These Highlights Do Not Include All The Information Needed To Use Orgovyx Safely And Effectively. See Full Prescribing Information For Orgovyx.
077a92f6-9f1b-479a-87c7-c92b5db6aa9c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ORGOVYX is indicated for the treatment of adult patients with advanced prostate cancer.
Indications and Usage
ORGOVYX is indicated for the treatment of adult patients with advanced prostate cancer.
Dosage and Administration
Recommended Dosage: A loading dose of 360 mg on the first day of treatment followed by 120 mg taken orally once daily, at approximately the same time each day ( 2.1 ). ORGOVYX can be taken with or without food ( 2.1 , 12.3 ). Instruct patients to swallow tablets whole and not to crush or chew tablets ( 2.1 ).
Warnings and Precautions
QT/QTc Interval Prolongation: Androgen deprivation therapy may prolong the QT interval ( 5.1 ). Hypersensitivity: ORGOVYX can cause hypersensitivity reactions, including angioedema. Withhold ORGOVYX in patients who experience symptoms of hypersensitivity. Discontinue ORGOVYX for severe hypersensitivity reactions and manage as clinically indicated ( 5.2 ). Embryo-Fetal Toxicity: ORGOVYX can cause fetal harm. Advise males with female partners of reproductive potential to use effective contraception ( 5.3 , 8.1 , 8.3 ).
Contraindications
ORGOVYX is contraindicated in patients with severe hypersensitivity to relugolix or to any of the product components.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: QT/QTc Interval Prolongation [see Warnings and Precautions ( 5.1 )] .
Drug Interactions
P-gp Inhibitors: Avoid co-administration. If unavoidable, take ORGOVYX first, separate dosing by at least 6 hours, and monitor patients more frequently for adverse reactions ( 2.2 , 7.1 ). Combined P-gp and Strong CYP3A Inducers: Avoid co-administration. If unavoidable, increase the ORGOVYX dose to 240 mg once daily ( 2.3 , 7.1 ).
Medication Information
Warnings and Precautions
QT/QTc Interval Prolongation: Androgen deprivation therapy may prolong the QT interval ( 5.1 ). Hypersensitivity: ORGOVYX can cause hypersensitivity reactions, including angioedema. Withhold ORGOVYX in patients who experience symptoms of hypersensitivity. Discontinue ORGOVYX for severe hypersensitivity reactions and manage as clinically indicated ( 5.2 ). Embryo-Fetal Toxicity: ORGOVYX can cause fetal harm. Advise males with female partners of reproductive potential to use effective contraception ( 5.3 , 8.1 , 8.3 ).
Indications and Usage
ORGOVYX is indicated for the treatment of adult patients with advanced prostate cancer.
Dosage and Administration
Recommended Dosage: A loading dose of 360 mg on the first day of treatment followed by 120 mg taken orally once daily, at approximately the same time each day ( 2.1 ). ORGOVYX can be taken with or without food ( 2.1 , 12.3 ). Instruct patients to swallow tablets whole and not to crush or chew tablets ( 2.1 ).
Dosage Forms and Strengths
How Supplied
The 120 mg tablets are film-coated, light red, almond shaped, and debossed with “R” on one side and “120” on the other side and are supplied in two configurations, bottles and blister packs. Each bottle (NDC 72974-120-01) contains 30 tablets and a desiccant and is closed with a child resistant induction seal cap. The blister cards contain nine tablets packaged in a carton (NDC 72974-120-02). Each ORGOVYX tablet contains 120 mg of relugolix.
Contraindications
ORGOVYX is contraindicated in patients with severe hypersensitivity to relugolix or to any of the product components.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: QT/QTc Interval Prolongation [see Warnings and Precautions ( 5.1 )] .
Drug Interactions
P-gp Inhibitors: Avoid co-administration. If unavoidable, take ORGOVYX first, separate dosing by at least 6 hours, and monitor patients more frequently for adverse reactions ( 2.2 , 7.1 ). Combined P-gp and Strong CYP3A Inducers: Avoid co-administration. If unavoidable, increase the ORGOVYX dose to 240 mg once daily ( 2.3 , 7.1 ).
Description
ORGOVYX is indicated for the treatment of adult patients with advanced prostate cancer.
Section 42229-5
P-gp Inhibitors
Relugolix is a P-gp substrate. Co-administration of ORGOVYX with an oral P-gp inhibitor increases relugolix exposure [see Clinical Pharmacology (12.3)], which may increase the risk of adverse reactions associated with ORGOVYX.
Avoid co-administration of ORGOVYX with oral P-gp inhibitors.
If co-administration with an oral P-gp inhibitor cannot be avoided, take ORGOVYX first and separate dosing by at least 6 hours. Monitor patients for increased adverse reactions [see Dosage and Administration (2.2)].
Treatment with ORGOVYX may be interrupted for up to two weeks if a short course of treatment with a P-gp inhibitor is required. Resume ORGOVYX after the P-gp inhibitor is discontinued. If treatment with ORGOVYX is interrupted for greater than 7 days, restart ORGOVYX with a loading dose of 360 mg on the first day and continue with a dose of 120 mg once daily.
Section 42230-3
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 08/2023 |
|||
|
PATIENT INFORMATION
ORGOVYX (or-GO-vix) (relugolix) Tablets |
||||
|
What is ORGOVYX?
