Freshkote ®
049e9cc9-0663-53bc-e063-6294a90a2c4f
34390-5
HUMAN OTC DRUG LABEL
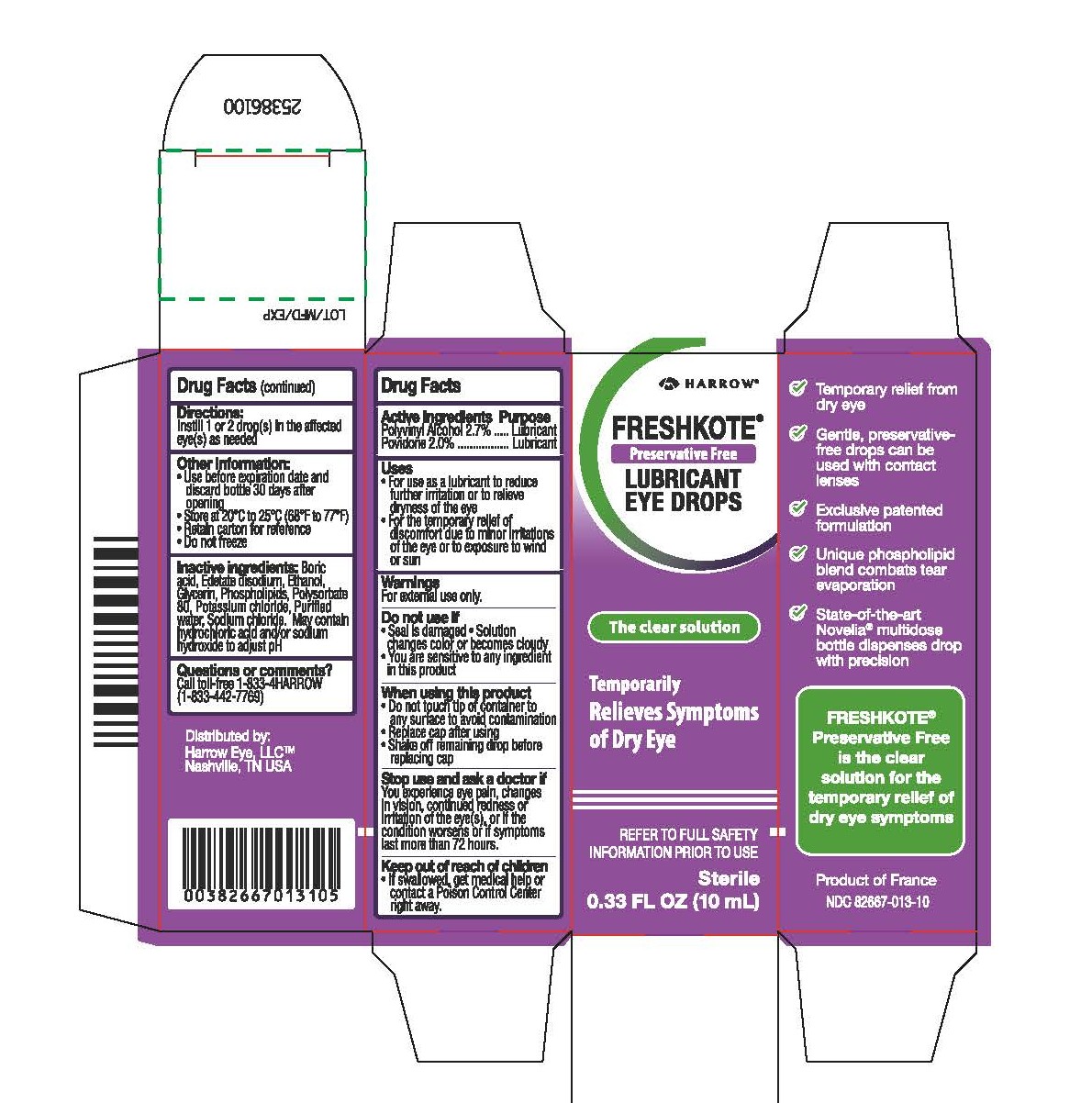
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredients Purpose Polyvinyl Alcohol 2.7%...... Lubricant Povidone 2.0%...................Lubricant
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Uses
For use as a lubricant to reduce further irritation or to relieve dryness of the eye
For the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
Dosage and Administration
Directions:
Instill 1 or 2 drop(s) in the affected eye(s) as needed
Description
Active Ingredients Purpose Polyvinyl Alcohol 2.7%...... Lubricant Povidone 2.0%...................Lubricant
Section 44425-7
Other information:
Use before expiration date and discard bottle 30 days after opening
Store at 20°C to 25°C (68°F to 77°F)
Retain carton for reference
Do not freeze
Section 50565-1
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9
Stop use and ask a doctor if
You experience eye pain, changes in vision, continued redness or irritation of the eye(s), or if the condition worsens or if symptoms last more than 72 hours.
