These Highlights Do Not Include All The Information Needed To Use Solosec®
056485b6-8bba-4a21-ae00-cdb4e7ae4bfa
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
SOLOSEC ® is a nitroimidazole antimicrobial indicated for: Treatment of bacterial vaginosis in female patients 12 years of age and older. ( 1.1 ) Treatment of trichomoniasis in patients 12 years of age and older. ( 1.2 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.3 )
Indications and Usage
SOLOSEC ® is a nitroimidazole antimicrobial indicated for: Treatment of bacterial vaginosis in female patients 12 years of age and older. ( 1.1 ) Treatment of trichomoniasis in patients 12 years of age and older. ( 1.2 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.3 )
Dosage and Administration
Bacterial Vaginosis (female patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. ( 2.1 ) Trichomoniasis (patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. Treat sexual partners with the same dose and at the same time. ( 2.2 ) Sprinkle entire contents of packet onto applesauce, yogurt or pudding and consume all of the mixture within 30 minutes without chewing or crunching the granules. A glass of water may be taken after the administration of SOLOSEC to aid in swallowing. ( 2.3 ) SOLOSEC is not intended to be dissolved in any liquid. ( 2.3 )
Warnings and Precautions
Vulvovaginal Candidiasis: This may develop with SOLOSEC and require treatment with an antifungal agent. ( 5.1 ) Potential Risk for Carcinogenicity : Carcinogenicity has been seen in mice and rats treated chronically with nitroimidazole derivatives, which are structurally related to secnidazole. It is unclear if the positive tumor findings in lifetime rodent studies indicate a risk to patients taking a single dose of SOLOSEC to treat bacterial vaginosis. Avoid chronic use. ( 5.2 )
Contraindications
SOLOSEC is contraindicated: In patients who have shown hypersensitivity to secnidazole, or other nitroimidazole derivatives. In patients with Cockayne syndrome: Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to secnidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2) ] .
Adverse Reactions
The following important adverse reactions are discussed in greater detail in other sections of labeling: Vulvovaginal Candidiasis [Warnings and Precautions (5.1)]
Storage and Handling
SOLOSEC (secnidazole) Oral Granules, 2 g, consists of off-white to slightly yellowish granules containing secnidazole. SOLOSEC is supplied in one unit-of-use child-resistant foil packet of granules in an individual carton. Each packet contains 4.8 g of granules containing 2 g secnidazole. SOLOSEC is supplied as follows: NDC 69751-400-01 carton containing one unit-of-use child-resistant 2 g foil packet.
How Supplied
SOLOSEC (secnidazole) Oral Granules, 2 g, consists of off-white to slightly yellowish granules containing secnidazole. SOLOSEC is supplied in one unit-of-use child-resistant foil packet of granules in an individual carton. Each packet contains 4.8 g of granules containing 2 g secnidazole. SOLOSEC is supplied as follows: NDC 69751-400-01 carton containing one unit-of-use child-resistant 2 g foil packet.
Medication Information
Warnings and Precautions
Vulvovaginal Candidiasis: This may develop with SOLOSEC and require treatment with an antifungal agent. ( 5.1 ) Potential Risk for Carcinogenicity : Carcinogenicity has been seen in mice and rats treated chronically with nitroimidazole derivatives, which are structurally related to secnidazole. It is unclear if the positive tumor findings in lifetime rodent studies indicate a risk to patients taking a single dose of SOLOSEC to treat bacterial vaginosis. Avoid chronic use. ( 5.2 )
Indications and Usage
SOLOSEC ® is a nitroimidazole antimicrobial indicated for: Treatment of bacterial vaginosis in female patients 12 years of age and older. ( 1.1 ) Treatment of trichomoniasis in patients 12 years of age and older. ( 1.2 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.3 )
Dosage and Administration
Bacterial Vaginosis (female patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. ( 2.1 ) Trichomoniasis (patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. Treat sexual partners with the same dose and at the same time. ( 2.2 ) Sprinkle entire contents of packet onto applesauce, yogurt or pudding and consume all of the mixture within 30 minutes without chewing or crunching the granules. A glass of water may be taken after the administration of SOLOSEC to aid in swallowing. ( 2.3 ) SOLOSEC is not intended to be dissolved in any liquid. ( 2.3 )
Contraindications
SOLOSEC is contraindicated: In patients who have shown hypersensitivity to secnidazole, or other nitroimidazole derivatives. In patients with Cockayne syndrome: Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to secnidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2) ] .
Adverse Reactions
The following important adverse reactions are discussed in greater detail in other sections of labeling: Vulvovaginal Candidiasis [Warnings and Precautions (5.1)]
Storage and Handling
SOLOSEC (secnidazole) Oral Granules, 2 g, consists of off-white to slightly yellowish granules containing secnidazole. SOLOSEC is supplied in one unit-of-use child-resistant foil packet of granules in an individual carton. Each packet contains 4.8 g of granules containing 2 g secnidazole. SOLOSEC is supplied as follows: NDC 69751-400-01 carton containing one unit-of-use child-resistant 2 g foil packet.
How Supplied
SOLOSEC (secnidazole) Oral Granules, 2 g, consists of off-white to slightly yellowish granules containing secnidazole. SOLOSEC is supplied in one unit-of-use child-resistant foil packet of granules in an individual carton. Each packet contains 4.8 g of granules containing 2 g secnidazole. SOLOSEC is supplied as follows: NDC 69751-400-01 carton containing one unit-of-use child-resistant 2 g foil packet.
Description
SOLOSEC ® is a nitroimidazole antimicrobial indicated for: Treatment of bacterial vaginosis in female patients 12 years of age and older. ( 1.1 ) Treatment of trichomoniasis in patients 12 years of age and older. ( 1.2 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. ( 1.3 )
Section 42229-5
Bacterial Vaginosis
The safety data described below reflect exposure to 629 patients, of whom 558 received a 2 g dose of SOLOSEC. SOLOSEC was evaluated in four clinical trials of female patients diagnosed with bacterial vaginosis: two placebo-controlled trials (Trial 1 n=215, Trial 2 n=189) and two uncontrolled safety trials (Trial 3 n=321, Trial 4 n=40).
