These Highlights Do Not Include All The Information Needed To Use Plegridy®
08f0ea03-4e6d-195d-aef4-886e32befa95
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
PLEGRIDY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
Dosage and Administration
For subcutaneous or intramuscular use only ( 2.1 ) Recommended dose: 125 micrograms every 14 days ( 2.1 ) PLEGRIDY dose should be titrated, starting with 63 micrograms on day 1, 94 micrograms on day 15, and 125 micrograms (full dose) on day 29 ( 2.1 ) A healthcare professional should train patients in the proper technique for self-administering subcutaneous injections using the prefilled pen or syringe or intramuscular injections using the prefilled syringe ( 2.2 ) Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms ( 2.3 )
Contraindications
PLEGRIDY is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta or peginterferon, or any other component of PLEGRIDY [see Warnings and Precautions ( 5.3 )] .
Warnings and Precautions
Hepatic injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of PLEGRIDY if hepatic injury occurs ( 5.1 ) Depression and suicide: advise patients to report immediately any symptom of depression or suicidal ideation to their healthcare provider; consider discontinuation of PLEGRIDY if depression occurs ( 5.2 ) Anaphylaxis and other allergic reactions: Discontinue PLEGRIDY if a serious allergic reaction occurs ( 5.3 ) Injection site reactions: Do not administer PLEGRIDY into affected area until fully healed; if multiple lesions occur, change injection site or discontinue PLEGRIDY until healing of skin lesions ( 5.4 ) Congestive heart failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms ( 5.5 ) Decreased peripheral blood counts: monitor complete blood counts ( 5.6 ) Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported with interferon beta products. Discontinue PLEGRIDY if clinical symptoms and laboratory findings consistent with TMA occur ( 5.7 ) Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including PLEGRIDY. Discontinue PLEGRIDY if PAH is diagnosed ( 5.8 ) Autoimmune disorders: consider discontinuation of PLEGRIDY if a new autoimmune disorder occurs ( 5.9 )
Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of labeling: Hepatic Injury [see Warnings and Precautions ( 5.1 )] Depression and Suicide [see Warnings and Precautions ( 5.2 )] Anaphylaxis and Other Allergic Reactions [see Warnings and Precautions ( 5.3 )] Injection Site Reactions Including Necrosis [see Warnings and Precautions ( 5.4 )] Congestive Heart Failure [see Warnings and Precautions (Section 5.5 )] Decreased Peripheral Blood Counts [see Warnings and Precautions ( 5.6 )] Thrombotic Microangiopathy [see Warnings and Precautions ( 5.7 )] Pulmonary Arterial Hypertension [see Warnings and Precautions ( 5.8 )] Autoimmune Disorders [see Warnings and Precautions ( 5.9 )] Seizures [see Warnings and Precautions ( 5.10 )]
How Supplied
PLEGRIDY (peginterferon beta-1a) injection is a sterile, preservative-free, clear to slightly opalescent and colorless to slightly yellow solution supplied as a 0.5 mL single-dose prefilled pen or a 0.5 mL single-dose prefilled syringe.
Storage and Handling
Store PLEGRIDY prefilled pens and prefilled syringes in a refrigerator between 2°C to 8°C (36°F to 46°F) in the closed original carton to protect from light until ready for injection. Do not freeze. Discard if frozen. If refrigeration is unavailable, PLEGRIDY may be stored at room temperature up to 25°C (77°F) for a period up to 30 days, protected from light. PLEGRIDY can be removed from, and returned to, a refrigerator if necessary. The total combined time out of refrigeration should not exceed 30 days. PLEGRIDY prefilled syringe for intramuscular administration contains natural rubber latex which may cause allergic reactions. Dispose in a sharps-bin container or other hard plastic or metal sealable container. Always follow local regulations for disposal.
Description
Warnings and Precautions ( 5.8 ) 7/2023
Medication Information
Warnings and Precautions
Hepatic injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of PLEGRIDY if hepatic injury occurs ( 5.1 ) Depression and suicide: advise patients to report immediately any symptom of depression or suicidal ideation to their healthcare provider; consider discontinuation of PLEGRIDY if depression occurs ( 5.2 ) Anaphylaxis and other allergic reactions: Discontinue PLEGRIDY if a serious allergic reaction occurs ( 5.3 ) Injection site reactions: Do not administer PLEGRIDY into affected area until fully healed; if multiple lesions occur, change injection site or discontinue PLEGRIDY until healing of skin lesions ( 5.4 ) Congestive heart failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms ( 5.5 ) Decreased peripheral blood counts: monitor complete blood counts ( 5.6 ) Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported with interferon beta products. Discontinue PLEGRIDY if clinical symptoms and laboratory findings consistent with TMA occur ( 5.7 ) Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including PLEGRIDY. Discontinue PLEGRIDY if PAH is diagnosed ( 5.8 ) Autoimmune disorders: consider discontinuation of PLEGRIDY if a new autoimmune disorder occurs ( 5.9 )
Indications and Usage
PLEGRIDY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
Dosage and Administration
For subcutaneous or intramuscular use only ( 2.1 ) Recommended dose: 125 micrograms every 14 days ( 2.1 ) PLEGRIDY dose should be titrated, starting with 63 micrograms on day 1, 94 micrograms on day 15, and 125 micrograms (full dose) on day 29 ( 2.1 ) A healthcare professional should train patients in the proper technique for self-administering subcutaneous injections using the prefilled pen or syringe or intramuscular injections using the prefilled syringe ( 2.2 ) Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms ( 2.3 )
Contraindications
PLEGRIDY is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta or peginterferon, or any other component of PLEGRIDY [see Warnings and Precautions ( 5.3 )] .
Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of labeling: Hepatic Injury [see Warnings and Precautions ( 5.1 )] Depression and Suicide [see Warnings and Precautions ( 5.2 )] Anaphylaxis and Other Allergic Reactions [see Warnings and Precautions ( 5.3 )] Injection Site Reactions Including Necrosis [see Warnings and Precautions ( 5.4 )] Congestive Heart Failure [see Warnings and Precautions (Section 5.5 )] Decreased Peripheral Blood Counts [see Warnings and Precautions ( 5.6 )] Thrombotic Microangiopathy [see Warnings and Precautions ( 5.7 )] Pulmonary Arterial Hypertension [see Warnings and Precautions ( 5.8 )] Autoimmune Disorders [see Warnings and Precautions ( 5.9 )] Seizures [see Warnings and Precautions ( 5.10 )]
Storage and Handling
Store PLEGRIDY prefilled pens and prefilled syringes in a refrigerator between 2°C to 8°C (36°F to 46°F) in the closed original carton to protect from light until ready for injection. Do not freeze. Discard if frozen. If refrigeration is unavailable, PLEGRIDY may be stored at room temperature up to 25°C (77°F) for a period up to 30 days, protected from light. PLEGRIDY can be removed from, and returned to, a refrigerator if necessary. The total combined time out of refrigeration should not exceed 30 days. PLEGRIDY prefilled syringe for intramuscular administration contains natural rubber latex which may cause allergic reactions. Dispose in a sharps-bin container or other hard plastic or metal sealable container. Always follow local regulations for disposal.
