These Highlights Do Not Include All The Information Needed To Use Inmazeb Safely And Effectively. See Full Prescribing Information For Inmazeb.
0536b6fe-6fe5-4fd6-8cc6-1b481945c1fa
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
INMAZEB is indicated for the treatment of infection caused by Orthoebolavirus zairense in adult and pediatric patients, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection [see Dosage and Administration (2.2) , and Clinical Studies (14) ] .
Indications and Usage
INMAZEB is indicated for the treatment of infection caused by Orthoebolavirus zairense in adult and pediatric patients, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection [see Dosage and Administration (2.2) , and Clinical Studies (14) ] .
Dosage and Administration
The recommended dosage of INMAZEB is 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg diluted and administered as a single intravenous infusion. ( 2.1 ) Refer to the Full Prescribing Information for information on preparation and administration. ( 2.2 )
Warnings and Precautions
Hypersensitivity Reactions Including Infusion-Associated Events: Hypersensitivity reactions including infusion-associated events have been reported during and post-infusion with INMAZEB. These may include acute, life-threatening reactions during and after the infusion. Monitor patients and in the case of severe or life-threatening hypersensitivity reactions, discontinue the administration of INMAZEB immediately and administer appropriate emergency care. ( 5.1 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions Including Infusion-Associated Events [see Warnings and Precautions (5.1) ]
Drug Interactions
Interaction with live vaccine indicated for prevention of Orthoebolavirus zairense infection: No vaccine interaction studies have been performed. INMAZEB may reduce the efficacy of the live vaccine. The interval between live vaccination following initiation of INMAZEB therapy should be in accordance with current vaccination guidelines. ( 7.1 )
Storage and Handling
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection is a clear to slightly opalescent and colorless to pale yellow solution. It is supplied in a carton containing one single dose vial of: 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) (NDC 61755-018-01) 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) (NDC 61755-019-01)
How Supplied
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection is a clear to slightly opalescent and colorless to pale yellow solution. It is supplied in a carton containing one single dose vial of: 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) (NDC 61755-018-01) 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) (NDC 61755-019-01)
Medication Information
Warnings and Precautions
Hypersensitivity Reactions Including Infusion-Associated Events: Hypersensitivity reactions including infusion-associated events have been reported during and post-infusion with INMAZEB. These may include acute, life-threatening reactions during and after the infusion. Monitor patients and in the case of severe or life-threatening hypersensitivity reactions, discontinue the administration of INMAZEB immediately and administer appropriate emergency care. ( 5.1 )
Indications and Usage
INMAZEB is indicated for the treatment of infection caused by Orthoebolavirus zairense in adult and pediatric patients, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection [see Dosage and Administration (2.2) , and Clinical Studies (14) ] .
Dosage and Administration
The recommended dosage of INMAZEB is 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg diluted and administered as a single intravenous infusion. ( 2.1 ) Refer to the Full Prescribing Information for information on preparation and administration. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions Including Infusion-Associated Events [see Warnings and Precautions (5.1) ]
Drug Interactions
Interaction with live vaccine indicated for prevention of Orthoebolavirus zairense infection: No vaccine interaction studies have been performed. INMAZEB may reduce the efficacy of the live vaccine. The interval between live vaccination following initiation of INMAZEB therapy should be in accordance with current vaccination guidelines. ( 7.1 )
Storage and Handling
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection is a clear to slightly opalescent and colorless to pale yellow solution. It is supplied in a carton containing one single dose vial of: 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) (NDC 61755-018-01) 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) (NDC 61755-019-01)
How Supplied
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection is a clear to slightly opalescent and colorless to pale yellow solution. It is supplied in a carton containing one single dose vial of: 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) (NDC 61755-018-01) 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) (NDC 61755-019-01)
Description
INMAZEB is indicated for the treatment of infection caused by Orthoebolavirus zairense in adult and pediatric patients, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection [see Dosage and Administration (2.2) , and Clinical Studies (14) ] .
Section 42229-5
Limitations of Use
The efficacy of INMAZEB has not been established for other species of the Orthoebolavirus and Orthomarburgvirus genera.
Orthoebolavirus zairense can change over time, and factors such as emergence of resistance, or changes in viral virulence could diminish the clinical benefit of antiviral drugs. Consider available information on drug susceptibility patterns for circulating Orthoebolavirus zairense strains when deciding whether to use INMAZEB.
Section 44425-7
Prior to dilution
Store INMAZEB vial refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze or shake.
11 Description
Atoltivimab, maftivimab, and odesivimab-ebgn is a combination of Orthoebolavirus zairense glycoprotein (GP) directed recombinant human IgG1 human monoclonal antibodies of similar structure. The human monoclonal antibodies, atoltivimab, maftivimab, and odesivimab are produced by recombinant DNA technology in Chinese hamster ovary (CHO) cell suspension culture and have an approximate molecular weight of 145 kDa, 146 kDa and 144 kDa, respectively.
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection for intravenous use is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution, that is free from visible particulates.
INMAZEB injection is supplied as two strength presentations:
- 16.67 mg/16.67 mg/16.67 mg per mL: Each vial contains 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab in 14.5 mL. Each mL contains 16.67 mg of atoltivimab, 16.67 mg of maftivimab, 16.67 mg of odesivimab, and L-histidine (0.74 mg), L-histidine monohydrochloride monohydrate (1.09 mg), polysorbate 80 (1 mg), sucrose (100 mg), and Water for Injection, USP with a pH of 6.0.