ORGOVYX is a prescription medicine used in adults for the treatment of advanced prostate cancer. It is not known if ORGOVYX is safe or effective in females. It is not known if ORGOVYX is safe or effective in children. |
||||
| Do not take ORGOVYX if you have had a severe allergic reaction to relugolix or any of the ingredients in ORGOVYX. See the end of this Patient Information for a complete list of the ingredients in ORGOVYX. | ||||
Before taking ORGOVYX, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking ORGOVYX with certain other medicines can affect how ORGOVYX works or may cause side effects. You should not start or stop any medicine before you talk with your healthcare provider who prescribed ORGOVYX. Know the medicines you take. Keep a list of them with you to show to your healthcare provider and pharmacist when you get a new medicine. |
||||
How should I take ORGOVYX?
|
||||
|
What are the possible side effects of ORGOVYX?
ORGOVYX may cause serious side effects, including:
|
||||
|
The most common side effects of ORGOVYX include:
|
||||
|
|
|||
| Other side effects include weight gain, decreased sex drive, and erectile function problems. ORGOVYX may cause fertility problems in males, which may affect your ability to father children. Talk to your healthcare provider if this is a concern for you. These are not all the possible side effects of ORGOVYX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store ORGOVYX?
|
||||
| Keep ORGOVYX and all medicines out of the reach of children. | ||||
|
General information about the safe and effective use of ORGOVYX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ORGOVYX for a condition for which it was not prescribed. Do not give ORGOVYX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ORGOVYX that is written for health professionals. |
||||
|
What are the ingredients in ORGOVYX?
Active ingredient: relugolix Inactive ingredients: mannitol, sodium starch glycolate, hydroxypropyl cellulose, magnesium stearate, hypromellose, titanium dioxide, ferric oxide red, and carnauba wax. Manufactured for: Sumitomo Pharma America, Inc., Marlborough, MA 01752 For more information, go to www.orgovyx.com or call 1-833-696-8268. |
214621-MS-005
Section 44425-7
Storage and Handling
- Store ORGOVYX at room temperature. Do not store above 30°C (86°F).
- Dispense to patients in original container only.
- For bottles, keep container tightly closed after first opening.
- Keep out of reach of children.
11 Description
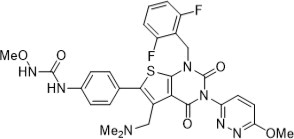
Relugolix is a nonpeptide small molecule, GnRH receptor antagonist. The chemical name is N-(4-{1-[(2,6-difluorophenyl)methyl]-5-[(dimethylamino)methyl]-3-(6-methoxypyridazin-3-yl)-2,4-dioxo-1,2,3,4-tetrahydrothieno[2,3-d]pyrimidin-6-yl}phenyl)-N'-methoxyurea.
The molecular weight is 623.63 daltons and the molecular formula is C29H27F2N7O5S. The structural formula is:
Relugolix is a white to off-white to slightly yellow solid with a solubility of 0.04 mg per mL in water at 25°C.
ORGOVYX is provided as film-coated tablets for oral administration. Each tablet contains 120 mg of relugolix. The inactive ingredients are mannitol, sodium starch glycolate, hydroxypropyl cellulose, magnesium stearate, hypromellose, titanium dioxide, ferric oxide red, and carnauba wax.
8.4 Pediatric Use
The safety and efficacy of ORGOVYX in pediatric patients have not been established.
8.5 Geriatric Use
Of the 622 patients who received ORGOVYX in the HERO study, 81% were 65 years of age or older, while 35% were 75 years of age or older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. There was no clinically relevant impact of age on the pharmacokinetics of ORGOVYX or testosterone response based on population pharmacokinetic and pharmacokinetic/pharmacodynamic analyses in men 45 to 91 years of age.
4 Contraindications
ORGOVYX is contraindicated in patients with severe hypersensitivity to relugolix or to any of the product components.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- QT/QTc Interval Prolongation [see Warnings and Precautions (5.1)].
7 Drug Interactions
P-gp Inhibitors: Avoid co-administration. If unavoidable, take ORGOVYX first, separate dosing by at least 6 hours, and monitor patients more frequently for adverse reactions (2.2, 7.1).
Combined P-gp and Strong CYP3A Inducers: Avoid co-administration. If unavoidable, increase the ORGOVYX dose to 240 mg once daily (2.3, 7.1).
12.3 Pharmacokinetics
After administration of single doses ranging from 60 mg to 360 mg (0.17 to 1 times the recommended loading dose), total systemic exposure (AUC) and the maximum concentration (Cmax) of relugolix increases in an approximately dose proportional manner. After administration of multiple doses of relugolix once daily, the AUC of relugolix increases in an approximately dose proportional manner while the Cmax increase is greater than dose proportional for doses from 20 mg to 180 mg (0.17 to 1.5 times the recommended daily dose).
After administration of a single 360 mg loading dose, the mean (± standard deviation [± SD]) AUC and Cmax of relugolix are 985 (± 742) ng.hr/mL and 215 (± 184) ng/mL, respectively. After administration of 120 mg once daily, the mean (± SD) AUC and Cmax of relugolix at steady-state are 407 (± 168) ng.hr/mL and 70 (± 65) ng/mL, respectively. The accumulation of relugolix upon once daily administration is approximately 2-fold.
