Mychelle®
0821857c-d67f-4da1-af66-5e7f64cce59b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
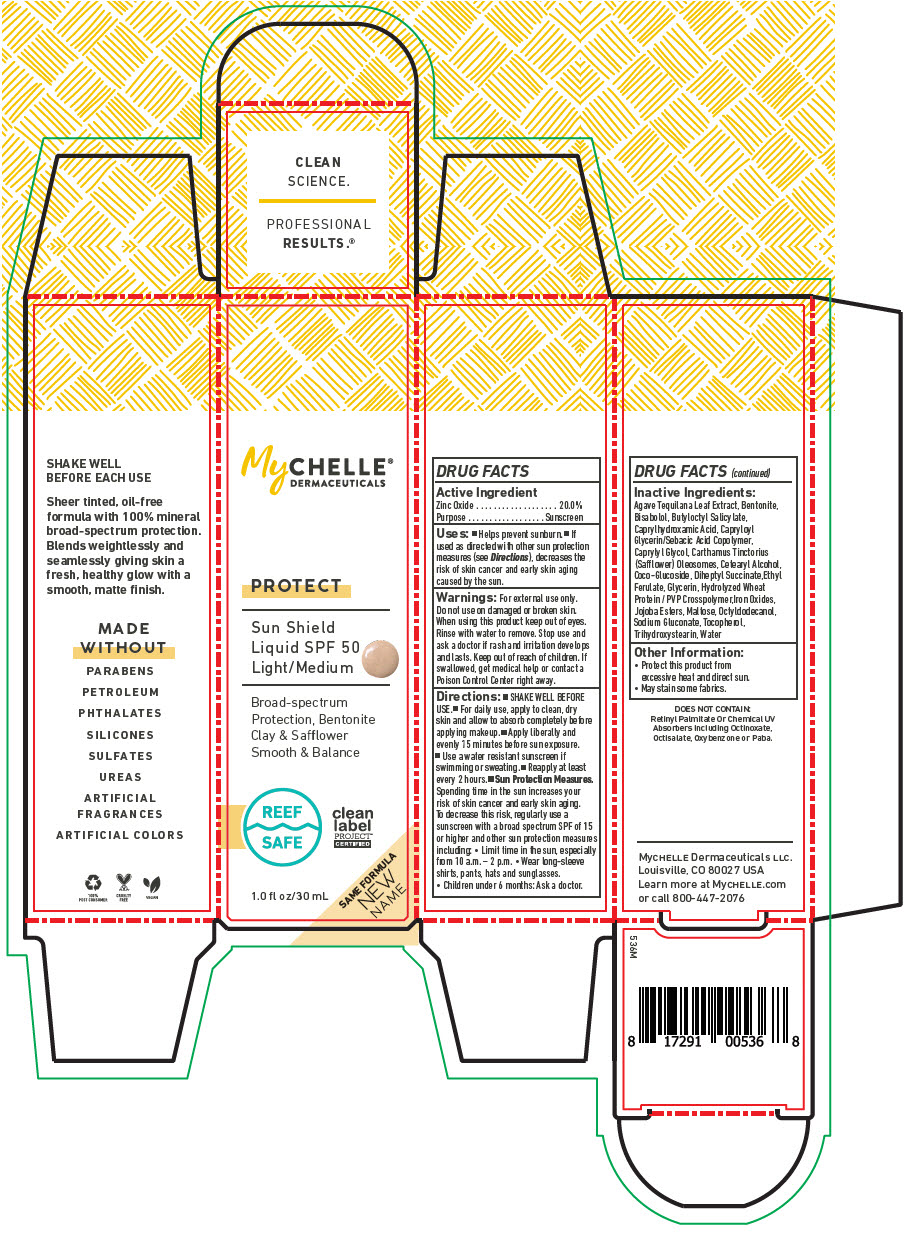
DRUG FACTS
Purpose
Sunscreen
Medication Information
Purpose
Sunscreen
Description
DRUG FACTS
Uses
- Helps prevent sunburn.
- If used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun.
Section 42229-5
DRUG FACTS
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7
When using this product keep out of eyes.