Section 50567-7
When using this product
Do not touch tip of container to any surface to avoid contamination
Replace cap after using
Shake off remaining drop before replacing cap
Section 50570-1
Do not use if
Seal is damaged
Solution changes color or becomes cloudy
You are sensitive to any ingredient in this product
Section 51727-6
Inactive ingredients
Boric acid, Edetate disodium, Ethanol, Glycerin, Phospholipids, Polysorbate 80, Potassium chloride, Purified water, Sodium chloride. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
Section 51945-4
HARROW
FRESHKOTE ®
Preservative Free
LUBRICANT EYE DROPS
The clear solution
Temporarily Relieves Symptoms of Dry Eye
REFER TO FULL SAFETY INFORMATION PRIOR TO USE
Sterile
0.33 FL OZ (10mL)
Temporary relief from dry eye
Gentle, preservative-free drops can be used with contact lenses
Exclusive patented formulation
Unique phospholipid blend combats tear evaporation
State-of-the-art Novelia ® multidose bottle dispenses drop with precision
FRESHKOTE ®
Preservative Free
is the clear
solution for the
temporary relief of
dry eye symptoms
Product of France
NDC 82667-013-10
Section 53413-1
Questions or comments?
Call toll-free +1-833-4HARROW (+1-833-442-7769)
Section 55105-1
FRESHKOTE® Preservative Free is the clear solution for the temporary relief of dry eye symptoms
Section 55106-9
Active Ingredients Purpose
Polyvinyl Alcohol 2.7%...... Lubricant
Povidone 2.0%...................Lubricant
Structured Label Content
Indications and Usage (34067-9)
Uses
For use as a lubricant to reduce further irritation or to relieve dryness of the eye
For the temporary relief of discomfort due to minor irritations of the eye or to exposure to wind or sun
Dosage and Administration (34068-7)
Directions:
Instill 1 or 2 drop(s) in the affected eye(s) as needed
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 44425-7 (44425-7)
Other information:
Use before expiration date and discard bottle 30 days after opening
Store at 20°C to 25°C (68°F to 77°F)
Retain carton for reference
Do not freeze
Section 50565-1 (50565-1)
Keep out of reach of children
If swallowed, get medical help or contact a Poison Control Center right away
Section 50566-9 (50566-9)
Stop use and ask a doctor if
You experience eye pain, changes in vision, continued redness or irritation of the eye(s), or if the condition worsens or if symptoms last more than 72 hours.
Section 50567-7 (50567-7)
When using this product
Do not touch tip of container to any surface to avoid contamination
Replace cap after using
Shake off remaining drop before replacing cap
Section 50570-1 (50570-1)
Do not use if
Seal is damaged
Solution changes color or becomes cloudy
You are sensitive to any ingredient in this product
Section 51727-6 (51727-6)
Inactive ingredients
Boric acid, Edetate disodium, Ethanol, Glycerin, Phospholipids, Polysorbate 80, Potassium chloride, Purified water, Sodium chloride. May contain hydrochloric acid and/or sodium hydroxide to adjust pH.
Section 51945-4 (51945-4)
HARROW
FRESHKOTE ®
Preservative Free
LUBRICANT EYE DROPS
The clear solution
Temporarily Relieves Symptoms of Dry Eye
REFER TO FULL SAFETY INFORMATION PRIOR TO USE
Sterile
0.33 FL OZ (10mL)
Temporary relief from dry eye
Gentle, preservative-free drops can be used with contact lenses
Exclusive patented formulation
Unique phospholipid blend combats tear evaporation
State-of-the-art Novelia ® multidose bottle dispenses drop with precision
FRESHKOTE ®
Preservative Free
is the clear
solution for the
temporary relief of
dry eye symptoms
Product of France
NDC 82667-013-10
Section 53413-1 (53413-1)
Questions or comments?
Call toll-free +1-833-4HARROW (+1-833-442-7769)
Section 55105-1 (55105-1)
FRESHKOTE® Preservative Free is the clear solution for the temporary relief of dry eye symptoms
Section 55106-9 (55106-9)
Active Ingredients Purpose
Polyvinyl Alcohol 2.7%...... Lubricant
Povidone 2.0%...................Lubricant
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:20.969622 · Updated: 2026-03-14T23:09:05.302478