2.1 Recommended Dosage
Initiate treatment of ORGOVYX with a loading dose of 360 mg on the first day and continue treatment with a 120 mg dose taken orally once daily at approximately the same time each day.
ORGOVYX can be taken with or without food [see Clinical Pharmacology (12.3)]. Instruct patients to swallow tablets whole and not to crush or chew tablets.
Advise patients to take a missed dose of ORGOVYX as soon as they remember. If the dose was missed by more than 12 hours, patients should not take the missed dose and resume with the next scheduled dose.
If treatment with ORGOVYX is interrupted for greater than 7 days, restart ORGOVYX with a loading dose of 360 mg on the first day, and continue with a dose of 120 mg once daily.
In patients treated with GnRH receptor agonists and antagonists for prostate cancer, treatment is usually continued upon development of nonmetastatic or metastatic castration-resistant prostate cancer.
5.4 Laboratory Testing
Therapy with ORGOVYX results in suppression of the pituitary gonadal system. Results of diagnostic tests of the pituitary gonadotropic and gonadal functions conducted during and after ORGOVYX may be affected. The therapeutic effect of ORGOVYX should be monitored by measuring serum concentrations of prostate specific antigen (PSA) periodically. If PSA increases, serum concentrations of testosterone should be measured.
1 Indications and Usage
ORGOVYX is indicated for the treatment of adult patients with advanced prostate cancer.
12.1 Mechanism of Action
Relugolix is a nonpeptide GnRH receptor antagonist that competitively binds to pituitary GnRH receptors, thereby, reducing the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), and consequently testosterone.
5.3 Embryo Fetal Toxicity
The safety and efficacy of ORGOVYX have not been established in females. Based on findings in animals and mechanism of action, ORGOVYX can cause fetal harm and loss of pregnancy when administered to a pregnant female. In an animal reproduction study, oral administration of relugolix to pregnant rabbits during the period of organogenesis caused embryo-fetal lethality at maternal exposures that were 0.3 times the human exposure at the recommended dose of 120 mg daily based on area under the curve (AUC). Advise males with female partners of reproductive potential to use effective contraception during treatment and for 2 weeks after the last dose of ORGOVYX [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
- QT/QTc Interval Prolongation: Androgen deprivation therapy may prolong the QT interval (5.1).
- Hypersensitivity: ORGOVYX can cause hypersensitivity reactions, including angioedema. Withhold ORGOVYX in patients who experience symptoms of hypersensitivity. Discontinue ORGOVYX for severe hypersensitivity reactions and manage as clinically indicated (5.2).
- Embryo-Fetal Toxicity: ORGOVYX can cause fetal harm. Advise males with female partners of reproductive potential to use effective contraception (5.3, 8.1, 8.3).
2 Dosage and Administration
- Recommended Dosage: A loading dose of 360 mg on the first day of treatment followed by 120 mg taken orally once daily, at approximately the same time each day (2.1).
- ORGOVYX can be taken with or without food (2.1, 12.3). Instruct patients to swallow tablets whole and not to crush or chew tablets (2.1).
3 Dosage Forms and Strengths
Tablets: 120 mg, light red, almond-shaped, film-coated, and debossed with “R” on one side and “120” on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ORGOVYX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: hypersensitivity, including angioedema and urticaria.
5.2 Hypersensitivity Reactions
ORGOVYX is contraindicated in patients with severe hypersensitivity to relugolix or any of the product components [see Contraindications (4)]. Hypersensitivity reactions, including pharyngeal edema and other serious cases of angioedema, have been reported postmarketing in patients treated with ORGOVYX.
In HERO, patients treated with relugolix reported angioedema (0.2%) [see Clinical Trials Experience (6.1)].
Advise patients who experience any symptoms of hypersensitivity to temporarily discontinue ORGOVYX and promptly seek medical care.
Discontinue ORGOVYX for severe hypersensitivity reactions and manage as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ORGOVYX was evaluated in HERO, a randomized (2:1), open-label, clinical study in patients with advanced prostate cancer [see Clinical Studies (14)]. Patients received orally administered ORGOVYX as a loading dose of 360 mg on the first day followed by 120 mg taken orally once daily (n = 622) or received leuprolide acetate administered by depot injection at doses of 22.5 mg (n = 264) or 11.25 mg (n = 44) per local guidelines every 12 weeks (n = 308). Leuprolide acetate 11.25 mg is a dosing regimen that is not recommended for this indication in the US. Among patients who received ORGOVYX, 91% were exposed for at least 48 weeks. Ninety-nine (16%) patients received concomitant radiotherapy and 17 (3%) patients received concomitant enzalutamide with ORGOVYX.
Serious adverse reactions occurred in 12% of patients receiving ORGOVYX. Serious adverse reactions in ≥ 0.5% of patients included myocardial infarction (0.8%), acute kidney injury (0.6%), arrhythmia (0.6%), hemorrhage (0.6%), and urinary tract infection (0.5%). Fatal adverse reactions occurred in 0.8% of patients receiving ORGOVYX including metastatic lung cancer (0.3%), myocardial infarction (0.3%), and acute kidney injury (0.2%). Fatal and non-fatal myocardial infarction and stroke were reported in 2.7% of patients receiving ORGOVYX.