How Supplied
PLEGRIDY (peginterferon beta-1a) injection is a sterile, preservative-free, clear to slightly opalescent and colorless to slightly yellow solution supplied as a 0.5 mL single-dose prefilled pen or a 0.5 mL single-dose prefilled syringe.
Description
Warnings and Precautions ( 5.8 ) 7/2023
Section 42229-5
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised: 7/2023 |
|
Medication Guide PLEGRIDY® (PLEGG-rih-dee) (peginterferon beta-1a) injection, for subcutaneous or intramuscular use |
|
| Read this Medication Guide before you start using PLEGRIDY, and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is the most important information I should know about PLEGRIDY? |
|
PLEGRIDY can cause serious side effects, including:
|
|
What is PLEGRIDY?
|
|
Do not take PLEGRIDY if you:
|
|
Before using PLEGRIDY, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |
How should I use PLEGRIDY?
|
|
| What are the possible side effects of PLEGRIDY? | |
|
See “What is the most important information I should know about PLEGRIDY?”
PLEGRIDY may cause serious side effects, including:
|
|
|
Flu-like symptoms or other common side effects of PLEGRIDY may include: headache, muscle and joint aches, fever, chills, or tiredness. These are not all the possible side effects of PLEGRIDY. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
| General Information about the safe and effective use of PLEGRIDY. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use PLEGRIDY for a condition for which it was not prescribed. Do not give PLEGRIDY to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about PLEGRIDY that is written for health professionals. | |
| What are the ingredients in PLEGRIDY? | |
| Active ingredient: peginterferon beta-1a. | |
| Inactive ingredients: | |
|
|
| For more information, go to www.plegridy.com or call 1-800-456-2255. Manufactured by: Biogen Inc., Cambridge, MA 02142, U.S. License # 1697 PLEGRIDY is a registered trademark of Biogen. ©2013-2023 Biogen |
Section 43683-2
| Warnings and Precautions (5.8) | 7/2023 |
Section 51945-4
Principal Display Panel - Carton Label
NDC 64406-012-01
Plegridy-Pen
(peginterferon beta-1a)
Injection
63. mcg/0.5 mL
94 mcg/0.5 mL
For subcutaneous use only
Starter Pack
Contents:
Dose 1: 1 Single-Dose 63 mcg/0.5 mL
Prefilled Autoinjector
Dose 2: 1 Single-Dose 94 mcg/0.5 mL
Prefilled Autoinjector
Dispense with enclosed Medicaton Guide
Rx Only
Biogen ®
Section 59845-8
Instructions for Use
PLEGRIDY ® (PLEGG-rih-dee)
(peginterferon beta-1a) injection, for Subcutaneous Use
Single-Dose Prefilled Pen
Starter Pack
Important: Do not remove the Plegridy Pen cap until you are ready to inject.
How to Inject your Plegridy Pen
Read this Instructions for Use before you start using Plegridy and each time you get a refill of your prescription. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Note:
- Before you use your Plegridy Pen for the first time, your healthcare provider should show you or your caregiver how to prepare and inject your Plegridy Pen the right way.
- If you experience difficulty or have questions, call 1-800-456-2255.
- Plegridy Pen is for use under the skin only (subcutaneous).
- Each Plegridy Pen can be used 1 time only. Do not share your Plegridy Pen with anyone else. By sharing the Pen, you may give an infection to them or get an infection from them.
- Take your Plegridy Pen out of the refrigerator and let it come to room temperature for at least 30 minutes before your injection.
- Do not use more than 1 Plegridy Pen every 14 days.
- Do not use your Plegridy Pen if it has been dropped or is visibly damaged.
How should I store Plegridy?
- Store Plegridy in the refrigerator between 36°F to 46°F (2°C to 8°C).
- If a refrigerator is not available, PLEGRIDY may be stored at room temperature up to 77°F (25°C) for up to 30 days in total.
- Keep Plegridy in the original carton to protect it from light.
- Do not freeze Plegridy.
- Keep Plegridy Pens, needles, and all medicines out of the reach of children.
Supplies needed for your Starter Pack for Plegridy Pen injection (See Figure A):
- 1 Starter Pack for Plegridy Pen which contains:
- 1 Plegridy 63 mcg Pen (orange pen)
- 1 Plegridy 94 mcg Pen (blue pen)
Additional supplies which are not included in the pack (See Figure B):
- alcohol wipe
- gauze pad
- adhesive bandage
- 1 sharps container for throwing away used needles and Plegridy Pens. See “Disposing of your used Plegridy Pens” at the end of these instructions.
- a well-lit area and a clean, flat surface to work on, like a table
Dose Schedule
- Choose the right Plegridy Pen for your dose. The Starter Pack for Plegridy Pen contains your first 2 injections to slowly adjust your dose.
| When | Which Dose | Choose |
|
Day 1
(63 mcg) |
First Dose:
63 mcg |
Orange Pen (See Figure C) |
| When | Which Dose | Choose |
|
Day 15
(94 mcg) |
Second Dose:
94 mcg |
Blue Pen (See Figure D) |
- Make sure you use the 63 mcg orange pen for your first dose (on day 1).
- Make sure you use the 94 mcg blue pen for your second dose (on day 15).
Ask your healthcare provider which Plegridy Pen you should use for your correct dose if you are not sure.
Before Use – Parts of your Plegridy Pen before use (See Figure E):
Important: Do not remove the Plegridy Pen cap until you are ready to inject. If you remove the cap before you are ready to inject, do not re-cap the pen. Re-capping could cause the pen to lock.
Preparing for your injection:
Step 1: Take your Plegridy Pen out of the refrigerator and let it come to room temperature for at least 30 minutes.
- Do not use external heat sources such as hot water to warm your Plegridy Pen.
Step 2: Collect your supplies and wash your hands
- Find a well-lit area and a clean, flat surface like a table and collect all the supplies you will need to give yourself or to receive an injection.
- Wash your hands with soap and water.
Step 3: Check your Plegridy Pen (see Figure F)
|
|
|
|
|
| Step 4: Choose and clean your injection site | |
|
|
|
Giving your injection: Step 5: Remove the Plegridy Pen cap |
|
|
Figure H |
| Step 6: Give your injection | |
|
|
|
If you do not hear clicking sounds, or you do not see green checkmarks in the injection status window after trying to give your injection, your Plegridy Pen may have locked and you should call 1-800-456-2255 for help.
Step 7: Remove your Plegridy Pen from your injection site |
|
|
|
| Step 8: Check to make sure you have received your full dose of Plegridy (see Figure M) | |
|
|
After your injection:
After Use – Parts of your Plegridy Pen (Figure N)
Important: Do not touch the needle cover in any way. You could get a needle stick injury.
Note: After the pen has been removed from the injection site, the needle cover will lock to protect against needle stick injury. Do not recap your Plegridy Pen.
Step 9: Disposing of your used Plegridy Pens
- Put your used Plegridy Pens in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) Plegridy Pens in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and pens. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- Do not recap your Plegridy Pen.
Step 10: Check your injection site
- Plegridy may commonly cause redness, pain or swelling of your skin at the injection site.
- After 2 hours check your injection site for redness, swelling or tenderness.
- Call your healthcare provider right away if your injection site becomes swollen and painful or the area looks infected and does not heal within a few days.
