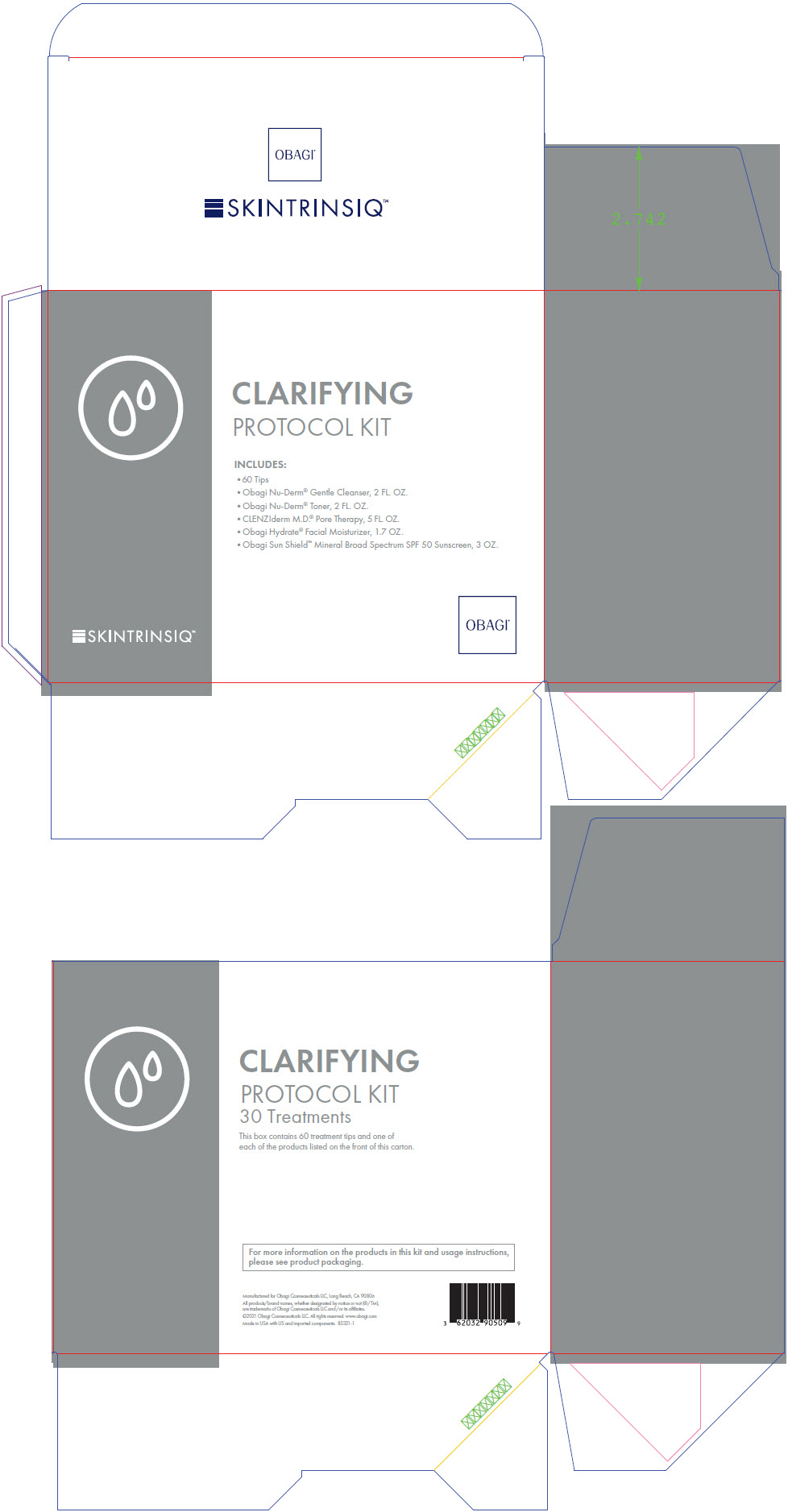
[{"name": "ALOE VERA LEAF", "unii": "ZY81Z83H0X", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "APRICOT KERNEL OIL", "unii": "54JB35T06A", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "BORAGO OFFICINALIS SEED", "unii": "2GXJ790US0", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "BUTYLPARABEN", "unii": "3QPI1U3FV8", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED)", "unii": "59TL3WG5CO", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "COCAMIDOPROPYL BETAINE", "unii": "5OCF3O11KX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DIETHYLENE GLYCOL MONOETHYL ETHER", "unii": "A1A1I8X02B", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYLPARABEN", "unii": "14255EXE39", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C YELLOW NO. 5", "unii": "I753WB2F1M", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERETH-7", "unii": "3D2Y91QZ2H", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ISOBUTYLPARABEN", "unii": "0QQJ25X58G", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "METHYLPARABEN", "unii": "A2I8C7HI9T", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "OLEYL LACTATE", "unii": "B3AWW0N3GM", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PANTHENOL", "unii": "WV9CM0O67Z", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PHENOXYETHANOL", "unii": "HIE492ZZ3T", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPYLPARABEN", "unii": "Z8IX2SC1OH", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SAGE", "unii": "065C5D077J", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM LAURETH-3 SULFATE", "unii": "BPV390UAP0", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM LAUROYL OAT AMINO ACIDS", "unii": "FSW2K9B9N5", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TROLAMINE", "unii": "9O3K93S3TK", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALLANTOIN", "unii": "344S277G0Z", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALOE VERA LEAF", "unii": "ZY81Z83H0X", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "BORAGO OFFICINALIS SEED", "unii": "2GXJ790US0", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CALENDULA OFFICINALIS FLOWER", "unii": "P0M7O4Y7YD", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DMDM HYDANTOIN", "unii": "BYR0546TOW", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C BLUE NO. 1", "unii": "H3R47K3TBD", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "HAMAMELIS VIRGINIANA TOP WATER", "unii": "NT00Y05A2V", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "IODOPROPYNYL BUTYLCARBAMATE", "unii": "603P14DHEB", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POLYSORBATE 80", "unii": "6OZP39ZG8H", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POTASSIUM ALUM", "unii": "1L24V9R23S", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SAGE", "unii": "065C5D077J", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM PYRROLIDONE CARBOXYLATE", "unii": "469OTG57A2", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "3-((L-MENTHYL)OXY)PROPANE-1,2-DIOL", "unii": "KD6TZ2QICH", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALCOHOL", "unii": "3K9958V90M", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "BENZYL ALCOHOL", "unii": "LKG8494WBH", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CHAMOMILE", "unii": "FGL3685T2X", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DIETHYLENE GLYCOL MONOETHYL ETHER", "unii": "A1A1I8X02B", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "EXT. D&C VIOLET NO. 2", "unii": "G5UX3K0728", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "MENTHOL, UNSPECIFIED FORM", "unii": "L7T10EIP3A", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "MENTHYL LACTATE, (-)-", "unii": "2BF9E65L7I", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SALICYLIC ACID", "unii": "O414PZ4LPZ", "classCode": "ACTIB", "strengthNumerator": "20", "strengthDenominator": "1", "strengthNumeratorUnit": "mg", "strengthDenominatorUnit": "mL"}, {"name": "ALLANTOIN", "unii": "344S277G0Z", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "AVOCADO OIL", "unii": "6VNO72PFC1", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CAESALPINIA SPINOSA RESIN", "unii": "WL3883U2PO", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CAPRYLYL GLYCOL", "unii": "00YIU5438U", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE", "unii": "0A5MM307FC", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CETYL