Permanent discontinuation of ORGOVYX due to an adverse reaction occurred in 3.5% of patients. Adverse reactions which resulted in permanent discontinuation of ORGOVYX in ≥ 0.3 % of patients included atrioventricular block (0.3%), cardiac failure (0.3%), hemorrhage (0.3%), increased transaminases (0.3%), abdominal pain (0.3%), and pneumonia (0.3%).
Dosage interruptions of ORGOVYX due to an adverse reaction occurred in 2.7% of patients. Adverse reactions which required dosage interruption in ≥ 0.3% of patients included fracture (0.3%).
The most common adverse reactions (≥ 10%) and laboratory abnormalities (≥ 15%) were hot flush (54%), glucose increased (44%), triglycerides increased (35%), musculoskeletal pain (30%), hemoglobin decreased (28%), alanine aminotransferase increased (ALT) (27%), fatigue (26%), aspartate aminotransferase increased (AST) (18%), constipation (12%), and diarrhea (12%).
Table 1 summarizes the adverse reactions in HERO.
|
a Includes arthralgia, back pain, pain in extremity, musculoskeletal pain, myalgia, bone pain, neck pain, arthritis, musculoskeletal stiffness, non-cardiac chest pain, musculoskeletal chest pain, spinal pain, and musculoskeletal discomfort. |
||||
|
b Includes fatigue and asthenia. |
||||
|
c Includes diarrhea and colitis. |
||||
| Adverse Reaction |
ORGOVYX N = 622 |
Leuprolide Acetate
N = 308 |
||
|
All Grades
(%) |
Grade 3-4
(%) |
All Grades
(%) |
Grade 3-4
(%) |
|
| Vascular disorders | ||||
| Hot flush | 54 | 0.6 | 52 | 0 |
| Musculoskeletal and connective tissue disorders | ||||
| Musculoskeletal paina | 30 | 1.1 | 29 | 1.6 |
| General | ||||
| Fatigueb | 26 | 0.3 | 24 | 0 |
| Gastrointestinal disorders | ||||
| Diarrheac | 12 | 0.2 | 7 | 0 |
| Constipation | 12 | 0 | 10 | 0 |
Clinically relevant adverse reactions in < 10% of patients who received ORGOVYX included increased weight, insomnia, gynecomastia, hyperhidrosis, depression, decreased libido, and angioedema.
Table 2 summarizes the laboratory abnormalities in HERO.
| Laboratory Test | ORGOVYX a | Leuprolide Acetate a | ||
|---|---|---|---|---|
|
All Grades
(%) |
Grade 3-4
(%) |
All Grades
(%) |
Grade 3-4
(%) |
|
|
a The denominator used to calculate the rate varied from 611 to 619 in the ORGOVYX arm and from 301 to 306 in the leuprolide arm based on the number of patients with a baseline value and at least one post-treatment value. |
||||
| Chemistry | ||||
| Glucose increased | 44 | 2.9 | 54 | 6 |
| Triglycerides increased | 35 | 2 | 36 | 0.7 |
| ALT increased | 27 | 0.3 | 28 | 0 |
| AST increased | 18 | 0 | 19 | 0.3 |
| Hematology | ||||
| Hemoglobin decreased | 28 | 0.5 | 29 | 0.7 |
5.1 Qt/qtc Interval Prolongation
Androgen deprivation therapy, such as ORGOVYX, may prolong the QT/QTc interval. Providers should consider whether the benefits of androgen deprivation therapy outweigh the potential risks in patients with congenital long QT syndrome, congestive heart failure, or frequent electrolyte abnormalities and in patients taking drugs known to prolong the QT interval. Electrolyte abnormalities should be corrected. Consider periodic monitoring of electrocardiograms and electrolytes [see Clinical Pharmacology (12.2)].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
13.2 Animal Toxicology And/or Pharmacology
Phospholipidosis (intracellular phospholipid accumulation) was observed in multiple organs and tissues (e.g., liver, pancreas, spleen, kidney, lymph nodes, lung, bone marrow, gastrointestinal tract or testes) after repeated oral administration of relugolix in rats and monkeys. In a rat 26-week toxicity study, phospholipidosis was observed at doses ≥ 100 mg/kg (approximately 18 times the human exposure at the recommended dose based on AUC). In a monkey 39-week toxicity study, this effect was observed at doses ≥ 1.5 mg/kg (approximately 0.6 times the human exposure at the recommended dose based on AUC) and demonstrated evidence of reversibility after cessation of treatment. The significance of this finding in humans is unknown.
2.2 Dosage Modifications for P Gp Inhibitors
Avoid co-administration of ORGOVYX with oral P-gp inhibitors. If co-administration is unavoidable, take ORGOVYX first and separate dosing by at least 6 hours [ see Drug Interactions (7.1) and Clinical Pharmacology (12.3) ]. Monitor patients for increased adverse reactions.
Treatment with ORGOVYX may be interrupted for up to two weeks if a short course of treatment with a P-gp inhibitor is required. Resume ORGOVYX after the P-gp inhibitor is discontinued. If treatment with ORGOVYX is interrupted for greater than 7 days, restart ORGOVYX with a loading dose of 360 mg on the first day and continue with a dose of 120 mg once daily.
