These Highlights Do Not Include All The Information Needed To Use Edurant Safely And Effectively. See Full Prescribing Information For Edurant.
03880372-2c68-45c6-a53a-f420c49541d6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1.1 ) 03/2024 Dosage and Administration ( 2.1 , 2.3 , 2.4 , 2.5 ) 03/2024 Warnings and Precautions ( 5.6 ) 03/2024
Indications and Usage
EDURANT and EDURANT PED are a human immunodeficiency virus type 1 (HIV-1) specific, non-nucleoside reverse transcriptase inhibitor (NNRTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in treatment-naïve patients 2 years of age and older and weighing at least 14 kg with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1 ). Limitations of Use: More EDURANT treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA ≥50 copies/mL) compared to EDURANT treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1 , 14 ) EDURANT is indicated in combination with VOCABRIA (cabotegravir), for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine. ( 1.2 )
Dosage and Administration
One 25 mg EDURANT tablet taken once daily with a meal for patients weighing at least 25 kg. ( 2.2 ) Pediatric patients 2 years of age and older and weighing at least 14 kg to less than 25 kg: Dosage of EDURANT PED is based on body weight. ( 2.3 ) EDURANT PED must be dispersed in drinking water and taken with a meal. ( 2.4 ) Do not substitute EDURANT tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. ( 2.1 , 5.6 ) See full prescribing information for dosing information when used in combination with cabotegravir. ( 2.6 ) For pregnant patients who are already on a stable EDURANT regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing more than 25 kg is one 25 mg tablet once daily taken orally with a meal. ( 2.5 , 12.3 ) Rifabutin coadministration: Take two 25 mg tablets of EDURANT once daily with a meal for the duration of the rifabutin coadministration. ( 2.7 )
Warnings and Precautions
Skin and Hypersensitivity Reactions: Severe skin and hypersensitivity reactions have been reported during postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. Immediately discontinue treatment if hypersensitivity or rash with systemic symptoms or elevations in hepatic serum biochemistries develop and closely monitor clinical status, including hepatic serum biochemistries. ( 5.1 ) Hepatotoxicity: Hepatic adverse events have been reported in patients with underlying liver disease, including hepatitis B or C virus co-infection, or in patients with elevated baseline transaminases. A few cases of hepatotoxicity have occurred in patients with no pre-existing hepatic disease. Monitor liver function tests before and during treatment with EDURANT or EDURANT PED in patients with underlying hepatic disease, such as hepatitis B or C virus co-infection, or marked elevations in transaminase. Also consider monitoring liver functions tests in patients without pre-existing hepatic dysfunction or other risk factors. ( 5.2 ) Depressive Disorders: Severe depressive disorders have been reported. Immediate medical evaluation is recommended for severe depressive disorders. ( 5.3 ) Patients may develop immune reconstitution syndrome. ( 5.5 )
Contraindications
EDURANT and EDURANT PED are contraindicated for coadministration with the drugs in Table 2 for which significant decreases in rilpivirine plasma concentrations may occur due to CYP3A enzyme induction or gastric pH increase, which may result in loss of virologic response and possible resistance to EDURANT or EDURANT PED or to the class of NNRTIs [see Drug Interactions (7) and Clinical Pharmacology (12.3) ] . Table 2: Drugs That are Contraindicated with EDURANT and EDURANT PED Drug Class Contraindicated Drugs in Class Clinical Comment Anticonvulsants Carbamazepine Oxcarbazepine Phenobarbital Phenytoin Potential for significant decreases in rilpivirine plasma concentrations due to CYP3A enzyme induction, which may result in loss of virologic response. Antimycobacterials Rifampin Rifapentine Glucocorticoid (systemic) Dexamethasone (more than a single-dose treatment) Herbal Products St. John's wort ( Hypericum perforatum ) Proton Pump Inhibitors e.g., Esomeprazole Lansoprazole Omeprazole Pantoprazole Rabeprazole Potential for significant decreases in rilpivirine plasma concentrations due to gastric pH increase, which may result in loss of virologic response.