Section 42230-3
|
PATIENT INFORMATION
SOLOSEC ® (SO-lo-sec) (secnidazole) oral granules |
||
|
What is SOLOSEC?
SOLOSEC is a prescription medicine used to treat:
|
||
Do not take SOLOSEC if you:
|
||
Before taking SOLOSEC, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take SOLOSEC?
|
||
|
What should I avoid while taking SOLOSEC?
Avoid drinking alcohol during treatment with SOLOSEC and for 2 days (48 hours) after you take SOLOSEC because the following side effects may happen: |
||
|
|
|
|
What are the possible side effects of SOLOSEC? SOLOSEC can cause side effects including vaginal yeast infections. Symptoms of a vaginal yeast infection include white or yellowish discharge (discharge may be lumpy or look like cottage cheese) and vaginal itching. The most common side effects of SOLOSEC include headache, nausea, vomiting, diarrhea, abdominal pain, vaginal itching and a bad, bitter or metallic taste in your mouth (dysgeusia). These are not all of the possible side effects of SOLOSEC. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about the safe and effective use of SOLOSEC.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SOLOSEC for a condition for which it was not prescribed. Do not give SOLOSEC to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about SOLOSEC that is written for health professionals. |
||
|
What are the ingredients in SOLOSEC?
Active ingredient: secnidazole Inactive ingredients: Eudragit NE30D (ethyl acrylate methyl methacrylate copolymer), polyethylene glycol 4000, povidone, sugar spheres, and talc. Contains no ingredient made from a gluten-containing grain (wheat, barley, or rye). For more information, visit www.solosec.com or contact Evofem at 1-833-EVFMBIO (1-833-383-6246). |
Section 44425-7
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room temperature].
1.3 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
7.2 Alcohol
Alcoholic beverages and preparations containing ethanol or propylene glycol should be avoided during SOLOSEC therapy and for 2 days after treatment is stopped.
Nausea, vomiting, diarrhea, abdominal pain, dizziness, and headache have been reported when SOLOSEC was taken concomitantly with alcohol [see Dosage and Administration (2.3), Adverse Reactions (6.2) and Clinical Pharmacology (12.3)].
15 References
- Özbilgin A, Özbel Y, Alkan MZ et al. Trichomoniasis in non-gonococcic urethritis among male patients. J Egypt Soc Parasitol. 1994; 24(3):621-625.
- Dyudyun AD, Polyon NM, Gorbuntsov VV. Secnidazole in complex treatment of patients with urogenital trichomoniasis. Dermatovenerology Cosmetology Sexopathology. 2016;1(4): 287-292.
- Siboulet A, Catalan F, Videau D, Niel G. Urogenital trichomoniasis. Trials with a long half-life imidazole: secnidazole. Med Mal Infect. 1977;7(9):400-409.
- Videau D, Niel G, Siboulet A, Catalan F. Secnidazole: A 5-nitroimidazole derivative with a long half-life. Br J Vener Dis. 1978;54(2):77-80.
- Krieger JN, Verdon M, Siegel N, Holmes KK. Natural history of urogenital trichomoniasis in men. J Urol. 1993 Jun;149(6):1455-8.
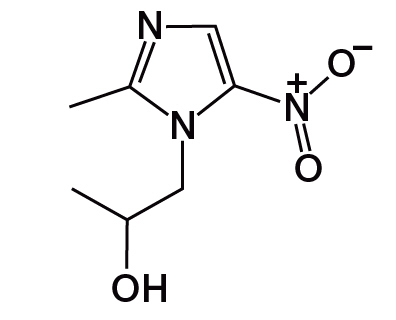
11 Description
The active ingredient in SOLOSEC Oral Granules is secnidazole (also named 1-(2-hydroxypropyl)-2-methyl-5-nitroimidazole and 1-(2-methyl-5-nitro-1H-imidazol-1-yl) propan-2-ol), a nitroimidazole antimicrobial.
The molecular formula of secnidazole is C7H11N3O3, the molecular weight is 185.18 and the chemical structure is:
| Figure 1: Structure of Secnidazole |
Each packet of SOLOSEC contains 4.8 g of off-white to slightly yellowish granules, which contain 2 g of secnidazole and the following inactive ingredients: Eudragit NE30D (ethyl acrylate methyl methacrylate copolymer), polyethylene glycol 4000, povidone, sugar spheres, and talc. Contains no ingredient made from a gluten-containing grain (wheat, barley, or rye).
8.4 Pediatric Use
The safety and effectiveness of SOLOSEC for the treatment of bacterial vaginosis have been established in pediatric patients aged 12 to 17 years old. Use of SOLOSEC in this age group is supported by evidence from a multicenter, open-label safety study in 40 pediatric female patients with bacterial vaginosis [see Adverse Reactions (6.1)] and evidence from adequate and well-controlled studies in adult women [see Clinical Studies (14.1)].
The safety and effectiveness of SOLOSEC for the treatment of trichomoniasis have been established in pediatric patients aged 12 to 17 years old. Use of SOLOSEC in this group is based on the extrapolation of clinical trial data from adult women with trichomoniasis, four open-label trials in males with trichomoniasis, and an open-label safety study in pediatric female patients with bacterial vaginosis [see Adverse Reactions (6.1) and Clinical Studies (14.2)].The safety and effectiveness of SOLOSEC in pediatric patients below the age of 12 years have not been established.
8.5 Geriatric Use
Clinical studies with secnidazole did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
1.2 Trichomoniasis
SOLOSEC is indicated for the treatment of trichomoniasis caused by Trichomonas vaginalis in patients 12 years of age and older. Because trichomoniasis is a sexually transmitted disease with potentially serious sequelae, treat partners of infected patients simultaneously in order to prevent reinfection [see Dosage and Administration (2.2) and Clinical Studies (14.2)].