Principal Display Panel 120 Mg Tablet Bottle Label
NDC 72974-120-01
Rx only
ORGOVYX®
(relugolix) tablets
120 mg
30 tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted in mice at oral relugolix doses up to 100 mg/kg/day and in rats at doses up to 600 mg/kg/day. Relugolix was not carcinogenic in mice or rats at exposures up to approximately 75 or 224 times, respectively, the human exposure at the recommended dose of 120 mg daily based on AUC.
Relugolix was not mutagenic in the in vitro bacterial reverse mutation (Ames) assay or clastogenic in the in vitro chromosomal aberration assay in Chinese hamster lung cells or the in vivo rat bone marrow micronucleus assay.
In human GnRH-receptor knock-in male mice, oral administration of relugolix decreased prostate and seminal vesicle weights at doses ≥ 3 mg/kg twice daily for 28 days. The effects of relugolix were reversible, except for testis weight, which did not fully recover within 28 days after drug withdrawal. In a 39-week repeat-dose toxicity study in monkeys, there were no significant effects on male reproductive organs at oral relugolix doses up to 50 mg/kg/day (approximately 53 times the human exposure at the recommended dose of 120 mg daily based on AUC).
2.3 Dosage Modifications for Combined P Gp and Strong Cyp3a Inducers
Avoid co-administration of ORGOVYX with combined P-gp and strong CYP3A inducers. If co-administration is unavoidable, increase the ORGOVYX dose to 240 mg once daily. After discontinuation of the combined P-gp and strong CYP3A inducer, resume the recommended ORGOVYX dose of 120 mg once daily [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
Structured Label Content
Dosage Forms and Strengths (34069-5)
How Supplied
The 120 mg tablets are film-coated, light red, almond shaped, and debossed with “R” on one side and “120” on the other side and are supplied in two configurations, bottles and blister packs. Each bottle (NDC 72974-120-01) contains 30 tablets and a desiccant and is closed with a child resistant induction seal cap. The blister cards contain nine tablets packaged in a carton (NDC 72974-120-02). Each ORGOVYX tablet contains 120 mg of relugolix.
Section 42229-5 (42229-5)
P-gp Inhibitors
Relugolix is a P-gp substrate. Co-administration of ORGOVYX with an oral P-gp inhibitor increases relugolix exposure [see Clinical Pharmacology (12.3)], which may increase the risk of adverse reactions associated with ORGOVYX.
Avoid co-administration of ORGOVYX with oral P-gp inhibitors.
If co-administration with an oral P-gp inhibitor cannot be avoided, take ORGOVYX first and separate dosing by at least 6 hours. Monitor patients for increased adverse reactions [see Dosage and Administration (2.2)].
Treatment with ORGOVYX may be interrupted for up to two weeks if a short course of treatment with a P-gp inhibitor is required. Resume ORGOVYX after the P-gp inhibitor is discontinued. If treatment with ORGOVYX is interrupted for greater than 7 days, restart ORGOVYX with a loading dose of 360 mg on the first day and continue with a dose of 120 mg once daily.
Section 42230-3 (42230-3)
|
This Patient Information has been approved by the U.S. Food and Drug Administration. |
Revised: 08/2023 |
|||
|
PATIENT INFORMATION
ORGOVYX (or-GO-vix) (relugolix) Tablets |
||||
|
What is ORGOVYX?
ORGOVYX is a prescription medicine used in adults for the treatment of advanced prostate cancer. It is not known if ORGOVYX is safe or effective in females. It is not known if ORGOVYX is safe or effective in children. |
||||
| Do not take ORGOVYX if you have had a severe allergic reaction to relugolix or any of the ingredients in ORGOVYX. See the end of this Patient Information for a complete list of the ingredients in ORGOVYX. | ||||
Before taking ORGOVYX, tell your healthcare provider about all of your medical conditions, including if you:
|
||||
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking ORGOVYX with certain other medicines can affect how ORGOVYX works or may cause side effects. You should not start or stop any medicine before you talk with your healthcare provider who prescribed ORGOVYX. Know the medicines you take. Keep a list of them with you to show to your healthcare provider and pharmacist when you get a new medicine. |
||||
How should I take ORGOVYX?
|
||||
|
What are the possible side effects of ORGOVYX?
ORGOVYX may cause serious side effects, including:
|
||||
|
The most common side effects of ORGOVYX include:
|
||||
|
|
|||
| Other side effects include weight gain, decreased sex drive, and erectile function problems. ORGOVYX may cause fertility problems in males, which may affect your ability to father children. Talk to your healthcare provider if this is a concern for you. These are not all the possible side effects of ORGOVYX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store ORGOVYX?
|
||||
| Keep ORGOVYX and all medicines out of the reach of children. | ||||
|
General information about the safe and effective use of ORGOVYX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ORGOVYX for a condition for which it was not prescribed. Do not give ORGOVYX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ORGOVYX that is written for health professionals. |
||||
|
What are the ingredients in ORGOVYX?
