Mycozyl Al
07edd043-4ac4-b2de-e063-6394a90af25d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
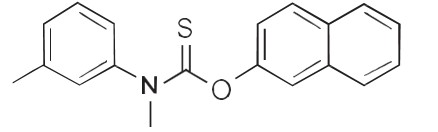
Each gram of Mycozyl AL™ contains 10 mg of tolnaftate in a vehicle consisting of: Apple Cider Vinegar, Argania Spinosa (Argan) Kernel Oil, Benzyl Alcohol, DMSO (Dimethyl Sulfoxide), Eucalyptus Globulus (Eucalyptus) Leaf Oil, Glycerin, Laureth-4, Lavandula Angustifolia (Lavender) Oil, Melaleuca Alternifolia (Tea Tree) Leaf Oil, PEG-8, DL-alpha-tocopheryl Acetate. Chemically, tolnaftate molecular formula is C19H17NOS and molecular weight 314.5 and is represented by the following structure formula:
Indications and Usage
Mycozyl AL™ is effective in the treatment of most skin infections such as athlete's foot (tinea pedis) and ringworm (tinea corporis). Mycozyl AL™ has been designed to reach skin areas around and under the nails while it relives burning, cracking, scaling and discomfort which accompany these conditions. Mycozyl AL™ is an antifungal that works by preventing and eliminating the growth of fungus on fingers, toes and around the nails. It eliminates and helps stop the spread of fungal infections on cuticles around nail edges and under the nail tips where reachable with applicator brush. Mycozyl AL™ cures and prevents fungal infections from coming back with daily use.
Dosage and Administration
• Clean the affected area with soap and warm water and dry thoroughly • Apply a thin layer of product over affected area twice daily (morning and night) paying special attention to the edges of the nail, cuticles and skin around the nails or as directed by a doctor • The brush applicator allows for easy application on skin around the nail and cuticle areas. • For athlete's foot, pay special attention to spaces between the toes, wear well-fitting,ventilated shoes, and change shoes and socks at least once daily. • For athlete's foot and ringworm, use daily for 4 weeks. • Supervise children in the use of this product. Use under the direction of a licensed medical practitioner. Call your doctor about side effects. To report side effects, call PureTek Corporation at 1-877-921-7873 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Contraindications
Tolnaftate topically applied is not likely to affect drugs taken or injected, but many drugs can interact with each other.
Adverse Reactions
For athlete’s foot and ringworm - if irritation occurs or there is no improvement within 4 weeks. For prevention of athlete’s foot – if irritation occurs, discontinue use and consult a doctor.
How Supplied
Mycozyl AL™ is supplied in: 10 mL glass bottle with a screw cap fitted with a brush applicator (NDC 59088-443-01). Store at 20º-25ºC (68º-77ºF) [see USP Controlled Room Temperature]. Protect from freezing and excessive heat. Keep container tightly closed. Do not use if package is damaged. Keep out of reach of children. Manufactured by: PureTek Corporation Panorama City, CA 91402 For questions or information call toll-free: 877-921-7873
Medication Information
Indications and Usage
Mycozyl AL™ is effective in the treatment of most skin infections such as athlete's foot (tinea pedis) and ringworm (tinea corporis). Mycozyl AL™ has been designed to reach skin areas around and under the nails while it relives burning, cracking, scaling and discomfort which accompany these conditions. Mycozyl AL™ is an antifungal that works by preventing and eliminating the growth of fungus on fingers, toes and around the nails. It eliminates and helps stop the spread of fungal infections on cuticles around nail edges and under the nail tips where reachable with applicator brush. Mycozyl AL™ cures and prevents fungal infections from coming back with daily use.
Dosage and Administration
• Clean the affected area with soap and warm water and dry thoroughly • Apply a thin layer of product over affected area twice daily (morning and night) paying special attention to the edges of the nail, cuticles and skin around the nails or as directed by a doctor • The brush applicator allows for easy application on skin around the nail and cuticle areas. • For athlete's foot, pay special attention to spaces between the toes, wear well-fitting,ventilated shoes, and change shoes and socks at least once daily. • For athlete's foot and ringworm, use daily for 4 weeks. • Supervise children in the use of this product. Use under the direction of a licensed medical practitioner. Call your doctor about side effects. To report side effects, call PureTek Corporation at 1-877-921-7873 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Contraindications
Tolnaftate topically applied is not likely to affect drugs taken or injected, but many drugs can interact with each other.
Adverse Reactions
For athlete’s foot and ringworm - if irritation occurs or there is no improvement within 4 weeks. For prevention of athlete’s foot – if irritation occurs, discontinue use and consult a doctor.