Adverse Reactions
The concomitant use of EDURANT or EDURANT PED and other drugs may result in potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.7) , Contraindications (4) , and Drug Interactions (7) ] : Loss of therapeutic effect of EDURANT or EDURANT PED and possible development of resistance. In healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in EDURANT) have been shown to prolong the QTc interval of the electrocardiogram. Consider alternatives to EDURANT or EDURANT PED when coadministered with a drug that is known to have a risk of torsade de pointes [see Drug Interactions (7) and Clinical Pharmacology (12.2) ] . See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during EDURANT or EDURANT PED therapy and review concomitant medications during therapy.
Drug Interactions
The concomitant use of EDURANT or EDURANT PED and other drugs may result in potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.7) , Contraindications (4) , and Drug Interactions (7) ] : Loss of therapeutic effect of EDURANT or EDURANT PED and possible development of resistance. In healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in EDURANT) have been shown to prolong the QTc interval of the electrocardiogram. Consider alternatives to EDURANT or EDURANT PED when coadministered with a drug that is known to have a risk of torsade de pointes [see Drug Interactions (7) and Clinical Pharmacology (12.2) ] . See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during EDURANT or EDURANT PED therapy and review concomitant medications during therapy.
Medication Information
Warnings and Precautions
Skin and Hypersensitivity Reactions: Severe skin and hypersensitivity reactions have been reported during postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. Immediately discontinue treatment if hypersensitivity or rash with systemic symptoms or elevations in hepatic serum biochemistries develop and closely monitor clinical status, including hepatic serum biochemistries. ( 5.1 ) Hepatotoxicity: Hepatic adverse events have been reported in patients with underlying liver disease, including hepatitis B or C virus co-infection, or in patients with elevated baseline transaminases. A few cases of hepatotoxicity have occurred in patients with no pre-existing hepatic disease. Monitor liver function tests before and during treatment with EDURANT or EDURANT PED in patients with underlying hepatic disease, such as hepatitis B or C virus co-infection, or marked elevations in transaminase. Also consider monitoring liver functions tests in patients without pre-existing hepatic dysfunction or other risk factors. ( 5.2 ) Depressive Disorders: Severe depressive disorders have been reported. Immediate medical evaluation is recommended for severe depressive disorders. ( 5.3 ) Patients may develop immune reconstitution syndrome. ( 5.5 )
Indications and Usage
EDURANT and EDURANT PED are a human immunodeficiency virus type 1 (HIV-1) specific, non-nucleoside reverse transcriptase inhibitor (NNRTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in treatment-naïve patients 2 years of age and older and weighing at least 14 kg with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1 ). Limitations of Use: More EDURANT treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA ≥50 copies/mL) compared to EDURANT treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1 , 14 ) EDURANT is indicated in combination with VOCABRIA (cabotegravir), for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine. ( 1.2 )
Dosage and Administration
One 25 mg EDURANT tablet taken once daily with a meal for patients weighing at least 25 kg. ( 2.2 ) Pediatric patients 2 years of age and older and weighing at least 14 kg to less than 25 kg: Dosage of EDURANT PED is based on body weight. ( 2.3 ) EDURANT PED must be dispersed in drinking water and taken with a meal. ( 2.4 ) Do not substitute EDURANT tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. ( 2.1 , 5.6 ) See full prescribing information for dosing information when used in combination with cabotegravir. ( 2.6 ) For pregnant patients who are already on a stable EDURANT regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing more than 25 kg is one 25 mg tablet once daily taken orally with a meal. ( 2.5 , 12.3 ) Rifabutin coadministration: Take two 25 mg tablets of EDURANT once daily with a meal for the duration of the rifabutin coadministration. ( 2.7 )