Active ingredient: relugolix Inactive ingredients: mannitol, sodium starch glycolate, hydroxypropyl cellulose, magnesium stearate, hypromellose, titanium dioxide, ferric oxide red, and carnauba wax. Manufactured for: Sumitomo Pharma America, Inc., Marlborough, MA 01752 For more information, go to www.orgovyx.com or call 1-833-696-8268. |
214621-MS-005
Section 44425-7 (44425-7)
Storage and Handling
- Store ORGOVYX at room temperature. Do not store above 30°C (86°F).
- Dispense to patients in original container only.
- For bottles, keep container tightly closed after first opening.
- Keep out of reach of children.
11 Description (11 DESCRIPTION)
Relugolix is a nonpeptide small molecule, GnRH receptor antagonist. The chemical name is N-(4-{1-[(2,6-difluorophenyl)methyl]-5-[(dimethylamino)methyl]-3-(6-methoxypyridazin-3-yl)-2,4-dioxo-1,2,3,4-tetrahydrothieno[2,3-d]pyrimidin-6-yl}phenyl)-N'-methoxyurea.
The molecular weight is 623.63 daltons and the molecular formula is C29H27F2N7O5S. The structural formula is:
Relugolix is a white to off-white to slightly yellow solid with a solubility of 0.04 mg per mL in water at 25°C.
ORGOVYX is provided as film-coated tablets for oral administration. Each tablet contains 120 mg of relugolix. The inactive ingredients are mannitol, sodium starch glycolate, hydroxypropyl cellulose, magnesium stearate, hypromellose, titanium dioxide, ferric oxide red, and carnauba wax.
8.4 Pediatric Use
The safety and efficacy of ORGOVYX in pediatric patients have not been established.
8.5 Geriatric Use
Of the 622 patients who received ORGOVYX in the HERO study, 81% were 65 years of age or older, while 35% were 75 years of age or older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. There was no clinically relevant impact of age on the pharmacokinetics of ORGOVYX or testosterone response based on population pharmacokinetic and pharmacokinetic/pharmacodynamic analyses in men 45 to 91 years of age.
4 Contraindications (4 CONTRAINDICATIONS)
ORGOVYX is contraindicated in patients with severe hypersensitivity to relugolix or to any of the product components.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- QT/QTc Interval Prolongation [see Warnings and Precautions (5.1)].
7 Drug Interactions (7 DRUG INTERACTIONS)
P-gp Inhibitors: Avoid co-administration. If unavoidable, take ORGOVYX first, separate dosing by at least 6 hours, and monitor patients more frequently for adverse reactions (2.2, 7.1).
Combined P-gp and Strong CYP3A Inducers: Avoid co-administration. If unavoidable, increase the ORGOVYX dose to 240 mg once daily (2.3, 7.1).
12.3 Pharmacokinetics
After administration of single doses ranging from 60 mg to 360 mg (0.17 to 1 times the recommended loading dose), total systemic exposure (AUC) and the maximum concentration (Cmax) of relugolix increases in an approximately dose proportional manner. After administration of multiple doses of relugolix once daily, the AUC of relugolix increases in an approximately dose proportional manner while the Cmax increase is greater than dose proportional for doses from 20 mg to 180 mg (0.17 to 1.5 times the recommended daily dose).
After administration of a single 360 mg loading dose, the mean (± standard deviation [± SD]) AUC and Cmax of relugolix are 985 (± 742) ng.hr/mL and 215 (± 184) ng/mL, respectively. After administration of 120 mg once daily, the mean (± SD) AUC and Cmax of relugolix at steady-state are 407 (± 168) ng.hr/mL and 70 (± 65) ng/mL, respectively. The accumulation of relugolix upon once daily administration is approximately 2-fold.
2.1 Recommended Dosage
Initiate treatment of ORGOVYX with a loading dose of 360 mg on the first day and continue treatment with a 120 mg dose taken orally once daily at approximately the same time each day.
ORGOVYX can be taken with or without food [see Clinical Pharmacology (12.3)]. Instruct patients to swallow tablets whole and not to crush or chew tablets.
Advise patients to take a missed dose of ORGOVYX as soon as they remember. If the dose was missed by more than 12 hours, patients should not take the missed dose and resume with the next scheduled dose.
If treatment with ORGOVYX is interrupted for greater than 7 days, restart ORGOVYX with a loading dose of 360 mg on the first day, and continue with a dose of 120 mg once daily.
In patients treated with GnRH receptor agonists and antagonists for prostate cancer, treatment is usually continued upon development of nonmetastatic or metastatic castration-resistant prostate cancer.
5.4 Laboratory Testing
Therapy with ORGOVYX results in suppression of the pituitary gonadal system. Results of diagnostic tests of the pituitary gonadotropic and gonadal functions conducted during and after ORGOVYX may be affected. The therapeutic effect of ORGOVYX should be monitored by measuring serum concentrations of prostate specific antigen (PSA) periodically. If PSA increases, serum concentrations of testosterone should be measured.
1 Indications and Usage (1 INDICATIONS AND USAGE)
ORGOVYX is indicated for the treatment of adult patients with advanced prostate cancer.
12.1 Mechanism of Action
Relugolix is a nonpeptide GnRH receptor antagonist that competitively binds to pituitary GnRH receptors, thereby, reducing the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH), and consequently testosterone.