14.2 Trichomoniasis
The efficacy of a single 2-gram oral dose of SOLOSEC for the treatment of trichomoniasis was evaluated in a multi-center, prospective, randomized, placebo-controlled, delayed treatment, double-blind, trial (Trial 5, NCT03935217). A total of 147 female patients from the United States aged 15 to 65 years were enrolled and randomized 1:1 to receive either SOLOSEC or placebo. The modified intent-to-treat (mITT) population included all randomized patients who were culture positive for T. vaginalis and negative for other sexually transmitted infections. Of the 131 female patients in the mITT population, the median age was 36 years, and 90.8% were African American. Baseline clinical symptoms of vaginal itching, discharge, or odor were reported in 111 (84.7%) patients. Following initial dosing, the test of cure (TOC) visit occurred 6 to 12 days later. At the TOC visit, patients received the opposite treatment (placebo patients received SOLOSEC and vice versa) with a return visit 7 to 12 days later.
Results for microbiological cure, defined as testing negative for T. vaginalis, for the mITT population are presented in Table 5. The microbiological cure rate at the TOC visit was significantly higher in the SOLOSEC treatment group compared to the placebo group.
| Endpoint | SOLOSEC 2 g n/N (%) N: number of patients in treatment group. For microbiological cure, included all subjects in the modified intent-to-treat (mITT) population, defined as all randomized patients who were culture positive for Trichomonas vaginalis and negative for other sexually transmitted infections.
|
Placebo n/N (%) , n placebo subjects with positive T. vaginalis culture at TOC, receiving delayed SOLOSEC treatment showed a comparable microbiological cure rate 7-12 days later (56/63 (88.9%), 95% CI: 78.4%, 95.4%) to the 92.2% cure rate for the initially SOLOSEC treated subjects.
|
Treatment Difference (95% CI) |
|---|---|---|---|
| CI = confidence interval; TOC = Test of Cure | |||
| Microbiological Cure InPouch™ TV test negative for T. vaginalis.
,
Subjects with no test results are assumed to be positive (1 SOLOSEC and 3 placebo subjects).
|
59/64 (92.2) | 1/67 (1.5) | 90.7 (80.7, 96.5) Exact CI from the Score Method.
,
P-value <0.001 versus placebo from a CMH test adjusted for clinical symptoms (present/absent) of trichomoniasis at baseline.
|
The single oral 2 g secnidazole dose was also assessed in four open-label trials in males (one comparative study with metronidazole and ornidazole in males only1 and three single-arm studies in males and females2,3,4). Parasitological evaluation was performed both pre- and post-treatment and reported cure rates ranged from 91.7% (165/180) to 100% (30/30) at time points ranging from 2 to 20 days (n=437, 211 males and 226 females). In addition, the natural history of trichomoniasis in men was evaluated in one study.5 The spontaneous resolution during a mean follow-up of 16 ± 12 days was noted in 36% (5/14) (95% CI: 12.8%, 64.9%) of untreated men.
6 Adverse Reactions
The following important adverse reactions are discussed in greater detail in other sections of labeling:
- Vulvovaginal Candidiasis [Warnings and Precautions (5.1)]
4. Contraindications
SOLOSEC is contraindicated:
- In patients who have shown hypersensitivity to secnidazole, or other nitroimidazole derivatives.
- In patients with Cockayne syndrome: Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to secnidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2)].
Instructions for Use
SOLOSEC ®
(SO-lo-sec)
(secnidazole) oral granules
For oral use (by mouth) only.
How to take SOLOSEC?
- Open the SOLOSEC packet by folding over the corner marked by an arrow (see diagram) and tearing across the top.
- Sprinkle the entire contents of the SOLOSEC packet onto applesauce, yogurt or pudding. The granules will not dissolve. Take within 30 minutes without chewing or crunching the granules.
- You can drink a glass of water after taking SOLOSEC to help with swallowing.
- You should not try to dissolve the medicine in water or any other liquid.
Important Information
- SOLOSEC may be taken before, after or during a meal.
- Take 1 entire packet of SOLOSEC at one time. Do not take only part of the medicine and save a portion for later.
- Avoid drinking alcohol during treatment with SOLOSEC and for 2 days (48 hours) after you take SOLOSEC.
How should I store SOLOSEC?
- Store SOLOSEC at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep SOLOSEC and all medicines out of the reach of children.
This Patient Information and Instructions for Use have been approved by the US Food and Drug Administration. Issued: 10/2024
Manufactured for Evofem, Inc., a wholly owned subsidiary of Evofem Biosciences, Inc.
San Diego, CA 92130
© 2024 Evofem, Inc. All Rights Reserved.
PN-5055 R02
12.2 Pharmacodynamics
Secnidazole exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
A single oral dose of 2 g of SOLOSEC in healthy adult female subjects, following an overnight fast and admixed with (4 oz) of applesauce, resulted in a mean (SD) secnidazole peak plasma concentration (Cmax) of 45.4 (7.64) mcg/mL and mean (SD) systemic exposure (AUC0-inf) of 1331.6 (230.16) mcg∙hr/mL. Median (range) time to peak concentration (Tmax) was 4.0 (3.0-4.0) hours. Following administration of the 2 g dose, mean secnidazole plasma concentrations decreased to 22.1 mcg/mL at 24 hours, 9.2 mcg/mL at 48 hours, 3.8 mcg/mL at 72 hours, and 1.4 mcg/mL at 96 hours.
1 Indications and Usage
SOLOSEC® is a nitroimidazole antimicrobial indicated for:
- Treatment of bacterial vaginosis in female patients 12 years of age and older. (1.1)
- Treatment of trichomoniasis in patients 12 years of age and older. (1.2)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.3)
1.1 Bacterial Vaginosis
SOLOSEC is indicated for the treatment of bacterial vaginosis in female patients 12 years of age and older [see Use in Specific Populations (8.1) and Clinical Studies (14)].
7.1 Oral Contraceptives
There was no clinically significant drug interaction between secnidazole and the combination oral contraceptive, ethinyl estradiol plus norethindrone [see Clinical Pharmacology (12.3)]. SOLOSEC can be co-administered with combination oral contraceptives (e.g., ethinyl estradiol plus norethindrone).
12.1 Mechanism of Action
SOLOSEC is a nitroimidazole antimicrobial drug [see Microbiology (12.4)].