Questions?
For product or service-related questions, call 1-800-456-2255 or go to www.plegridy.com.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by: Biogen Inc., Cambridge, MA 02142, U.S. License # 1697
©2013-2022 Biogen. All rights reserved. 1-800-456-2255
Issued: 03/2020
43839-03
5.10 Seizures
Seizures are associated with the use of interferon beta.
The incidence of seizures in multiple sclerosis clinical studies was less than 1% in patients receiving PLEGRIDY and placebo.
Exercise caution when administering PLEGRIDY to patients with a seizure disorder.
11 Description
Peginterferon beta-1a is a covalent conjugate of recombinant interferon beta-1a (approximate molecular weight [MW] 20,000 daltons) with a single, linear methoxy poly(ethyleneglycol)-O-2-methylpropionaldehyde molecule (approximate MW 20,000 daltons). Interferon beta-1a is produced as a glycosylated protein using genetically-engineered Chinese hamster ovary cells into which the human interferon beta gene has been introduced. The amino acid sequence of recombinant interferon beta-1a is identical to that of the human interferon beta counterpart.
The molecular weight of peginterferon beta-1a is approximately 44,000 daltons, consistent with the mass of the protein, the carbohydrate moieties (approximately 2,500 daltons), and the attached poly(ethylene glycol).
Peginterferon beta-1a 125 mcg contains 125 mcg of interferon beta-1a plus 125 mcg of poly(ethylene glycol). Using the World Health Organization International Standard for interferon beta, peginterferon beta-1a has a specific antiviral activity of approximately 100 million International Units (MIU) per mg of protein as determined using an in vitro cytopathic effect assay. Peginterferon beta-1a 125 mcg contains approximately 12 MIU of antiviral activity.
16.1 How Supplied
PLEGRIDY (peginterferon beta-1a) injection is a sterile, preservative-free, clear to slightly opalescent and colorless to slightly yellow solution supplied as a 0.5 mL single-dose prefilled pen or a 0.5 mL single-dose prefilled syringe.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of PLEGRIDY did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
5.1 Hepatic Injury
Severe hepatic injury, including hepatitis, autoimmune hepatitis, and rare cases of severe hepatic failure, have been reported with interferon beta. Asymptomatic elevation of hepatic transaminases has also been reported, and in some patients has recurred upon rechallenge with interferon beta.
Elevations in hepatic enzymes and hepatic injury have been observed with the use of PLEGRIDY in clinical studies. The incidence of increases in hepatic transaminases was greater in patients taking PLEGRIDY than in those taking placebo. The incidence of elevations of alanine aminotransferase above 5 times the upper limit of normal was 1% in placebo-treated patients and 2% in PLEGRIDY-treated patients. The incidence of elevations of aspartate aminotransferase above 5 times the upper limit of normal was less than 1% in placebo-treated patients and less than 1% in PLEGRIDY-treated patients. Elevations of serum hepatic transaminases combined with elevated bilirubin occurred in 2 patients. Both cases resolved following discontinuation of PLEGRIDY.
Cases of noninfectious hepatitis have been reported in the postmarketing setting with use of PLEGRIDY.
Monitor patients for signs and symptoms of hepatic injury.
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity.
The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies in the studies described below with the incidence of antibodies in other studies or to other interferon beta-1a products may be misleading.
In Study 1, fewer than 1% of patients treated with PLEGRIDY SC every 14 days for 1 year developed neutralizing antibodies. Approximately 7% of PLEGRIDY SC-treated patients developed antibodies to the polyethylene glycol moiety.
No formal studies have been conducted with regards to immunogenicity of the intramuscular route of administration of PLEGRIDY.
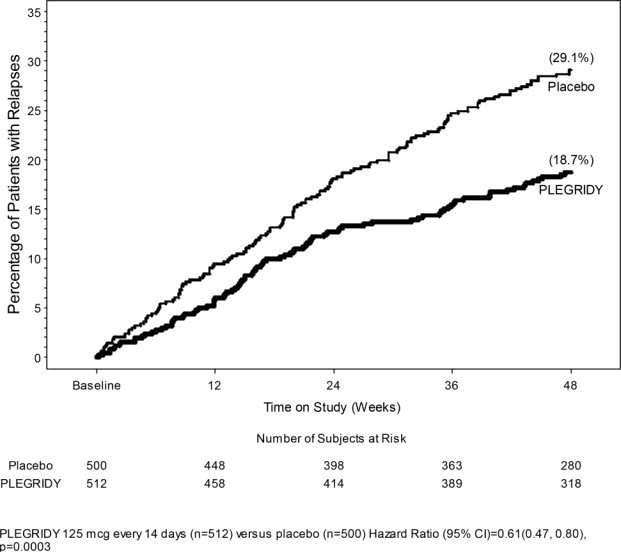
14 Clinical Studies
The efficacy of PLEGRIDY was demonstrated in the randomized, double-blind, and placebo-controlled phase (year 1) of Study 1. The trial compared clinical and MRI outcomes at 48 weeks in patients who received PLEGRIDY 125 micrograms (n=512) or placebo (n=500) by the subcutaneous route, once every 14 days.
Study 1 enrolled patients who had a baseline Expanded Disability Status Scale (EDSS) score from 0 to 5, who had experienced at least 2 relapses within the previous three years, and had experienced at least 1 relapse in the previous year. The trial excluded patients with progressive forms of multiple sclerosis. The mean age of the study population was 37 years, the mean disease duration was 3.6 years, and the mean EDSS score at baseline was 2.46. The majority of the patients were women (71%).
The trial scheduled neurological evaluations at baseline, every 12 weeks, and at the time of a suspected relapse. Brain MRI evaluations were scheduled at baseline, week 24, and week 48.
The primary outcome was the annualized relapse rate over 1 year. Secondary outcomes included the proportion of patients relapsing, number of new or newly enlarging T2 hyperintense lesions, and time to confirmed disability progression. Confirmed disability progression was defined as follows: if the baseline EDSS score was 0, a sustained 12-week increase in EDSS score of 1.5 points was required; if the baseline EDSS score was greater than 0, a sustained 12-week increase in EDSS score of 1 point was required. Table 4 and Figure 1 show the results of Study 1.
| Endpoint |
PLEGRIDY
125 micrograms every 14 days |
Placebo | p-value |
|---|---|---|---|
| Clinical outcomes at 48 weeks | N=512 | N=500 | |
| Annualized relapse rate | 0.26 | 0.40 | 0.0007 |
| Relative reduction | 36% | ||
| Proportion of patients with relapses | 0.19 | 0.29 | 0.0003 |
| Relative risk reduction | 39% | ||
| Proportion of patients with disability progression | 0.07 | 0.11 | 0.0383 |
| Relative risk reduction | 38% | ||
| MRI outcomes at 48 weeks | N=457 | N=476 | |
| Mean number of new or newly enlarging T2 hyperintense lesions | 3.6 | 10.9 | <0.0001 |
| Relative reduction | 67% | ||
| Mean number of Gd enhancing lesions | 0.2 | 1.4 | <0.0001 |
| Relative reduction | 86% |
Figure 1: Time to first relapse
4 Contraindications
PLEGRIDY is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta or peginterferon, or any other component of PLEGRIDY [see Warnings and Precautions ( 5.3 )].
