Metronidazole Gel Usp (topical), 0.75%
05707735-e676-412a-9a77-ebd003afb16c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
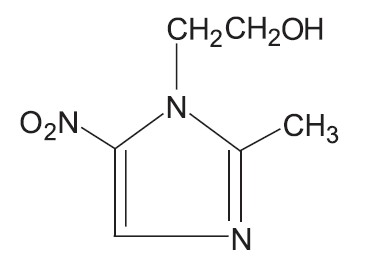
Metronidazole gel USP (topical), 0.75%, contains metronidazole, at a concentration of 7.5 mg per gram (0.75%) in a gel consisting of carbopol 980, edetate disodium, methylparaben, propylene glycol, propylparaben, purified water, and sodium hydroxide. Metronidazole is classified therapeutically as an antiprotozoal and anti-bacterial agent. Chemically, metronidazole is named 2-methyl-5-nitro-1H-imidazole-1- ethanol and has the following structure:
Indications and Usage
Metronidazole gel USP (topical), 0.75%, is indicated for topical application in the treatment of inflammatory papules and pustules of rosacea.
Dosage and Administration
Apply and rub in a thin film of metronidazole gel USP (topical), 0.75%, twice daily, morning and evening, to entire affected areas after washing. Areas to be treated should be cleansed before application of metronidazole gel USP (topical), 0.75%. Patients may use cosmetics after application of metronidazole gel USP (topical), 0.75%.
Contraindications
Metronidazole gel USP (topical), 0.75%, is contraindicated in individuals with a history of hypersensitivity lo metronidazole, parabens, or other ingredients of the formulation.
Adverse Reactions
The following adverse experiences have been reported with the topical use of metronidazole: burning, skin irritation, dryness, transient redness, metallic taste, tingling or numbness of extremities and nausea.
Drug Interactions
Oral metronidazole has been reported to potentiate the anticoagulant effect of coumarin and warfarin resulting in a prolongation of prothrombin time. The effect of topical metronidazole on prothrombin time is not known.
How Supplied
Metronidazole gel USP (topical), 0.75%, is supplied as the following: NDC 21922-070-06: 45 g aluminum lube Storage conditions: Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. To report SUSPECTED ADVERSE REACTIONS, contact Encube Ethicals Private Limited at 1-833-285 4151 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch Manufactured by: Encube Ethicals Pvt. Ltd. Plot No. C-1, Madkaim Industrial Estate, Madkaim, Post: Mardol, Panda, Goa - 403 404, India. Distributed by: Encube Ethicals, Inc. 200 Meredith Drive, Suite 202Durham, NC 27713 USA Revised: 05/2023
Medication Information
Indications and Usage
Metronidazole gel USP (topical), 0.75%, is indicated for topical application in the treatment of inflammatory papules and pustules of rosacea.
Dosage and Administration
Apply and rub in a thin film of metronidazole gel USP (topical), 0.75%, twice daily, morning and evening, to entire affected areas after washing. Areas to be treated should be cleansed before application of metronidazole gel USP (topical), 0.75%. Patients may use cosmetics after application of metronidazole gel USP (topical), 0.75%.
Contraindications
Metronidazole gel USP (topical), 0.75%, is contraindicated in individuals with a history of hypersensitivity lo metronidazole, parabens, or other ingredients of the formulation.
Adverse Reactions
The following adverse experiences have been reported with the topical use of metronidazole: burning, skin irritation, dryness, transient redness, metallic taste, tingling or numbness of extremities and nausea.
Drug Interactions
Oral metronidazole has been reported to potentiate the anticoagulant effect of coumarin and warfarin resulting in a prolongation of prothrombin time. The effect of topical metronidazole on prothrombin time is not known.
How Supplied
Metronidazole gel USP (topical), 0.75%, is supplied as the following:
NDC 21922-070-06: 45 g aluminum lube
Storage conditions: Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
To report SUSPECTED ADVERSE REACTIONS, contact Encube Ethicals Private Limited at 1-833-285 4151 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Manufactured by:
Encube Ethicals Pvt. Ltd.
Plot No. C-1, Madkaim Industrial Estate,
Madkaim, Post: Mardol, Panda,
Goa - 403 404, India.
Distributed by:
Encube Ethicals, Inc.