Rinse with water to remove. Stop use and ask a doctor if rash and irritation develops and lasts.
Section 50570-1
Do not use on damaged or broken skin.
Warnings
For external use only.
Directions
- SHAKE WELL BEFORE USE.
- For daily use, apply to clean, dry skin and allow to absorb completely before applying makeup.
- Apply liberally and evenly 15 minutes before sun exposure.
- Use a water resistant sunscreen if swimming or sweating.
- Reapply at least every 2 hours.
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10 a.m. – 2 p.m.
- Wear long-sleeve shirts, pants, hats and sunglasses.
- Children under 6 months: Ask a doctor.
Active Ingredient
Zinc Oxide 20.0%
Other Information
- Protect this product from excessive heat and direct sun.
- May stain some fabrics.
Inactive Ingredients
Agave Tequilana Leaf Extract, Bentonite, Bisabolol, Butyloctyl Salicylate, Caprylhydroxamic Acid, Capryloyl Glycerin/Sebacic Acid Copolymer, Caprylyl Glycol, Carthamus Tinctorius (Safflower) Oleosomes, Cetearyl Alcohol, Coco-Glucoside, Diheptyl Succinate,Ethyl Ferulate, Glycerin, Hydrolyzed Wheat Protein / PVP Crosspolymer,Iron Oxides, Jojoba Esters, Maltose, Octyldodecanol, Sodium Gluconate, Tocopherol, Trihydroxystearin, Water
Principal Display Panel 30 Ml Bottle Box
MyCHELLE®
DERMACEUTICALS
PROTECT
Sun Shield
Liquid SPF 50
Light/Medium
Broad-spectrum
Protection, Bentonite
Clay & Safflower
Smooth & Balance
REEF
SAFE
clean
label
PROJECT®
CERTIFIED
SAME FORMULA
NEW
NAME
1.0 fl oz/30 mL
Structured Label Content
Uses
- Helps prevent sunburn.
- If used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun.
Section 42229-5 (42229-5)
DRUG FACTS
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50567-7 (50567-7)
When using this product keep out of eyes.
Rinse with water to remove. Stop use and ask a doctor if rash and irritation develops and lasts.
Section 50570-1 (50570-1)
Do not use on damaged or broken skin.
Purpose
Sunscreen
Warnings
For external use only.
Directions
- SHAKE WELL BEFORE USE.
- For daily use, apply to clean, dry skin and allow to absorb completely before applying makeup.
- Apply liberally and evenly 15 minutes before sun exposure.
- Use a water resistant sunscreen if swimming or sweating.
- Reapply at least every 2 hours.
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10 a.m. – 2 p.m.
- Wear long-sleeve shirts, pants, hats and sunglasses.
- Children under 6 months: Ask a doctor.
Active Ingredient
Zinc Oxide 20.0%
Other Information
- Protect this product from excessive heat and direct sun.
- May stain some fabrics.
Inactive Ingredients
Agave Tequilana Leaf Extract, Bentonite, Bisabolol, Butyloctyl Salicylate, Caprylhydroxamic Acid, Capryloyl Glycerin/Sebacic Acid Copolymer, Caprylyl Glycol, Carthamus Tinctorius (Safflower) Oleosomes, Cetearyl Alcohol, Coco-Glucoside, Diheptyl Succinate,Ethyl Ferulate, Glycerin, Hydrolyzed Wheat Protein / PVP Crosspolymer,Iron Oxides, Jojoba Esters, Maltose, Octyldodecanol, Sodium Gluconate, Tocopherol, Trihydroxystearin, Water
Principal Display Panel 30 Ml Bottle Box (PRINCIPAL DISPLAY PANEL - 30 mL Bottle Box)
MyCHELLE®
DERMACEUTICALS
PROTECT
Sun Shield
Liquid SPF 50
Light/Medium
Broad-spectrum
Protection, Bentonite
Clay & Safflower
Smooth & Balance
REEF
SAFE
clean
label
PROJECT®
CERTIFIED
SAME FORMULA
NEW
NAME
1.0 fl oz/30 mL
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:29.116599 · Updated: 2026-03-14T22:59:28.137987