5.3 Embryo Fetal Toxicity (5.3 Embryo-Fetal Toxicity)
The safety and efficacy of ORGOVYX have not been established in females. Based on findings in animals and mechanism of action, ORGOVYX can cause fetal harm and loss of pregnancy when administered to a pregnant female. In an animal reproduction study, oral administration of relugolix to pregnant rabbits during the period of organogenesis caused embryo-fetal lethality at maternal exposures that were 0.3 times the human exposure at the recommended dose of 120 mg daily based on area under the curve (AUC). Advise males with female partners of reproductive potential to use effective contraception during treatment and for 2 weeks after the last dose of ORGOVYX [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- QT/QTc Interval Prolongation: Androgen deprivation therapy may prolong the QT interval (5.1).
- Hypersensitivity: ORGOVYX can cause hypersensitivity reactions, including angioedema. Withhold ORGOVYX in patients who experience symptoms of hypersensitivity. Discontinue ORGOVYX for severe hypersensitivity reactions and manage as clinically indicated (5.2).
- Embryo-Fetal Toxicity: ORGOVYX can cause fetal harm. Advise males with female partners of reproductive potential to use effective contraception (5.3, 8.1, 8.3).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Recommended Dosage: A loading dose of 360 mg on the first day of treatment followed by 120 mg taken orally once daily, at approximately the same time each day (2.1).
- ORGOVYX can be taken with or without food (2.1, 12.3). Instruct patients to swallow tablets whole and not to crush or chew tablets (2.1).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 120 mg, light red, almond-shaped, film-coated, and debossed with “R” on one side and “120” on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ORGOVYX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: hypersensitivity, including angioedema and urticaria.
5.2 Hypersensitivity Reactions
ORGOVYX is contraindicated in patients with severe hypersensitivity to relugolix or any of the product components [see Contraindications (4)]. Hypersensitivity reactions, including pharyngeal edema and other serious cases of angioedema, have been reported postmarketing in patients treated with ORGOVYX.
In HERO, patients treated with relugolix reported angioedema (0.2%) [see Clinical Trials Experience (6.1)].
Advise patients who experience any symptoms of hypersensitivity to temporarily discontinue ORGOVYX and promptly seek medical care.
Discontinue ORGOVYX for severe hypersensitivity reactions and manage as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ORGOVYX was evaluated in HERO, a randomized (2:1), open-label, clinical study in patients with advanced prostate cancer [see Clinical Studies (14)]. Patients received orally administered ORGOVYX as a loading dose of 360 mg on the first day followed by 120 mg taken orally once daily (n = 622) or received leuprolide acetate administered by depot injection at doses of 22.5 mg (n = 264) or 11.25 mg (n = 44) per local guidelines every 12 weeks (n = 308). Leuprolide acetate 11.25 mg is a dosing regimen that is not recommended for this indication in the US. Among patients who received ORGOVYX, 91% were exposed for at least 48 weeks. Ninety-nine (16%) patients received concomitant radiotherapy and 17 (3%) patients received concomitant enzalutamide with ORGOVYX.
Serious adverse reactions occurred in 12% of patients receiving ORGOVYX. Serious adverse reactions in ≥ 0.5% of patients included myocardial infarction (0.8%), acute kidney injury (0.6%), arrhythmia (0.6%), hemorrhage (0.6%), and urinary tract infection (0.5%). Fatal adverse reactions occurred in 0.8% of patients receiving ORGOVYX including metastatic lung cancer (0.3%), myocardial infarction (0.3%), and acute kidney injury (0.2%). Fatal and non-fatal myocardial infarction and stroke were reported in 2.7% of patients receiving ORGOVYX.
Permanent discontinuation of ORGOVYX due to an adverse reaction occurred in 3.5% of patients. Adverse reactions which resulted in permanent discontinuation of ORGOVYX in ≥ 0.3 % of patients included atrioventricular block (0.3%), cardiac failure (0.3%), hemorrhage (0.3%), increased transaminases (0.3%), abdominal pain (0.3%), and pneumonia (0.3%).
Dosage interruptions of ORGOVYX due to an adverse reaction occurred in 2.7% of patients. Adverse reactions which required dosage interruption in ≥ 0.3% of patients included fracture (0.3%).
The most common adverse reactions (≥ 10%) and laboratory abnormalities (≥ 15%) were hot flush (54%), glucose increased (44%), triglycerides increased (35%), musculoskeletal pain (30%), hemoglobin decreased (28%), alanine aminotransferase increased (ALT) (27%), fatigue (26%), aspartate aminotransferase increased (AST) (18%), constipation (12%), and diarrhea (12%).
Table 1 summarizes the adverse reactions in HERO.
|
a Includes arthralgia, back pain, pain in extremity, musculoskeletal pain, myalgia, bone pain, neck pain, arthritis, musculoskeletal stiffness, non-cardiac chest pain, musculoskeletal chest pain, spinal pain, and musculoskeletal discomfort. |
||||
|
b Includes fatigue and asthenia. |
||||
|
c Includes diarrhea and colitis. |
||||
| Adverse Reaction |
ORGOVYX N = 622 |
Leuprolide Acetate
N = 308 |
||
|
All Grades
(%) |
Grade 3-4
(%) |
All Grades
(%) |
Grade 3-4
(%) |
|
| Vascular disorders | ||||
| Hot flush | 54 | 0.6 | 52 | 0 |
| Musculoskeletal and connective tissue disorders | ||||
| Musculoskeletal paina | 30 | 1.1 | 29 | 1.6 |
| General | ||||
| Fatigueb | 26 | 0.3 | 24 | 0 |
| Gastrointestinal disorders | ||||
| Diarrheac | 12 | 0.2 | 7 | 0 |
| Constipation | 12 | 0 | 10 | 0 |
Clinically relevant adverse reactions in < 10% of patients who received ORGOVYX included increased weight, insomnia, gynecomastia, hyperhidrosis, depression, decreased libido, and angioedema.
