drug facts
056ec3cf-edcf-4a9a-8cfa-7cf9c4a9a138
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Stool softener laxative
Description
Docusate Sodium USP, 100 mg
Medication Information
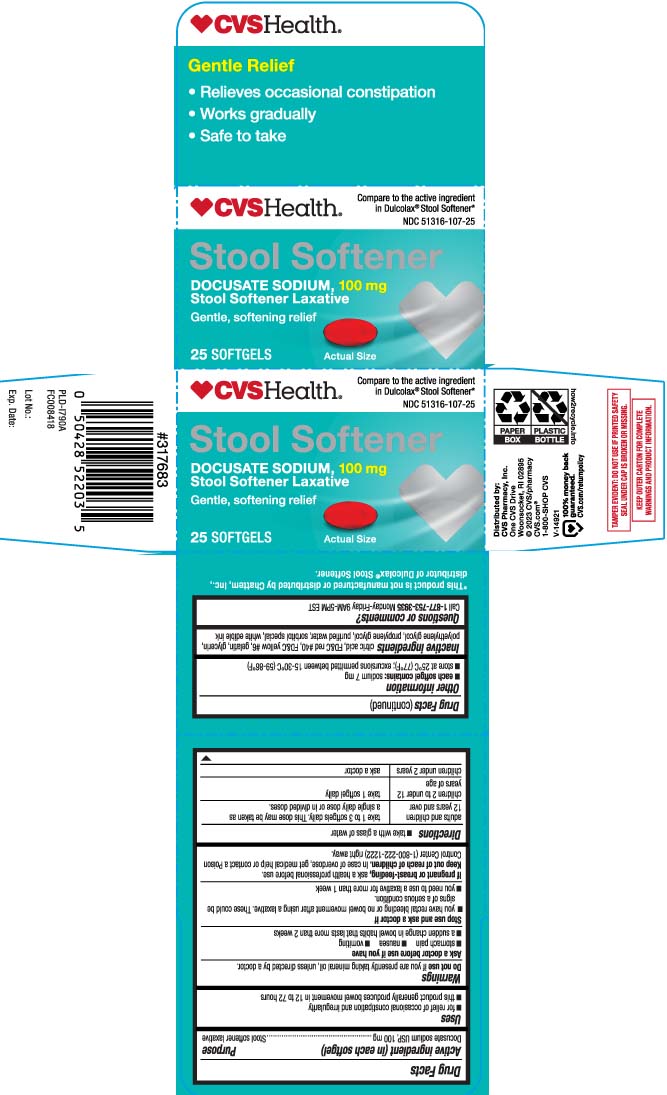
Uses
- for relief of occasional constipation and irregularity
- this product generally produces bowel movement in 12 to 72 hours
Purpose
Stool softener laxative
Directions
- take with a glass of water
| adults and children 12 years and over | take 1-3 softgels daily. This dose may be taken as a single daily dose or in divided doses |
| children 2 to under 12 years of age | take 1 softgel daily |
| children under 2 years | ask a doctor |
Other Information
- each softgel contains: sodium 7 mg
- store at 25ºC (77ºF); excursion permitted between 15-30ºC (59-86ºF)
Inactive Ingredients
citric acid, FD&C Red #40, FD&C Yellow #6, gelatin, glycerin, polyethylene glycol, propylene glycol, purified water, sorbitol special, white edible ink
Description
Docusate Sodium USP, 100 mg
Principal Display Panel
Compare to the active ingredient in Dulcolax® Stool Softener*
Stool Softener
DOCUSATE SODIUM 100 mg
Stool Softener Laxative
- Relief of occasional constipation
- Works gradually
- Safe to take
Gentle, softening relief
SOFTGELS
*This product is not manufactured or distributed by Chattem, Inc., distributor of Dulcolax® Stool Softener.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
Distributed by:
CVS Pharmacy, Inc.
One CVS Drive
Woonsocket, RI 02895
Do Not Use
if you are presently taking mineral oil, unless told to do so by a doctor.
Product Label
Questions Or Comments?
Call 1-877-753-3935 Monday-Friday 9AM-5PM EST
Ask A Doctor Before Use If
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that lasts over 2 weeks
Stop Use and Ask A Doctor If
- you have rectal bleeding or fail to have a bowel movement after using a laxative. These could be a sign of a serious condition.
- you need to use a stool softener laxative for more than 1 week
If Pregnant Or Breast Feeding,
ask a health care professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center (1-800-2222-1222) right away.
Active Ingredient (in Each Softgel)
Docusate Sodium USP, 100 mg
Structured Label Content
Uses
- for relief of occasional constipation and irregularity
- this product generally produces bowel movement in 12 to 72 hours
Purpose
Stool softener laxative
Directions
- take with a glass of water
| adults and children 12 years and over | take 1-3 softgels daily. This dose may be taken as a single daily dose or in divided doses |
| children 2 to under 12 years of age | take 1 softgel daily |
| children under 2 years | ask a doctor |
Other Information (Other information)
- each softgel contains: sodium 7 mg
- store at 25ºC (77ºF); excursion permitted between 15-30ºC (59-86ºF)
Inactive Ingredients (Inactive ingredients)
citric acid, FD&C Red #40, FD&C Yellow #6, gelatin, glycerin, polyethylene glycol, propylene glycol, purified water, sorbitol special, white edible ink
Principal Display Panel
Compare to the active ingredient in Dulcolax® Stool Softener*
Stool Softener
DOCUSATE SODIUM 100 mg
Stool Softener Laxative
- Relief of occasional constipation
- Works gradually
- Safe to take
Gentle, softening relief
SOFTGELS
*This product is not manufactured or distributed by Chattem, Inc., distributor of Dulcolax® Stool Softener.
TAMPER EVIDENT: DO NOT USE IF PRINTED SAFETY SEAL UNDER CAP IS BROKEN OR MISSING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
Distributed by:
CVS Pharmacy, Inc.
One CVS Drive
Woonsocket, RI 02895
Do Not Use (Do not use)
if you are presently taking mineral oil, unless told to do so by a doctor.
Product Label
Questions Or Comments? (Questions or comments?)
Call 1-877-753-3935 Monday-Friday 9AM-5PM EST
Ask A Doctor Before Use If (Ask a doctor before use if)
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that lasts over 2 weeks
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you have rectal bleeding or fail to have a bowel movement after using a laxative. These could be a sign of a serious condition.
- you need to use a stool softener laxative for more than 1 week
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health care professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center (1-800-2222-1222) right away.
Active Ingredient (in Each Softgel) (Active ingredient (in each softgel))
Docusate Sodium USP, 100 mg
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:51.823677 · Updated: 2026-03-14T23:02:39.250584