200 Meredith Drive,
Suite 202Durham,
NC 27713 USA
Revised: 05/2023
Description
Metronidazole gel USP (topical), 0.75%, contains metronidazole, at a concentration of 7.5 mg per gram (0.75%) in a gel consisting of carbopol 980, edetate disodium, methylparaben, propylene glycol, propylparaben, purified water, and sodium hydroxide. Metronidazole is classified therapeutically as an antiprotozoal and anti-bacterial agent. Chemically, metronidazole is named 2-methyl-5-nitro-1H-imidazole-1- ethanol and has the following structure:
General
Metronidazole gel USP (topical), 0.75%, has been reported to cause tearing of the eyes. Therefore, contact with the eyes should be avoided. If a reaction suggesting local irritation occurs, patients should be directed to use the medication less frequently or discontinue use. Metronidazole is a nitroimidazole and should be used with care in patients with evidence of, or history of blood dyscrasia.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Nursing Mothers
After oral administration, metronidazole is secreted in breast milk in concentrations similar to those found in the plasma. Even though metronidazole gel USP (topical), 0.75%, blood levels are significantly lower than those achieved after oral metronidazole, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Teratogenic Effects
Teratogenic effects: Pregnancy category B: There has been no experience to date with the use of metronidazole gel USP (topical), 0.75%, in pregnant patients. Metronidazole crosses the placental barrier and enters the fetal circulation rapidly. No fetotoxicity was observed after oral metronidazole in rats or mice. However, because animal reproduction studies are not always predictive of human response and since oral metronidazole has been shown to be a carcinogen in some rodents, this drug should be used during pregnancy only if clearly needed.
Clinical Pharmacology
Bioavailability studies on the topical administration of 1 gram of metronidazole gel USP (topical), 0.75%, (7.5 mg of metronidazole) to the face of 10 rosacea patients showed a maximum serum concentration of 66 nanograms per milliliter in one patient. This concentration is approximately 100 times less than concentrations afforded by a single 250 mg oral tablet. The serum metronidazole concentrations were below the detectable limits of the assay at the majority of time points in all patients. Three of the patients had no detectable serum concentrations of metronidazole at any time point. The mean dose of gel applied during clinical studies was 600 mg which represents 4.5 mg of metronidazole per application. Therefore, under normal usage levels, the formulation affords minimal serum concentrations of metronidazole. The mechanisms by which metronidazole gel USP (topical), 0.75%, acts in the treatment of rosacea are unknown, but appear to include an anti-inflammatory effect.
Information for Patients
This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
Package Label.principal Display Panel
Carton Label
NDC 21922-070-06
Metronidazole Gel USP (Topical), 0.75%
FOR TOPICAL USE ONLY. NOT FOR OPHTHALMIC USE.
Keep this and all medications out of the reach of children.
NET WT.45g
Rx only
Carcinogenesis, Mutagenesis, Impairment of Fertility
Metronidazole has shown evidence of carcinogenic activity in a number of studies involving chronic, oral administration in mice and rats but not in studies involving hamsters.
Metronidazole has shown evidence of mutagenic activity in several in vitro bacterial assay systems. In addition, a dose-response increase in the frequency of micronuclei was observed in mice after intraperitoneal injections and an increase in chromosome aberrations have been reported in patients with Crohn's disease who were treated with 200-1200 mg/day of metronidazole for 1 to 24 months. However, no excess chromosomal aberrations in circulating human lymphocytes have been observed in patients treated for 8 months.
Structured Label Content
General
Metronidazole gel USP (topical), 0.75%, has been reported to cause tearing of the eyes. Therefore, contact with the eyes should be avoided. If a reaction suggesting local irritation occurs, patients should be directed to use the medication less frequently or discontinue use. Metronidazole is a nitroimidazole and should be used with care in patients with evidence of, or history of blood dyscrasia.
Description (DESCRIPTION)
Metronidazole gel USP (topical), 0.75%, contains metronidazole, at a concentration of 7.5 mg per gram (0.75%) in a gel consisting of carbopol 980, edetate disodium, methylparaben, propylene glycol, propylparaben, purified water, and sodium hydroxide. Metronidazole is classified therapeutically as an antiprotozoal and anti-bacterial agent. Chemically, metronidazole is named 2-methyl-5-nitro-1H-imidazole-1- ethanol and has the following structure:
How Supplied (HOW SUPPLIED)
Metronidazole gel USP (topical), 0.75%, is supplied as the following:
NDC 21922-070-06: 45 g aluminum lube
Storage conditions: Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
To report SUSPECTED ADVERSE REACTIONS, contact Encube Ethicals Private Limited at 1-833-285 4151 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
Manufactured by:
Encube Ethicals Pvt. Ltd.
