These Highlights Do Not Include All The Information Needed To Use Dopamine Hydrochloride In Dextrose Injection Safely And Effectively. See Full Prescribing Information For Dopamine Hydrochloride In Dextrose Injection.
08f23f6e-150d-45ea-098e-f2edf64c21a1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dopamine Hydrochloride in Dextrose Injection is indicated to improve hemodynamic status in patients in distributive shock, or shock due to reduced cardiac output.
Indications and Usage
Dopamine Hydrochloride in Dextrose Injection is indicated to improve hemodynamic status in patients in distributive shock, or shock due to reduced cardiac output.
Dosage and Administration
• Correct hypovolemia, acidosis, and hypoxia prior to use. ( 2.1 ) • Administer in a large vein with an infusion pump preferably in an intensive care setting. ( 2.1 ) • Recommended starting dosage in adults and pediatric patients is 2 to 5 mcg/kg/minute as a continuous intravenous infusion. Titrate in 5 to 10 mcg/kg/minute increments based on hemodynamic response and tolerability, up to not more than 50 mcg/kg/minute. ( 2.2 ) • See the Full Prescribing Information for important preparation instructions and drug incompatibilities. ( 2.3 )
Warnings and Precautions
• Tissue ischemia : Severe peripheral and visceral vasoconstriction can occur. Address hypovolemia prior to use, monitor extremities, and infuse into large vein. ( 5.1 ) • Cardiac arrhythmias : Monitor closely. ( 5.2 ) • Hypotension after abrupt discontinuation : Gradually reduce infusion rate while expanding blood volume with intravenous fluids. ( 5.3 ) • Severe hypersensitivity reactions due to sodium metabisulfite excipient : May cause anaphylaxis including life-threatening or less severe asthmatic episodes in susceptible individuals. ( 5.4 )
Contraindications
Dopamine is contraindicated in patients with pheochromocytoma.
Adverse Reactions
The following adverse reactions are described elsewhere in the labeling: • Tissue Ischemia [see Warnings and Precautions (5.1) ] • Cardiac Arrhythmias [see Warnings and Precautions (5.2) ] • Hypotension [see Warnings and Precautions (5.3) ] • Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4) ] The following adverse reactions have been identified during post-approval use of dopamine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Cardiac Disorders : anginal pain, palpitation Gastrointestinal Disorders : nausea, vomiting Metabolism and Nutrition Disorders : azotemia Nervous System Disorders : headache, anxiety Respiratory Disorders : dyspnea Skin and Subcutaneous Tissue Disorders : piloerection Vascular Disorders : hypertension
Drug Interactions
See Table 1 for clinically significant drug interactions with dopamine. Table 1: Clinically Significant Drug Interactions with Dopamine Halogenated Anesthetics Clinical Impact: Concomitant use may increase cardiac autonomic irritability and can sensitize the myocardium to the action of dopamine which may lead to ventricular arrhythmias and hypertension. Intervention: Monitor cardiac rhythm. Examples: desflurane, enflurane, isoflurane, and sevoflurane. MAO Inhibitors Clinical Impact: Because dopamine is metabolized by monoamine oxidase (MAO), inhibition of this enzyme prolongs and potentiates the effect of dopamine which may result in severe hypertension and cardiac arrhythmia. Intervention: Reduce the recommended starting dosage to no greater than one-tenth (1/10) of the recommended dose in patients who have been treated with MAO inhibitors within two to three weeks prior to the administration of Dopamine HCl in Dextrose Injection. Examples: isocarboxazid, phenelzine, tranylcypromine, rasagiline, selegiline, linezolid. Tricyclic Antidepressants Clinical Impact: Concomitant use may potentiate the cardiovascular effects of dopamine (e.g., hypertension). Intervention: Monitor blood pressure. Examples: amitriptyline, desipramine, doxepin, imipramine, nortriptyline. Vasopressors Clinical Impact: Concomitant use may result in severe hypertension. Intervention: Monitor blood pressure. Examples: norepinephrine, epinephrine, oxytocin.
Storage and Handling
Dopamine Hydrochloride in 5% Dextrose Injection, USP, is supplied in 250 and 500 mL single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance) as follows. Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection. Unit of Sale Total Strength/Total Volume (Concentration) NDC 0409-7809-22 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-0042-12 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-7809-24 Case of 12 single-dose flexible containers 800 mg/500 mL (1,600 mcg/mL) NDC 0409-7810-22 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL) NDC 0409-1858-12 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL)
How Supplied
Dopamine Hydrochloride in 5% Dextrose Injection, USP, is supplied in 250 and 500 mL single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance) as follows. Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection. Unit of Sale Total Strength/Total Volume (Concentration) NDC 0409-7809-22 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-0042-12 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-7809-24 Case of 12 single-dose flexible containers 800 mg/500 mL (1,600 mcg/mL) NDC 0409-7810-22 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL) NDC 0409-1858-12 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL)
Medication Information
Warnings and Precautions
• Tissue ischemia : Severe peripheral and visceral vasoconstriction can occur. Address hypovolemia prior to use, monitor extremities, and infuse into large vein. ( 5.1 ) • Cardiac arrhythmias : Monitor closely. ( 5.2 ) • Hypotension after abrupt discontinuation : Gradually reduce infusion rate while expanding blood volume with intravenous fluids. ( 5.3 ) • Severe hypersensitivity reactions due to sodium metabisulfite excipient : May cause anaphylaxis including life-threatening or less severe asthmatic episodes in susceptible individuals. ( 5.4 )
Indications and Usage
Dopamine Hydrochloride in Dextrose Injection is indicated to improve hemodynamic status in patients in distributive shock, or shock due to reduced cardiac output.
Dosage and Administration
• Correct hypovolemia, acidosis, and hypoxia prior to use. ( 2.1 ) • Administer in a large vein with an infusion pump preferably in an intensive care setting. ( 2.1 ) • Recommended starting dosage in adults and pediatric patients is 2 to 5 mcg/kg/minute as a continuous intravenous infusion. Titrate in 5 to 10 mcg/kg/minute increments based on hemodynamic response and tolerability, up to not more than 50 mcg/kg/minute. ( 2.2 ) • See the Full Prescribing Information for important preparation instructions and drug incompatibilities. ( 2.3 )
Contraindications
Dopamine is contraindicated in patients with pheochromocytoma.