6 Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of labeling:
- Hepatic Injury [see Warnings and Precautions ( 5.1 )]
- Depression and Suicide [see Warnings and Precautions ( 5.2 )]
- Anaphylaxis and Other Allergic Reactions [see Warnings and Precautions ( 5.3 )]
- Injection Site Reactions Including Necrosis [see Warnings and Precautions ( 5.4 )]
- Congestive Heart Failure [see Warnings and Precautions (Section 5.5 )]
- Decreased Peripheral Blood Counts [see Warnings and Precautions ( 5.6 )]
- Thrombotic Microangiopathy [see Warnings and Precautions ( 5.7 )]
- Pulmonary Arterial Hypertension [see Warnings and Precautions (5.8)]
- Autoimmune Disorders [see Warnings and Precautions (5.9)]
- Seizures [see Warnings and Precautions (5.10)]
8.6 Renal Impairment
Monitor for adverse reactions due to increased drug exposure in patients with severe renal impairment [see Clinical Pharmacology ( 12.3 )].
12.2 Pharmacodynamics
There is no biochemical or physiologic effect known to relate directly to the clinical effect of PLEGRIDY.
12.3 Pharmacokinetics
After single-dose or multiple-dose subcutaneous administration of PLEGRIDY to healthy subjects, serum PLEGRIDY peak concentration (Cmax) and total exposure over time (area under the curve, or AUC) increased in proportion to doses from 63 to 188 micrograms. PLEGRIDY did not accumulate in the serum after multiple doses of 125 micrograms every 14 days. Pharmacokinetic parameters for PLEGRIDY, including Cmax and AUC, did not differ significantly between healthy volunteers and multiple sclerosis patients or between single-dose and multiple-dose administrations. However, the coefficient of variation between individual patients for AUC, Cmax, and half-life was high (41% to 68%, 74% to 89%, and 45% to 93%, respectively).
2.1 Dosing Information
PLEGRIDY may only be administered subcutaneously (SC) or intramuscularly (IM).
1 Indications and Usage
PLEGRIDY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
12.1 Mechanism of Action
The mechanism by which PLEGRIDY exerts its effects in patients with multiple sclerosis is unknown.
5.9 Autoimmune Disorders
Autoimmune disorders of multiple target organs including idiopathic thrombocytopenia, hyper- and hypothyroidism, and autoimmune hepatitis have been reported with interferon beta.
In clinical studies, the incidence of autoimmune disorders was less than 1% in both PLEGRIDY and placebo treatment groups.
If patients develop a new autoimmune disorder, consider stopping PLEGRIDY.
16.2 Storage and Handling
Store PLEGRIDY prefilled pens and prefilled syringes in a refrigerator between 2°C to 8°C (36°F to 46°F) in the closed original carton to protect from light until ready for injection. Do not freeze. Discard if frozen.
If refrigeration is unavailable, PLEGRIDY may be stored at room temperature up to 25°C (77°F) for a period up to 30 days, protected from light. PLEGRIDY can be removed from, and returned to, a refrigerator if necessary. The total combined time out of refrigeration should not exceed 30 days.
PLEGRIDY prefilled syringe for intramuscular administration contains natural rubber latex which may cause allergic reactions.
Dispose in a sharps-bin container or other hard plastic or metal sealable container. Always follow local regulations for disposal.
5 Warnings and Precautions
- Hepatic injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of PLEGRIDY if hepatic injury occurs (5.1)
- Depression and suicide: advise patients to report immediately any symptom of depression or suicidal ideation to their healthcare provider; consider discontinuation of PLEGRIDY if depression occurs (5.2)
- Anaphylaxis and other allergic reactions: Discontinue PLEGRIDY if a serious allergic reaction occurs (5.3)
- Injection site reactions: Do not administer PLEGRIDY into affected area until fully healed; if multiple lesions occur, change injection site or discontinue PLEGRIDY until healing of skin lesions (5.4)
- Congestive heart failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms (5.5)
- Decreased peripheral blood counts: monitor complete blood counts (5.6)
- Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported with interferon beta products. Discontinue PLEGRIDY if clinical symptoms and laboratory findings consistent with TMA occur (5.7)
- Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including PLEGRIDY. Discontinue PLEGRIDY if PAH is diagnosed (5.8)
- Autoimmune disorders: consider discontinuation of PLEGRIDY if a new autoimmune disorder occurs (5.9)
5.2 Depression and Suicide
Depression, suicidal ideation, and suicide occur more frequently in patients receiving interferon beta than in patients receiving placebo.
In clinical studies, the overall incidence of adverse events related to depression and suicidal ideation in multiple sclerosis patients was 8% in both the PLEGRIDY and placebo groups. The incidence of serious events related to depression and suicidal ideation was similar and less than 1% in both groups.
Advise patients to report immediately any symptom of depression or suicidal ideation to their healthcare provider. If a patient develops depression or other severe psychiatric symptoms, consider stopping treatment with PLEGRIDY.
2 Dosage and Administration
- For subcutaneous or intramuscular use only (2.1)
- Recommended dose: 125 micrograms every 14 days (2.1)
- PLEGRIDY dose should be titrated, starting with 63 micrograms on day 1, 94 micrograms on day 15, and 125 micrograms (full dose) on day 29 (2.1)
- A healthcare professional should train patients in the proper technique for self-administering subcutaneous injections using the prefilled pen or syringe or intramuscular injections using the prefilled syringe (2.2)
- Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms (2.3)
3 Dosage Forms and Strengths
PLEGRIDY is a clear to slightly opalescent and colorless to slightly yellow solution.
5.5 Congestive Heart Failure
Congestive heart failure, cardiomyopathy, and cardiomyopathy with congestive heart failure occur in patients receiving interferon beta.
In clinical studies, the incidence of cardiovascular events was 7% in both PLEGRIDY and placebo treatment groups. No serious cardiovascular events were reported in the PLEGRIDY group.
Monitor patients with significant cardiac disease for worsening of their cardiac condition during initiation and continuation of treatment with PLEGRIDY.
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of PLEGRIDY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
5.7 Thrombotic Microangiopathy
Cases of thrombotic microangiopathy (TMA), including thrombotic thrombocytopenic purpura and hemolytic uremic syndrome, some fatal, have been reported with interferon beta products. Cases have been reported several weeks to years after starting interferon beta products. Discontinue PLEGRIDY if clinical symptoms and laboratory findings consistent with TMA occur, and manage as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of PLEGRIDY cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
5.6 Decreased Peripheral Blood Counts
Interferon beta can cause decreased peripheral blood counts in all cell lines, including rare instances of pancytopenia and severe thrombocytopenia.
In clinical studies, decreases in white blood cell counts below 3.0 x 109/L occurred in 7% of patients receiving PLEGRIDY and in 1% receiving placebo. There is no apparent association between decreases in white blood cell counts and an increased risk of infections or serious infections. The incidence of clinically significant decreases in lymphocyte counts (below 0.5 x 109/L), neutrophil counts (below 1.0 x 109/L), and platelet counts (below 100 x 109/L) were all less than 1% and similar in both placebo and PLEGRIDY groups. Two serious cases were reported in patients treated with PLEGRIDY: one patient (less than 1%) experienced severe thrombocytopenia (defined as a platelet count less than or equal to 10 x 109/L), and another patient (less than 1%) experienced severe neutropenia (defined as a neutrophil count less than or equal to 0.5 x 109/L). In both patients, cell counts recovered after discontinuation of PLEGRIDY. Compared to placebo, there were no significant differences in red blood cell counts in patients treated with PLEGRIDY.
