could cause gagging or choking (see Figure G). |
|
Figure G |
|
| Step 10. | Do not give using a bottle or "sippy cup" because the Oral Pellets will not pass through the opening. The oral pellets will not dissolve in liquid. |
| Step 11. | Give water or an age-appropriate liquid such as breast milk or infant formula after the dose is taken. |
| Step 12. | Repeat Steps 8 and 9 until the entire dose has been given. |
| Step 13. | Check to make sure all of the liquid and pellet mixture has been swallowed. |
| Step 14. | Dispose of (throw away) the empty Oral Pellet shells in the trash. |
Taking BYLVAY Capsules
- Take BYLVAY Capsules along with your morning meal. Swallow BYLVAY Capsules whole with a glass of water. Do not chew or crush the Capsules.
- For children unable to swallow BYLVAY Capsules whole, follow instructions under Preparing to Give BYLVAY above.
How should I store BYLVAY Capsules or Oral Pellets?
Store BYLVAY at room temperature between 68°F to 77°F (20°C to 25°C).
Disposing (throwing away) of BYLVAY Capsules or Oral Pellets shells.
Dispose of (throw away) the empty BYLVAY Capsule or Oral Pellets shells in the household trash.
What are the ingredients in BYLVAY?
Active ingredient: odevixibat.
Inactive ingredients: hypromellose and microcrystalline cellulose.
Manufactured for:
Ipsen Biopharmaceuticals, Inc.
One Main Street,
Cambridge, MA 02142
© 2024 Ipsen Biopharmaceuticals, Inc. All rights reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 01/2024
12.2 Pharmacodynamics
Odevixibat reduces serum bile acids in patients with PFIC and ALGS.
In Trial 1, a 24-week, randomized, double-blind, placebo-controlled trial conducted in 62 patients with a confirmed diagnosis of PFIC type 1 or type 2, the majority of patients (88.7%) had elevated serum bile acids above 100 mcmol/L at baseline [see Clinical Studies (14.1)]. Serum bile acids concentrations were reduced from baseline within 4-8 weeks of odevixibat treatment compared to placebo treatment. The decreased concentrations of serum bile acids fluctuated over time but generally were maintained during the treatment over 24 weeks. The extent of decrease in serum bile acids was similar between 40 and 120 mcg/kg.
Trial 3 is a 24-week, randomized, double-blind, placebo-controlled trial conducted in 52 patients with a confirmed diagnosis of ALGS who were administered treatment with BYLVAY 120 mcg/kg once daily [see Clinical Studies (14.2)]. At baseline, serum bile acids were variable ranging from 93 to 510 mcmol/L. Serum bile acid concentrations were reduced from baseline as early as Week 4 of odevixibat treatment and the reduction was generally maintained during treatment over 24 weeks.
12.3 Pharmacokinetics
In pediatric patients with PFIC, 6 months to 17 years of age who received BYLVAY 40 mcg/kg or 120 mcg/kg once daily with food in the morning, the measurable odevixibat concentrations ranged from 0.06 to 0.72 ng/mL, and odevixibat concentrations were below the limit of quantification (0.05 ng/mL) in the majority of plasma samples.
In pediatric patients with ALGS who received BYLVAY 120 mcg/kg once daily with food in the morning, the measurable odevixibat concentrations ranged from 0.05 to 3.4 ng/mL.
Following single and repeated oral administration of odevixibat from 0.1 to 3 mg in healthy adults, plasma concentrations of odevixibat were mostly below the limit of quantification (0.05 ng/mL); therefore, AUC and peak plasma concentration (Cmax) could not be calculated.
Following a single administration of odevixibat 7.2 mg in healthy adults, the mean (%CV) Cmax and AUC0-24h were 0.47 ng/mL (34.8) and 2.19 ng∙h/mL (36.2), respectively. No accumulation of odevixibat was observed following once-daily dosing.
12.5 Pharmacogenomics
PFIC is a heterogenous disease caused by homozygous or compound heterozygous variants, with different PFIC subtypes occurring in the general population. PFIC1 is caused by variants in the ATP8B1 gene, which encodes FIC1. PFIC2 is caused by variants in the ABCB11 gene, which encodes BSEP. PFIC2 is further categorized into BSEP subgroups based on specific variants. The BSEP-1 subgroup includes patients with at least one p.D482G (c.1445A>G) or p.E297G (c.890A>G) variant, BSEP-2 includes patients with at least one missense variant other than p.D482G or p.E297G (non BSEP-1), and BSEP-3 includes patients with variants that are predicted to encode a non-functional protein. PFIC3 is caused by variants in the ABCB4 gene, which encodes MDR3. PFIC4 is caused by variants in the TJP2 gene, which encodes TJP2. PFIC6 is caused by variants in the MYO5B gene, which encodes MYO5B. Patients can be clinically diagnosed with PFIC without a known pathogenic variant.
