Kanka®
078fcdab-38a1-4a9a-bdd3-e17446591ea8
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Description
Drug Facts
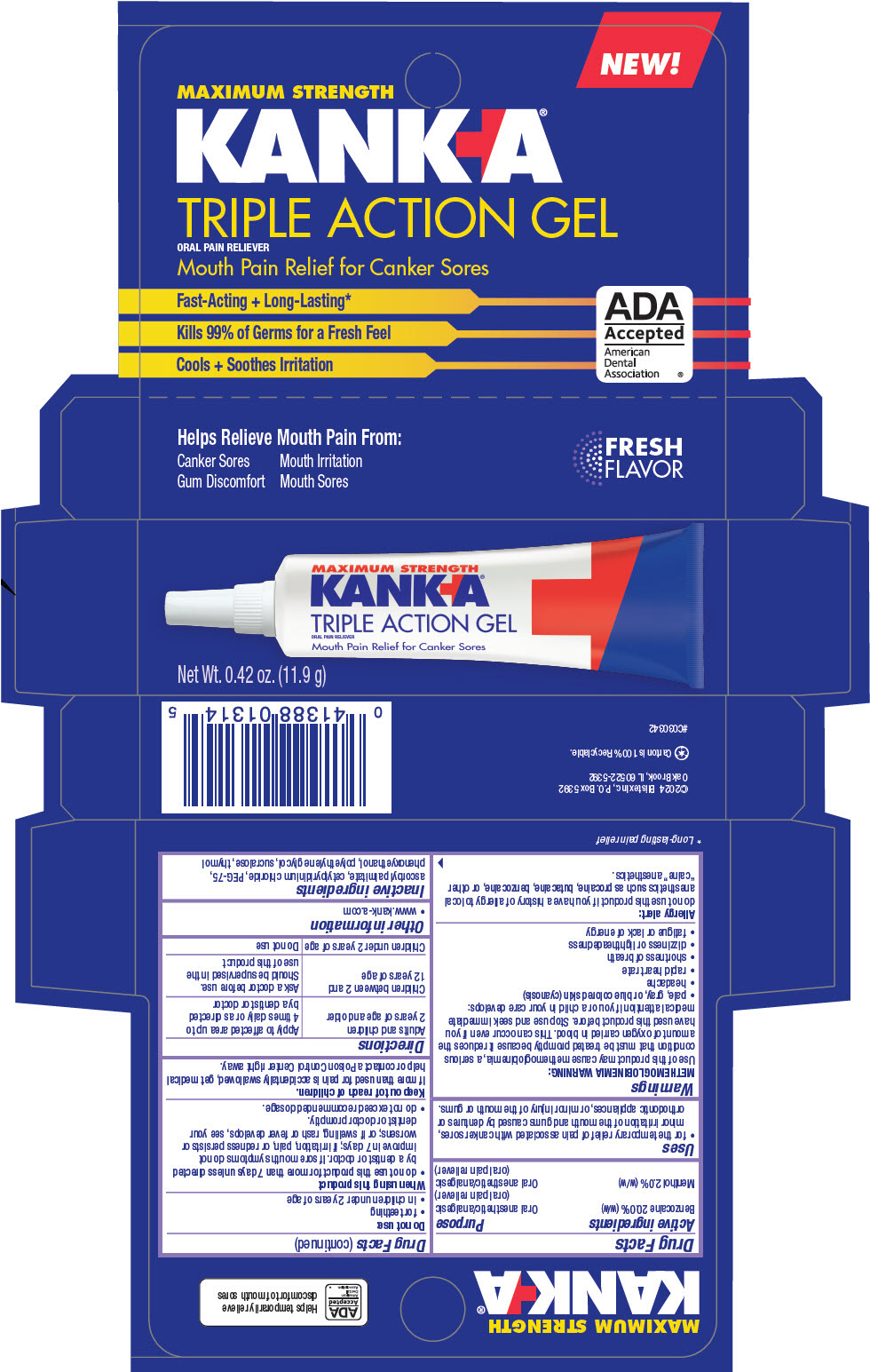
Uses
- for the temporary relief of pain associated with canker sores, minor irritation of the mouth and gums caused by dentures or orthodontic appliances, or minor injury of the mouth or gums.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
If more than used for pain is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Section 55105-1
| Active ingredients | Purpose |
|---|---|
| Benzocaine 20.0% (w/w) | Oral anesthetic/analgesic (oral pain reliever) |
| Menthol 2.0% (w/w) | Oral anesthetic/analgesic (oral pain reliever) |
Directions
| Adults and children 2 years of age and older | Apply to affected area up to 4 times daily or as directed by a dentist or doctor |
| Children between 2 and 12 years of age | Ask a doctor before use. Should be supervised in the use of this product |
| Children under 2 years of age | Do not use |
Do Not Use
- for teething
- in children under 2 years of age
Allergy Alert
do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or other "caine" anesthetics.