- 33.33 mg/33.33 mg/33.33 mg per mL: Each vial contains 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab in 14.5 mL. Each mL contains 33.33 mg of atoltivimab, 33.33 mg of maftivimab, 33.33 mg of odesivimab, and L-histidine (0.74 mg), L-histidine monohydrochloride monohydrate (1.09 mg), polysorbate 80 (1 mg), sucrose (100 mg), and Water for Injection, USP with a pH of 6.0.
8.4 Pediatric Use
The safety and effectiveness of INMAZEB for the treatment of infection caused by Orthoebolavirus zairense have been established in pediatric patients from birth to less than 18 years of age. Use of INMAZEB for this indication is supported by evidence from a multi-center, open label, randomized controlled trial of INMAZEB in adults and pediatric subjects that included 39 pediatric subjects birth to less than 18 years of age, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection. The 28-day mortality and safety in adult and pediatric subjects treated with INMAZEB were similar [see Adverse Reactions (6.1) and Clinical Studies (14)]. An additional 38 pediatric subjects from birth to less than 18 years of age received INMAZEB in an expanded access program.
8.5 Geriatric Use
Clinical studies of INMAZEB did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Of the 154 subjects with Orthoebolavirus zairense infection who received INMAZEB in the randomized controlled trial, 5 (3.2%) were 65 years or older. The limited clinical experience has not identified differences in responses between the elderly and younger subjects.
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies in the study described below with the incidence of antibodies in other studies or to other atoltivimab, maftivimab, and odesivimab products may be misleading.
The development of anti-atoltivimab, anti-maftivimab, and anti-odesivimab antibodies was evaluated in 24 healthy adults in a single dose, randomized, double-blind, placebo-controlled, dose escalation study. Immunogenic responses against atoltivimab, maftivimab, and odesivimab were not detected at baseline or through 168 days post-dose in any subjects.
14 Clinical Studies
The efficacy of INMAZEB was evaluated in PALM, a multi-center, open-label, randomized controlled trial sponsored by the National Institute of Allergy and Infectious Diseases (NIAID; NCT03719586). The trial was conducted in the Democratic Republic of Congo, where an outbreak began in August 2018, and enrolled 681 subjects of all ages, including pregnant women, with documented Orthoebolavirus zairense infection and symptoms of any duration who were receiving optimized standard of care (oSOC). Subjects were randomized to receive INMAZEB (50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg) intravenously as a single infusion, an investigational control 50 mg/kg intravenously every third day, for a total of 3 doses, or other investigational drugs. Eligible subjects had a positive reverse transcriptase-polymerase chain reaction (RT-PCR) for the nucleoprotein (NP) gene of Orthoebolavirus zairense and had not received other investigational treatments (with the exception of experimental vaccines) within the previous 30 days. Neonates ≤7 days of age were eligible if the mother had documented infection. Neonates born to a mother who had cleared Orthoebolavirus zairense following a course of her assigned investigational medication were also eligible to be enrolled at investigator discretion regarding the likelihood that the neonate was infected. Randomization was stratified by reverse transcription-PCR cycle threshold calculated using NP targets (CtNP ≤22.0 vs >22.0; corresponding to high and low viral load, respectively) and Ebola Treatment Unit (ETU) site. All subjects received oSOC consisting of a minimum of intravenous fluids, daily clinical laboratory testing, correction of hypoglycemia and electrolyte imbalances, and broad-spectrum antibiotics and antimalarials, as indicated.
The primary efficacy endpoint was 28-day mortality. The primary analysis population includes all subjects who were randomized and concurrently eligible to receive either INMAZEB or the investigational control during the same time period of the trial.
The demographics and baseline characteristics are provided in Table 10 below.
| Parameter | INMAZEB (N=154) |