Adverse Reactions
The following adverse reactions are described elsewhere in the labeling: • Tissue Ischemia [see Warnings and Precautions (5.1) ] • Cardiac Arrhythmias [see Warnings and Precautions (5.2) ] • Hypotension [see Warnings and Precautions (5.3) ] • Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4) ] The following adverse reactions have been identified during post-approval use of dopamine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Cardiac Disorders : anginal pain, palpitation Gastrointestinal Disorders : nausea, vomiting Metabolism and Nutrition Disorders : azotemia Nervous System Disorders : headache, anxiety Respiratory Disorders : dyspnea Skin and Subcutaneous Tissue Disorders : piloerection Vascular Disorders : hypertension
Drug Interactions
See Table 1 for clinically significant drug interactions with dopamine. Table 1: Clinically Significant Drug Interactions with Dopamine Halogenated Anesthetics Clinical Impact: Concomitant use may increase cardiac autonomic irritability and can sensitize the myocardium to the action of dopamine which may lead to ventricular arrhythmias and hypertension. Intervention: Monitor cardiac rhythm. Examples: desflurane, enflurane, isoflurane, and sevoflurane. MAO Inhibitors Clinical Impact: Because dopamine is metabolized by monoamine oxidase (MAO), inhibition of this enzyme prolongs and potentiates the effect of dopamine which may result in severe hypertension and cardiac arrhythmia. Intervention: Reduce the recommended starting dosage to no greater than one-tenth (1/10) of the recommended dose in patients who have been treated with MAO inhibitors within two to three weeks prior to the administration of Dopamine HCl in Dextrose Injection. Examples: isocarboxazid, phenelzine, tranylcypromine, rasagiline, selegiline, linezolid. Tricyclic Antidepressants Clinical Impact: Concomitant use may potentiate the cardiovascular effects of dopamine (e.g., hypertension). Intervention: Monitor blood pressure. Examples: amitriptyline, desipramine, doxepin, imipramine, nortriptyline. Vasopressors Clinical Impact: Concomitant use may result in severe hypertension. Intervention: Monitor blood pressure. Examples: norepinephrine, epinephrine, oxytocin.
Storage and Handling
Dopamine Hydrochloride in 5% Dextrose Injection, USP, is supplied in 250 and 500 mL single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance) as follows. Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection. Unit of Sale Total Strength/Total Volume (Concentration) NDC 0409-7809-22 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-0042-12 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-7809-24 Case of 12 single-dose flexible containers 800 mg/500 mL (1,600 mcg/mL) NDC 0409-7810-22 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL) NDC 0409-1858-12 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL)
How Supplied
Dopamine Hydrochloride in 5% Dextrose Injection, USP, is supplied in 250 and 500 mL single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance) as follows. Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection. Unit of Sale Total Strength/Total Volume (Concentration) NDC 0409-7809-22 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-0042-12 Case of 12 single-dose flexible containers 400 mg/250 mL (1,600 mcg/mL) NDC 0409-7809-24 Case of 12 single-dose flexible containers 800 mg/500 mL (1,600 mcg/mL) NDC 0409-7810-22 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL) NDC 0409-1858-12 Case of 12 single-dose flexible containers 800 mg/250 mL (3,200 mcg/mL)
Description
Dopamine Hydrochloride in Dextrose Injection is indicated to improve hemodynamic status in patients in distributive shock, or shock due to reduced cardiac output.
Section 42229-5
Correct Hypovolemia, Acidosis, and Hypoxia
Address hypovolemia, acidosis, and hypoxia before initiating Dopamine HCl in Dextrose Injection. If patient does not respond to therapy, suspect occult hypovolemia. Acidosis may reduce the effectiveness of dopamine [see Warnings and Precautions (5.1)].
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing. Discard unused portion.
10 Overdosage
Manifestations of overdosage include excessive blood pressure elevation.
In the case of accidental overdosage, reduce rate of Dopamine HCl in Dextrose Injection infusion, or temporarily discontinue the Dopamine HCl in Dextrose Injection infusion until the overdosage related adverse reactions resolves. Since dopamine's duration of action is short, no additional remedial measures are usually necessary. If these measures fail to resolve the overdosage related adverse reactions, consider using an alpha-adrenergic blocking agent (e.g., phentolamine).
11 Description
Dopamine Hydrochloride in 5% Dextrose Injection, USP is a sterile, nonpyrogenic, premixed solution of dopamine hydrochloride in 5% dextrose injection for intravenous infusion.
Each 100 mL contains 160 mg (1,600 mcg/mL) or 320 mg (3,200 mcg/mL) of dopamine HCl; 5 grams of hydrous dextrose, in Water for Injection, and 50 mg of sodium metabisulfite (a stabilizer); pH = 2.5 to 4.5, and the following osmolar concentrations: 269, or 286 mOsmol/liter, respectively. May contain hydrochloric acid and/or sodium hydroxide for pH adjustment.
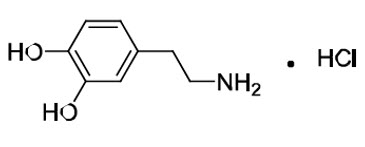
Dopamine HCl is chemically designated 3, 4-dihydroxyphenethylamine hydrochloride (C8H11NO2 ∙ HCl), a white crystalline powder freely soluble in water. Dopamine HCl has a molecular weight of 189.64 and it has the following structural formula:
Dopamine (also referred to as 3-hydroxytyramine) is a naturally occurring endogenous catecholamine.
Dextrose, USP is chemically designated D-glucose monohydrate (C6H12O6 ∙ H2O), a hexose sugar freely soluble in water. The molecular weight of dextrose (D-glucose) monohydrate is 198.17 and it has the following structural formula:
Water for Injection, USP is chemically designated H2O.
8.4 Pediatric Use
Dopamine HCl infusions have been used in pediatric patients from birth through adolescence. Most reports in pediatric patients describe dosing that is similar (on a mcg/kg/minute basis) to that used in adults [see Dosage and Administration (2.2)]. Except for vasoconstrictive effects caused by inadvertent infusion of dopamine into the umbilical artery, adverse reactions unique to pediatric patients have not been identified, nor have adverse reactions identified in adults been found to be more common in pediatric patients.
8.5 Geriatric Use
Clinical studies of dopamine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should start at the low end of the dosing range, reflecting the frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications
Dopamine is contraindicated in patients with pheochromocytoma.
5.1 Tissue Ischemia
Administration of dopamine to patients who are hypotensive from hypovolemia can result in severe peripheral and visceral vasoconstriction, decreased renal perfusion and hypouresis, tissue hypoxia, lactic acidosis, and poor systemic blood flow despite "normal" blood pressure. Address hypovolemia prior to initiating Dopamine HCl in Dextrose Injection [see Dosage and Administration (2.2)].
Gangrene of the extremities has occurred in patients with occlusive vascular disease or who received prolonged or high dose infusions. Monitor for changes to the skin of the extremities in susceptible patients.
Extravasation of Dopamine HCl in Dextrose Injection may cause necrosis and sloughing of surrounding tissue. To reduce the risk of extravasation, infuse into a large vein [see Dosage and Administration (2.1)], check the infusion site frequently for free flow, and monitor for signs of extravasation.
