Drug Facts
04eb9e98-1323-353f-3fd7-2d317dfac526
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Budesonide (glucocorticoid) 32 mcg
Purpose
Nasal allergy symptom reliever
Medication Information
Purpose
Nasal allergy symptom reliever
Description
Budesonide (glucocorticoid) 32 mcg
Use
Temporarily relieves these symptoms of hay fever or other upper respiratory allergies:
• nasal congestion • runny nose • itchy nose • sneezing
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Warnings
Directions
- get a new bottle ready (primed) before first use
- prime bottle again if not used for two days
- use the spray
- clean the spray nozzle
|
ADULTS AND CHILDREN 12 YEARS OF AGE AND OLDER |
|
|
adults and children 12 years of age and older |
|
| CHILDREN 6 TO UNDER 12 YEARS OF AGE | |
|
|
| children 6 to under 12 years of age |
|
| children under 6 years of age |
|
|
Do Not Use
- in children under 6 years of age
- if you have ever had an allergic reaction to any of the ingredients
Other Information
- do not use if the safety seal labeled "sealed for your protection" is broken or missing.
- keep package and insert. They contain important information.
- store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Do not freeze. Protect from light.
Inactive Ingredients
carboxymethyl cellulose sodium, dextrose anhydrous, edetate disodium dihydrate, hydrochloric acid (for pH adjustment), microcrystalline cellulose, polysorbate 80, potassium sorbate, purified water
Questions Or Comments?
call toll free 1-800-645-2158
When Using This Product
- the growth rate of some children may be slower
- some symptoms may get better on the first day of treatment. It may take up to two weeks of daily use to feel the most symptom relief.
- do not share this bottle with anyone else as this may spread germs
- remember to tell your doctor about all the medicines you take, including this one
Stop Use and Ask A Doctor If
- you have, or come into contact with someone who has, chickenpox, measles or tuberculosis
- you have or develop symptoms of an infection such as persistent fever
- you have any change in vision
- you have severe or frequent nosebleeds
Ask A Doctor Before Use If You
- have had recent nose ulcers or nose surgery
- have had a nose injury that has not healed
- are using a steroid medicine for asthma, allergies or skin rash
- have an eye infection
- have or had glaucoma or cataracts
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Active Ingredient (in Each Spray)
Budesonide (glucocorticoid) 32 mcg
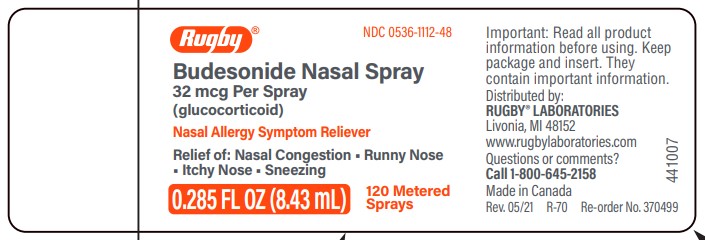
Principal Display Panel Bottle 120ms
BOTTLE LABEL - PRINCIPAL DISPLAY PANEL - 32 mcg per spray
Rugby Laboratories NDC 0536-1112-48
Budesonide Nasal Spray
Allergy Spray
120 sprays
Principal Display Panel Carton 120ms
CARTON LABEL - PRINCIPAL DISPLAY PANEL - 32 mcg per spray
Rugby Laboratories NDC 0536-1112-48
Budesonide Nasal Spray
Allergy Spray
120 sprays
Relief of:
- Nasal Congestion
- Runny Nose
- Itchy Nose
- Sneezing
Structured Label Content
Use
Temporarily relieves these symptoms of hay fever or other upper respiratory allergies:
• nasal congestion • runny nose • itchy nose • sneezing
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Nasal allergy symptom reliever
Warnings
Directions
- get a new bottle ready (primed) before first use
- prime bottle again if not used for two days
- use the spray
- clean the spray nozzle
|
ADULTS AND CHILDREN 12 YEARS OF AGE AND OLDER |
|
|
adults and children 12 years of age and older |
|
| CHILDREN 6 TO UNDER 12 YEARS OF AGE | |
|
|
| children 6 to under 12 years of age |
|
| children under 6 years of age |
|
|
Do Not Use (Do not use)
- in children under 6 years of age
- if you have ever had an allergic reaction to any of the ingredients
Other Information (Other information)
- do not use if the safety seal labeled "sealed for your protection" is broken or missing.
- keep package and insert. They contain important information.
- store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Do not freeze. Protect from light.
Inactive Ingredients (Inactive ingredients)
carboxymethyl cellulose sodium, dextrose anhydrous, edetate disodium dihydrate, hydrochloric acid (for pH adjustment), microcrystalline cellulose, polysorbate 80, potassium sorbate, purified water
Questions Or Comments? (Questions or comments?)
call toll free 1-800-645-2158
When Using This Product (When using this product)
- the growth rate of some children may be slower
- some symptoms may get better on the first day of treatment. It may take up to two weeks of daily use to feel the most symptom relief.
- do not share this bottle with anyone else as this may spread germs
- remember to tell your doctor about all the medicines you take, including this one
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- you have, or come into contact with someone who has, chickenpox, measles or tuberculosis
- you have or develop symptoms of an infection such as persistent fever
- you have any change in vision
- you have severe or frequent nosebleeds
Ask A Doctor Before Use If You (Ask a doctor before use if you)
- have had recent nose ulcers or nose surgery
- have had a nose injury that has not healed
- are using a steroid medicine for asthma, allergies or skin rash
- have an eye infection
- have or had glaucoma or cataracts
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Active Ingredient (in Each Spray) (Active ingredient (in each spray))
Budesonide (glucocorticoid) 32 mcg
Principal Display Panel Bottle 120ms (Principal Display Panel - Bottle 120ms)
BOTTLE LABEL - PRINCIPAL DISPLAY PANEL - 32 mcg per spray
Rugby Laboratories NDC 0536-1112-48
Budesonide Nasal Spray
Allergy Spray
120 sprays
Principal Display Panel Carton 120ms (Principal Display Panel - Carton 120ms)
CARTON LABEL - PRINCIPAL DISPLAY PANEL - 32 mcg per spray
Rugby Laboratories NDC 0536-1112-48
Budesonide Nasal Spray
Allergy Spray
120 sprays
Relief of:
- Nasal Congestion
- Runny Nose
- Itchy Nose
- Sneezing
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:40.788087 · Updated: 2026-03-14T23:13:57.106193