Control (N=153) |
|---|---|---|
| CtNP = cycle threshold calculated using NP targets; IQR = interquartile range; AST=Aspartate aminotransferase; ALT=Alanine aminotransferase; ETU=Ebola treatment unit | ||
| Mean age (years) | 28 | 31 |
| Age <1 month (%) | 1 (1%) | 2 (1%) |
| Age 1 month to <1 year (%) | 4 (3%) | 1 (1%) |
| Age 1 year to < 6 years (%) | 18 (12%) | 13 (8%) |
| Age 6 years to <12 years (%) | 8 (5%) | 4 (3%) |
| Age 12 years to <18 years (%) | 8 (5%) | 8 (5%) |
| Age 18 years to <50 years (%) | 93 (60%) | 105 (69%) |
| Age 50 years to <65 years (%) | 17 (11%) | 18 (12%) |
| Age ≥65 years (%) | 5 (3%) | 2 (1%) |
| Female (%) | 90 (58%) | 80 (52%) |
| Positive result on pregnancy test Pregnancy positive test was calculated based on subjects who had pregnancy test result. , n (%) |
2/67 (3%) | 4/61 (7%) |
| RT-PCR CtNP cycle threshold ≤22, n | 66 | 64 |
| Median RT-PCR CtNP (IQR) | 22.7 (20.1, 28.1) | 22.9 (18.8, 26.4) |
| Median creatinine (IQR) | 1.0 (0.7, 4.0) | 1.1 (0.7, 3.2) |
| Median AST (IQR) | 225.5 (98.0, 941.0) | 351.0 (109, 1404.0) |
| Median ALT (IQR) | 165.0 (56.0, 418.0) | 223.5 (47.0, 564.0) |
| Median days from onset of symptoms to randomization (IQR) | 5.0 (3.0, 7.0) | 5.0 (3.0, 7.0) |
| Reported Vaccination with rVSV-ZEBOV vaccine, n (%) | 34 (22%) | 41 (27%) |
| <10 days before ETU admission | 20/34 (59%) | 21/41 (51%) |
| ≥10 days before ETU admission | 14/34 (41%) | 18/41 (44%) |
| Timing unknown | 0/34 (0%) | 2/41 (5%) |
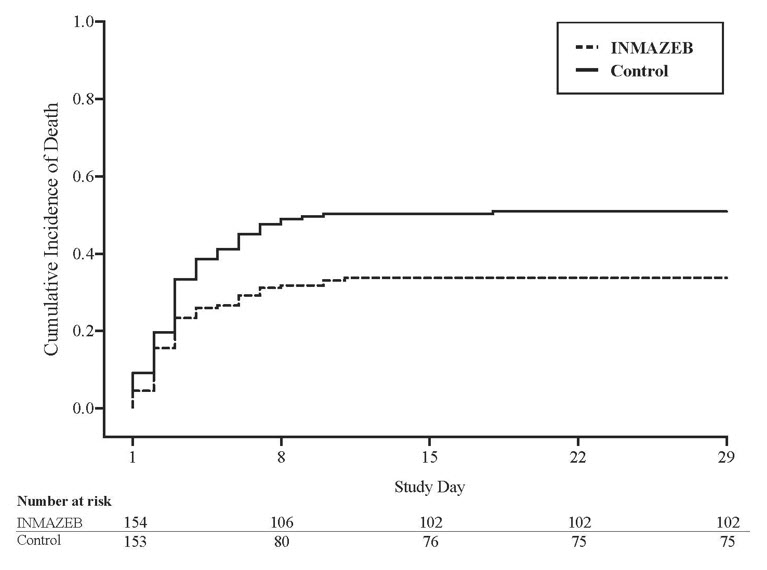
The PALM trial was stopped early on the basis of a pre-specified interim analysis showing a statistically significant reduction in mortality for INMAZEB compared to control.
Mortality efficacy results are shown in Table 11.
| Efficacy Endpoints | INMAZEB Both INMAZEB and Control were administered with optimized standard of care
(N=154) |
Control
(N=153) |
|---|---|---|
| Overall | ||
| 28-day mortality, n (%) | 52 (34%) | 78 (51%) |
| Mortality rate difference relative to control (95% CI) | -17.2 (-28.4, -2.6) | |
| p-Value The result is significant according to the interim stopping boundary, p<0.028
|
0.0024 | |
| Baseline Viral Load | ||
|
High viral load (CtNP ≤ 22)
Cepheid GeneXpert Ebola® Assay used for detection of Orthoebolavirus zairense RNA
|
n=66 | n=64 |
| 28-day mortality, n (%) | 42 (64%) | 56 (88%) |
| Mortality rate difference relative to control (95% CI) | -23.9 (-43.8, -6.4) | |
| Low viral load (CtNP > 22) | n=88 | n=88 |
| 28-day mortality, n (%) | 10 (11%) | 22 (25%) |
| Mortality rate difference relative to control (95% CI) | -13.6 (-31.8, -1.4) | |
| Age group | ||
| Adults (age ≥18 years) | 39/115 (34%) | 67/125 (54%) |
| 12 to < 18 years of age | 2/8 (25%) | 4/8 (50%) |
| 6 to < 12 years of age | 1/8 (13%) | 1/4 (25%) |
| < 6 years of age | 10/23 (43%) | 6/16 (38%) |
| Sex | ||
| Male | 21/64 (33%) | 31/73 (42%) |
| Female | 31/90 (34%) | 47/80 (59%) |
Figure 1: Kaplan-Meier Curve for Overall Mortality
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions Including Infusion-Associated Events [see Warnings and Precautions (5.1)]
7 Drug Interactions
Interaction with live vaccine indicated for prevention of Orthoebolavirus zairense infection: No vaccine interaction studies have been performed. INMAZEB may reduce the efficacy of the live vaccine. The interval between live vaccination following initiation of INMAZEB therapy should be in accordance with current vaccination guidelines. (7.1)
12.2 Pharmacodynamics
Atoltivimab, maftivimab, and odesivimab exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
No pharmacokinetic data are available in patients with Orthoebolavirus zairense infection. The pharmacokinetics of atoltivimab, maftivimab, and odesivimab in 18 healthy subjects 21 to 60 years of age are linear and dose-proportional over the range of 1 mg of atoltivimab, 1 mg of maftivimab, and 1 mg of odesivimab per kg to 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg (0.02 to 1 times the approved recommended dosage) of INMAZEB following a single intravenous (IV) infusion. Pharmacokinetic parameters for the individual antibodies of INMAZEB are provided in Table 6.
| Atoltivimab 50 mg/kg INMAZEB was administered at a total dose of 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg in a 1:1:1 ratio.