6 Adverse Reactions
The following adverse reactions are described elsewhere in the labeling:
-
•Tissue Ischemia [see Warnings and Precautions (5.1)]
-
•Cardiac Arrhythmias [see Warnings and Precautions (5.2)]
-
•Hypotension [see Warnings and Precautions (5.3)]
-
•Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4)]
The following adverse reactions have been identified during post-approval use of dopamine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: anginal pain, palpitation
Gastrointestinal Disorders: nausea, vomiting
Metabolism and Nutrition Disorders: azotemia
Nervous System Disorders: headache, anxiety
Respiratory Disorders: dyspnea
Skin and Subcutaneous Tissue Disorders: piloerection
Vascular Disorders: hypertension
7 Drug Interactions
See Table 1 for clinically significant drug interactions with dopamine.
|
Halogenated Anesthetics |
|
|
Clinical Impact: |
Concomitant use may increase cardiac autonomic irritability and can sensitize the myocardium to the action of dopamine which may lead to ventricular arrhythmias and hypertension. |
|
Intervention: |
Monitor cardiac rhythm. |
|
Examples: |
desflurane, enflurane, isoflurane, and sevoflurane. |
|
MAO Inhibitors |
|
|
Clinical Impact: |
Because dopamine is metabolized by monoamine oxidase (MAO), inhibition of this enzyme prolongs and potentiates the effect of dopamine which may result in severe hypertension and cardiac arrhythmia. |
|
Intervention: |
Reduce the recommended starting dosage to no greater than one-tenth (1/10) of the recommended dose in patients who have been treated with MAO inhibitors within two to three weeks prior to the administration of Dopamine HCl in Dextrose Injection. |
|
Examples: |
isocarboxazid, phenelzine, tranylcypromine, rasagiline, selegiline, linezolid. |
|
Tricyclic Antidepressants |
|
|
Clinical Impact: |
Concomitant use may potentiate the cardiovascular effects of dopamine (e.g., hypertension). |
|
Intervention: |
Monitor blood pressure. |
|
Examples: |
amitriptyline, desipramine, doxepin, imipramine, nortriptyline. |
|
Vasopressors |
|
|
Clinical Impact: |
Concomitant use may result in severe hypertension. |
|
Intervention: |
Monitor blood pressure. |
|
Examples: |
norepinephrine, epinephrine, oxytocin. |
12.2 Pharmacodynamics
Dopamine's onset of action occurs within five minutes of intravenous administration and the duration of action is less than about ten minutes. Dopamine effects are dosage-dependent.
-
•At <5 mcg/kg/minute, dopamine HCl activates dopamine D1 and D2 receptors in the renal, mesenteric, and coronary vasculature causing vasodilation.
-
•At 5 to 10 mcg/kg/minute, dopamine HCl activates beta-1 receptors enhancing heart rate and contractility.
-
•At >10 mcg/kg/minute, dopamine HCl activates alpha-1 receptors causing vasoconstriction and increased blood pressure
2.2 Recommended Dosage
The recommended starting dosage in adults and pediatric patients is 2 to 5 mcg/kg/minute as a continuous intravenous infusion [see Dosage and Administration (2.3)]. Titrate the infusion rate in 5 to 10 mcg/kg/minute increments based on hemodynamic response and tolerability, up to but not more than 50 mcg/kg/minute.
Infusion rates may be calculated using the following formula:
|
Infusion Rate (mL/hour) = |
[Dose (mcg/kg/minute) × Weight (kg) × 60 (minutes/hour)]
|
Example calculations for infusion rates are as follows:
Example 1: for a 60 kg person at the recommended initial dose of 2 mcg/kg/minute using a 1,600 mcg/mL concentration, the infusion rate would be as follows:
|
Infusion Rate (mL/hour) = |
[2 (mcg/kg/minute) × 60 (kg) × 60 (minutes/hour)] |
= 4.5 (mL/hour) |
|
1,600 (mcg/mL) |
Example 2: for a 70 kg person at a dose of 5 mcg/kg/minute using a 3,200 mcg/mL concentration, the infusion rate would be as follows:
|
Infusion Rate (mL/hour) = |
[5 (mcg/kg/minute) × 70 (kg) × 60 (minutes/hour)] |
= 6.56 (mL/hour) |
|
3,200 (mcg/mL) |
1 Indications and Usage
Dopamine Hydrochloride in Dextrose Injection is indicated to improve hemodynamic status in patients in distributive shock, or shock due to reduced cardiac output.
5.2 Cardiac Arrhythmias
Dopamine may cause arrhythmias. Monitor patients with arrhythmias and treat appropriately.
12.1 Mechanism of Action
Dopamine is a natural catecholamine formed by the decarboxylation of 3,4-dihydroxyphenylalanine (DOPA). It is a precursor to norepinephrine in noradrenergic nerves and is also a neurotransmitter in certain areas of the central nervous system, especially in the nigrostriatal tract, and in a few peripheral sympathetic nerves.
Dopamine elicits its pharmacological action by activating dopamine D1 and D2 receptors, beta-1 receptors and alpha-1 receptors. The activation of different receptors leading to its effects are dependent on dopamine dose.
2.3 Drug Incompatibilities
Dopamine HCl in Dextrose Injection is incompatible with the following products; therefore, avoid simultaneous administration (through the same infusion set):
-
•Sodium bicarbonate or other alkalinizing substances, because dopamine is inactivated in alkaline solution.
-
•Blood, because of the risk of pseudoagglutination of red cells
-
•Iron salts
Do not add additional medications in the premixed infusion solution.
5 Warnings and Precautions
-
•Tissue ischemia: Severe peripheral and visceral vasoconstriction can occur. Address hypovolemia prior to use, monitor extremities, and infuse into large vein. (5.1)
-
•Cardiac arrhythmias: Monitor closely. (5.2)
-
•Hypotension after abrupt discontinuation: Gradually reduce infusion rate while expanding blood volume with intravenous fluids. (5.3)
-
•Severe hypersensitivity reactions due to sodium metabisulfite excipient: May cause anaphylaxis including life-threatening or less severe asthmatic episodes in susceptible individuals. (5.4)
2 Dosage and Administration
-
•Correct hypovolemia, acidosis, and hypoxia prior to use. (2.1)
-
•Administer in a large vein with an infusion pump preferably in an intensive care setting. (2.1)
-
•Recommended starting dosage in adults and pediatric patients is 2 to 5 mcg/kg/minute as a continuous intravenous infusion. Titrate in 5 to 10 mcg/kg/minute increments based on hemodynamic response and tolerability, up to not more than 50 mcg/kg/minute. (2.2)
-
•See the Full Prescribing Information for important preparation instructions and drug incompatibilities. (2.3)
3 Dosage Forms and Strengths
The following strengths of Dopamine Hydrochloride in 5% Dextrose Injection, USP, are supplied in single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance):
-
•1,600 mcg/mL (250 or 500 mL)
-
•3,200 mcg/mL (250 mL)
Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection.