Plot No. C-1, Madkaim Industrial Estate,
Madkaim, Post: Mardol, Panda,
Goa - 403 404, India.
Distributed by:
Encube Ethicals, Inc.
200 Meredith Drive,
Suite 202Durham,
NC 27713 USA
Revised: 05/2023
Pediatric Use (Pediatric use)
Safety and effectiveness in pediatric patients have not been established.
Nursing Mothers (Nursing mothers)
After oral administration, metronidazole is secreted in breast milk in concentrations similar to those found in the plasma. Even though metronidazole gel USP (topical), 0.75%, blood levels are significantly lower than those achieved after oral metronidazole, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Adverse Reactions (ADVERSE REACTIONS)
The following adverse experiences have been reported with the topical use of metronidazole: burning, skin irritation, dryness, transient redness, metallic taste, tingling or numbness of extremities and nausea.
Contraindications (CONTRAINDICATIONS)
Metronidazole gel USP (topical), 0.75%, is contraindicated in individuals with a history of hypersensitivity lo metronidazole, parabens, or other ingredients of the formulation.
Drug Interactions (Drug interactions)
Oral metronidazole has been reported to potentiate the anticoagulant effect of coumarin and warfarin resulting in a prolongation of prothrombin time. The effect of topical metronidazole on prothrombin time is not known.
Teratogenic Effects (Teratogenic effects)
Teratogenic effects: Pregnancy category B: There has been no experience to date with the use of metronidazole gel USP (topical), 0.75%, in pregnant patients. Metronidazole crosses the placental barrier and enters the fetal circulation rapidly. No fetotoxicity was observed after oral metronidazole in rats or mice. However, because animal reproduction studies are not always predictive of human response and since oral metronidazole has been shown to be a carcinogen in some rodents, this drug should be used during pregnancy only if clearly needed.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Bioavailability studies on the topical administration of 1 gram of metronidazole gel USP (topical), 0.75%, (7.5 mg of metronidazole) to the face of 10 rosacea patients showed a maximum serum concentration of 66 nanograms per milliliter in one patient. This concentration is approximately 100 times less than concentrations afforded by a single 250 mg oral tablet. The serum metronidazole concentrations were below the detectable limits of the assay at the majority of time points in all patients. Three of the patients had no detectable serum concentrations of metronidazole at any time point. The mean dose of gel applied during clinical studies was 600 mg which represents 4.5 mg of metronidazole per application. Therefore, under normal usage levels, the formulation affords minimal serum concentrations of metronidazole. The mechanisms by which metronidazole gel USP (topical), 0.75%, acts in the treatment of rosacea are unknown, but appear to include an anti-inflammatory effect.
Indications and Usage (INDICATIONS AND USAGE)
Metronidazole gel USP (topical), 0.75%, is indicated for topical application in the treatment of inflammatory papules and pustules of rosacea.
Information for Patients (Information for patients)
This medication is to be used as directed by the physician. It is for external use only. Avoid contact with the eyes.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Apply and rub in a thin film of metronidazole gel USP (topical), 0.75%, twice daily, morning and evening, to entire affected areas after washing. Areas to be treated should be cleansed before application of metronidazole gel USP (topical), 0.75%. Patients may use cosmetics after application of metronidazole gel USP (topical), 0.75%.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Carton Label
NDC 21922-070-06
Metronidazole Gel USP (Topical), 0.75%
FOR TOPICAL USE ONLY. NOT FOR OPHTHALMIC USE.
Keep this and all medications out of the reach of children.
NET WT.45g
Rx only
Carcinogenesis, Mutagenesis, Impairment of Fertility (Carcinogenesis, mutagenesis, impairment of fertility)
Metronidazole has shown evidence of carcinogenic activity in a number of studies involving chronic, oral administration in mice and rats but not in studies involving hamsters.
Metronidazole has shown evidence of mutagenic activity in several in vitro bacterial assay systems. In addition, a dose-response increase in the frequency of micronuclei was observed in mice after intraperitoneal injections and an increase in chromosome aberrations have been reported in patients with Crohn's disease who were treated with 200-1200 mg/day of metronidazole for 1 to 24 months. However, no excess chromosomal aberrations in circulating human lymphocytes have been observed in patients treated for 8 months.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:32.092383 · Updated: 2026-03-14T22:33:31.162945