Contraindications
EDURANT and EDURANT PED are contraindicated for coadministration with the drugs in Table 2 for which significant decreases in rilpivirine plasma concentrations may occur due to CYP3A enzyme induction or gastric pH increase, which may result in loss of virologic response and possible resistance to EDURANT or EDURANT PED or to the class of NNRTIs [see Drug Interactions (7) and Clinical Pharmacology (12.3) ] . Table 2: Drugs That are Contraindicated with EDURANT and EDURANT PED Drug Class Contraindicated Drugs in Class Clinical Comment Anticonvulsants Carbamazepine Oxcarbazepine Phenobarbital Phenytoin Potential for significant decreases in rilpivirine plasma concentrations due to CYP3A enzyme induction, which may result in loss of virologic response. Antimycobacterials Rifampin Rifapentine Glucocorticoid (systemic) Dexamethasone (more than a single-dose treatment) Herbal Products St. John's wort ( Hypericum perforatum ) Proton Pump Inhibitors e.g., Esomeprazole Lansoprazole Omeprazole Pantoprazole Rabeprazole Potential for significant decreases in rilpivirine plasma concentrations due to gastric pH increase, which may result in loss of virologic response.
Adverse Reactions
The concomitant use of EDURANT or EDURANT PED and other drugs may result in potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.7) , Contraindications (4) , and Drug Interactions (7) ] : Loss of therapeutic effect of EDURANT or EDURANT PED and possible development of resistance. In healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in EDURANT) have been shown to prolong the QTc interval of the electrocardiogram. Consider alternatives to EDURANT or EDURANT PED when coadministered with a drug that is known to have a risk of torsade de pointes [see Drug Interactions (7) and Clinical Pharmacology (12.2) ] . See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during EDURANT or EDURANT PED therapy and review concomitant medications during therapy.
Drug Interactions
The concomitant use of EDURANT or EDURANT PED and other drugs may result in potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.7) , Contraindications (4) , and Drug Interactions (7) ] : Loss of therapeutic effect of EDURANT or EDURANT PED and possible development of resistance. In healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in EDURANT) have been shown to prolong the QTc interval of the electrocardiogram. Consider alternatives to EDURANT or EDURANT PED when coadministered with a drug that is known to have a risk of torsade de pointes [see Drug Interactions (7) and Clinical Pharmacology (12.2) ] . See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during EDURANT or EDURANT PED therapy and review concomitant medications during therapy.
Description
Indications and Usage ( 1.1 ) 03/2024 Dosage and Administration ( 2.1 , 2.3 , 2.4 , 2.5 ) 03/2024 Warnings and Precautions ( 5.6 ) 03/2024
Section 42229-5
Limitations of Use
- More EDURANT treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA ≥50 copies/mL) compared to EDURANT treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL [see Clinical Studies (14.1)] .
Section 42230-3
| PATIENT INFORMATION | ||||
|---|---|---|---|---|
| EDURANT
® (ee dur ant)
(rilpivirine) tablets, for oral use |
EDURANT
® PED (ee dur ant ped)
(rilpivirine) tablets, for oral suspension |
|||
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2024 | |||
What are EDURANT and EDURANT PED?
If you take EDURANT in combination with oral VOCABRIA (cabotegravir), you should also read the Patient Information that comes with oral VOCABRIA (cabotegravir). It is not known if EDURANT or EDURANT PED is safe and effective in children less than 2 years of age or who weigh less than 31 Ibs (14 kg). |
||||
| Do not take EDURANT or EDURANT PED if you are taking any of the following medicines: | ||||
|
|
|||
Before taking EDURANT or EDURANT PED, tell your healthcare provider about all your medical conditions, including if you:
|
||||
|
How should I take EDURANT or EDURANT PED?
See the " Instructions for Use" for detailed instructions on how to prepare and give a dose of EDURANT PED tablets for oral suspension.
|
||||
|
What are the possible side effects of EDURANT and EDURANT PED?