14.1 Bacterial Vaginosis
Two randomized placebo-controlled clinical trials (Trial 1 and Trial 2) with similar designs were conducted to evaluate the efficacy of SOLOSEC 2 gram for the treatment of bacterial vaginosis. A diagnosis of bacterial vaginosis was defined as all of (a) the presence of an off-white (milky or gray), thin, homogeneous vaginal discharge; (b) a vaginal pH ≥ 4.7; (c) the presence of Clue cells ≥ 20% of the total epithelial cells on a microscopic examination of the vaginal saline wet mount; (d) a positive "whiff" test (detection of amine odor on addition of 10% KOH solution to a sample of the vaginal discharge); and (e) a Nugent score ≥ 4.
Trial 1 enrolled 144 non-pregnant female patients aged 19 to 54 years and Trial 2 enrolled 189 non-pregnant females aged 18 to 54 years. Black or African American subjects in both trials were 54%. Efficacy was assessed by clinical outcome evaluated 21 to 30 days following a single dose of SOLOSEC. A clinical responder was defined as "normal" vaginal discharge, negative "whiff" test, and clue cells <20%. Additional endpoints included Nugent score cure (Nugent score of 0-3) and therapeutic outcome. A therapeutic responder was defined as a clinical responder with a Nugent score cure. In Trial 2, the endpoints were also assessed at Day 7-14.
In both trials, a statistically significantly greater percentage of patients experienced clinical response, Nugent score cure, and therapeutic response at 21 to 30 days following a single dose of SOLOSEC compared to placebo. Statistically significant results for the endpoints were also achieved at Day 7-14 in Trial 2.
The percentage of patients with clinical response was also consistently higher in both trials in the SOLOSEC arm compared to placebo among all subsets of patients: number of prior episodes of bacterial vaginosis (≤ 3 episodes and ≥ 4 episodes) in past 12 months, baseline Nugent score (score 4-6 and score 7-10), and race (Black/African American and White). Tables 3 and 4 describe the efficacy of SOLOSEC in the treatment of bacterial vaginosis.
| Trial 1 | Trial 2 | |||
|---|---|---|---|---|
| SOLOSEC (N=62) N=number of patients in treatment group (modified intent-to-treat population defined as all patients randomized who had a baseline Nugent score ≥4 and were negative for other sexually transmitted infections at baseline).
n (%) |
Placebo (N=62) n (%) |
SOLOSEC (N=107) n (%) |
Placebo (N=57) n (%) |
|
|
Clinical Responder Patients missing one or more of the clinical assessments were considered as non-responders/not cured.
|
42 (67.7) | 11 (17.7) | 57 (53.3) | 11 (19.3) |
| 50.0 (33.4, 66.7) Difference in response (SOLOSEC – placebo) and 95% confidence interval
|
34.0 (18.7, 49.3) | |||
| p<0.001 | p<0.001 | |||
|
Nugent Score Cure Patients with missing Nugent scores were considered Nugent score failures.
|
25 (40.3) | 4 (6.5) | 47 (43.9) | 3 (5.3) |
| 33.8 (18.5, 49.1) | 38.6 (26.2, 51.0) | |||
| p<0.001 | p<0.001 | |||
| Therapeutic Responder | 25 (40.3) | 4 (6.5) | 37 (34.6) | 2 (3.5) |
| 33.8 (18.5, 49.1) | 31.1 (19.6, 42.6) | |||
| p<0.001 | p<0.001 |
| Trial 2 | ||
|---|---|---|
| SOLOSEC (N=107) N=number of patients in treatment group (modified intent-to-treat population defined as all patients randomized who had a baseline Nugent score ≥4 and were negative for other sexually transmitted infections at baseline).
n (%) |
Placebo (N=57) n (%) |
|
|
Clinical Responder
Patients missing one or more of the clinical assessments were considered as non-responders/not cured.
|
62 (57.9) | 14 (24.6) |
| 33.3 (17.4, 49.2) Difference in response (SOLOSEC – placebo) and 95% confidence interval
|
||
| p<0.001 | ||
|
Nugent Score Cure
Patients with missing Nugent scores were considered Nugent score failures.
|
49 (45.8) | 2 (3.5) |
| 42.3 (30.4, 54.2) | ||
| p<0.001 | ||
| Therapeutic Responder | 37 (34.6) | 2 (3.5) |
| 31.1 (19.6, 42.6) | ||
| p<0.001 |
5 Warnings and Precautions
- Vulvovaginal Candidiasis: This may develop with SOLOSEC and require treatment with an antifungal agent. (5.1)
- Potential Risk for Carcinogenicity: Carcinogenicity has been seen in mice and rats treated chronically with nitroimidazole derivatives, which are structurally related to secnidazole. It is unclear if the positive tumor findings in lifetime rodent studies indicate a risk to patients taking a single dose of SOLOSEC to treat bacterial vaginosis. Avoid chronic use. (5.2)
2 Dosage and Administration
- Bacterial Vaginosis (female patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. (2.1)
- Trichomoniasis (patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. Treat sexual partners with the same dose and at the same time. (2.2)
- Sprinkle entire contents of packet onto applesauce, yogurt or pudding and consume all of the mixture within 30 minutes without chewing or crunching the granules. A glass of water may be taken after the administration of SOLOSEC to aid in swallowing. (2.3)
- SOLOSEC is not intended to be dissolved in any liquid. (2.3)
5.1 Vulvovaginal Candidiasis
The use of SOLOSEC may result in vulvovaginal candidiasis. In controlled clinical trials of non-pregnant women with bacterial vaginosis, vulvovaginal candidiasis developed in 19/197 (9.6%) of patients who received 2 g SOLOSEC and 4/136 (2.9%) subjects who received placebo. In a controlled clinical trial of non-pregnant female patients with trichomoniasis, vulvovaginal candidiasis developed in 2/74 (2.7%) of patients who received 2 g SOLOSEC and 0/73 (0%) subjects who received placebo [see Adverse Reactions (6.1)]. Symptomatic vulvovaginal candidiasis may require treatment with an antifungal agent.
6.2 Postmarketing Experience
The following adverse reactions have been reported during use of SOLOSEC and other 2 g formulations of secnidazole outside of the United States. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
3. Dosage Forms and Strengths
Oral Granules: 2 g, of off-white to slightly yellowish granules with 4.8 g net weight, packed in a unit-of-use child-resistant foil packet.