How Supplied
Mycozyl AL™ is supplied in: 10 mL glass bottle with a screw cap fitted with a brush applicator (NDC 59088-443-01). Store at 20º-25ºC (68º-77ºF) [see USP Controlled Room Temperature]. Protect from freezing and excessive heat. Keep container tightly closed. Do not use if package is damaged. Keep out of reach of children. Manufactured by: PureTek Corporation Panorama City, CA 91402 For questions or information call toll-free: 877-921-7873
Description
Each gram of Mycozyl AL™ contains 10 mg of tolnaftate in a vehicle consisting of: Apple Cider Vinegar, Argania Spinosa (Argan) Kernel Oil, Benzyl Alcohol, DMSO (Dimethyl Sulfoxide), Eucalyptus Globulus (Eucalyptus) Leaf Oil, Glycerin, Laureth-4, Lavandula Angustifolia (Lavender) Oil, Melaleuca Alternifolia (Tea Tree) Leaf Oil, PEG-8, DL-alpha-tocopheryl Acetate. Chemically, tolnaftate molecular formula is C19H17NOS and molecular weight 314.5 and is represented by the following structure formula:
Section 42228-7
Teratogenic effects - There is currently no published human data available on the teratogenic and fetotoxic effects of tolnaftate.
General:
Do not start, stop, or change the dosage of any medicine before checking with your doctor, health care provider or pharmacist first.
Warnings:
For External Use Only. Not For Ophthalmic Use
Overdosage:
(See PRECAUTIONS).
Description:
Each gram of
Mycozyl AL™ contains 10 mg of tolnaftate in a vehicle consisting of: Apple Cider Vinegar, Argania
Spinosa (Argan) Kernel Oil, Benzyl Alcohol, DMSO (Dimethyl Sulfoxide), Eucalyptus Globulus (Eucalyptus) Leaf
Oil, Glycerin, Laureth-4, Lavandula Angustifolia (Lavender) Oil, Melaleuca Alternifolia (Tea Tree) Leaf Oil, PEG-8,
DL-alpha-tocopheryl Acetate.
Chemically, tolnaftate molecular formula is C19H17NOS and molecular weight 314.5 and is represented by the
following structure formula:
Precautions:
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. Avoid contact with eyes, lips and mucous membranes.
How Supplied:
Mycozyl AL™ is supplied in:
10 mL glass bottle with a screw cap fitted with a brush applicator (NDC 59088-443-01).
Store at 20º-25ºC (68º-77ºF) [see USP Controlled Room Temperature]. Protect from freezing and excessive heat. Keep
container tightly closed.
Do not use if package is damaged. Keep out of reach of children.
Manufactured by:
PureTek Corporation
Panorama City, CA 91402
For questions or information
call toll-free:
877-921-7873
Mycozyl Al™
Pediatric Use:
Safety and effectiveness have not been established in pediatric patients less than 2 years of age.
Nursing Mothers:
Before using this medication while breastfeeding, it is recommended to consult with a healthcare professional.
Pharmacokinetics:
Tolnaftate is only given topically, and there are no documented reports on its pharmacokinetics and systemic metabolism.
Adverse Reactions:
For athlete’s foot and ringworm - if irritation occurs or there is no improvement within 4 weeks.
For prevention of athlete’s foot – if irritation occurs, discontinue use and consult a doctor.
Contraindications:
Tolnaftate topically applied is not likely to affect drugs taken or injected, but many drugs can interact with each other.
Clinical Pharmacology:
Tolnaftate has antifungal properties.
Indications and Usage:
Mycozyl AL™ is effective in the treatment of most skin infections such as athlete's foot (tinea pedis) and ringworm (tinea corporis). Mycozyl AL™ has been designed to reach skin areas around and under the nails while it relives burning, cracking, scaling and discomfort which accompany these conditions. Mycozyl AL™ is an antifungal that works by preventing and eliminating the growth of fungus on fingers, toes and around the nails. It eliminates and helps stop the spread of fungal infections on cuticles around nail edges and under the nail tips where reachable with applicator brush. Mycozyl AL™ cures and prevents fungal infections from coming back with daily use.
Dosage and Administration:
• Clean the affected area with soap and warm water and dry thoroughly
• Apply a thin layer of product over affected area twice daily (morning and night) paying special attention to the edges of the nail, cuticles and skin around the nails or as directed by a doctor
• The brush applicator allows for easy application on skin around the nail and cuticle areas.
• For athlete's foot, pay special attention to spaces between the toes, wear well-fitting,ventilated shoes, and change shoes and socks at least once daily.
• For athlete's foot and ringworm, use daily for 4 weeks.