Monitor patients for infections, bleeding, and symptoms of anemia. Monitor complete blood cell counts, differential white blood cell counts, and platelet counts during treatment with PLEGRIDY. Patients with myelosuppression may require more intensive monitoring of blood cell counts.
2.3 Premedication for Flu Like Symptoms
Prophylactic and concurrent use of analgesics and/or antipyretics may prevent or ameliorate flu-like symptoms sometimes experienced during treatment with PLEGRIDY.
5.3 Anaphylaxis and Other Allergic Reactions
Serious allergic reactions are rare complications of treatment with interferon beta; anaphylaxis has been reported with use of PLEGRIDY in the postmarketing setting.
Less than 1% of PLEGRIDY-treated patients experienced a serious allergic reaction such as angioedema or urticaria. Those who did have serious allergic reactions recovered promptly after treatment with antihistamines or corticosteroids. Discontinue PLEGRIDY if a serious allergic reaction occurs.
The protective rubber cover of the PLEGRIDY prefilled syringe for intramuscular administration contains natural rubber latex which may cause allergic reactions and should not be handled by latex-sensitive individuals. The safe use of PLEGRIDY prefilled syringe in latex-sensitive individuals has not been studied.
5.4 Injection Site Reactions Including Necrosis
Injection site reactions, including injection site necrosis, can occur with the use of interferon beta, including PLEGRIDY.
In clinical studies of subcutaneous PLEGRIDY, the incidence of injection site reactions (e.g., injection site erythema, pain, pruritus, or edema) was 66% in the PLEGRIDY group and 11% in the placebo group; the incidence of severe injection site reactions was 3% in the PLEGRIDY group and 0% in the placebo group. One patient out of 1468 patients who received PLEGRIDY in clinical studies experienced injection site necrosis. The injury resolved with standard medical treatment.
In Study 3, which compared single doses of intramuscular and subcutaneous PLEGRIDY [see Adverse Reactions (6.1)], the incidence of injection site reactions (e.g., injection site erythema, pain, pruritus, or edema) was 14% in the intramuscular PLEGRIDY group and 32% in the subcutaneous PLEGRIDY group.
Injection site abscesses and cellulitis have been reported in the postmarketing setting with use of interferon beta. Some cases required treatment with hospitalization for surgical drainage and intravenous antibiotics.
Periodically evaluate patient understanding and use of aseptic self-injection techniques and procedures, particularly if injection site necrosis has occurred.
Decisions to discontinue therapy following necrosis at a single injection site should be based on the extent of the necrosis. For patients who continue therapy with PLEGRIDY after injection site necrosis has occurred, avoid administration of PLEGRIDY near the affected area until it is fully healed. If multiple lesions occur, change injection site or discontinue PLEGRIDY until healing occurs.
2.2 Important Administration Instructions (all Dosage Forms)
Healthcare professionals should train patients in the proper technique for self-administering subcutaneous injections using the prefilled pen or syringe or intramuscular injections using the prefilled syringe. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Advise patients and caregivers to rotate injection sites with each administration to minimize the likelihood of severe injection site reactions, including necrosis or localized infection [see Warnings and Precautions (5.4)].
Once removed from the refrigerator, PLEGRIDY should be allowed to warm to room temperature (about 30 minutes) prior to injection. Do not use external heat sources such as hot water to warm PLEGRIDY.
Each PLEGRIDY pen and syringe for subcutaneous injection is provided with the needle pre-attached. PLEGRIDY prefilled syringe for intramuscular injection is supplied as a prefilled syringe with a separate needle. Both intramuscular and subcutaneous prefilled syringes and subcutaneously administered prefilled pens are for one-time use in one patient only and should be discarded after use.
Structured Label Content
Section 42229-5 (42229-5)
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised: 7/2023 |
|
Medication Guide PLEGRIDY® (PLEGG-rih-dee) (peginterferon beta-1a) injection, for subcutaneous or intramuscular use |
|
| Read this Medication Guide before you start using PLEGRIDY, and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |
|
What is the most important information I should know about PLEGRIDY? |
|
PLEGRIDY can cause serious side effects, including:
|
|
What is PLEGRIDY?
|
|
Do not take PLEGRIDY if you:
|
|
Before using PLEGRIDY, tell your healthcare provider about all of your medical conditions, including if you:
|
|
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |
How should I use PLEGRIDY?
|
|
| What are the possible side effects of PLEGRIDY? | |
|
See “What is the most important information I should know about PLEGRIDY?”
PLEGRIDY may cause serious side effects, including:
|
|
|
Flu-like symptoms or other common side effects of PLEGRIDY may include: headache, muscle and joint aches, fever, chills, or tiredness. These are not all the possible side effects of PLEGRIDY. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
| General Information about the safe and effective use of PLEGRIDY. | |
| Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use PLEGRIDY for a condition for which it was not prescribed. Do not give PLEGRIDY to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about PLEGRIDY that is written for health professionals. | |
| What are the ingredients in PLEGRIDY? | |
| Active ingredient: peginterferon beta-1a. | |
| Inactive ingredients: | |
|
|
| For more information, go to www.plegridy.com or call 1-800-456-2255. Manufactured by: Biogen Inc., Cambridge, MA 02142, U.S. License # 1697 PLEGRIDY is a registered trademark of Biogen. ©2013-2023 Biogen |
Section 43683-2 (43683-2)
| Warnings and Precautions (5.8) | 7/2023 |
Section 51945-4 (51945-4)
Principal Display Panel - Carton Label
NDC 64406-012-01
Plegridy-Pen
(peginterferon beta-1a)
Injection
63. mcg/0.5 mL
94 mcg/0.5 mL
For subcutaneous use only
Starter Pack
Contents:
Dose 1: 1 Single-Dose 63 mcg/0.5 mL
Prefilled Autoinjector
Dose 2: 1 Single-Dose 94 mcg/0.5 mL
Prefilled Autoinjector
Dispense with enclosed Medicaton Guide
Rx Only
Biogen ®
Section 59845-8 (59845-8)
Instructions for Use
PLEGRIDY ® (PLEGG-rih-dee)
(peginterferon beta-1a) injection, for Subcutaneous Use
Single-Dose Prefilled Pen
Starter Pack
Important: Do not remove the Plegridy Pen cap until you are ready to inject.
How to Inject your Plegridy Pen
Read this Instructions for Use before you start using Plegridy and each time you get a refill of your prescription. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Note:
- Before you use your Plegridy Pen for the first time, your healthcare provider should show you or your caregiver how to prepare and inject your Plegridy Pen the right way.
- If you experience difficulty or have questions, call 1-800-456-2255.
- Plegridy Pen is for use under the skin only (subcutaneous).
