08da6353-9625-4e8f-8575-a2b49f5d2ac6
08da6353-9625-4e8f-8575-a2b49f5d2ac6
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
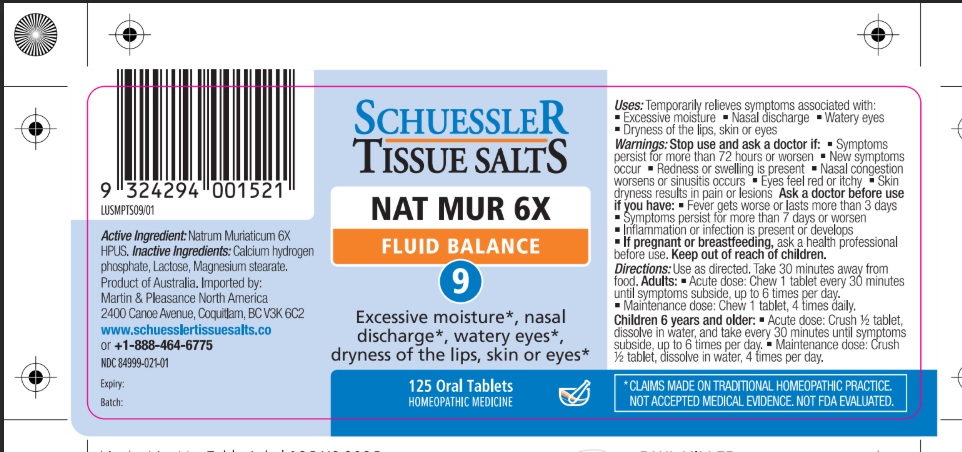
Natrum Muriaticum 6X HPUS excessive moisture, nasal discharge, watery eyes, dryness of the lips, skin, or eyes " HPUS " is the Homeopathic Pharmacopoeia of the United States
Description
Natrum Muriaticum 6X HPUS excessive moisture, nasal discharge, watery eyes, dryness of the lips, skin, or eyes " HPUS " is the Homeopathic Pharmacopoeia of the United States
Medication Information
Warnings
Stop use and ask a doctor if:
- Symptoms persist for more than 72 hours or worsen
- New symptoms occur
- Redness or swelling is present
- Nasal congestion worsens or sinusitis occurs
- Eyes feel red or itchy
- Skin dryness results in pain or lesions
If pregnant or breastfeeding: Ask a health professional before use.
Ask a doctor before use if you have:
• Fever get worse or lasts more than 3 days
• Symptoms persist for more than 7 days or worsen
• Inflammation or inflection is present or develops.
Keep out of reach of children.
Uses
Temporarily relieves symptoms associated with
- Excessive moisture
- Nasal discharge
- Watery eyes
- Dryness of the lips, skin, or eyes
Purpose
Natrum Muriaticum 6X HPUS excessive moisture, nasal discharge, watery eyes, dryness of the lips, skin, or eyes " HPUS " is the Homeopathic Pharmacopoeia of the United States
Directions
- Use as directed
- Take 30 minutes away from food
Adults: Acute dose: Chew 1 tablet every 30 minutes until symptoms subside up to 6 times per day.
Maintenance dose: Chew 1 tablet, 4 times daily.
Children 6 years and older: Acute dose: Crush 1/2 tablet, dissolve in water, and take every 30 minutes until symptoms subside, up to six times per day.
Maintenance dose :Crush 1/2 tablet, dissolve in water, 4 times per day.
Other Information
Store below 86°F
Inactive Ingredients
Calcium hydrogen phosphate, Lactose, Magnesium stearate
Description
Natrum Muriaticum 6X HPUS excessive moisture, nasal discharge, watery eyes, dryness of the lips, skin, or eyes " HPUS " is the Homeopathic Pharmacopoeia of the United States
Section 42229-5
Questions? www.schuesslertissuesalts.co or + 1-888-464-6775
Section 50565-1
Section 55105-1
Product Label
Active Ingredients purpose
Natrum Muriaticum 6X HPUS excessive moisture, nasal discharge, watery eyes, dryness of the lips, skin, or eyes
" HPUS" is the Homeopathic Pharmacopoeia of the United States
Structured Label Content
Warnings
Stop use and ask a doctor if:
- Symptoms persist for more than 72 hours or worsen
- New symptoms occur
- Redness or swelling is present
- Nasal congestion worsens or sinusitis occurs
- Eyes feel red or itchy
- Skin dryness results in pain or lesions
If pregnant or breastfeeding: Ask a health professional before use.
Ask a doctor before use if you have:
• Fever get worse or lasts more than 3 days
• Symptoms persist for more than 7 days or worsen
• Inflammation or inflection is present or develops.
Keep out of reach of children.
Uses
Temporarily relieves symptoms associated with
- Excessive moisture
- Nasal discharge
- Watery eyes
- Dryness of the lips, skin, or eyes
Directions
- Use as directed
- Take 30 minutes away from food
Adults: Acute dose: Chew 1 tablet every 30 minutes until symptoms subside up to 6 times per day.
Maintenance dose: Chew 1 tablet, 4 times daily.
Children 6 years and older: Acute dose: Crush 1/2 tablet, dissolve in water, and take every 30 minutes until symptoms subside, up to six times per day.
Maintenance dose :Crush 1/2 tablet, dissolve in water, 4 times per day.
Other Information
Store below 86°F
Inactive Ingredients
Calcium hydrogen phosphate, Lactose, Magnesium stearate
Section 42229-5 (42229-5)
Questions? www.schuesslertissuesalts.co or + 1-888-464-6775
Section 50565-1 (50565-1)
Section 55105-1 (55105-1)
Product Label (Product label)
Active Ingredients purpose (Active Ingredients Purpose)
Natrum Muriaticum 6X HPUS excessive moisture, nasal discharge, watery eyes, dryness of the lips, skin, or eyes
" HPUS" is the Homeopathic Pharmacopoeia of the United States
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:05.494632 · Updated: 2026-03-14T23:17:27.453851