16 How Supplied/storage and Handling
Dopamine Hydrochloride in 5% Dextrose Injection, USP, is supplied in 250 and 500 mL single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance) as follows. Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection.
| Unit of Sale |
Total Strength/Total Volume
(Concentration) |
|---|---|
|
NDC 0409-7809-22
|
400 mg/250 mL |
|
NDC 0409-0042-12
|
400 mg/250 mL |
|
NDC 0409-7809-24
|
800 mg/500 mL |
|
NDC 0409-7810-22
|
800 mg/250 mL |
|
NDC 0409-1858-12
|
800 mg/250 mL |
5.3 Hypotension After Abrupt Discontinuation
Sudden cessation of the infusion rate may result in marked hypotension. Gradually reduce the infusion rate while expanding blood volume with intravenous fluids.
Principal Display Panel 250 Ml Bag Label 12726 02
250 mL
NDC 0409-0042-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
Each 100 mL contains dopamine hydrochloride 160 mg;
dextrose, hydrous 5 g in water for injection; sodium
metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
269 mOsmol/liter (calc.) pH 2.5 to 4.5
Single-dose container. Discard unused portion. For
intravenous use only. Recommended Dosage: See
Prescribing Information. Warning: contains sulfites.
Drug additives should not be made to this solution.
Sterile, nonpyrogenic.
Use only if solution is clear and container is undamaged.
Not made with natural rubber latex.
Rx only
5
OTHER
MADE IN GERMANY
Hospira
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
12726-02
Principal Display Panel 250 Ml Bag Label 12727 02
250 mL
NDC 0409-1858-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
Each 100 mL contains dopamine hydrochloride 320 mg;
dextrose, hydrous 5 g in water for injection; sodium
metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
286 mOsmol/liter (calc.) pH 2.5 to 4.5
Single-dose container.
Discard unused portion. For
intravenous use only. Recommended Dosage: See
Prescribing Information. Warning: contains sulfites
Drug additives should not be made to this solution.
Sterile, nonpyrogenic.
Use only if solution is clear and container is undamaged.
Not made with natural rubber latex.
Rx only
5
OTHER
MADE IN GERMANY
Hospira
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
12727-02
Principal Display Panel 250 Ml Bag Label Im 4355
250 mL
NDC 0409-7809-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
EACH 100 mL CONTAINS
DOPAMINE HYDROCHLORIDE
160 mg; DEXTROSE, HYDROUS 5 g IN
WATER FOR INJECTION; SODIUM
METABISULFITE ADDED 50 mg. MAY
CONTAIN HYDROCHLORIC ACID
AND/OR SODIUM HYDROXIDE FOR
pH ADJUSTMENT.
269 mOsmol/LITER (CALC.) pH 3.8
(2.5 to 4.5). SINGLE-DOSE
CONTAINER. DISCARD UNUSED
PORTION. FOR INTRAVENOUS
USE. USUAL DOSAGE: SEE INSERT.
WARNING: CONTAINS SULFITES.
DRUG ADDITIVES SHOULD NOT
BE MADE TO THIS SOLUTION.
STERILE NONPYROGENIC.
USE ONLY IF SOLUTION IS CLEAR
AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES
CONNECTIONS.
Rx ONLY
7
OTHER
Hospira
IM-4355
DISTRIBUTED BY HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Principal Display Panel 250 Ml Bag Label Im 4357
250 mL
NDC 0409-7810-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
EACH 100 mL CONTAINS DOPAMINE
HYDROCHLORIDE 320 mg; DEXTROSE,
HYDROUS 5 g IN WATER FOR
INJECTION; SODIUM METABISULFITE
ADDED 50 mg. MAY CONTAIN
HYDROCHLORIC ACID AND/OR
SODIUM HYDROXIDE FOR pH
ADJUSTMENT. 286 mOsmol/LITER
(CALC.) pH 3.8 (2.5 to 4.5). SINGLE-
DOSE CONTAINER. DISCARD UNUSED
PORTION. FOR INTRAVENOUS USE.
USUAL DOSAGE: SEE INSERT.
WARNING: CONTAINS SULFITES.
DRUG ADDITIVES SHOULD NOT
BE MADE TO THIS SOLUTION.
STERILE NONPYROGENIC.
USE ONLY IF SOLUTION IS CLEAR
AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES
CONNECTIONS.
Rx ONLY
7
OTHER
IM-4357
Hospira
DISTRIBUTED BY HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Principal Display Panel 250 Ml Bag Label Wr 1540
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 160 mg; dextrose,
hydrous 5 g in water for injection; sodium metabisulfite added 50 mg.
May contain hydrochloric acid and/or sodium hydroxide for pH
adjustment.
269 mOsmol/liter (calc.)
ph 3.8 (2.5 to 4.5).
Single-dose container. Discard unused portion. For intravenous use.
Usual dosage: see insert. WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution. The overwrap is a
moisture and oxygen barrier. Do not remove unit from overwrap until
ready for use. Visually inspect overwrap for tears or holes. Discard
unit if overwrap is damaged or if solution is darker than slightly yellow
or discolored in any other way. Use unit promptly when overwrap is
opened. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room
Temperature.] Protect from freezing. See insert. After removing the
overwrap, check for minute leaks by squeezing container firmly. If
leaks are found, discard solution as sterility may be impaired.
Rx only
250 mL
NDC 0409-7809-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
Single-dose container
F WR-1540
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 250 Ml Bag Label Wr 1544
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 320 mg; dextrose,
hydrous 5 g in water for injection; sodium metabisulfite added 50 mg.
May contain hydrochloric acid and/or sodium hydroxide for pH
adjustment.
286 mOsmol/liter (calc.)
ph 3.8 (2.5 to 4.5).
Single-dose container. Discard unused portion. For intravenous use.
Usual dosage: see insert. WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution. The overwrap is a
moisture and oxygen barrier. Do not remove unit from overwrap until
ready for use. Visually inspect overwrap for tears or holes. Discard
unit if overwrap is damaged or if solution is darker than slightly yellow
or discolored in any other way. Use unit promptly when overwrap is
opened. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room
Temperature.] Protect from freezing. See insert. After removing the
overwrap, check for minute leaks by squeezing container firmly. If
leaks are found, discard solution as sterility may be impaired.
Rx only
250 mL
NDC 0409-7810-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
Single-dose container
F WR-1544
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 250 Ml Bag Label 13444 01
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 160 mg; dextrose, hydrous 5 g in
water for injection; sodium metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
269 mOsmol/liter (calc.)
pH 2.5 to 4.5
Single-dose container. Discard unused portion. For intravenous use only.