PFIC2 is the most common subtype accounting for 37-90% of PFIC patients. The prevalence of BSEP-1, BSEP-2, and BSEP-3 subgroups are approximately 27%, 52%, and 21%, respectively, based on data from a global consortium characterizing the natural history of severe BSEP deficiency.
8.6 Hepatic Impairment
Patients with PFIC and ALGS may have impaired hepatic function. The efficacy and safety of BYLVAY in PFIC and ALGS patients with clinically significant portal hypertension, and in patients with decompensated cirrhosis have not been established [see Dosage and Administration (2.3), Contraindications (4), Warning and Precautions (5.1), and Clinical Studies (14)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
BYLVAY is an ileal bile acid transporter (IBAT) inhibitor indicated for:
Progressive Familial Intrahepatic Cholestasis (PFIC)
- the treatment of pruritus in patients 3 months of age and older with progressive familial intrahepatic cholestasis (PFIC). (1.1)
Limitation of Use:
BYLVAY is not recommended in a subgroup of PFIC type 2 patients with specific ABCB11 variants resulting in non-functional or complete absence of the bile salt export pump protein. (12.5, 14.1)
Alagille Syndrome (ALGS)
- the treatment of cholestatic pruritus in patients 12 months of age and older with Alagille syndrome (ALGS). (1.2)
12.1 Mechanism of Action
Odevixibat is a reversible inhibitor of the ileal bile acid transporter (IBAT). It decreases the reabsorption of bile acids (primarily the salt forms) from the terminal ileum.
Pruritus is a common symptom in patients with PFIC and ALGS; the pathophysiology of pruritus in patients with PFIC is not completely understood. Although the complete mechanism by which odevixibat improves pruritus in both PFIC and ALGS patients is unknown, it may involve inhibition of the IBAT, which results in decreased reuptake of bile salts, as observed by a decrease in serum bile acids [see Clinical Pharmacology (12.2)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatoxicity: Obtain baseline liver tests and monitor patients frequently for the first 6 to 8 months after starting therapy, and as clinically indicated thereafter during treatment. If liver test abnormalities or signs of clinical hepatitis occur, consider dose reduction or treatment interruption. For persistent or recurrent liver test abnormalities relative to baseline, discontinue BYLVAY. Monitor patients with compensated cirrhosis or portal hypertension more frequently. Permanently discontinue BYLVAY if hepatic decompensation occurs. (2.3, 5.1)
- Diarrhea: Treat dehydration. Treatment interruption or discontinuation may be required for persistent diarrhea. (5.2)
-
Fat-Soluble Vitamin (FSV) Deficiency: Obtain baseline levels and monitor during treatment. Supplement with FSV if deficiency is observed. If FSV deficiency persists or worsens despite FSV supplementation, consider discontinuing BYLVAY treatment.
- Fracture: Consider interrupting BYLVAY treatment. Supplement with FSV if indicated.
- Bleeding: Interrupt treatment with BYLVAY. Optimize treatment of FSV deficiency and consider restarting BYLVAY once the patient is clinically stable. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Recommended Dosage
PFIC:
- Patients 3 months and older: 40 mcg/kg taken orally once daily. (2.1)
- If there is no improvement in pruritus after 3 months, the dosage may be increased in 40 mcg/kg increments up to 120 mcg/kg once daily, not to exceed a daily dosage of 6 mg/day. (2.1)
ALGS:
- Patients 12 months and older: 120 mcg/kg taken orally once daily. (2.2)
- Preparation and Administration Instructions
1.2 Alagille Syndrome (algs) (1.2 Alagille Syndrome (ALGS))
BYLVAY is indicated for the treatment of cholestatic pruritus in patients 12 months of age and older with ALGS.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Oral Pellets:
- 200 mcg: capsule with ivory opaque cap and white opaque body; imprinted "A200" (black ink).
- 600 mcg: capsule with ivory opaque cap and body; imprinted "A600" (black ink).
- Capsules:
- 400 mcg: capsule with medium orange opaque cap and white opaque body; imprinted "A400" (black ink).
- 1200 mcg: capsule with medium orange opaque cap and body; imprinted "A1200" (black ink).