8 Use in Specific Populations
Lactation: Breastfeeding is not recommended. Discontinue breastfeeding for 96 hours after administration of SOLOSEC. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied / Storage and Handling
SOLOSEC (secnidazole) Oral Granules, 2 g, consists of off-white to slightly yellowish granules containing secnidazole. SOLOSEC is supplied in one unit-of-use child-resistant foil packet of granules in an individual carton. Each packet contains 4.8 g of granules containing 2 g secnidazole. SOLOSEC is supplied as follows:
NDC 69751-400-01 carton containing one unit-of-use child-resistant 2 g foil packet.
5.2 Potential Risk for Carcinogenicity
Carcinogenicity has been seen in mice and rats treated chronically with nitroimidazole derivatives which are structurally related to secnidazole. It is unclear if the positive tumor findings in lifetime rodent studies of these nitroimidazoles indicate a risk to patients taking a single dose of SOLOSEC to treat bacterial vaginosis. Avoid chronic use of SOLOSEC [see Nonclinical Toxicology (13.1)]
2.2 Recommended Dosage for Trichomoniasis
The recommended dosage of SOLOSEC for the treatment of trichomoniasis in patients 12 years of age and older is a single 2-gram packet of granules taken once orally, without regard to the timing of meals [see Clinical Pharmacology (12.3)]. Since trichomoniasis is a sexually transmitted disease, treat sexual partners with the same dose and at the same time [see Indications and Usage (1.2)].
5.3 Risk of Development of Drug Resistance
Prescribing SOLOSEC in the absence of proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Principal Display Panel 2 G Packet Carton
NDC 69751-400-01
Rx Only
solosec® 2g
secnidazole
Oral Granules
1 Unit-of-Use Packet
EVOFEM
BIOSCIENCES®
2.1 Recommended Dosage for Bacterial Vaginosis
The recommended dosage of SOLOSEC for the treatment of bacterial vaginosis in female patients 12 years of age and older is a single 2-gram packet of granules taken once orally, without regard to the timing of meals [see Clinical Pharmacology (12.3)].
2.3 Instructions for the Preparation and Administration of Solosec
- Open the SOLOSEC packet by folding over the corner (marked by an arrow) and tearing across the top.
- Sprinkle the entire contents of the SOLOSEC packet onto applesauce, yogurt or pudding [see Clinical Pharmacology (12.3)]. The granules will not dissolve. Consume all of the mixture within 30 minutes without chewing or crunching the granules. A glass of water may be taken after the administration of SOLOSEC to aid in swallowing.
- The granules are not intended to be dissolved in any liquid.
- Avoid consumption of alcoholic beverages and preparations containing ethanol or propylene glycol during treatment with SOLOSEC and for at least 2 days after completing therapy [see Adverse Reactions (6.2), Drug Interactions (7.2), and Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Bacterial Vaginosis
The safety data described below reflect exposure to 629 patients, of whom 558 received a 2 g dose of SOLOSEC. SOLOSEC was evaluated in four clinical trials of female patients diagnosed with bacterial vaginosis: two placebo-controlled trials (Trial 1 n=215, Trial 2 n=189) and two uncontrolled safety trials (Trial 3 n=321, Trial 4 n=40).
Section 42230-3 (42230-3)
|
PATIENT INFORMATION
SOLOSEC ® (SO-lo-sec) (secnidazole) oral granules |
||
|
What is SOLOSEC?
SOLOSEC is a prescription medicine used to treat:
|
||
Do not take SOLOSEC if you:
|
||
Before taking SOLOSEC, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take SOLOSEC?
|
||
|
What should I avoid while taking SOLOSEC?
Avoid drinking alcohol during treatment with SOLOSEC and for 2 days (48 hours) after you take SOLOSEC because the following side effects may happen: |
||
|
|
|
|
What are the possible side effects of SOLOSEC? SOLOSEC can cause side effects including vaginal yeast infections. Symptoms of a vaginal yeast infection include white or yellowish discharge (discharge may be lumpy or look like cottage cheese) and vaginal itching. The most common side effects of SOLOSEC include headache, nausea, vomiting, diarrhea, abdominal pain, vaginal itching and a bad, bitter or metallic taste in your mouth (dysgeusia). These are not all of the possible side effects of SOLOSEC. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about the safe and effective use of SOLOSEC.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SOLOSEC for a condition for which it was not prescribed. Do not give SOLOSEC to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about SOLOSEC that is written for health professionals. |
||
|
What are the ingredients in SOLOSEC?
Active ingredient: secnidazole Inactive ingredients: Eudragit NE30D (ethyl acrylate methyl methacrylate copolymer), polyethylene glycol 4000, povidone, sugar spheres, and talc. Contains no ingredient made from a gluten-containing grain (wheat, barley, or rye). For more information, visit www.solosec.com or contact Evofem at 1-833-EVFMBIO (1-833-383-6246). |
Section 44425-7 (44425-7)
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room temperature].
1.3 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
7.2 Alcohol
Alcoholic beverages and preparations containing ethanol or propylene glycol should be avoided during SOLOSEC therapy and for 2 days after treatment is stopped.
Nausea, vomiting, diarrhea, abdominal pain, dizziness, and headache have been reported when SOLOSEC was taken concomitantly with alcohol [see Dosage and Administration (2.3), Adverse Reactions (6.2) and Clinical Pharmacology (12.3)].
15 References (15 REFERENCES)
- Özbilgin A, Özbel Y, Alkan MZ et al. Trichomoniasis in non-gonococcic urethritis among male patients. J Egypt Soc Parasitol. 1994; 24(3):621-625.
- Dyudyun AD, Polyon NM, Gorbuntsov VV. Secnidazole in complex treatment of patients with urogenital trichomoniasis. Dermatovenerology Cosmetology Sexopathology. 2016;1(4): 287-292.
- Siboulet A, Catalan F, Videau D, Niel G. Urogenital trichomoniasis. Trials with a long half-life imidazole: secnidazole. Med Mal Infect. 1977;7(9):400-409.
- Videau D, Niel G, Siboulet A, Catalan F. Secnidazole: A 5-nitroimidazole derivative with a long half-life. Br J Vener Dis. 1978;54(2):77-80.