Recommended Dosage: See Prescribing Information
WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution.
The overwrap is a moisture and oxygen barrier. Do not remove unit from overwrap
until ready for use. Visually inspect overwrap for tears or holes. Discard unit if
overwrap is damaged or if solution is darker than slightly yellow or discolored in
any other way. Use unit promptly when overwrap is opened. Store at 20°C to 25°C
(68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
After removing the overwrap, check for minute leaks by squeezing container firmly.
If leaks are found, discard solution as sterility may be impaired.
Not made with natural rubber latex.
Rx only
7
OTHER
250 mL
NDC 0409-0042-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
Single-dose container
13444-01
Hospira
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Principal Display Panel 250 Ml Bag Label 13445 01
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 320mg; dextrose, hydrous 5 g in
water for injection; sodium metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
286 mOsmol/liter (calc.)
pH 2.5 to 4.5
Single-dose container. Discard unused portion. For intravenous use only.
Recommended Dosage: See Prescribing Information.
WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution.
The overwrap is a moisture and oxygen barrier. Do not remove unit from overwrap
until ready for use. Visually inspect overwrap for tears or holes. Discard unit if
overwrap is damaged or if solution is darker than slightly yellow or discolored in
any other way. Use unit promptly when overwrap is opened. Store at 20°C to 25°C
(68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
After removing the overwrap, check for minute leaks by squeezing container
firmly. If leaks are found, discard solution as sterility may be impaired.
Not made with natural rubber latex.
Rx only
7
OTHER
250 mL
NDC 0409-1858-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
Single-dose container
13445-01
Hospira
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Principal Display Panel 500 Ml Bag Label Im 4356
500 mL
NDC 0409-7809-31
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/500 mL
(1,600 mcg/mL)
EACH 100 mL CONTAINS DOPAMINE
HYDROCHLORIDE 160 mg; DEXTROSE,
HYDROUS 5 g IN WATER FOR INJECTION;
SODIUM METABISULFITE ADDED 50 mg.
MAY CONTAIN HYDROCHLORIC ACID
AND/OR SODIUM HYDROXIDE FOR pH
ADJUSTMENT.
269 mOsmol/LITER (CALC.) pH 3.8 (2.5 to
4.5). SINGLE-DOSE CONTAINER. DISCARD
UNUSED PORTION. FOR INTRAVENOUS
USE. USUAL DOSAGE: SEE INSERT.
WARNING: CONTAINS SULFITES.
DRUG ADDITIVES SHOULD NOT
BE MADE TO THIS SOLUTION.
STERILE NONPYROGENIC.
USE ONLY IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED. MUST NOT
BE USED IN SERIES CONNECTIONS.
Rx ONLY
7
OTHER
Hospira
IM-4356
DISTRIBUTED BY HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Principal Display Panel 500 Ml Bag Label Wr 1543
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 160 mg; dextrose,
hydrous 5 g in water for injection; sodium metabisulfite added 50 mg.
May contain hydrochloric acid and/or sodium hydroxide for pH
adjustment.
269 mOsmol/liter (calc.)
ph 3.8 (2.5 to 4.5).
Single-dose container. Discard unused portion. For intravenous use.
Usual dosage: see insert. WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution. The overwrap is a
moisture and oxygen barrier. Do not remove unit from overwrap until
ready for use. Visually inspect overwrap for tears or holes. Discard
unit if overwrap is damaged or if solution is darker than slightly yellow
or discolored in any other way. Use unit promptly when overwrap is
opened. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room
Temperature.] Protect from freezing. See insert. After removing the
overwrap, check for minute leaks by squeezing container firmly. If
leaks are found, discard solution as sterility may be impaired.
Rx only
500 mL
NDC 0409-7809-31
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/500 mL
(1,600 mcg/mL)
Single-dose container
F WR-1543
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
5.4 Severe Hypersensitivity Reactions Due to Sodium Metabisulfite Excipient
Dopamine HCl in Dextrose Injection, contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people.
Structured Label Content
Section 42229-5 (42229-5)
Correct Hypovolemia, Acidosis, and Hypoxia
Address hypovolemia, acidosis, and hypoxia before initiating Dopamine HCl in Dextrose Injection. If patient does not respond to therapy, suspect occult hypovolemia. Acidosis may reduce the effectiveness of dopamine [see Warnings and Precautions (5.1)].
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing. Discard unused portion.
10 Overdosage (10 OVERDOSAGE)
Manifestations of overdosage include excessive blood pressure elevation.
In the case of accidental overdosage, reduce rate of Dopamine HCl in Dextrose Injection infusion, or temporarily discontinue the Dopamine HCl in Dextrose Injection infusion until the overdosage related adverse reactions resolves. Since dopamine's duration of action is short, no additional remedial measures are usually necessary. If these measures fail to resolve the overdosage related adverse reactions, consider using an alpha-adrenergic blocking agent (e.g., phentolamine).
11 Description (11 DESCRIPTION)
Dopamine Hydrochloride in 5% Dextrose Injection, USP is a sterile, nonpyrogenic, premixed solution of dopamine hydrochloride in 5% dextrose injection for intravenous infusion.
Each 100 mL contains 160 mg (1,600 mcg/mL) or 320 mg (3,200 mcg/mL) of dopamine HCl; 5 grams of hydrous dextrose, in Water for Injection, and 50 mg of sodium metabisulfite (a stabilizer); pH = 2.5 to 4.5, and the following osmolar concentrations: 269, or 286 mOsmol/liter, respectively. May contain hydrochloric acid and/or sodium hydroxide for pH adjustment.
Dopamine HCl is chemically designated 3, 4-dihydroxyphenethylamine hydrochloride (C8H11NO2 ∙ HCl), a white crystalline powder freely soluble in water. Dopamine HCl has a molecular weight of 189.64 and it has the following structural formula:
Dopamine (also referred to as 3-hydroxytyramine) is a naturally occurring endogenous catecholamine.
Dextrose, USP is chemically designated D-glucose monohydrate (C6H12O6 ∙ H2O), a hexose sugar freely soluble in water. The molecular weight of dextrose (D-glucose) monohydrate is 198.17 and it has the following structural formula:
Water for Injection, USP is chemically designated H2O.
8.4 Pediatric Use
Dopamine HCl infusions have been used in pediatric patients from birth through adolescence. Most reports in pediatric patients describe dosing that is similar (on a mcg/kg/minute basis) to that used in adults [see Dosage and Administration (2.2)]. Except for vasoconstrictive effects caused by inadvertent infusion of dopamine into the umbilical artery, adverse reactions unique to pediatric patients have not been identified, nor have adverse reactions identified in adults been found to be more common in pediatric patients.