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of BYLVAY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders: gastrointestinal hemorrhage, gingival hemorrhage, liver transplant
Investigations: gamma-glutamyltransferase increased, hemoglobin decreased
Nervous system disorders: extra-axial hemorrhage (subdural hemorrhage)
Respiratory, thoracic, and mediastinal disorders: epistaxis
7.1 Bile Acid Binding Resins
Administer bile acid binding resins (e.g., cholestyramine, colesevelam, or colestipol) at least 4 hours before or 4 hours after administration of BYLVAY [see Dosage and Administration (2.3)]. Bile acid binding resins may bind odevixibat in the gut, which may reduce BYLVAY efficacy.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: Based on animal data, may cause cardiac malformations (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
5.3 Fat Soluble Vitamin Deficiency (5.3 Fat-Soluble Vitamin Deficiency)
BYLVAY may adversely affect absorption of fat-soluble vitamins (FSV). FSV include vitamin A, D, E, and K (measured using INR levels). PFIC and ALGS patients can have FSV deficiency at baseline and are frequently supplemented with FSV.
In Trial 1 in PFIC patients, new onset or worsening of existing FSV deficiency was reported in 1 (5%) placebo-treated patient, and 3 (16%) BYLVAY-treated 120 mcg/kg/day patients; none of the patients treated with BYLVAY dosage 40 mcg/kg/day had new onset or worsening of existing FSV deficiency.
In Trial 3 in ALGS patients, new or worsening of existing FSV deficiency was reported in 3 (17.6%) placebo- treated patients and 3 (8.6%) BYLVAY-treated patients [see Adverse Reactions (6.1)].
Obtain serum FSV levels at baseline and monitor during treatment, along with any clinical manifestations of FSV deficiency. If FSV deficiency is diagnosed, supplement with FSV. If FSV deficiency persists or worsens despite adequate FSV supplementation, consider permanent discontinuation of BYLVAY depending on the benefit and risk balance.
If complications of FSV deficiency occur, consider interrupting BYLVAY treatment and reassess to ensure adequate supplementation with FSV. Consider restarting BYLVAY once the patient is clinically stable.
2.4 Preparation and Administration Instructions
- For patients taking bile acid binding resins, take BYLVAY at least 4 hours before or 4 hours after taking a bile acid binding resin [see Drug Interactions (7.1)].
- Do not crush or chew capsules.
Oral Pellets:
- Mix the contents of the shell containing Oral Pellets into soft food or a liquid (as described below).
- Discard the emptied shells. Do not let your child swallow the unopened shells containing the Oral Pellets.
Administration Instructions for patients capable of swallowing soft food:
- Administer BYLVAY with the first morning meal.
- Place a small amount (up to 2 tablespoons [30 mL]) of soft food (apple sauce, oatmeal, banana or carrot puree, chocolate or rice pudding) in a bowl. Keep the soft food at, or cooler than, room temperature.
- Open the shell containing Oral Pellets and empty the contents into the bowl of soft food. Gently tap the Oral Pellet shell to ensure that all contents have been dispersed.
- If the dose requires more than one shell of Oral Pellets, repeat Step 3.
- Gently mix until well dispersed and administer the entire dose immediately.
- Follow the dose with breast milk, infant formula, or other age-appropriate liquid.
- Do not store the mixture for future use.
- For patients unable to swallow soft food, see the instructions below.
Administration Instructions with liquids (Using an oral dosing syringe):
- Administer BYLVAY with the first morning meal.
- Open the shell containing Oral Pellets and empty the contents into a small mixing cup. Gently tap the shell containing Oral Pellets to ensure that all contents have been emptied into the mixing cup.
- If the dose requires more than one shell of Oral Pellets, repeat Step 2.
- Add 1 teaspoon (5 mL) of an age-appropriate liquid (for example, breast milk, infant formula, or water).
- Let the pellets sit in the liquid for approximately 5 minutes to allow complete wetting REMINDER: The Oral Pellets will not dissolve in the liquid.
- After 5 minutes, place the tip of the oral syringe completely into the mixing cup. Pull the plunger of the syringe up slowly to withdraw the liquid/pellet mixture into the syringe. Gently push the plunger down again to expel the liquid/pellet mixture back into the mixing cup. Do this 2 to 3 times to ensure complete mixing of the pellets into the liquid.
- Withdraw the entire contents into the oral syringe by pulling the plunger on the end of the syringe.
- Place the tip of the syringe into the front of the patient's mouth between the tongue and the side of the mouth, and then gently push the plunger down to squirt the liquid/pellet mixture between your child's tongue and the side of the mouth. Do not squirt liquid/pellet mixture in the back of the child's throat because this could cause gagging or choking.