ALCOHOL", "unii": "936JST6JCN", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CYCLOMETHICONE 5", "unii": "0THT5PCI0R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DIMETHICONE, UNSPECIFIED", "unii": "92RU3N3Y1O", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE)", "unii": "9E4CO0W6C5", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYLHEXYLGLYCERIN", "unii": "147D247K3P", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL MONOSTEARATE", "unii": "230OU9XXE4", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "HEXYLENE GLYCOL", "unii": "KEH0A3F75J", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "LAURETH-12", "unii": "OAH19558U1", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "LEVOMENOL", "unii": "24WE03BX2T", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "MANGIFERA INDICA SEED BUTTER", "unii": "4OXD9M35X2", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "MEDIUM-CHAIN TRIGLYCERIDES", "unii": "C9H2L21V7U", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PANTHENOL", "unii": "WV9CM0O67Z", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PHENOXYETHANOL", "unii": "HIE492ZZ3T", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SACCHARIDE ISOMERATE", "unii": "W8K377W98I", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SHEA BUTTER", "unii": "K49155WL9Y", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM HYDROXIDE", "unii": "55X04QC32I", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM STEAROYL GLUTAMATE", "unii": "65A9F4P024", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SOY STEROL", "unii": "PL360EPO9J", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SOYBEAN", "unii": "L7HT8F1ZOD", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "STEARIC ACID", "unii": "4ELV7Z65AP", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TETRAHYDRODIFERULOYLMETHANE", "unii": "00U0645U03", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TOCOPHEROL", "unii": "R0ZB2556P8", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALUMINUM OXIDE", "unii": "LMI26O6933", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CAPRYLHYDROXAMIC ACID", "unii": "UPY805K99W", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CAPRYLYL GLYCOL", "unii": "00YIU5438U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CETEARYL GLUCOSIDE", "unii": "09FUA47KNA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CETOSTEARYL ALCOHOL", "unii": "2DMT128M1S", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DICAPRYLYL CARBONATE", "unii": "609A3V1SUA", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCYRRHIZINATE DIPOTASSIUM", "unii": "CA2Y0FE3FX", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "MEDIUM-CHAIN TRIGLYCERIDES", "unii": "C9H2L21V7U", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PENTYLENE GLYCOL", "unii": "50C1307PZG", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POLYHYDROXYSTEARIC ACID (2300 MW)", "unii": "YXH47AOU0F", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPANEDIOL", "unii": "5965N8W85T", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SILICON DIOXIDE", "unii": "ETJ7Z6XBU4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM STEAROYL GLUTAMATE", "unii": "65A9F4P024", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "STEARETH-2", "unii": "V56DFE46J5", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "STEARETH-21", "unii": "53J3F32P58", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "STEARIC ACID", "unii": "4ELV7Z65AP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TRIETHOXYCAPRYLYLSILANE", "unii": "LDC331P08E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TRISODIUM ETHYLENEDIAMINE DISUCCINATE", "unii": "YA22H34H9Q", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TROPAEOLUM MAJUS FLOWERING TOP", "unii": "RGT30824HY", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "XANTHAN GUM", "unii": "TTV12P4NEE", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TITANIUM DIOXIDE", "unii": "15FIX9V2JP", "classCode": "ACTIB", "strengthNumerator": "6.2", "strengthDenominator": "100", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}, {"name": "ZINC OXIDE", "unii": "SOI2LOH54Z", "classCode": "ACTIB", "strengthNumerator": "15.6", "strengthDenominator": "100", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "g"}]