|
Maftivimab 50 mg/kg |
Odesivimab 50 mg/kg |
|
|---|---|---|---|
| Systemic Exposure (n=6) | |||
| Mean (SD) Cmax, mg/L | 1,220 (101) | 1,280 (68.0) | 1,260 (81.2) |
| Mean (SD) AUCinf, mg day/L | 17,100 (4,480) | 18,700 (4,100) | 25,600 (5,040) |
| Distribution | |||
| Mean (SD) Volume of Distribution at Steady State, mL/kg | 58.2 (2.66) | 57.6 (3.89) | 56.0 (3.16) |
| Elimination | |||
| Mean (SD) Elimination Half-Life (days) | 21.2 (3.36) | 22.3 (3.09) | 25.3 (3.86) |
| Mean (SD) Clearance (mL/day/kg) | 3.08 (0.719) | 2.78 (0.558) | 2.02 (0.374) |
2.1 Recommended Dosage
INMAZEB is a combination of three human monoclonal antibodies co-formulated in a 1:1:1 ratio of atoltivimab, maftivimab, and odesivimab. INMAZEB is available as two different strength presentations, containing either 16.67 mg of each antibody per mL or 33.33 mg of each antibody per mL [see Dosage Forms and Strengths (3)].
The recommended dosage of INMAZEB is 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg diluted and administered as a single intravenous infusion as shown in Table 1 [see Dosage and Administration (2.2)].
1 Indications and Usage
INMAZEB is indicated for the treatment of infection caused by Orthoebolavirus zairense in adult and pediatric patients, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection [see Dosage and Administration (2.2), and Clinical Studies (14)].
12.1 Mechanism of Action
INMAZEB is an antiviral drug combination of three recombinant human IgG1κ monoclonal antibodies (atoltivimab, maftivimab, and odesivimab) that inhibit Orthoebolavirus zairense [see Microbiology (12.4) ].
7.1 Vaccine Interactions
No vaccine-therapeutic interaction studies have been performed in human subjects using INMAZEB. However, because of the potential for INMAZEB to inhibit replication of a live vaccine virus indicated for prevention of Orthoebolavirus zairense infection and possibly reduce the efficacy of the vaccine, avoid the concurrent administration of a live vaccine during treatment with INMAZEB. The interval between live vaccination following initiation of INMAZEB therapy should be in accordance with current vaccination guidelines. The efficacy of INMAZEB among subjects who reported receipt of a recombinant live vaccine prior to their enrollment in the PALM clinical trial was similar to subjects who did not receive a vaccine.
5 Warnings and Precautions
Hypersensitivity Reactions Including Infusion-Associated Events: Hypersensitivity reactions including infusion-associated events have been reported during and post-infusion with INMAZEB. These may include acute, life-threatening reactions during and after the infusion. Monitor patients and in the case of severe or life-threatening hypersensitivity reactions, discontinue the administration of INMAZEB immediately and administer appropriate emergency care. (5.1)
2 Dosage and Administration
3 Dosage Forms and Strengths
INMAZEB is a clear to slightly opalescent and colorless to pale yellow solution available as:
- Injection: 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) in a single-dose vial.
- Injection: 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) in a single-dose vial.
8 Use in Specific Populations
Lactation: Patients infected with Orthoebolavirus zairense should be instructed not to breastfeed due to the potential for Orthoebolavirus zairense transmission. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates may not reflect the rates observed in practice.
Overall, 382 adult and pediatric subjects with Orthoebolavirus zairense infection received INMAZEB in one clinical trial (the PALM trial) and as part of an expanded access program conducted in the Democratic Republic of Congo during a Orthoebolavirus zairense outbreak in 2018-2019. In the PALM trial, the safety of INMAZEB was evaluated in a multi-center, open-label, randomized controlled trial, in which 154 subjects (115 adult subjects and 39 pediatric subjects) received INMAZEB [50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg (3 mL/kg)] intravenously as a single infusion and 168 subjects received an investigational control [see Clinical Studies (14)]. All subjects received optimized standard of care treatment. During the same outbreak, INMAZEB [50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg (3 mL/kg)] was given to 228 subjects (190 adult subjects and 38 pediatric subjects) in the expanded access program.
The safety data described below is derived from the PALM trial.
Table 4 summarizes adverse events that were reported during INMAZEB infusion. The evaluation of adverse events in subjects who received INMAZEB may have been confounded by the signs and symptoms of the underlying Orthoebolavirus zairense infection. The most common adverse events reported in at least 20% of subjects who received INMAZEB were pyrexia (or elevation in fever), chills, tachycardia, tachypnea, and vomiting. The adverse event profile in adult and pediatric subjects treated with INMAZEB was similar.
| Adverse Event Adverse events in this table were reported as preferred terms from a list of pre-defined or other adverse events that occurred on the day of infusion, and included signs and symptoms that occurred during or immediately after infusion
|
INMAZEB (N=154) % |
Control Investigational therapy administered as three separate infusions
(N=168) % |
|---|---|---|
| Pyrexia (Elevation in fever) | 54 | 58 |
| Chills | 39 | 33 |
| Tachycardia | 20 | 32 |
| Tachypnea | 19 | 28 |
| Vomiting Adverse events that were not pre-specified
|
19 | 23 |
| Hypotension | 15 | 31 |
| Diarrhea | 11 | 18 |
| Hypoxia | 10 | 11 |
The following pre-specified symptoms, which were assessed on a daily basis while admitted to the treatment unit, were reported in 40% or more of subjects who received INMAZEB: diarrhea, pyrexia, and vomiting. Evaluation of these symptoms may have been confounded by the underlying Orthoebolavirus zairense infection.