• Supervise children in the use of this product.
Use under the direction of a licensed medical practitioner.
Call your doctor about side effects. To report side effects, call PureTek Corporation at
1-877-921-7873 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Carcinogenesis, Mutagenesis, and Impairment of Fertility:
Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of Tolnaftate.
Structured Label Content
Section 42228-7 (42228-7)
Teratogenic effects - There is currently no published human data available on the teratogenic and fetotoxic effects of tolnaftate.
General:
Do not start, stop, or change the dosage of any medicine before checking with your doctor, health care provider or pharmacist first.
Warnings: (WARNINGS:)
For External Use Only. Not For Ophthalmic Use
Overdosage: (OVERDOSAGE:)
(See PRECAUTIONS).
Description: (DESCRIPTION:)
Each gram of
Mycozyl AL™ contains 10 mg of tolnaftate in a vehicle consisting of: Apple Cider Vinegar, Argania
Spinosa (Argan) Kernel Oil, Benzyl Alcohol, DMSO (Dimethyl Sulfoxide), Eucalyptus Globulus (Eucalyptus) Leaf
Oil, Glycerin, Laureth-4, Lavandula Angustifolia (Lavender) Oil, Melaleuca Alternifolia (Tea Tree) Leaf Oil, PEG-8,
DL-alpha-tocopheryl Acetate.
Chemically, tolnaftate molecular formula is C19H17NOS and molecular weight 314.5 and is represented by the
following structure formula:
Precautions: (PRECAUTIONS:)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away. Avoid contact with eyes, lips and mucous membranes.
How Supplied: (HOW SUPPLIED:)
Mycozyl AL™ is supplied in:
10 mL glass bottle with a screw cap fitted with a brush applicator (NDC 59088-443-01).
Store at 20º-25ºC (68º-77ºF) [see USP Controlled Room Temperature]. Protect from freezing and excessive heat. Keep
container tightly closed.
Do not use if package is damaged. Keep out of reach of children.
Manufactured by:
PureTek Corporation
Panorama City, CA 91402
For questions or information
call toll-free:
877-921-7873
Mycozyl Al™ (Mycozyl AL™)
Pediatric Use:
Safety and effectiveness have not been established in pediatric patients less than 2 years of age.
Nursing Mothers:
Before using this medication while breastfeeding, it is recommended to consult with a healthcare professional.
Pharmacokinetics:
Tolnaftate is only given topically, and there are no documented reports on its pharmacokinetics and systemic metabolism.
Adverse Reactions: (ADVERSE REACTIONS:)
For athlete’s foot and ringworm - if irritation occurs or there is no improvement within 4 weeks.
For prevention of athlete’s foot – if irritation occurs, discontinue use and consult a doctor.
Contraindications: (CONTRAINDICATIONS:)
Tolnaftate topically applied is not likely to affect drugs taken or injected, but many drugs can interact with each other.
Clinical Pharmacology: (CLINICAL PHARMACOLOGY:)
Tolnaftate has antifungal properties.
Indications and Usage: (INDICATIONS AND USAGE:)
Mycozyl AL™ is effective in the treatment of most skin infections such as athlete's foot (tinea pedis) and ringworm (tinea corporis). Mycozyl AL™ has been designed to reach skin areas around and under the nails while it relives burning, cracking, scaling and discomfort which accompany these conditions. Mycozyl AL™ is an antifungal that works by preventing and eliminating the growth of fungus on fingers, toes and around the nails. It eliminates and helps stop the spread of fungal infections on cuticles around nail edges and under the nail tips where reachable with applicator brush. Mycozyl AL™ cures and prevents fungal infections from coming back with daily use.
Dosage and Administration: (DOSAGE AND ADMINISTRATION:)
• Clean the affected area with soap and warm water and dry thoroughly
• Apply a thin layer of product over affected area twice daily (morning and night) paying special attention to the edges of the nail, cuticles and skin around the nails or as directed by a doctor
• The brush applicator allows for easy application on skin around the nail and cuticle areas.
• For athlete's foot, pay special attention to spaces between the toes, wear well-fitting,ventilated shoes, and change shoes and socks at least once daily.
• For athlete's foot and ringworm, use daily for 4 weeks.
• Supervise children in the use of this product.
Use under the direction of a licensed medical practitioner.
Call your doctor about side effects. To report side effects, call PureTek Corporation at
1-877-921-7873 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Carcinogenesis, Mutagenesis, and Impairment of Fertility:
Long-term animal studies have not been performed to evaluate the carcinogenic potential or the effect on fertility of Tolnaftate.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:05.519979 · Updated: 2026-03-14T21:56:34.647389