8.5 Geriatric Use
Clinical studies of dopamine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should start at the low end of the dosing range, reflecting the frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
Dopamine is contraindicated in patients with pheochromocytoma.
5.1 Tissue Ischemia
Administration of dopamine to patients who are hypotensive from hypovolemia can result in severe peripheral and visceral vasoconstriction, decreased renal perfusion and hypouresis, tissue hypoxia, lactic acidosis, and poor systemic blood flow despite "normal" blood pressure. Address hypovolemia prior to initiating Dopamine HCl in Dextrose Injection [see Dosage and Administration (2.2)].
Gangrene of the extremities has occurred in patients with occlusive vascular disease or who received prolonged or high dose infusions. Monitor for changes to the skin of the extremities in susceptible patients.
Extravasation of Dopamine HCl in Dextrose Injection may cause necrosis and sloughing of surrounding tissue. To reduce the risk of extravasation, infuse into a large vein [see Dosage and Administration (2.1)], check the infusion site frequently for free flow, and monitor for signs of extravasation.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described elsewhere in the labeling:
-
•Tissue Ischemia [see Warnings and Precautions (5.1)]
-
•Cardiac Arrhythmias [see Warnings and Precautions (5.2)]
-
•Hypotension [see Warnings and Precautions (5.3)]
-
•Severe Hypersensitivity Reactions [see Warnings and Precautions (5.4)]
The following adverse reactions have been identified during post-approval use of dopamine. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: anginal pain, palpitation
Gastrointestinal Disorders: nausea, vomiting
Metabolism and Nutrition Disorders: azotemia
Nervous System Disorders: headache, anxiety
Respiratory Disorders: dyspnea
Skin and Subcutaneous Tissue Disorders: piloerection
Vascular Disorders: hypertension
7 Drug Interactions (7 DRUG INTERACTIONS)
See Table 1 for clinically significant drug interactions with dopamine.
|
Halogenated Anesthetics |
|
|
Clinical Impact: |
Concomitant use may increase cardiac autonomic irritability and can sensitize the myocardium to the action of dopamine which may lead to ventricular arrhythmias and hypertension. |
|
Intervention: |
Monitor cardiac rhythm. |
|
Examples: |
desflurane, enflurane, isoflurane, and sevoflurane. |
|
MAO Inhibitors |
|
|
Clinical Impact: |
Because dopamine is metabolized by monoamine oxidase (MAO), inhibition of this enzyme prolongs and potentiates the effect of dopamine which may result in severe hypertension and cardiac arrhythmia. |
|
Intervention: |
Reduce the recommended starting dosage to no greater than one-tenth (1/10) of the recommended dose in patients who have been treated with MAO inhibitors within two to three weeks prior to the administration of Dopamine HCl in Dextrose Injection. |
|
Examples: |
isocarboxazid, phenelzine, tranylcypromine, rasagiline, selegiline, linezolid. |
|
Tricyclic Antidepressants |
|
|
Clinical Impact: |
Concomitant use may potentiate the cardiovascular effects of dopamine (e.g., hypertension). |
|
Intervention: |
Monitor blood pressure. |
|
Examples: |
amitriptyline, desipramine, doxepin, imipramine, nortriptyline. |
|
Vasopressors |
|
|
Clinical Impact: |
Concomitant use may result in severe hypertension. |
|
Intervention: |
Monitor blood pressure. |
|
Examples: |
norepinephrine, epinephrine, oxytocin. |
12.2 Pharmacodynamics
Dopamine's onset of action occurs within five minutes of intravenous administration and the duration of action is less than about ten minutes. Dopamine effects are dosage-dependent.
-
•At <5 mcg/kg/minute, dopamine HCl activates dopamine D1 and D2 receptors in the renal, mesenteric, and coronary vasculature causing vasodilation.
-
•At 5 to 10 mcg/kg/minute, dopamine HCl activates beta-1 receptors enhancing heart rate and contractility.
-
•At >10 mcg/kg/minute, dopamine HCl activates alpha-1 receptors causing vasoconstriction and increased blood pressure
2.2 Recommended Dosage
The recommended starting dosage in adults and pediatric patients is 2 to 5 mcg/kg/minute as a continuous intravenous infusion [see Dosage and Administration (2.3)]. Titrate the infusion rate in 5 to 10 mcg/kg/minute increments based on hemodynamic response and tolerability, up to but not more than 50 mcg/kg/minute.
Infusion rates may be calculated using the following formula:
|
Infusion Rate (mL/hour) = |
[Dose (mcg/kg/minute) × Weight (kg) × 60 (minutes/hour)]
|
Example calculations for infusion rates are as follows:
Example 1: for a 60 kg person at the recommended initial dose of 2 mcg/kg/minute using a 1,600 mcg/mL concentration, the infusion rate would be as follows:
|
Infusion Rate (mL/hour) = |
[2 (mcg/kg/minute) × 60 (kg) × 60 (minutes/hour)] |
= 4.5 (mL/hour) |
|
1,600 (mcg/mL) |
Example 2: for a 70 kg person at a dose of 5 mcg/kg/minute using a 3,200 mcg/mL concentration, the infusion rate would be as follows:
|
Infusion Rate (mL/hour) = |
[5 (mcg/kg/minute) × 70 (kg) × 60 (minutes/hour)] |
= 6.56 (mL/hour) |
|
3,200 (mcg/mL) |
1 Indications and Usage (1 INDICATIONS AND USAGE)
Dopamine Hydrochloride in Dextrose Injection is indicated to improve hemodynamic status in patients in distributive shock, or shock due to reduced cardiac output.
5.2 Cardiac Arrhythmias
Dopamine may cause arrhythmias. Monitor patients with arrhythmias and treat appropriately.
12.1 Mechanism of Action
Dopamine is a natural catecholamine formed by the decarboxylation of 3,4-dihydroxyphenylalanine (DOPA). It is a precursor to norepinephrine in noradrenergic nerves and is also a neurotransmitter in certain areas of the central nervous system, especially in the nigrostriatal tract, and in a few peripheral sympathetic nerves.
Dopamine elicits its pharmacological action by activating dopamine D1 and D2 receptors, beta-1 receptors and alpha-1 receptors. The activation of different receptors leading to its effects are dependent on dopamine dose.
2.3 Drug Incompatibilities
Dopamine HCl in Dextrose Injection is incompatible with the following products; therefore, avoid simultaneous administration (through the same infusion set):
-
•Sodium bicarbonate or other alkalinizing substances, because dopamine is inactivated in alkaline solution.