2.2 Preparation and Administration
INMAZEB must be prepared and administered under the supervision of a healthcare provider. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. INMAZEB should be clear to slightly opalescent, colorless to pale yellow solution that is free from visible particulates. Discard the vial if the solution is cloudy, discolored or contains particulate matter.
16 How Supplied/storage and Handling
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection is a clear to slightly opalescent and colorless to pale yellow solution. It is supplied in a carton containing one single dose vial of:
- 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) (NDC 61755-018-01)
- 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) (NDC 61755-019-01)
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity, genotoxicity, and fertility studies have not been conducted with INMAZEB.
Principal Display Panel 14.5 Ml Vial Carton 61755 018
NDC 61755-018-01
Rx only
Inmazeb®
(atoltivimab, maftivimab,
and odesivimab – ebgn)
Injection
241.7 mg / 241.7 mg / 241.7 mg per 14.5 mL
(16.67 mg / 16.67 mg / 16.67 mg per mL)
For Intravenous Infusion after Dilution
Single-Dose Vial
Discard unused portion.
Do not use vial if seal is broken or missing.
One 14.5 mL Vial
Principal Display Panel 14.5 Ml Vial Carton 61755 019
NDC 61755-019-01
Rx only
Inmazeb®
(atoltivimab, maftivimab,
and odesivimab – ebgn)
Injection
483.3 mg / 483.3 mg / 483.3 mg per 14.5 mL
(33.33 mg / 33.33 mg / 33.33 mg per mL)
For Intravenous Infusion after Dilution
Single-Dose Vial
Discard unused portion.
Do not use vial if seal is broken or missing.
One 14.5 mL Vial
5.1 Hypersensitivity Reactions Including Infusion Associated Events
Hypersensitivity reactions including infusion-associated events have been reported during and post-infusion with INMAZEB. These may include acute, life-threatening reactions during and after the infusion. Monitor all patients for signs and symptoms including, but not limited to, hypotension, chills and elevation of fever, during and following INMAZEB infusion. In the case of severe or life-threatening hypersensitivity reactions, discontinue the administration of INMAZEB immediately and administer appropriate emergency care [see Adverse Reactions (6.1)].
Infusion could not be completed in 1% of subjects who received INMAZEB due to infusion-associated adverse events. The rate of infusion of INMAZEB may be slowed or interrupted if the patient develops any signs of infusion-associated events or other adverse events [see Adverse Reactions (6.1)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
The efficacy of INMAZEB has not been established for other species of the Orthoebolavirus and Orthomarburgvirus genera.
Orthoebolavirus zairense can change over time, and factors such as emergence of resistance, or changes in viral virulence could diminish the clinical benefit of antiviral drugs. Consider available information on drug susceptibility patterns for circulating Orthoebolavirus zairense strains when deciding whether to use INMAZEB.
Section 44425-7 (44425-7)
Prior to dilution
Store INMAZEB vial refrigerated at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze or shake.
11 Description (11 DESCRIPTION)
Atoltivimab, maftivimab, and odesivimab-ebgn is a combination of Orthoebolavirus zairense glycoprotein (GP) directed recombinant human IgG1 human monoclonal antibodies of similar structure. The human monoclonal antibodies, atoltivimab, maftivimab, and odesivimab are produced by recombinant DNA technology in Chinese hamster ovary (CHO) cell suspension culture and have an approximate molecular weight of 145 kDa, 146 kDa and 144 kDa, respectively.
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection for intravenous use is a sterile, preservative-free, clear to slightly opalescent, colorless to pale yellow solution, that is free from visible particulates.
INMAZEB injection is supplied as two strength presentations:
- 16.67 mg/16.67 mg/16.67 mg per mL: Each vial contains 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab in 14.5 mL. Each mL contains 16.67 mg of atoltivimab, 16.67 mg of maftivimab, 16.67 mg of odesivimab, and L-histidine (0.74 mg), L-histidine monohydrochloride monohydrate (1.09 mg), polysorbate 80 (1 mg), sucrose (100 mg), and Water for Injection, USP with a pH of 6.0.
- 33.33 mg/33.33 mg/33.33 mg per mL: Each vial contains 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab in 14.5 mL. Each mL contains 33.33 mg of atoltivimab, 33.33 mg of maftivimab, 33.33 mg of odesivimab, and L-histidine (0.74 mg), L-histidine monohydrochloride monohydrate (1.09 mg), polysorbate 80 (1 mg), sucrose (100 mg), and Water for Injection, USP with a pH of 6.0.
8.4 Pediatric Use
The safety and effectiveness of INMAZEB for the treatment of infection caused by Orthoebolavirus zairense have been established in pediatric patients from birth to less than 18 years of age. Use of INMAZEB for this indication is supported by evidence from a multi-center, open label, randomized controlled trial of INMAZEB in adults and pediatric subjects that included 39 pediatric subjects birth to less than 18 years of age, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection. The 28-day mortality and safety in adult and pediatric subjects treated with INMAZEB were similar [see Adverse Reactions (6.1) and Clinical Studies (14)]. An additional 38 pediatric subjects from birth to less than 18 years of age received INMAZEB in an expanded access program.
8.5 Geriatric Use
Clinical studies of INMAZEB did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Of the 154 subjects with Orthoebolavirus zairense infection who received INMAZEB in the randomized controlled trial, 5 (3.2%) were 65 years or older. The limited clinical experience has not identified differences in responses between the elderly and younger subjects.
6.2 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies in the study described below with the incidence of antibodies in other studies or to other atoltivimab, maftivimab, and odesivimab products may be misleading.