EDURANT and EDURANT PED can cause serious side effects including:
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
|
The most common side effects of EDURANT or EDURANT PED include depression, headache, trouble sleeping (insomnia) and rash.
These are not all the possible side effects of EDURANT or EDURANT PED. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store EDURANT or EDURANT PED?
|
||||
|
General information about the safe and effective use of EDURANT and EDURANT PED.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use EDURANT or EDURANT PED for a condition for which it was not prescribed. Do not give EDURANT or EDURANT PED to other people even if they have the same condition you have. It may harm them. You can ask your healthcare provider or pharmacist for information about EDURANT or EDURANT PED that is written for health professionals. |
||||
|
What are the ingredients in EDURANT and EDURANT PED?
Active ingredient: rilpivirine. Inactive ingredients: EDURANT 25 mg tablets: croscarmellose sodium, lactose monohydrate, magnesium stearate, polysorbate 20, povidone K30 and silicified microcrystalline cellulose. The tablet coating contains hypromellose 2910 6 mPa.s, lactose monohydrate, PEG 3000, titanium dioxide and triacetin. EDURANT PED 2.5 mg tablets for oral suspension: croscarmellose sodium, lactose monohydrate, mannitol, microcrystalline cellulose, povidone K30, polysorbate 20, sodium lauryl sulfate and sodium stearyl fumarate. Manufactured for: Janssen Products, LP, Horsham PA 19044, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2011, 2024 For more information go to www.EDURANT.com or call 1-800-526-7736 |
Section 43683-2
Section 44425-7
EDURANT Tablets
EDURANT ® (rilpivirine) 25 mg tablets are supplied as white to off-white, film-coated, round, biconvex, 6.4 mm tablets. Each tablet contains 27.5 mg of rilpivirine hydrochloride, which is equivalent to 25 mg of rilpivirine. Each tablet is debossed with "TMC" on one side and "25" on the other side.
EDURANT 25 mg tablets are packaged in bottles in the following configuration: 25 mg tablets-bottles of 30 (NDC 59676-278-01).
Store EDURANT tablets in the original bottle in order to protect from light. Store EDURANT tablets at 20° to 25°C (68° to 77°F); with excursions permitted to 15° to 30°C (59° to 86°F) [see USP controlled room temperature].
Section 59845-8
|
INSTRUCTIONS FOR USE
EDURANT ® PED ( ee dur ant ped) (rilpivirine) tablets, for oral suspension |
|
|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: 11/2024 |
| This Instructions for Use contains information on how to prepare and give EDURANT PED. Read this Instructions for Use before your child starts taking EDURANT PED for the first time and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your child's medical condition or treatment.
Important information you need to know before giving EDURANT PED:
|
|
Supplies needed to prepare and give EDURANT PED:
|
|
| Step 1: Prepare EDURANT PED | |
|
|
|
| Step 2: Place the tablets in a small cup | |
|
|
|
|
|
|
| If you spill any medicine, clean up the spill. Throw away the rest of the prepared medicine and make a new dose.
You must give the prepared medicine right away. If you do not give the prepared medicine right away, throw away the mixture and prepare a new dose of medicine. |
|
| Step 3: Give EDURANT PED | |
|
|
Step 4: Clean the dosing items
|
|
How should I store EDURANT PED?
|
|
| Manufactured for: Janssen Products, LP, Horsham PA 19044, USA
For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2011, 2024 For more information go to www.EDURANT.com or call 1-800-526-7736 |
10 Overdosage
There is no specific antidote for overdose with EDURANT or EDURANT PED. Human experience of overdose with EDURANT or EDURANT PED is limited. Treatment of overdose with EDURANT or EDURANT PED consists of general supportive measures including monitoring of vital signs and ECG (QT interval) as well as observation of the clinical status of the patient. It is advisable to contact a poison control center to obtain the latest recommendations for the management of an overdose. Since rilpivirine is highly bound to plasma protein, dialysis is unlikely to result in significant removal of the active substance.