- Each Plegridy Pen can be used 1 time only. Do not share your Plegridy Pen with anyone else. By sharing the Pen, you may give an infection to them or get an infection from them.
- Take your Plegridy Pen out of the refrigerator and let it come to room temperature for at least 30 minutes before your injection.
- Do not use more than 1 Plegridy Pen every 14 days.
- Do not use your Plegridy Pen if it has been dropped or is visibly damaged.
How should I store Plegridy?
- Store Plegridy in the refrigerator between 36°F to 46°F (2°C to 8°C).
- If a refrigerator is not available, PLEGRIDY may be stored at room temperature up to 77°F (25°C) for up to 30 days in total.
- Keep Plegridy in the original carton to protect it from light.
- Do not freeze Plegridy.
- Keep Plegridy Pens, needles, and all medicines out of the reach of children.
Supplies needed for your Starter Pack for Plegridy Pen injection (See Figure A):
- 1 Starter Pack for Plegridy Pen which contains:
- 1 Plegridy 63 mcg Pen (orange pen)
- 1 Plegridy 94 mcg Pen (blue pen)
Additional supplies which are not included in the pack (See Figure B):
- alcohol wipe
- gauze pad
- adhesive bandage
- 1 sharps container for throwing away used needles and Plegridy Pens. See “Disposing of your used Plegridy Pens” at the end of these instructions.
- a well-lit area and a clean, flat surface to work on, like a table
Dose Schedule
- Choose the right Plegridy Pen for your dose. The Starter Pack for Plegridy Pen contains your first 2 injections to slowly adjust your dose.
| When | Which Dose | Choose |
|
Day 1
(63 mcg) |
First Dose:
63 mcg |
Orange Pen (See Figure C) |
| When | Which Dose | Choose |
|
Day 15
(94 mcg) |
Second Dose:
94 mcg |
Blue Pen (See Figure D) |
- Make sure you use the 63 mcg orange pen for your first dose (on day 1).
- Make sure you use the 94 mcg blue pen for your second dose (on day 15).
Ask your healthcare provider which Plegridy Pen you should use for your correct dose if you are not sure.
Before Use – Parts of your Plegridy Pen before use (See Figure E):
Important: Do not remove the Plegridy Pen cap until you are ready to inject. If you remove the cap before you are ready to inject, do not re-cap the pen. Re-capping could cause the pen to lock.
Preparing for your injection:
Step 1: Take your Plegridy Pen out of the refrigerator and let it come to room temperature for at least 30 minutes.
- Do not use external heat sources such as hot water to warm your Plegridy Pen.
Step 2: Collect your supplies and wash your hands
- Find a well-lit area and a clean, flat surface like a table and collect all the supplies you will need to give yourself or to receive an injection.
- Wash your hands with soap and water.
Step 3: Check your Plegridy Pen (see Figure F)
|
|
|
|
|
| Step 4: Choose and clean your injection site | |
|
|
|
Giving your injection: Step 5: Remove the Plegridy Pen cap |
|
|
Figure H |
| Step 6: Give your injection | |
|
|
|
If you do not hear clicking sounds, or you do not see green checkmarks in the injection status window after trying to give your injection, your Plegridy Pen may have locked and you should call 1-800-456-2255 for help.
Step 7: Remove your Plegridy Pen from your injection site |
|
|
|
| Step 8: Check to make sure you have received your full dose of Plegridy (see Figure M) | |
|
|
After your injection:
After Use – Parts of your Plegridy Pen (Figure N)
Important: Do not touch the needle cover in any way. You could get a needle stick injury.
Note: After the pen has been removed from the injection site, the needle cover will lock to protect against needle stick injury. Do not recap your Plegridy Pen.
Step 9: Disposing of your used Plegridy Pens
- Put your used Plegridy Pens in a FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) Plegridy Pens in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and pens. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal.
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- Do not recap your Plegridy Pen.
Step 10: Check your injection site
- Plegridy may commonly cause redness, pain or swelling of your skin at the injection site.
- After 2 hours check your injection site for redness, swelling or tenderness.
- Call your healthcare provider right away if your injection site becomes swollen and painful or the area looks infected and does not heal within a few days.
Questions?
For product or service-related questions, call 1-800-456-2255 or go to www.plegridy.com.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by: Biogen Inc., Cambridge, MA 02142, U.S. License # 1697
©2013-2022 Biogen. All rights reserved. 1-800-456-2255
Issued: 03/2020
43839-03
5.10 Seizures
Seizures are associated with the use of interferon beta.
The incidence of seizures in multiple sclerosis clinical studies was less than 1% in patients receiving PLEGRIDY and placebo.
Exercise caution when administering PLEGRIDY to patients with a seizure disorder.
11 Description (11 DESCRIPTION)
Peginterferon beta-1a is a covalent conjugate of recombinant interferon beta-1a (approximate molecular weight [MW] 20,000 daltons) with a single, linear methoxy poly(ethyleneglycol)-O-2-methylpropionaldehyde molecule (approximate MW 20,000 daltons). Interferon beta-1a is produced as a glycosylated protein using genetically-engineered Chinese hamster ovary cells into which the human interferon beta gene has been introduced. The amino acid sequence of recombinant interferon beta-1a is identical to that of the human interferon beta counterpart.
The molecular weight of peginterferon beta-1a is approximately 44,000 daltons, consistent with the mass of the protein, the carbohydrate moieties (approximately 2,500 daltons), and the attached poly(ethylene glycol).
Peginterferon beta-1a 125 mcg contains 125 mcg of interferon beta-1a plus 125 mcg of poly(ethylene glycol). Using the World Health Organization International Standard for interferon beta, peginterferon beta-1a has a specific antiviral activity of approximately 100 million International Units (MIU) per mg of protein as determined using an in vitro cytopathic effect assay. Peginterferon beta-1a 125 mcg contains approximately 12 MIU of antiviral activity.
16.1 How Supplied
PLEGRIDY (peginterferon beta-1a) injection is a sterile, preservative-free, clear to slightly opalescent and colorless to slightly yellow solution supplied as a 0.5 mL single-dose prefilled pen or a 0.5 mL single-dose prefilled syringe.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of PLEGRIDY did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
5.1 Hepatic Injury
Severe hepatic injury, including hepatitis, autoimmune hepatitis, and rare cases of severe hepatic failure, have been reported with interferon beta. Asymptomatic elevation of hepatic transaminases has also been reported, and in some patients has recurred upon rechallenge with interferon beta.
Elevations in hepatic enzymes and hepatic injury have been observed with the use of PLEGRIDY in clinical studies. The incidence of increases in hepatic transaminases was greater in patients taking PLEGRIDY than in those taking placebo. The incidence of elevations of alanine aminotransferase above 5 times the upper limit of normal was 1% in placebo-treated patients and 2% in PLEGRIDY-treated patients. The incidence of elevations of aspartate aminotransferase above 5 times the upper limit of normal was less than 1% in placebo-treated patients and less than 1% in PLEGRIDY-treated patients. Elevations of serum hepatic transaminases combined with elevated bilirubin occurred in 2 patients. Both cases resolved following discontinuation of PLEGRIDY.
Cases of noninfectious hepatitis have been reported in the postmarketing setting with use of PLEGRIDY.
Monitor patients for signs and symptoms of hepatic injury.