- Krieger JN, Verdon M, Siegel N, Holmes KK. Natural history of urogenital trichomoniasis in men. J Urol. 1993 Jun;149(6):1455-8.
11 Description (11 DESCRIPTION)
The active ingredient in SOLOSEC Oral Granules is secnidazole (also named 1-(2-hydroxypropyl)-2-methyl-5-nitroimidazole and 1-(2-methyl-5-nitro-1H-imidazol-1-yl) propan-2-ol), a nitroimidazole antimicrobial.
The molecular formula of secnidazole is C7H11N3O3, the molecular weight is 185.18 and the chemical structure is:
| Figure 1: Structure of Secnidazole |
Each packet of SOLOSEC contains 4.8 g of off-white to slightly yellowish granules, which contain 2 g of secnidazole and the following inactive ingredients: Eudragit NE30D (ethyl acrylate methyl methacrylate copolymer), polyethylene glycol 4000, povidone, sugar spheres, and talc. Contains no ingredient made from a gluten-containing grain (wheat, barley, or rye).
8.4 Pediatric Use
The safety and effectiveness of SOLOSEC for the treatment of bacterial vaginosis have been established in pediatric patients aged 12 to 17 years old. Use of SOLOSEC in this age group is supported by evidence from a multicenter, open-label safety study in 40 pediatric female patients with bacterial vaginosis [see Adverse Reactions (6.1)] and evidence from adequate and well-controlled studies in adult women [see Clinical Studies (14.1)].
The safety and effectiveness of SOLOSEC for the treatment of trichomoniasis have been established in pediatric patients aged 12 to 17 years old. Use of SOLOSEC in this group is based on the extrapolation of clinical trial data from adult women with trichomoniasis, four open-label trials in males with trichomoniasis, and an open-label safety study in pediatric female patients with bacterial vaginosis [see Adverse Reactions (6.1) and Clinical Studies (14.2)].The safety and effectiveness of SOLOSEC in pediatric patients below the age of 12 years have not been established.
8.5 Geriatric Use
Clinical studies with secnidazole did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
1.2 Trichomoniasis
SOLOSEC is indicated for the treatment of trichomoniasis caused by Trichomonas vaginalis in patients 12 years of age and older. Because trichomoniasis is a sexually transmitted disease with potentially serious sequelae, treat partners of infected patients simultaneously in order to prevent reinfection [see Dosage and Administration (2.2) and Clinical Studies (14.2)].
14.2 Trichomoniasis
The efficacy of a single 2-gram oral dose of SOLOSEC for the treatment of trichomoniasis was evaluated in a multi-center, prospective, randomized, placebo-controlled, delayed treatment, double-blind, trial (Trial 5, NCT03935217). A total of 147 female patients from the United States aged 15 to 65 years were enrolled and randomized 1:1 to receive either SOLOSEC or placebo. The modified intent-to-treat (mITT) population included all randomized patients who were culture positive for T. vaginalis and negative for other sexually transmitted infections. Of the 131 female patients in the mITT population, the median age was 36 years, and 90.8% were African American. Baseline clinical symptoms of vaginal itching, discharge, or odor were reported in 111 (84.7%) patients. Following initial dosing, the test of cure (TOC) visit occurred 6 to 12 days later. At the TOC visit, patients received the opposite treatment (placebo patients received SOLOSEC and vice versa) with a return visit 7 to 12 days later.
Results for microbiological cure, defined as testing negative for T. vaginalis, for the mITT population are presented in Table 5. The microbiological cure rate at the TOC visit was significantly higher in the SOLOSEC treatment group compared to the placebo group.
| Endpoint | SOLOSEC 2 g n/N (%) N: number of patients in treatment group. For microbiological cure, included all subjects in the modified intent-to-treat (mITT) population, defined as all randomized patients who were culture positive for Trichomonas vaginalis and negative for other sexually transmitted infections.
|
Placebo n/N (%) , n placebo subjects with positive T. vaginalis culture at TOC, receiving delayed SOLOSEC treatment showed a comparable microbiological cure rate 7-12 days later (56/63 (88.9%), 95% CI: 78.4%, 95.4%) to the 92.2% cure rate for the initially SOLOSEC treated subjects.
|
Treatment Difference (95% CI) |
|---|---|---|---|
| CI = confidence interval; TOC = Test of Cure | |||
| Microbiological Cure InPouch™ TV test negative for T. vaginalis.
,
Subjects with no test results are assumed to be positive (1 SOLOSEC and 3 placebo subjects).
|
59/64 (92.2) | 1/67 (1.5) | 90.7 (80.7, 96.5) Exact CI from the Score Method.
,
P-value <0.001 versus placebo from a CMH test adjusted for clinical symptoms (present/absent) of trichomoniasis at baseline.
|
The single oral 2 g secnidazole dose was also assessed in four open-label trials in males (one comparative study with metronidazole and ornidazole in males only1 and three single-arm studies in males and females2,3,4). Parasitological evaluation was performed both pre- and post-treatment and reported cure rates ranged from 91.7% (165/180) to 100% (30/30) at time points ranging from 2 to 20 days (n=437, 211 males and 226 females). In addition, the natural history of trichomoniasis in men was evaluated in one study.5 The spontaneous resolution during a mean follow-up of 16 ± 12 days was noted in 36% (5/14) (95% CI: 12.8%, 64.9%) of untreated men.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are discussed in greater detail in other sections of labeling:
- Vulvovaginal Candidiasis [Warnings and Precautions (5.1)]
4. Contraindications (4. CONTRAINDICATIONS)
SOLOSEC is contraindicated:
- In patients who have shown hypersensitivity to secnidazole, or other nitroimidazole derivatives.
- In patients with Cockayne syndrome: Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole, another nitroimidazole drug, structurally related to secnidazole, in patients with Cockayne syndrome [see Adverse Reactions (6.2)].
Instructions for Use (INSTRUCTIONS FOR USE)
SOLOSEC ®
(SO-lo-sec)
(secnidazole) oral granules
For oral use (by mouth) only.
How to take SOLOSEC?
- Open the SOLOSEC packet by folding over the corner marked by an arrow (see diagram) and tearing across the top.