11 Description
EDURANT ® (rilpivirine) is a non-nucleoside reverse transcriptase inhibitor (NNRTI) of human immunodeficiency virus type 1 (HIV-1).
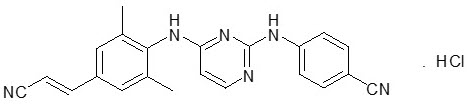
The chemical name for rilpivirine hydrochloride is 4-[[4-[[4-[(E)-2-cyanoethenyl]-2,6-dimethylphenyl]amino]-2-pyrimidinyl]amino]benzonitrile monohydrochloride. Its molecular formula is C 22H 18N 6∙ HCl and its molecular weight is 402.88. Rilpivirine hydrochloride has the following structural formula:
Rilpivirine hydrochloride is a white to almost white powder. Rilpivirine hydrochloride is practically insoluble in water over a wide pH range.
EDURANT 25 mg tablets are available as a white to off-white, film-coated, round, biconvex, 6.4 mm tablet for oral administration. Each tablet contains 27.5 mg of rilpivirine hydrochloride, which is equivalent to 25 mg of rilpivirine. Each EDURANT 25 mg tablet also contains the inactive ingredients croscarmellose sodium, lactose monohydrate, magnesium stearate, polysorbate 20, povidone K30 and silicified microcrystalline cellulose. The tablet coating contains hypromellose 2910 6 mPa.s, lactose monohydrate, PEG 3000, titanium dioxide and triacetin.
EDURANT PED 2.5 mg tablets for oral suspension are available as white to almost white, round 6.5 mm tablet, debossed with "TMC" on one side and "PED" on the other side. Each tablet for oral suspension contains 2.75 mg of rilpivirine hydrochloride equivalent to 2.5 mg rilpivirine. Each tablet for oral suspension also contains the inactive ingredients croscarmellose sodium, lactose monohydrate, mannitol, microcrystalline cellulose, polysorbate 20, povidone K30, sodium lauryl sulfate and sodium stearyl fumarate.
8.4 Pediatric Use
The safety and effectiveness of EDURANT and EDURANT PED has been established for the treatment of HIV-1 infection in treatment-naïve pediatric patients 2 years of age and older and weighing at least 14 kg. Use of EDURANT or EDURANT PED in this population is supported by three trials: TMC278-C213, TMC278HTX2002 and MOCHA.
8.5 Geriatric Use
Clinical studies of EDURANT did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, caution should be exercised in the administration and monitoring of EDURANT in elderly patients reflecting the greater frequency of decreased renal and hepatic function, and of concomitant disease or other drug therapy.
5.2 Hepatotoxicity
Hepatic adverse events have been reported in patients receiving a rilpivirine-containing regimen. Patients with underlying hepatitis B or C virus infection, or marked elevations in transaminases prior to treatment may be at increased risk for worsening or development of transaminase elevations with use of EDURANT or EDURANT PED. A few cases of hepatic toxicity have been reported in adult patients receiving a rilpivirine-containing regimen who had no pre-existing hepatic disease or other identifiable risk factors. Appropriate laboratory testing prior to initiating therapy and monitoring for hepatotoxicity during therapy with EDURANT or EDURANT PED is recommended in patients with underlying hepatic disease such as hepatitis B or C virus infection, or in patients with marked elevations in transaminases prior to treatment initiation. Liver enzyme monitoring should also be considered for patients without pre-existing hepatic dysfunction or other risk factors.