6.2 Immunogenicity
As with all therapeutic proteins, there is a potential for immunogenicity.
The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies in the studies described below with the incidence of antibodies in other studies or to other interferon beta-1a products may be misleading.
In Study 1, fewer than 1% of patients treated with PLEGRIDY SC every 14 days for 1 year developed neutralizing antibodies. Approximately 7% of PLEGRIDY SC-treated patients developed antibodies to the polyethylene glycol moiety.
No formal studies have been conducted with regards to immunogenicity of the intramuscular route of administration of PLEGRIDY.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of PLEGRIDY was demonstrated in the randomized, double-blind, and placebo-controlled phase (year 1) of Study 1. The trial compared clinical and MRI outcomes at 48 weeks in patients who received PLEGRIDY 125 micrograms (n=512) or placebo (n=500) by the subcutaneous route, once every 14 days.
Study 1 enrolled patients who had a baseline Expanded Disability Status Scale (EDSS) score from 0 to 5, who had experienced at least 2 relapses within the previous three years, and had experienced at least 1 relapse in the previous year. The trial excluded patients with progressive forms of multiple sclerosis. The mean age of the study population was 37 years, the mean disease duration was 3.6 years, and the mean EDSS score at baseline was 2.46. The majority of the patients were women (71%).
The trial scheduled neurological evaluations at baseline, every 12 weeks, and at the time of a suspected relapse. Brain MRI evaluations were scheduled at baseline, week 24, and week 48.
The primary outcome was the annualized relapse rate over 1 year. Secondary outcomes included the proportion of patients relapsing, number of new or newly enlarging T2 hyperintense lesions, and time to confirmed disability progression. Confirmed disability progression was defined as follows: if the baseline EDSS score was 0, a sustained 12-week increase in EDSS score of 1.5 points was required; if the baseline EDSS score was greater than 0, a sustained 12-week increase in EDSS score of 1 point was required. Table 4 and Figure 1 show the results of Study 1.
| Endpoint |
PLEGRIDY
125 micrograms every 14 days |
Placebo | p-value |
|---|---|---|---|
| Clinical outcomes at 48 weeks | N=512 | N=500 | |
| Annualized relapse rate | 0.26 | 0.40 | 0.0007 |
| Relative reduction | 36% | ||
| Proportion of patients with relapses | 0.19 | 0.29 | 0.0003 |
| Relative risk reduction | 39% | ||
| Proportion of patients with disability progression | 0.07 | 0.11 | 0.0383 |
| Relative risk reduction | 38% | ||
| MRI outcomes at 48 weeks | N=457 | N=476 | |
| Mean number of new or newly enlarging T2 hyperintense lesions | 3.6 | 10.9 | <0.0001 |
| Relative reduction | 67% | ||
| Mean number of Gd enhancing lesions | 0.2 | 1.4 | <0.0001 |
| Relative reduction | 86% |
Figure 1: Time to first relapse
4 Contraindications (4 CONTRAINDICATIONS)
PLEGRIDY is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta or peginterferon, or any other component of PLEGRIDY [see Warnings and Precautions ( 5.3 )].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in more detail in other sections of labeling:
- Hepatic Injury [see Warnings and Precautions ( 5.1 )]
- Depression and Suicide [see Warnings and Precautions ( 5.2 )]
- Anaphylaxis and Other Allergic Reactions [see Warnings and Precautions ( 5.3 )]
- Injection Site Reactions Including Necrosis [see Warnings and Precautions ( 5.4 )]
- Congestive Heart Failure [see Warnings and Precautions (Section 5.5 )]
- Decreased Peripheral Blood Counts [see Warnings and Precautions ( 5.6 )]
- Thrombotic Microangiopathy [see Warnings and Precautions ( 5.7 )]
- Pulmonary Arterial Hypertension [see Warnings and Precautions (5.8)]
- Autoimmune Disorders [see Warnings and Precautions (5.9)]
- Seizures [see Warnings and Precautions (5.10)]
8.6 Renal Impairment
Monitor for adverse reactions due to increased drug exposure in patients with severe renal impairment [see Clinical Pharmacology ( 12.3 )].
12.2 Pharmacodynamics
There is no biochemical or physiologic effect known to relate directly to the clinical effect of PLEGRIDY.
12.3 Pharmacokinetics
After single-dose or multiple-dose subcutaneous administration of PLEGRIDY to healthy subjects, serum PLEGRIDY peak concentration (Cmax) and total exposure over time (area under the curve, or AUC) increased in proportion to doses from 63 to 188 micrograms. PLEGRIDY did not accumulate in the serum after multiple doses of 125 micrograms every 14 days. Pharmacokinetic parameters for PLEGRIDY, including Cmax and AUC, did not differ significantly between healthy volunteers and multiple sclerosis patients or between single-dose and multiple-dose administrations. However, the coefficient of variation between individual patients for AUC, Cmax, and half-life was high (41% to 68%, 74% to 89%, and 45% to 93%, respectively).
2.1 Dosing Information
PLEGRIDY may only be administered subcutaneously (SC) or intramuscularly (IM).
1 Indications and Usage (1 INDICATIONS AND USAGE)
PLEGRIDY is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
12.1 Mechanism of Action
The mechanism by which PLEGRIDY exerts its effects in patients with multiple sclerosis is unknown.
5.9 Autoimmune Disorders
Autoimmune disorders of multiple target organs including idiopathic thrombocytopenia, hyper- and hypothyroidism, and autoimmune hepatitis have been reported with interferon beta.
In clinical studies, the incidence of autoimmune disorders was less than 1% in both PLEGRIDY and placebo treatment groups.
If patients develop a new autoimmune disorder, consider stopping PLEGRIDY.
16.2 Storage and Handling
Store PLEGRIDY prefilled pens and prefilled syringes in a refrigerator between 2°C to 8°C (36°F to 46°F) in the closed original carton to protect from light until ready for injection. Do not freeze. Discard if frozen.
If refrigeration is unavailable, PLEGRIDY may be stored at room temperature up to 25°C (77°F) for a period up to 30 days, protected from light. PLEGRIDY can be removed from, and returned to, a refrigerator if necessary. The total combined time out of refrigeration should not exceed 30 days.
PLEGRIDY prefilled syringe for intramuscular administration contains natural rubber latex which may cause allergic reactions.
Dispose in a sharps-bin container or other hard plastic or metal sealable container. Always follow local regulations for disposal.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hepatic injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of PLEGRIDY if hepatic injury occurs (5.1)
- Depression and suicide: advise patients to report immediately any symptom of depression or suicidal ideation to their healthcare provider; consider discontinuation of PLEGRIDY if depression occurs (5.2)
- Anaphylaxis and other allergic reactions: Discontinue PLEGRIDY if a serious allergic reaction occurs (5.3)
- Injection site reactions: Do not administer PLEGRIDY into affected area until fully healed; if multiple lesions occur, change injection site or discontinue PLEGRIDY until healing of skin lesions (5.4)
- Congestive heart failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms (5.5)
- Decreased peripheral blood counts: monitor complete blood counts (5.6)
- Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported with interferon beta products. Discontinue PLEGRIDY if clinical symptoms and laboratory findings consistent with TMA occur (5.7)
- Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including PLEGRIDY. Discontinue PLEGRIDY if PAH is diagnosed (5.8)
- Autoimmune disorders: consider discontinuation of PLEGRIDY if a new autoimmune disorder occurs (5.9)
5.2 Depression and Suicide
Depression, suicidal ideation, and suicide occur more frequently in patients receiving interferon beta than in patients receiving placebo.