Table 2 summarizes the laboratory abnormalities in HERO.
| Laboratory Test | ORGOVYX a | Leuprolide Acetate a | ||
|---|---|---|---|---|
|
All Grades
(%) |
Grade 3-4
(%) |
All Grades
(%) |
Grade 3-4
(%) |
|
|
a The denominator used to calculate the rate varied from 611 to 619 in the ORGOVYX arm and from 301 to 306 in the leuprolide arm based on the number of patients with a baseline value and at least one post-treatment value. |
||||
| Chemistry | ||||
| Glucose increased | 44 | 2.9 | 54 | 6 |
| Triglycerides increased | 35 | 2 | 36 | 0.7 |
| ALT increased | 27 | 0.3 | 28 | 0 |
| AST increased | 18 | 0 | 19 | 0.3 |
| Hematology | ||||
| Hemoglobin decreased | 28 | 0.5 | 29 | 0.7 |
5.1 Qt/qtc Interval Prolongation (5.1 QT/QTc Interval Prolongation)
Androgen deprivation therapy, such as ORGOVYX, may prolong the QT/QTc interval. Providers should consider whether the benefits of androgen deprivation therapy outweigh the potential risks in patients with congenital long QT syndrome, congestive heart failure, or frequent electrolyte abnormalities and in patients taking drugs known to prolong the QT interval. Electrolyte abnormalities should be corrected. Consider periodic monitoring of electrocardiograms and electrolytes [see Clinical Pharmacology (12.2)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Phospholipidosis (intracellular phospholipid accumulation) was observed in multiple organs and tissues (e.g., liver, pancreas, spleen, kidney, lymph nodes, lung, bone marrow, gastrointestinal tract or testes) after repeated oral administration of relugolix in rats and monkeys. In a rat 26-week toxicity study, phospholipidosis was observed at doses ≥ 100 mg/kg (approximately 18 times the human exposure at the recommended dose based on AUC). In a monkey 39-week toxicity study, this effect was observed at doses ≥ 1.5 mg/kg (approximately 0.6 times the human exposure at the recommended dose based on AUC) and demonstrated evidence of reversibility after cessation of treatment. The significance of this finding in humans is unknown.
2.2 Dosage Modifications for P Gp Inhibitors (2.2 Dosage Modifications for P-gp Inhibitors)
Avoid co-administration of ORGOVYX with oral P-gp inhibitors. If co-administration is unavoidable, take ORGOVYX first and separate dosing by at least 6 hours [ see Drug Interactions (7.1) and Clinical Pharmacology (12.3) ]. Monitor patients for increased adverse reactions.
Treatment with ORGOVYX may be interrupted for up to two weeks if a short course of treatment with a P-gp inhibitor is required. Resume ORGOVYX after the P-gp inhibitor is discontinued. If treatment with ORGOVYX is interrupted for greater than 7 days, restart ORGOVYX with a loading dose of 360 mg on the first day and continue with a dose of 120 mg once daily.
Principal Display Panel 120 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 120 mg Tablet Bottle Label)
NDC 72974-120-01
Rx only
ORGOVYX®
(relugolix) tablets
120 mg
30 tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenicity studies were conducted in mice at oral relugolix doses up to 100 mg/kg/day and in rats at doses up to 600 mg/kg/day. Relugolix was not carcinogenic in mice or rats at exposures up to approximately 75 or 224 times, respectively, the human exposure at the recommended dose of 120 mg daily based on AUC.
Relugolix was not mutagenic in the in vitro bacterial reverse mutation (Ames) assay or clastogenic in the in vitro chromosomal aberration assay in Chinese hamster lung cells or the in vivo rat bone marrow micronucleus assay.
In human GnRH-receptor knock-in male mice, oral administration of relugolix decreased prostate and seminal vesicle weights at doses ≥ 3 mg/kg twice daily for 28 days. The effects of relugolix were reversible, except for testis weight, which did not fully recover within 28 days after drug withdrawal. In a 39-week repeat-dose toxicity study in monkeys, there were no significant effects on male reproductive organs at oral relugolix doses up to 50 mg/kg/day (approximately 53 times the human exposure at the recommended dose of 120 mg daily based on AUC).
2.3 Dosage Modifications for Combined P Gp and Strong Cyp3a Inducers (2.3 Dosage Modifications for Combined P-gp and Strong CYP3A Inducers)
Avoid co-administration of ORGOVYX with combined P-gp and strong CYP3A inducers. If co-administration is unavoidable, increase the ORGOVYX dose to 240 mg once daily. After discontinuation of the combined P-gp and strong CYP3A inducer, resume the recommended ORGOVYX dose of 120 mg once daily [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:32.412701 · Updated: 2026-03-14T22:48:00.481820