-
•Blood, because of the risk of pseudoagglutination of red cells
-
•Iron salts
Do not add additional medications in the premixed infusion solution.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Tissue ischemia: Severe peripheral and visceral vasoconstriction can occur. Address hypovolemia prior to use, monitor extremities, and infuse into large vein. (5.1)
-
•Cardiac arrhythmias: Monitor closely. (5.2)
-
•Hypotension after abrupt discontinuation: Gradually reduce infusion rate while expanding blood volume with intravenous fluids. (5.3)
-
•Severe hypersensitivity reactions due to sodium metabisulfite excipient: May cause anaphylaxis including life-threatening or less severe asthmatic episodes in susceptible individuals. (5.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Correct hypovolemia, acidosis, and hypoxia prior to use. (2.1)
-
•Administer in a large vein with an infusion pump preferably in an intensive care setting. (2.1)
-
•Recommended starting dosage in adults and pediatric patients is 2 to 5 mcg/kg/minute as a continuous intravenous infusion. Titrate in 5 to 10 mcg/kg/minute increments based on hemodynamic response and tolerability, up to not more than 50 mcg/kg/minute. (2.2)
-
•See the Full Prescribing Information for important preparation instructions and drug incompatibilities. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
The following strengths of Dopamine Hydrochloride in 5% Dextrose Injection, USP, are supplied in single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance):
-
•1,600 mcg/mL (250 or 500 mL)
-
•3,200 mcg/mL (250 mL)
Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Dopamine Hydrochloride in 5% Dextrose Injection, USP, is supplied in 250 and 500 mL single-dose flexible plastic containers (the solutions are clear to slightly yellow in appearance) as follows. Each 100 mL contains 5 grams of hydrous dextrose in Water for Injection.
| Unit of Sale |
Total Strength/Total Volume
(Concentration) |
|---|---|
|
NDC 0409-7809-22
|
400 mg/250 mL |
|
NDC 0409-0042-12
|
400 mg/250 mL |
|
NDC 0409-7809-24
|
800 mg/500 mL |
|
NDC 0409-7810-22
|
800 mg/250 mL |
|
NDC 0409-1858-12
|
800 mg/250 mL |
5.3 Hypotension After Abrupt Discontinuation (5.3 Hypotension after Abrupt Discontinuation)
Sudden cessation of the infusion rate may result in marked hypotension. Gradually reduce the infusion rate while expanding blood volume with intravenous fluids.
Principal Display Panel 250 Ml Bag Label 12726 02 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label-12726-02)
250 mL
NDC 0409-0042-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
Each 100 mL contains dopamine hydrochloride 160 mg;
dextrose, hydrous 5 g in water for injection; sodium
metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
269 mOsmol/liter (calc.) pH 2.5 to 4.5
Single-dose container. Discard unused portion. For
intravenous use only. Recommended Dosage: See
Prescribing Information. Warning: contains sulfites.
Drug additives should not be made to this solution.
Sterile, nonpyrogenic.
Use only if solution is clear and container is undamaged.
Not made with natural rubber latex.
Rx only
5
OTHER
MADE IN GERMANY
Hospira
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
12726-02
Principal Display Panel 250 Ml Bag Label 12727 02 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label-12727-02)
250 mL
NDC 0409-1858-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
Each 100 mL contains dopamine hydrochloride 320 mg;
dextrose, hydrous 5 g in water for injection; sodium
metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
286 mOsmol/liter (calc.) pH 2.5 to 4.5
Single-dose container.
Discard unused portion. For
intravenous use only. Recommended Dosage: See
Prescribing Information. Warning: contains sulfites
Drug additives should not be made to this solution.
Sterile, nonpyrogenic.
Use only if solution is clear and container is undamaged.
Not made with natural rubber latex.
Rx only
5
OTHER
MADE IN GERMANY
Hospira
Distributed by Hospira, Inc.,
Lake Forest, IL 60045 USA
12727-02
Principal Display Panel 250 Ml Bag Label Im 4355 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label - IM-4355)
250 mL
NDC 0409-7809-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
EACH 100 mL CONTAINS
DOPAMINE HYDROCHLORIDE
160 mg; DEXTROSE, HYDROUS 5 g IN
WATER FOR INJECTION; SODIUM
METABISULFITE ADDED 50 mg. MAY
CONTAIN HYDROCHLORIC ACID
AND/OR SODIUM HYDROXIDE FOR
pH ADJUSTMENT.
269 mOsmol/LITER (CALC.) pH 3.8
(2.5 to 4.5). SINGLE-DOSE
CONTAINER. DISCARD UNUSED
PORTION. FOR INTRAVENOUS
USE. USUAL DOSAGE: SEE INSERT.
WARNING: CONTAINS SULFITES.
DRUG ADDITIVES SHOULD NOT
BE MADE TO THIS SOLUTION.
STERILE NONPYROGENIC.
USE ONLY IF SOLUTION IS CLEAR
AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES
CONNECTIONS.
Rx ONLY
7
OTHER
Hospira
IM-4355
DISTRIBUTED BY HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Principal Display Panel 250 Ml Bag Label Im 4357 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label - IM-4357)
250 mL
NDC 0409-7810-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
EACH 100 mL CONTAINS DOPAMINE
HYDROCHLORIDE 320 mg; DEXTROSE,
HYDROUS 5 g IN WATER FOR
INJECTION; SODIUM METABISULFITE
ADDED 50 mg. MAY CONTAIN
HYDROCHLORIC ACID AND/OR
SODIUM HYDROXIDE FOR pH
ADJUSTMENT. 286 mOsmol/LITER
(CALC.) pH 3.8 (2.5 to 4.5). SINGLE-
DOSE CONTAINER. DISCARD UNUSED
PORTION. FOR INTRAVENOUS USE.
USUAL DOSAGE: SEE INSERT.
WARNING: CONTAINS SULFITES.
DRUG ADDITIVES SHOULD NOT
BE MADE TO THIS SOLUTION.
STERILE NONPYROGENIC.
USE ONLY IF SOLUTION IS CLEAR
AND CONTAINER IS UNDAMAGED.
MUST NOT BE USED IN SERIES
CONNECTIONS.
Rx ONLY
7
OTHER
IM-4357
Hospira
DISTRIBUTED BY HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Principal Display Panel 250 Ml Bag Label Wr 1540 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label - WR-1540)
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 160 mg; dextrose,
hydrous 5 g in water for injection; sodium metabisulfite added 50 mg.
May contain hydrochloric acid and/or sodium hydroxide for pH
adjustment.
269 mOsmol/liter (calc.)
ph 3.8 (2.5 to 4.5).
Single-dose container. Discard unused portion. For intravenous use.
Usual dosage: see insert. WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution. The overwrap is a
moisture and oxygen barrier. Do not remove unit from overwrap until
ready for use. Visually inspect overwrap for tears or holes. Discard
unit if overwrap is damaged or if solution is darker than slightly yellow
or discolored in any other way. Use unit promptly when overwrap is
opened. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room
Temperature.] Protect from freezing. See insert. After removing the
overwrap, check for minute leaks by squeezing container firmly. If
leaks are found, discard solution as sterility may be impaired.