The development of anti-atoltivimab, anti-maftivimab, and anti-odesivimab antibodies was evaluated in 24 healthy adults in a single dose, randomized, double-blind, placebo-controlled, dose escalation study. Immunogenic responses against atoltivimab, maftivimab, and odesivimab were not detected at baseline or through 168 days post-dose in any subjects.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of INMAZEB was evaluated in PALM, a multi-center, open-label, randomized controlled trial sponsored by the National Institute of Allergy and Infectious Diseases (NIAID; NCT03719586). The trial was conducted in the Democratic Republic of Congo, where an outbreak began in August 2018, and enrolled 681 subjects of all ages, including pregnant women, with documented Orthoebolavirus zairense infection and symptoms of any duration who were receiving optimized standard of care (oSOC). Subjects were randomized to receive INMAZEB (50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg) intravenously as a single infusion, an investigational control 50 mg/kg intravenously every third day, for a total of 3 doses, or other investigational drugs. Eligible subjects had a positive reverse transcriptase-polymerase chain reaction (RT-PCR) for the nucleoprotein (NP) gene of Orthoebolavirus zairense and had not received other investigational treatments (with the exception of experimental vaccines) within the previous 30 days. Neonates ≤7 days of age were eligible if the mother had documented infection. Neonates born to a mother who had cleared Orthoebolavirus zairense following a course of her assigned investigational medication were also eligible to be enrolled at investigator discretion regarding the likelihood that the neonate was infected. Randomization was stratified by reverse transcription-PCR cycle threshold calculated using NP targets (CtNP ≤22.0 vs >22.0; corresponding to high and low viral load, respectively) and Ebola Treatment Unit (ETU) site. All subjects received oSOC consisting of a minimum of intravenous fluids, daily clinical laboratory testing, correction of hypoglycemia and electrolyte imbalances, and broad-spectrum antibiotics and antimalarials, as indicated.
The primary efficacy endpoint was 28-day mortality. The primary analysis population includes all subjects who were randomized and concurrently eligible to receive either INMAZEB or the investigational control during the same time period of the trial.
The demographics and baseline characteristics are provided in Table 10 below.
| Parameter | INMAZEB (N=154) |
Control (N=153) |
|---|---|---|
| CtNP = cycle threshold calculated using NP targets; IQR = interquartile range; AST=Aspartate aminotransferase; ALT=Alanine aminotransferase; ETU=Ebola treatment unit | ||
| Mean age (years) | 28 | 31 |
| Age <1 month (%) | 1 (1%) | 2 (1%) |
| Age 1 month to <1 year (%) | 4 (3%) | 1 (1%) |
| Age 1 year to < 6 years (%) | 18 (12%) | 13 (8%) |
| Age 6 years to <12 years (%) | 8 (5%) | 4 (3%) |
| Age 12 years to <18 years (%) | 8 (5%) | 8 (5%) |
| Age 18 years to <50 years (%) | 93 (60%) | 105 (69%) |
| Age 50 years to <65 years (%) | 17 (11%) | 18 (12%) |
| Age ≥65 years (%) | 5 (3%) | 2 (1%) |
| Female (%) | 90 (58%) | 80 (52%) |
| Positive result on pregnancy test Pregnancy positive test was calculated based on subjects who had pregnancy test result. , n (%) |
2/67 (3%) | 4/61 (7%) |
| RT-PCR CtNP cycle threshold ≤22, n | 66 | 64 |
| Median RT-PCR CtNP (IQR) | 22.7 (20.1, 28.1) | 22.9 (18.8, 26.4) |
| Median creatinine (IQR) | 1.0 (0.7, 4.0) | 1.1 (0.7, 3.2) |
| Median AST (IQR) | 225.5 (98.0, 941.0) | 351.0 (109, 1404.0) |
| Median ALT (IQR) | 165.0 (56.0, 418.0) | 223.5 (47.0, 564.0) |
| Median days from onset of symptoms to randomization (IQR) | 5.0 (3.0, 7.0) | 5.0 (3.0, 7.0) |
| Reported Vaccination with rVSV-ZEBOV vaccine, n (%) | 34 (22%) | 41 (27%) |
| <10 days before ETU admission | 20/34 (59%) | 21/41 (51%) |
| ≥10 days before ETU admission | 14/34 (41%) | 18/41 (44%) |
| Timing unknown | 0/34 (0%) | 2/41 (5%) |
The PALM trial was stopped early on the basis of a pre-specified interim analysis showing a statistically significant reduction in mortality for INMAZEB compared to control.
Mortality efficacy results are shown in Table 11.