4 Contraindications
EDURANT and EDURANT PED are contraindicated for coadministration with the drugs in Table 2 for which significant decreases in rilpivirine plasma concentrations may occur due to CYP3A enzyme induction or gastric pH increase, which may result in loss of virologic response and possible resistance to EDURANT or EDURANT PED or to the class of NNRTIs [see Drug Interactions (7)and Clinical Pharmacology (12.3)] .
| Drug Class | Contraindicated Drugs in Class | Clinical Comment |
|---|---|---|
| Anticonvulsants | Carbamazepine
Oxcarbazepine Phenobarbital Phenytoin |
Potential for significant decreases in rilpivirine plasma concentrations due to CYP3A enzyme induction, which may result in loss of virologic response. |
| Antimycobacterials | Rifampin
Rifapentine |
|
| Glucocorticoid
(systemic) |
Dexamethasone
(more than a single-dose treatment) |
|
| Herbal Products | St. John's wort
( Hypericum perforatum) |
|
| Proton Pump Inhibitors | e.g., Esomeprazole
Lansoprazole Omeprazole Pantoprazole Rabeprazole |
Potential for significant decreases in rilpivirine plasma concentrations due to gastric pH increase, which may result in loss of virologic response. |
6 Adverse Reactions
The following adverse reactions are discussed below and in other sections of the labeling:
- Skin and Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Depressive Disorders [see Warnings and Precautions (5.3)]
7 Drug Interactions
Rilpivirine is primarily metabolized by cytochrome P450 (CYP)3A, and drugs that induce or inhibit CYP3A may thus affect the clearance of rilpivirine. Coadministration of EDURANT or EDURANT PED and drugs that induce CYP3A may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance to rilpivirine or to the class of NNRTIs. Coadministration of EDURANT or EDURANT PED and drugs that inhibit CYP3A may result in increased plasma concentrations of rilpivirine. Coadministration of EDURANT or EDURANT PED with drugs that increase gastric pH may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance to rilpivirine or to the class of NNRTIs.
EDURANT or EDURANT PED at the recommended doses are not likely to have a clinically relevant effect on the exposure of drugs metabolized by CYP enzymes.
Table 6 shows the established and other potentially significant drug interactions based on which alterations in dose or regimen of EDURANT or EDURANT PED and/or coadministered drug may be recommended. Drugs that are not recommended for coadministration with EDURANT or EDURANT PED are also included in Table 6 [see Dosage and Administration (2), Contraindications (4), and Clinical Pharmacology (12.3)].
| Concomitant Drug Class:
Drug Name |
Effect on Concentration of Rilpivirine or Concomitant Drug | Clinical Comment |
|---|---|---|
| ↑=increase, ↓=decrease, ↔=no change | ||
|
Antacids:
antacids (e.g., aluminum or magnesium hydroxide, calcium carbonate) |
↔ rilpivirine
(antacids taken at least 2 hours before or at least 4 hours after rilpivirine) |
The combination of EDURANT or EDURANT PED and antacids should be used with caution as coadministration may cause significant decreases in rilpivirine plasma concentrations (increase in gastric pH). Antacids should only be administered either at least 2 hours before or at least 4 hours after EDURANT or EDURANT PED. |
| ↓ rilpivirine
(concomitant intake) |
||
|
Anticonvulsants:
carbamazepine oxcarbazepine phenobarbital phenytoin |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
|
Antimycobacterials:
rifampin rifapentine |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
|
Antimycobacterials:
rifabutin The interaction between EDURANT and the drug was evaluated in a clinical study. All other drug-drug interactions shown are predicted.
|
↓ rilpivirine | Concomitant use of EDURANT with rifabutin may cause a decrease in the plasma concentrations of rilpivirine (induction of CYP3A enzymes). Throughout coadministration of EDURANT with rifabutin, the EDURANT dose should be increased from 25 mg once daily to 50 mg once daily. When rifabutin coadministration is stopped, the EDURANT dose should be decreased to 25 mg once daily. |
|
Azole Antifungal Agents:
fluconazole itraconazole ketoconazole This interaction study has been performed with a dose higher than the recommended dose for EDURANT assessing the maximal effect on the coadministered drug. The dosing recommendation is applicable to the recommended doses of EDURANT once daily.