Rx only
250 mL
NDC 0409-7809-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
Single-dose container
F WR-1540
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 250 Ml Bag Label Wr 1544 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label - WR-1544)
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 320 mg; dextrose,
hydrous 5 g in water for injection; sodium metabisulfite added 50 mg.
May contain hydrochloric acid and/or sodium hydroxide for pH
adjustment.
286 mOsmol/liter (calc.)
ph 3.8 (2.5 to 4.5).
Single-dose container. Discard unused portion. For intravenous use.
Usual dosage: see insert. WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution. The overwrap is a
moisture and oxygen barrier. Do not remove unit from overwrap until
ready for use. Visually inspect overwrap for tears or holes. Discard
unit if overwrap is damaged or if solution is darker than slightly yellow
or discolored in any other way. Use unit promptly when overwrap is
opened. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room
Temperature.] Protect from freezing. See insert. After removing the
overwrap, check for minute leaks by squeezing container firmly. If
leaks are found, discard solution as sterility may be impaired.
Rx only
250 mL
NDC 0409-7810-11
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
Single-dose container
F WR-1544
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
Principal Display Panel 250 Ml Bag Label 13444 01 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label -13444-01)
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 160 mg; dextrose, hydrous 5 g in
water for injection; sodium metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
269 mOsmol/liter (calc.)
pH 2.5 to 4.5
Single-dose container. Discard unused portion. For intravenous use only.
Recommended Dosage: See Prescribing Information
WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution.
The overwrap is a moisture and oxygen barrier. Do not remove unit from overwrap
until ready for use. Visually inspect overwrap for tears or holes. Discard unit if
overwrap is damaged or if solution is darker than slightly yellow or discolored in
any other way. Use unit promptly when overwrap is opened. Store at 20°C to 25°C
(68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
After removing the overwrap, check for minute leaks by squeezing container firmly.
If leaks are found, discard solution as sterility may be impaired.
Not made with natural rubber latex.
Rx only
7
OTHER
250 mL
NDC 0409-0042-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
400 mg/250 mL
(1,600 mcg/mL)
Single-dose container
13444-01
Hospira
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Principal Display Panel 250 Ml Bag Label 13445 01 (PRINCIPAL DISPLAY PANEL - 250 mL Bag Label -13445-01)
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 320mg; dextrose, hydrous 5 g in
water for injection; sodium metabisulfite added 50 mg. May contain hydrochloric
acid and/or sodium hydroxide for pH adjustment.
286 mOsmol/liter (calc.)
pH 2.5 to 4.5
Single-dose container. Discard unused portion. For intravenous use only.
Recommended Dosage: See Prescribing Information.
WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution.
The overwrap is a moisture and oxygen barrier. Do not remove unit from overwrap
until ready for use. Visually inspect overwrap for tears or holes. Discard unit if
overwrap is damaged or if solution is darker than slightly yellow or discolored in
any other way. Use unit promptly when overwrap is opened. Store at 20°C to 25°C
(68°F to 77°F). [See USP Controlled Room Temperature.] Protect from freezing.
After removing the overwrap, check for minute leaks by squeezing container
firmly. If leaks are found, discard solution as sterility may be impaired.
Not made with natural rubber latex.
Rx only
7
OTHER
250 mL
NDC 0409-1858-01
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/250 mL
(3,200 mcg/mL)
Single-dose container
13445-01
Hospira
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Principal Display Panel 500 Ml Bag Label Im 4356 (PRINCIPAL DISPLAY PANEL - 500 mL Bag Label - IM-4356)
500 mL
NDC 0409-7809-31
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/500 mL
(1,600 mcg/mL)
EACH 100 mL CONTAINS DOPAMINE
HYDROCHLORIDE 160 mg; DEXTROSE,
HYDROUS 5 g IN WATER FOR INJECTION;
SODIUM METABISULFITE ADDED 50 mg.
MAY CONTAIN HYDROCHLORIC ACID
AND/OR SODIUM HYDROXIDE FOR pH
ADJUSTMENT.
269 mOsmol/LITER (CALC.) pH 3.8 (2.5 to
4.5). SINGLE-DOSE CONTAINER. DISCARD
UNUSED PORTION. FOR INTRAVENOUS
USE. USUAL DOSAGE: SEE INSERT.
WARNING: CONTAINS SULFITES.
DRUG ADDITIVES SHOULD NOT
BE MADE TO THIS SOLUTION.
STERILE NONPYROGENIC.
USE ONLY IF SOLUTION IS CLEAR AND
CONTAINER IS UNDAMAGED. MUST NOT
BE USED IN SERIES CONNECTIONS.
Rx ONLY
7
OTHER
Hospira
IM-4356
DISTRIBUTED BY HOSPIRA, INC., LAKE FOREST, IL 60045 USA
Principal Display Panel 500 Ml Bag Label Wr 1543 (PRINCIPAL DISPLAY PANEL - 500 mL Bag Label - WR-1543)
TO OPEN — TEAR AT NOTCH
Each 100 mL contains dopamine hydrochloride 160 mg; dextrose,
hydrous 5 g in water for injection; sodium metabisulfite added 50 mg.
May contain hydrochloric acid and/or sodium hydroxide for pH
adjustment.
269 mOsmol/liter (calc.)
ph 3.8 (2.5 to 4.5).
Single-dose container. Discard unused portion. For intravenous use.
Usual dosage: see insert. WARNING: CONTAINS SULFITES.
Drug additives should not be made to this solution. The overwrap is a
moisture and oxygen barrier. Do not remove unit from overwrap until
ready for use. Visually inspect overwrap for tears or holes. Discard
unit if overwrap is damaged or if solution is darker than slightly yellow
or discolored in any other way. Use unit promptly when overwrap is
opened. Store at 20 to 25°C (68 to 77°F). [See USP Controlled Room
Temperature.] Protect from freezing. See insert. After removing the
overwrap, check for minute leaks by squeezing container firmly. If
leaks are found, discard solution as sterility may be impaired.
Rx only
500 mL
NDC 0409-7809-31
DOPAMINE HCl
in 5% Dextrose Injection, USP
800 mg/500 mL
(1,600 mcg/mL)
Single-dose container
F WR-1543
Distributed by
Hospira, Inc., Lake Forest, IL 60045 USA
Hospira
5.4 Severe Hypersensitivity Reactions Due to Sodium Metabisulfite Excipient (5.4 Severe Hypersensitivity Reactions due to Sodium Metabisulfite Excipient)
Dopamine HCl in Dextrose Injection, contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:13.733098 · Updated: 2026-03-14T22:36:30.622003