| Efficacy Endpoints | INMAZEB Both INMAZEB and Control were administered with optimized standard of care
(N=154) |
Control
(N=153) |
|---|---|---|
| Overall | ||
| 28-day mortality, n (%) | 52 (34%) | 78 (51%) |
| Mortality rate difference relative to control (95% CI) | -17.2 (-28.4, -2.6) | |
| p-Value The result is significant according to the interim stopping boundary, p<0.028
|
0.0024 | |
| Baseline Viral Load | ||
|
High viral load (CtNP ≤ 22)
Cepheid GeneXpert Ebola® Assay used for detection of Orthoebolavirus zairense RNA
|
n=66 | n=64 |
| 28-day mortality, n (%) | 42 (64%) | 56 (88%) |
| Mortality rate difference relative to control (95% CI) | -23.9 (-43.8, -6.4) | |
| Low viral load (CtNP > 22) | n=88 | n=88 |
| 28-day mortality, n (%) | 10 (11%) | 22 (25%) |
| Mortality rate difference relative to control (95% CI) | -13.6 (-31.8, -1.4) | |
| Age group | ||
| Adults (age ≥18 years) | 39/115 (34%) | 67/125 (54%) |
| 12 to < 18 years of age | 2/8 (25%) | 4/8 (50%) |
| 6 to < 12 years of age | 1/8 (13%) | 1/4 (25%) |
| < 6 years of age | 10/23 (43%) | 6/16 (38%) |
| Sex | ||
| Male | 21/64 (33%) | 31/73 (42%) |
| Female | 31/90 (34%) | 47/80 (59%) |
Figure 1: Kaplan-Meier Curve for Overall Mortality
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions Including Infusion-Associated Events [see Warnings and Precautions (5.1)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Interaction with live vaccine indicated for prevention of Orthoebolavirus zairense infection: No vaccine interaction studies have been performed. INMAZEB may reduce the efficacy of the live vaccine. The interval between live vaccination following initiation of INMAZEB therapy should be in accordance with current vaccination guidelines. (7.1)
12.2 Pharmacodynamics
Atoltivimab, maftivimab, and odesivimab exposure-response relationships and the time course of pharmacodynamic response are unknown.
12.3 Pharmacokinetics
No pharmacokinetic data are available in patients with Orthoebolavirus zairense infection. The pharmacokinetics of atoltivimab, maftivimab, and odesivimab in 18 healthy subjects 21 to 60 years of age are linear and dose-proportional over the range of 1 mg of atoltivimab, 1 mg of maftivimab, and 1 mg of odesivimab per kg to 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg (0.02 to 1 times the approved recommended dosage) of INMAZEB following a single intravenous (IV) infusion. Pharmacokinetic parameters for the individual antibodies of INMAZEB are provided in Table 6.
| Atoltivimab 50 mg/kg INMAZEB was administered at a total dose of 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg in a 1:1:1 ratio.
|
Maftivimab 50 mg/kg |
Odesivimab 50 mg/kg |
|
|---|---|---|---|
| Systemic Exposure (n=6) | |||
| Mean (SD) Cmax, mg/L | 1,220 (101) | 1,280 (68.0) | 1,260 (81.2) |
| Mean (SD) AUCinf, mg day/L | 17,100 (4,480) | 18,700 (4,100) | 25,600 (5,040) |
| Distribution | |||
| Mean (SD) Volume of Distribution at Steady State, mL/kg | 58.2 (2.66) | 57.6 (3.89) | 56.0 (3.16) |
| Elimination | |||
| Mean (SD) Elimination Half-Life (days) | 21.2 (3.36) | 22.3 (3.09) | 25.3 (3.86) |
| Mean (SD) Clearance (mL/day/kg) | 3.08 (0.719) | 2.78 (0.558) | 2.02 (0.374) |
2.1 Recommended Dosage
INMAZEB is a combination of three human monoclonal antibodies co-formulated in a 1:1:1 ratio of atoltivimab, maftivimab, and odesivimab. INMAZEB is available as two different strength presentations, containing either 16.67 mg of each antibody per mL or 33.33 mg of each antibody per mL [see Dosage Forms and Strengths (3)].
The recommended dosage of INMAZEB is 50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg diluted and administered as a single intravenous infusion as shown in Table 1 [see Dosage and Administration (2.2)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
INMAZEB is indicated for the treatment of infection caused by Orthoebolavirus zairense in adult and pediatric patients, including neonates born to a mother who is RT-PCR positive for Orthoebolavirus zairense infection [see Dosage and Administration (2.2), and Clinical Studies (14)].
12.1 Mechanism of Action
INMAZEB is an antiviral drug combination of three recombinant human IgG1κ monoclonal antibodies (atoltivimab, maftivimab, and odesivimab) that inhibit Orthoebolavirus zairense [see Microbiology (12.4) ].
7.1 Vaccine Interactions
No vaccine-therapeutic interaction studies have been performed in human subjects using INMAZEB. However, because of the potential for INMAZEB to inhibit replication of a live vaccine virus indicated for prevention of Orthoebolavirus zairense infection and possibly reduce the efficacy of the vaccine, avoid the concurrent administration of a live vaccine during treatment with INMAZEB. The interval between live vaccination following initiation of INMAZEB therapy should be in accordance with current vaccination guidelines. The efficacy of INMAZEB among subjects who reported receipt of a recombinant live vaccine prior to their enrollment in the PALM clinical trial was similar to subjects who did not receive a vaccine.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Hypersensitivity Reactions Including Infusion-Associated Events: Hypersensitivity reactions including infusion-associated events have been reported during and post-infusion with INMAZEB. These may include acute, life-threatening reactions during and after the infusion. Monitor patients and in the case of severe or life-threatening hypersensitivity reactions, discontinue the administration of INMAZEB immediately and administer appropriate emergency care. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
INMAZEB is a clear to slightly opalescent and colorless to pale yellow solution available as:
- Injection: 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) in a single-dose vial.
- Injection: 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) in a single-dose vial.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Patients infected with Orthoebolavirus zairense should be instructed not to breastfeed due to the potential for Orthoebolavirus zairense transmission. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates may not reflect the rates observed in practice.