- Sprinkle the entire contents of the SOLOSEC packet onto applesauce, yogurt or pudding. The granules will not dissolve. Take within 30 minutes without chewing or crunching the granules.
- You can drink a glass of water after taking SOLOSEC to help with swallowing.
- You should not try to dissolve the medicine in water or any other liquid.
Important Information
- SOLOSEC may be taken before, after or during a meal.
- Take 1 entire packet of SOLOSEC at one time. Do not take only part of the medicine and save a portion for later.
- Avoid drinking alcohol during treatment with SOLOSEC and for 2 days (48 hours) after you take SOLOSEC.
How should I store SOLOSEC?
- Store SOLOSEC at room temperature between 68°F to 77°F (20°C to 25°C).
- Keep SOLOSEC and all medicines out of the reach of children.
This Patient Information and Instructions for Use have been approved by the US Food and Drug Administration. Issued: 10/2024
Manufactured for Evofem, Inc., a wholly owned subsidiary of Evofem Biosciences, Inc.
San Diego, CA 92130
© 2024 Evofem, Inc. All Rights Reserved.
PN-5055 R02
12.2 Pharmacodynamics
Secnidazole exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
A single oral dose of 2 g of SOLOSEC in healthy adult female subjects, following an overnight fast and admixed with (4 oz) of applesauce, resulted in a mean (SD) secnidazole peak plasma concentration (Cmax) of 45.4 (7.64) mcg/mL and mean (SD) systemic exposure (AUC0-inf) of 1331.6 (230.16) mcg∙hr/mL. Median (range) time to peak concentration (Tmax) was 4.0 (3.0-4.0) hours. Following administration of the 2 g dose, mean secnidazole plasma concentrations decreased to 22.1 mcg/mL at 24 hours, 9.2 mcg/mL at 48 hours, 3.8 mcg/mL at 72 hours, and 1.4 mcg/mL at 96 hours.
1 Indications and Usage (1 INDICATIONS AND USAGE)
SOLOSEC® is a nitroimidazole antimicrobial indicated for:
- Treatment of bacterial vaginosis in female patients 12 years of age and older. (1.1)
- Treatment of trichomoniasis in patients 12 years of age and older. (1.2)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of SOLOSEC and other antibacterial drugs, SOLOSEC should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. (1.3)
1.1 Bacterial Vaginosis
SOLOSEC is indicated for the treatment of bacterial vaginosis in female patients 12 years of age and older [see Use in Specific Populations (8.1) and Clinical Studies (14)].
7.1 Oral Contraceptives
There was no clinically significant drug interaction between secnidazole and the combination oral contraceptive, ethinyl estradiol plus norethindrone [see Clinical Pharmacology (12.3)]. SOLOSEC can be co-administered with combination oral contraceptives (e.g., ethinyl estradiol plus norethindrone).
12.1 Mechanism of Action
SOLOSEC is a nitroimidazole antimicrobial drug [see Microbiology (12.4)].
14.1 Bacterial Vaginosis
Two randomized placebo-controlled clinical trials (Trial 1 and Trial 2) with similar designs were conducted to evaluate the efficacy of SOLOSEC 2 gram for the treatment of bacterial vaginosis. A diagnosis of bacterial vaginosis was defined as all of (a) the presence of an off-white (milky or gray), thin, homogeneous vaginal discharge; (b) a vaginal pH ≥ 4.7; (c) the presence of Clue cells ≥ 20% of the total epithelial cells on a microscopic examination of the vaginal saline wet mount; (d) a positive "whiff" test (detection of amine odor on addition of 10% KOH solution to a sample of the vaginal discharge); and (e) a Nugent score ≥ 4.
Trial 1 enrolled 144 non-pregnant female patients aged 19 to 54 years and Trial 2 enrolled 189 non-pregnant females aged 18 to 54 years. Black or African American subjects in both trials were 54%. Efficacy was assessed by clinical outcome evaluated 21 to 30 days following a single dose of SOLOSEC. A clinical responder was defined as "normal" vaginal discharge, negative "whiff" test, and clue cells <20%. Additional endpoints included Nugent score cure (Nugent score of 0-3) and therapeutic outcome. A therapeutic responder was defined as a clinical responder with a Nugent score cure. In Trial 2, the endpoints were also assessed at Day 7-14.
In both trials, a statistically significantly greater percentage of patients experienced clinical response, Nugent score cure, and therapeutic response at 21 to 30 days following a single dose of SOLOSEC compared to placebo. Statistically significant results for the endpoints were also achieved at Day 7-14 in Trial 2.
The percentage of patients with clinical response was also consistently higher in both trials in the SOLOSEC arm compared to placebo among all subsets of patients: number of prior episodes of bacterial vaginosis (≤ 3 episodes and ≥ 4 episodes) in past 12 months, baseline Nugent score (score 4-6 and score 7-10), and race (Black/African American and White). Tables 3 and 4 describe the efficacy of SOLOSEC in the treatment of bacterial vaginosis.
| Trial 1 | Trial 2 | |||
|---|---|---|---|---|
| SOLOSEC (N=62) N=number of patients in treatment group (modified intent-to-treat population defined as all patients randomized who had a baseline Nugent score ≥4 and were negative for other sexually transmitted infections at baseline).
n (%) |
Placebo (N=62) n (%) |
SOLOSEC (N=107) n (%) |
Placebo (N=57) n (%) |
|
|
Clinical Responder Patients missing one or more of the clinical assessments were considered as non-responders/not cured.
|
42 (67.7) | 11 (17.7) | 57 (53.3) | 11 (19.3) |
| 50.0 (33.4, 66.7) Difference in response (SOLOSEC – placebo) and 95% confidence interval
|
34.0 (18.7, 49.3) | |||
| p<0.001 | p<0.001 | |||
|
Nugent Score Cure Patients with missing Nugent scores were considered Nugent score failures.
|
25 (40.3) | 4 (6.5) | 47 (43.9) | 3 (5.3) |
| 33.8 (18.5, 49.1) | 38.6 (26.2, 51.0) | |||
| p<0.001 | p<0.001 | |||
| Therapeutic Responder | 25 (40.3) | 4 (6.5) | 37 (34.6) | 2 (3.5) |
| 33.8 (18.5, 49.1) | 31.1 (19.6, 42.6) | |||
| p<0.001 | p<0.001 |
| Trial 2 | ||
|---|---|---|
| SOLOSEC (N=107) N=number of patients in treatment group (modified intent-to-treat population defined as all patients randomized who had a baseline Nugent score ≥4 and were negative for other sexually transmitted infections at baseline).
