Drug Facts
06328741-03e3-49cf-ae44-d4c2116e38a3
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dextromethorphan HBr 60 mg Guaifenesin 1200 mg
Purpose
Cough suppressant Expectorant
Medication Information
Purpose
Cough suppressant Expectorant
Description
Dextromethorphan HBr 60 mg Guaifenesin 1200 mg
Uses
-
•helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
-
•temporarily relieves:
-
•cough due to minor throat and bronchial irritation as may occur with the common cold or inhaled irritants
-
•the intensity of coughing
-
•the impulse to cough to help you get to sleep
Purposes
Cough suppressant
Expectorant
Warnings
Do not use
-
•for children under 12 years of age
-
•if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Directions
-
•do not crush, chew, or break extended-release tablet
-
•take with a full glass of water
-
•this product can be administered without regard for timing of meals
-
•adults and children 12 years and older: 1 extended-release tablet every 12 hours; not more than 2 extended-release tablets in 24 hours
-
•children under 12 years of age: do not use
Other Information
-
•Store between 20-25°C (68-77°F)
Inactive Ingredients
carbomer homopolymer, colloidal silicon dioxide, hypromellose, magnesium stearate, maltodextrin, microcrystalline cellulose, povidone (K-30), stearic acid
When Using This Product
-
•Do not use more than directed
Pregnancy Or Breast Feeding
If pregnant or breast-feeding, ask a health professional before use.
Stop Use and Ask A Doctor If
-
•cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
Keep Out of Reach of Children
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Before Use If You Have
-
•persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
-
•cough accompanied by too much phlegm (mucus)
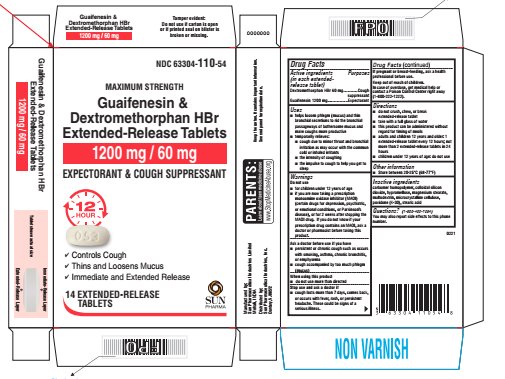
Package/label Principal Display Panel
NDC 63304-110-14
Maximum Strength
Guaifenesin & Dextromethorphan HBr Extended-Release Tablets
1200 mg/60 mg
Expectorant & Cough Suppressant
12 Hour
• Controls Cough
• Thins and Loosens Mucus
• Immediate and Extended Release
14 Extended-Release Tablets
SUN PHARMA
Active Ingredients (in Each Extended Release Tablet)
Dextromethorphan HBr 60 mg
Guaifenesin 1200 mg
Structured Label Content
Uses
-
•helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
-
•temporarily relieves:
-
•cough due to minor throat and bronchial irritation as may occur with the common cold or inhaled irritants
-
•the intensity of coughing
-
•the impulse to cough to help you get to sleep
Purposes
Cough suppressant
Expectorant
Warnings
Do not use
-
•for children under 12 years of age
-
•if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Directions
-
•do not crush, chew, or break extended-release tablet
-
•take with a full glass of water
-
•this product can be administered without regard for timing of meals
-
•adults and children 12 years and older: 1 extended-release tablet every 12 hours; not more than 2 extended-release tablets in 24 hours
-
•children under 12 years of age: do not use
Other Information (Other information)
-
•Store between 20-25°C (68-77°F)
Inactive Ingredients (Inactive ingredients)
carbomer homopolymer, colloidal silicon dioxide, hypromellose, magnesium stearate, maltodextrin, microcrystalline cellulose, povidone (K-30), stearic acid
When Using This Product (When using this product)
-
•Do not use more than directed
Pregnancy Or Breast Feeding (Pregnancy or Breast Feeding)
If pregnant or breast-feeding, ask a health professional before use.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
-
•cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache. These could be signs of a serious illness.
Keep Out of Reach of Children
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
-
•persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
-
•cough accompanied by too much phlegm (mucus)
Package/label Principal Display Panel (Package/Label Principal Display Panel)
NDC 63304-110-14
Maximum Strength
Guaifenesin & Dextromethorphan HBr Extended-Release Tablets
1200 mg/60 mg
Expectorant & Cough Suppressant
12 Hour
• Controls Cough
• Thins and Loosens Mucus
• Immediate and Extended Release
14 Extended-Release Tablets
SUN PHARMA
Active Ingredients (in Each Extended Release Tablet) (Active ingredients (in each extended-release tablet))
Dextromethorphan HBr 60 mg
Guaifenesin 1200 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:24.684305 · Updated: 2026-03-14T23:00:49.702127