Other Information
- www.kank-a.com
Inactive Ingredients
ascorbyl palmitate, cetylpyridinium chloride, PEG-75, phenoxyethanol, polyethylene glycol, sucralose, thymol
When Using This Product
- do not use this product for more than 7 days unless directed by a dentist or doctor. If sore mouth symptoms do not improve in 7 days; if irritation, pain, or redness persists or worsens; or if swelling, rash or fever develops, see your dentist or doctor promptly.
- do not exceed recommended dosage.
Methemoglobinemia Warning
Use of this product may cause methemoglobinemia, a serious condition that must be treated promptly because it reduces the amount of oxygen carried in blood. This can occur even if you have used this product before. Stop use and seek immediate medical attention if you or a child in your care develops:
- pale, gray, or blue colored skin (cyanosis)
- headache
- rapid heart rate
- shortness of breath
- dizziness or lightheadedness
- fatigue or lack of energy
Principal Display Panel 11.9 G Tube Carton
MAXIMUM STRENGTH
KANKA®
TRIPLE ACTION GEL
ORAL PAIN RELIEVER
Mouth Pain Relief for Canker Sores
Net Wt. 0.42 oz. (11.9 g)
Structured Label Content
Uses
- for the temporary relief of pain associated with canker sores, minor irritation of the mouth and gums caused by dentures or orthodontic appliances, or minor injury of the mouth or gums.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
If more than used for pain is accidentally swallowed, get medical help or contact a Poison Control Center right away.
Section 55105-1 (55105-1)
| Active ingredients | Purpose |
|---|---|
| Benzocaine 20.0% (w/w) | Oral anesthetic/analgesic (oral pain reliever) |
| Menthol 2.0% (w/w) | Oral anesthetic/analgesic (oral pain reliever) |
Directions
| Adults and children 2 years of age and older | Apply to affected area up to 4 times daily or as directed by a dentist or doctor |
| Children between 2 and 12 years of age | Ask a doctor before use. Should be supervised in the use of this product |
| Children under 2 years of age | Do not use |
Do Not Use (Do not use)
- for teething
- in children under 2 years of age
Allergy Alert (Allergy alert)
do not use this product if you have a history of allergy to local anesthetics such as procaine, butacaine, benzocaine, or other "caine" anesthetics.
Other Information (Other information)
- www.kank-a.com
Inactive Ingredients (Inactive ingredients)
ascorbyl palmitate, cetylpyridinium chloride, PEG-75, phenoxyethanol, polyethylene glycol, sucralose, thymol
When Using This Product (When using this product)
- do not use this product for more than 7 days unless directed by a dentist or doctor. If sore mouth symptoms do not improve in 7 days; if irritation, pain, or redness persists or worsens; or if swelling, rash or fever develops, see your dentist or doctor promptly.
- do not exceed recommended dosage.
Methemoglobinemia Warning (METHEMOGLOBINEMIA WARNING)
Use of this product may cause methemoglobinemia, a serious condition that must be treated promptly because it reduces the amount of oxygen carried in blood. This can occur even if you have used this product before. Stop use and seek immediate medical attention if you or a child in your care develops:
- pale, gray, or blue colored skin (cyanosis)
- headache
- rapid heart rate
- shortness of breath
- dizziness or lightheadedness
- fatigue or lack of energy
Principal Display Panel 11.9 G Tube Carton (PRINCIPAL DISPLAY PANEL - 11.9 g Tube Carton)
MAXIMUM STRENGTH
KANKA®
TRIPLE ACTION GEL
ORAL PAIN RELIEVER
Mouth Pain Relief for Canker Sores
Net Wt. 0.42 oz. (11.9 g)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:17.229683 · Updated: 2026-03-14T23:06:51.816479