n (%) |
Placebo (N=57) n (%) |
|
|
Clinical Responder
Patients missing one or more of the clinical assessments were considered as non-responders/not cured.
|
62 (57.9) | 14 (24.6) |
| 33.3 (17.4, 49.2) Difference in response (SOLOSEC – placebo) and 95% confidence interval
|
||
| p<0.001 | ||
|
Nugent Score Cure
Patients with missing Nugent scores were considered Nugent score failures.
|
49 (45.8) | 2 (3.5) |
| 42.3 (30.4, 54.2) | ||
| p<0.001 | ||
| Therapeutic Responder | 37 (34.6) | 2 (3.5) |
| 31.1 (19.6, 42.6) | ||
| p<0.001 |
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Vulvovaginal Candidiasis: This may develop with SOLOSEC and require treatment with an antifungal agent. (5.1)
- Potential Risk for Carcinogenicity: Carcinogenicity has been seen in mice and rats treated chronically with nitroimidazole derivatives, which are structurally related to secnidazole. It is unclear if the positive tumor findings in lifetime rodent studies indicate a risk to patients taking a single dose of SOLOSEC to treat bacterial vaginosis. Avoid chronic use. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Bacterial Vaginosis (female patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. (2.1)
- Trichomoniasis (patients 12 years of age and older): Administer a single 2-gram packet of granules once orally, without regard to the timing of meals. Treat sexual partners with the same dose and at the same time. (2.2)
- Sprinkle entire contents of packet onto applesauce, yogurt or pudding and consume all of the mixture within 30 minutes without chewing or crunching the granules. A glass of water may be taken after the administration of SOLOSEC to aid in swallowing. (2.3)
- SOLOSEC is not intended to be dissolved in any liquid. (2.3)
5.1 Vulvovaginal Candidiasis
The use of SOLOSEC may result in vulvovaginal candidiasis. In controlled clinical trials of non-pregnant women with bacterial vaginosis, vulvovaginal candidiasis developed in 19/197 (9.6%) of patients who received 2 g SOLOSEC and 4/136 (2.9%) subjects who received placebo. In a controlled clinical trial of non-pregnant female patients with trichomoniasis, vulvovaginal candidiasis developed in 2/74 (2.7%) of patients who received 2 g SOLOSEC and 0/73 (0%) subjects who received placebo [see Adverse Reactions (6.1)]. Symptomatic vulvovaginal candidiasis may require treatment with an antifungal agent.
6.2 Postmarketing Experience
The following adverse reactions have been reported during use of SOLOSEC and other 2 g formulations of secnidazole outside of the United States. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
3. Dosage Forms and Strengths (3. DOSAGE FORMS AND STRENGTHS)
Oral Granules: 2 g, of off-white to slightly yellowish granules with 4.8 g net weight, packed in a unit-of-use child-resistant foil packet.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Breastfeeding is not recommended. Discontinue breastfeeding for 96 hours after administration of SOLOSEC. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
16 How Supplied / Storage and Handling (16 HOW SUPPLIED / STORAGE AND HANDLING)
SOLOSEC (secnidazole) Oral Granules, 2 g, consists of off-white to slightly yellowish granules containing secnidazole. SOLOSEC is supplied in one unit-of-use child-resistant foil packet of granules in an individual carton. Each packet contains 4.8 g of granules containing 2 g secnidazole. SOLOSEC is supplied as follows:
NDC 69751-400-01 carton containing one unit-of-use child-resistant 2 g foil packet.
5.2 Potential Risk for Carcinogenicity
Carcinogenicity has been seen in mice and rats treated chronically with nitroimidazole derivatives which are structurally related to secnidazole. It is unclear if the positive tumor findings in lifetime rodent studies of these nitroimidazoles indicate a risk to patients taking a single dose of SOLOSEC to treat bacterial vaginosis. Avoid chronic use of SOLOSEC [see Nonclinical Toxicology (13.1)]
2.2 Recommended Dosage for Trichomoniasis
The recommended dosage of SOLOSEC for the treatment of trichomoniasis in patients 12 years of age and older is a single 2-gram packet of granules taken once orally, without regard to the timing of meals [see Clinical Pharmacology (12.3)]. Since trichomoniasis is a sexually transmitted disease, treat sexual partners with the same dose and at the same time [see Indications and Usage (1.2)].
5.3 Risk of Development of Drug Resistance
Prescribing SOLOSEC in the absence of proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Principal Display Panel 2 G Packet Carton (PRINCIPAL DISPLAY PANEL - 2 g Packet Carton)
NDC 69751-400-01
Rx Only
solosec® 2g
secnidazole
Oral Granules
1 Unit-of-Use Packet
EVOFEM
BIOSCIENCES®
2.1 Recommended Dosage for Bacterial Vaginosis
The recommended dosage of SOLOSEC for the treatment of bacterial vaginosis in female patients 12 years of age and older is a single 2-gram packet of granules taken once orally, without regard to the timing of meals [see Clinical Pharmacology (12.3)].
2.3 Instructions for the Preparation and Administration of Solosec (2.3 Instructions for the Preparation and Administration of SOLOSEC)
- Open the SOLOSEC packet by folding over the corner (marked by an arrow) and tearing across the top.
- Sprinkle the entire contents of the SOLOSEC packet onto applesauce, yogurt or pudding [see Clinical Pharmacology (12.3)]. The granules will not dissolve. Consume all of the mixture within 30 minutes without chewing or crunching the granules. A glass of water may be taken after the administration of SOLOSEC to aid in swallowing.
- The granules are not intended to be dissolved in any liquid.
- Avoid consumption of alcoholic beverages and preparations containing ethanol or propylene glycol during treatment with SOLOSEC and for at least 2 days after completing therapy [see Adverse Reactions (6.2), Drug Interactions (7.2), and Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:45:48.351443 · Updated: 2026-03-14T22:23:22.827754