Overall, 382 adult and pediatric subjects with Orthoebolavirus zairense infection received INMAZEB in one clinical trial (the PALM trial) and as part of an expanded access program conducted in the Democratic Republic of Congo during a Orthoebolavirus zairense outbreak in 2018-2019. In the PALM trial, the safety of INMAZEB was evaluated in a multi-center, open-label, randomized controlled trial, in which 154 subjects (115 adult subjects and 39 pediatric subjects) received INMAZEB [50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg (3 mL/kg)] intravenously as a single infusion and 168 subjects received an investigational control [see Clinical Studies (14)]. All subjects received optimized standard of care treatment. During the same outbreak, INMAZEB [50 mg of atoltivimab, 50 mg of maftivimab, and 50 mg of odesivimab per kg (3 mL/kg)] was given to 228 subjects (190 adult subjects and 38 pediatric subjects) in the expanded access program.
The safety data described below is derived from the PALM trial.
Table 4 summarizes adverse events that were reported during INMAZEB infusion. The evaluation of adverse events in subjects who received INMAZEB may have been confounded by the signs and symptoms of the underlying Orthoebolavirus zairense infection. The most common adverse events reported in at least 20% of subjects who received INMAZEB were pyrexia (or elevation in fever), chills, tachycardia, tachypnea, and vomiting. The adverse event profile in adult and pediatric subjects treated with INMAZEB was similar.
| Adverse Event Adverse events in this table were reported as preferred terms from a list of pre-defined or other adverse events that occurred on the day of infusion, and included signs and symptoms that occurred during or immediately after infusion
|
INMAZEB (N=154) % |
Control Investigational therapy administered as three separate infusions
(N=168) % |
|---|---|---|
| Pyrexia (Elevation in fever) | 54 | 58 |
| Chills | 39 | 33 |
| Tachycardia | 20 | 32 |
| Tachypnea | 19 | 28 |
| Vomiting Adverse events that were not pre-specified
|
19 | 23 |
| Hypotension | 15 | 31 |
| Diarrhea | 11 | 18 |
| Hypoxia | 10 | 11 |
The following pre-specified symptoms, which were assessed on a daily basis while admitted to the treatment unit, were reported in 40% or more of subjects who received INMAZEB: diarrhea, pyrexia, and vomiting. Evaluation of these symptoms may have been confounded by the underlying Orthoebolavirus zairense infection.
2.2 Preparation and Administration
INMAZEB must be prepared and administered under the supervision of a healthcare provider. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. INMAZEB should be clear to slightly opalescent, colorless to pale yellow solution that is free from visible particulates. Discard the vial if the solution is cloudy, discolored or contains particulate matter.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
INMAZEB (atoltivimab, maftivimab, and odesivimab-ebgn) injection is a clear to slightly opalescent and colorless to pale yellow solution. It is supplied in a carton containing one single dose vial of:
- 241.7 mg of atoltivimab, 241.7 mg of maftivimab, and 241.7 mg of odesivimab per 14.5 mL (16.67 mg/16.67 mg/16.67 mg per mL) (NDC 61755-018-01)
- 483.3 mg of atoltivimab, 483.3 mg of maftivimab, and 483.3 mg of odesivimab per 14.5 mL (33.33 mg/33.33 mg/33.33 mg per mL) (NDC 61755-019-01)
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity, genotoxicity, and fertility studies have not been conducted with INMAZEB.
Principal Display Panel 14.5 Ml Vial Carton 61755 018 (PRINCIPAL DISPLAY PANEL - 14.5 mL Vial Carton - 61755-018)
NDC 61755-018-01
Rx only
Inmazeb®
(atoltivimab, maftivimab,
and odesivimab – ebgn)
Injection
241.7 mg / 241.7 mg / 241.7 mg per 14.5 mL
(16.67 mg / 16.67 mg / 16.67 mg per mL)
For Intravenous Infusion after Dilution
Single-Dose Vial
Discard unused portion.
Do not use vial if seal is broken or missing.
One 14.5 mL Vial
Principal Display Panel 14.5 Ml Vial Carton 61755 019 (PRINCIPAL DISPLAY PANEL - 14.5 mL Vial Carton - 61755-019)
NDC 61755-019-01
Rx only
Inmazeb®
(atoltivimab, maftivimab,
and odesivimab – ebgn)
Injection
483.3 mg / 483.3 mg / 483.3 mg per 14.5 mL
(33.33 mg / 33.33 mg / 33.33 mg per mL)
For Intravenous Infusion after Dilution
Single-Dose Vial
Discard unused portion.
Do not use vial if seal is broken or missing.
One 14.5 mL Vial
5.1 Hypersensitivity Reactions Including Infusion Associated Events (5.1 Hypersensitivity Reactions Including Infusion-Associated Events)
Hypersensitivity reactions including infusion-associated events have been reported during and post-infusion with INMAZEB. These may include acute, life-threatening reactions during and after the infusion. Monitor all patients for signs and symptoms including, but not limited to, hypotension, chills and elevation of fever, during and following INMAZEB infusion. In the case of severe or life-threatening hypersensitivity reactions, discontinue the administration of INMAZEB immediately and administer appropriate emergency care [see Adverse Reactions (6.1)].
Infusion could not be completed in 1% of subjects who received INMAZEB due to infusion-associated adverse events. The rate of infusion of INMAZEB may be slowed or interrupted if the patient develops any signs of infusion-associated events or other adverse events [see Adverse Reactions (6.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:25.584576 · Updated: 2026-03-14T22:40:22.461215