- Do not administer via a bottle or "sippy cup" because the Oral Pellets will not pass through the opening. The oral pellets will not dissolve in liquid.
- Follow the dose with breast milk, infant formula, or other age-appropriate liquid.
- If any pellet/liquid mixture remains in the mixing cup, repeat Step 7 and Step 8 until the entire dose has been administered.
- Check the child's mouth to make sure all of the liquid/pellet mixture has been swallowed.
Capsules:
Administration Instructions:
- Take in the morning with a meal.
- Swallow the capsule whole with a glass of water.
- Alternatively, for patients unable to swallow the capsules whole, BYLVAY capsules may be opened, and sprinkled and mixed with a small amount of soft food, or mixed with an age-appropriate liquid. Follow directions above for oral pellets to prepare and administer such a mixture.
Principal Display Panel 200 Mcg Capsule Bottle Carton (PRINCIPAL DISPLAY PANEL - 200 mcg Capsule Bottle Carton)
NDC 15054-3301-1
Bylvay®
(odevixibat)
ORAL PELLETS
200 mcg
RX ONLY
30 Capsules
Principal Display Panel 400 Mcg Capsule Bottle Carton (PRINCIPAL DISPLAY PANEL - 400 mcg Capsule Bottle Carton)
NDC 15054-3302-1
Bylvay®
(odevixibat)
CAPSULES
400 mcg
RX ONLY
30 Capsules
Principal Display Panel 600 Mcg Capsule Bottle Carton (PRINCIPAL DISPLAY PANEL - 600 mcg Capsule Bottle Carton)
NDC 15054-3303-1
Bylvay®
(odevixibat)
ORAL PELLETS
600 mcg
RX ONLY
30 Capsules
1.1 Progressive Familial Intrahepatic Cholestasis (pfic) (1.1 Progressive Familial Intrahepatic Cholestasis (PFIC))
BYLVAY is indicated for the treatment of pruritus in patients 3 months of age and older with PFIC.
Principal Display Panel 1,200 Mcg Capsule Bottle Carton (PRINCIPAL DISPLAY PANEL - 1,200 mcg Capsule Bottle Carton)
NDC 15054-3304-1
Bylvay®
(odevixibat)
CAPSULES
1,200 mcg
RX ONLY
30 Capsules
2.2 Recommended Dosage for Alagille Syndrome (algs) in Patients Aged 12 Months and Older (2.2 Recommended Dosage for Alagille Syndrome (ALGS) in Patients Aged 12 Months and Older)
- The recommended dosage of BYLVAY is 120 mcg/kg taken orally once daily in the morning with a meal. Table 2 below shows the recommended once daily dosage by body weight.
- BYLVAY oral pellets are intended for use by patients weighing less than 19.5 kilograms.
- BYLVAY capsules are intended for use by patients weighing 19.5 kilograms or above.
| Body Weight (kg) | Once Daily Dosage (mcg) |
|---|---|
| 7.4 and below | 600 |
| 7.5 to 12.4 | 1,200 |
| 12.5 to 17.4 | 1,800 |
| 17.5 to 25.4 | 2,400 |
| 25.5 to 35.4 | 3,600 |
| 35.5 to 45.4 | 4,800 |
| 45.5 to 55.4 | 6,000 |
| 55.5 and above | 7,200 |
2.1 Recommended Dosage for Progressive Familial Intrahepatic Cholestasis (pfic) in Patients Aged 3 Months and Older (2.1 Recommended Dosage for Progressive Familial Intrahepatic Cholestasis (PFIC) in Patients Aged 3 Months and Older)
- The recommended dosage of BYLVAY is 40 mcg/kg taken orally once daily in the morning with a meal. Table 1 below shows the recommended once daily dosage by body weight.
- If there is no improvement in pruritus after 3 months, the dosage may be increased in 40 mcg/kg increments up to 120 mcg/kg once daily not to exceed a daily dosage of 6 mg/day.
- BYLVAY oral pellets are intended for use by patients weighing less than 19.5 kilograms.
- BYLVAY capsules are intended for use by patients weighing 19.5 kilograms or above.
| Body Weight (kg) | Once Daily Dosage (mcg) |
|---|---|
| 7.4 and below | 200 |
| 7.5 to 12.4 | 400 |
| 12.5 to 17.4 | 600 |
| 17.5 to 25.4 | 800 |
| 25.5 to 35.4 | 1,200 |
| 35.5 to 45.4 | 1,600 |
| 45.5 to 55.4 | 2,000 |
| 55.5 and above | 2,400 |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:42.172174 · Updated: 2026-03-14T22:41:13.455924