In clinical studies, the overall incidence of adverse events related to depression and suicidal ideation in multiple sclerosis patients was 8% in both the PLEGRIDY and placebo groups. The incidence of serious events related to depression and suicidal ideation was similar and less than 1% in both groups.
Advise patients to report immediately any symptom of depression or suicidal ideation to their healthcare provider. If a patient develops depression or other severe psychiatric symptoms, consider stopping treatment with PLEGRIDY.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For subcutaneous or intramuscular use only (2.1)
- Recommended dose: 125 micrograms every 14 days (2.1)
- PLEGRIDY dose should be titrated, starting with 63 micrograms on day 1, 94 micrograms on day 15, and 125 micrograms (full dose) on day 29 (2.1)
- A healthcare professional should train patients in the proper technique for self-administering subcutaneous injections using the prefilled pen or syringe or intramuscular injections using the prefilled syringe (2.2)
- Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
PLEGRIDY is a clear to slightly opalescent and colorless to slightly yellow solution.
5.5 Congestive Heart Failure
Congestive heart failure, cardiomyopathy, and cardiomyopathy with congestive heart failure occur in patients receiving interferon beta.
In clinical studies, the incidence of cardiovascular events was 7% in both PLEGRIDY and placebo treatment groups. No serious cardiovascular events were reported in the PLEGRIDY group.
Monitor patients with significant cardiac disease for worsening of their cardiac condition during initiation and continuation of treatment with PLEGRIDY.
6.3 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of PLEGRIDY. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.7 Thrombotic Microangiopathy
Cases of thrombotic microangiopathy (TMA), including thrombotic thrombocytopenic purpura and hemolytic uremic syndrome, some fatal, have been reported with interferon beta products. Cases have been reported several weeks to years after starting interferon beta products. Discontinue PLEGRIDY if clinical symptoms and laboratory findings consistent with TMA occur, and manage as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of PLEGRIDY cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
5.6 Decreased Peripheral Blood Counts
Interferon beta can cause decreased peripheral blood counts in all cell lines, including rare instances of pancytopenia and severe thrombocytopenia.
In clinical studies, decreases in white blood cell counts below 3.0 x 109/L occurred in 7% of patients receiving PLEGRIDY and in 1% receiving placebo. There is no apparent association between decreases in white blood cell counts and an increased risk of infections or serious infections. The incidence of clinically significant decreases in lymphocyte counts (below 0.5 x 109/L), neutrophil counts (below 1.0 x 109/L), and platelet counts (below 100 x 109/L) were all less than 1% and similar in both placebo and PLEGRIDY groups. Two serious cases were reported in patients treated with PLEGRIDY: one patient (less than 1%) experienced severe thrombocytopenia (defined as a platelet count less than or equal to 10 x 109/L), and another patient (less than 1%) experienced severe neutropenia (defined as a neutrophil count less than or equal to 0.5 x 109/L). In both patients, cell counts recovered after discontinuation of PLEGRIDY. Compared to placebo, there were no significant differences in red blood cell counts in patients treated with PLEGRIDY.
Monitor patients for infections, bleeding, and symptoms of anemia. Monitor complete blood cell counts, differential white blood cell counts, and platelet counts during treatment with PLEGRIDY. Patients with myelosuppression may require more intensive monitoring of blood cell counts.
2.3 Premedication for Flu Like Symptoms (2.3 Premedication for Flu-like Symptoms)
Prophylactic and concurrent use of analgesics and/or antipyretics may prevent or ameliorate flu-like symptoms sometimes experienced during treatment with PLEGRIDY.
5.3 Anaphylaxis and Other Allergic Reactions
Serious allergic reactions are rare complications of treatment with interferon beta; anaphylaxis has been reported with use of PLEGRIDY in the postmarketing setting.
Less than 1% of PLEGRIDY-treated patients experienced a serious allergic reaction such as angioedema or urticaria. Those who did have serious allergic reactions recovered promptly after treatment with antihistamines or corticosteroids. Discontinue PLEGRIDY if a serious allergic reaction occurs.
The protective rubber cover of the PLEGRIDY prefilled syringe for intramuscular administration contains natural rubber latex which may cause allergic reactions and should not be handled by latex-sensitive individuals. The safe use of PLEGRIDY prefilled syringe in latex-sensitive individuals has not been studied.
5.4 Injection Site Reactions Including Necrosis
Injection site reactions, including injection site necrosis, can occur with the use of interferon beta, including PLEGRIDY.
In clinical studies of subcutaneous PLEGRIDY, the incidence of injection site reactions (e.g., injection site erythema, pain, pruritus, or edema) was 66% in the PLEGRIDY group and 11% in the placebo group; the incidence of severe injection site reactions was 3% in the PLEGRIDY group and 0% in the placebo group. One patient out of 1468 patients who received PLEGRIDY in clinical studies experienced injection site necrosis. The injury resolved with standard medical treatment.
In Study 3, which compared single doses of intramuscular and subcutaneous PLEGRIDY [see Adverse Reactions (6.1)], the incidence of injection site reactions (e.g., injection site erythema, pain, pruritus, or edema) was 14% in the intramuscular PLEGRIDY group and 32% in the subcutaneous PLEGRIDY group.
Injection site abscesses and cellulitis have been reported in the postmarketing setting with use of interferon beta. Some cases required treatment with hospitalization for surgical drainage and intravenous antibiotics.
Periodically evaluate patient understanding and use of aseptic self-injection techniques and procedures, particularly if injection site necrosis has occurred.
Decisions to discontinue therapy following necrosis at a single injection site should be based on the extent of the necrosis. For patients who continue therapy with PLEGRIDY after injection site necrosis has occurred, avoid administration of PLEGRIDY near the affected area until it is fully healed. If multiple lesions occur, change injection site or discontinue PLEGRIDY until healing occurs.
2.2 Important Administration Instructions (all Dosage Forms) (2.2 Important Administration Instructions (All Dosage Forms))
Healthcare professionals should train patients in the proper technique for self-administering subcutaneous injections using the prefilled pen or syringe or intramuscular injections using the prefilled syringe. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Advise patients and caregivers to rotate injection sites with each administration to minimize the likelihood of severe injection site reactions, including necrosis or localized infection [see Warnings and Precautions (5.4)].
Once removed from the refrigerator, PLEGRIDY should be allowed to warm to room temperature (about 30 minutes) prior to injection. Do not use external heat sources such as hot water to warm PLEGRIDY.
Each PLEGRIDY pen and syringe for subcutaneous injection is provided with the needle pre-attached. PLEGRIDY prefilled syringe for intramuscular injection is supplied as a prefilled syringe with a separate needle. Both intramuscular and subcutaneous prefilled syringes and subcutaneously administered prefilled pens are for one-time use in one patient only and should be discarded after use.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:35.432268 · Updated: 2026-03-14T22:37:58.050044