posaconazole voriconazole |
↑ rilpivirine
↓ ketoconazole |
Concomitant use of EDURANT or EDURANT PED with azole antifungal agents may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No rilpivirine dose adjustment is required when EDURANT or EDURANT PED is coadministered with azole antifungal agents. Clinically monitor for breakthrough fungal infections when azole antifungals are coadministered with EDURANT or EDURANT PED. |
|
Glucocorticoid (systemic):
dexamethasone (more than a single-dose treatment) |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
|
H
2-Receptor Antagonists:
cimetidine famotidine nizatidine ranitidine |
↔ rilpivirine
(famotidine taken 12 hours before rilpivirine or 4 hours after rilpivirine) |
The combination of EDURANT or EDURANT PED and H 2-receptor antagonists should be used with caution as coadministration may cause significant decreases in rilpivirine plasma concentrations (increase in gastric pH). H 2-receptor antagonists should only be administered at least 12 hours before or at least 4 hours after EDURANT or EDURANT PED. |
| ↓ rilpivirine
(famotidine taken 2 hours before rilpivirine) |
||
|
Herbal Products:
St. John's wort ( Hypericum perforatum) |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
| HIV-Antiviral Agents: Non-nucleoside Reverse Transcriptase Inhibitors (NNRTIs) | ||
| NNRTI
(delavirdine) |
↑ rilpivirine
↔ delavirdine |
It is not recommended to coadminister EDURANT or EDURANT PED with delavirdine and other NNRTIs. |
| Other NNRTIs
(efavirenz, etravirine, nevirapine) |
↓ rilpivirine
↔ other NNRTIs |
|
| HIV-Antiviral Agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) | ||
| didanosine | ↔ rilpivirine
↔ didanosine |
No dose adjustment is required when EDURANT or EDURANT PED is coadministered with didanosine. Didanosine is to be administered on an empty stomach and at least two hours before or at least four hours after EDURANT or EDURANT PED (which should be administered with a meal). |
| HIV-Antiviral Agents: Protease Inhibitors (PIs)-Boosted (i.e., with coadministration of low-dose ritonavir) or Unboosted (i.e., without coadministration of low-dose ritonavir) | ||
| darunavir/ritonavir | ↑ rilpivirine
↔ boosted darunavir |
Concomitant use of EDURANT or EDURANT PED with darunavir/ritonavir may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No dose adjustment is required when EDURANT or EDURANT PED is coadministered with darunavir/ritonavir. |
| Lopinavir/ritonavir | ↑ rilpivirine
↔ boosted lopinavir |
Concomitant use of EDURANT or EDURANT PED with lopinavir/ritonavir may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No dose adjustment is required when EDURANT or EDURANT PED is coadministered with lopinavir/ritonavir. |
| Other boosted PIs
(atazanavir/ritonavir, fosamprenavir/ritonavir, saquinavir/ritonavir, tipranavir/ritonavir) |
↑ rilpivirine
↔ boosted PI |
Concomitant use of EDURANT or EDURANT PED with boosted PIs may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). EDURANT or EDURANT PED is not expected to affect the plasma concentrations of coadministered PIs. |
| Unboosted PIs (atazanavir, fosamprenavir, indinavir, nelfinavir) | ↑ rilpivirine
↔ unboosted PI |
Concomitant use of EDURANT or EDURANT PED with unboosted PIs may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). EDURANT or EDURANT PED is not expected to affect the plasma concentrations of coadministered PIs. |
|
Macrolide or ketolide antibiotics:
azithromycin clarithromycin erythromycin |
↑ rilpivirine
↔ azithromycin ↔ clarithromycin ↔ erythromycin |
Macrolides are expected to increase concentrations of rilpivirine and are associated with a risk of Torsade de Pointes [Warnings and Precautions (5.4)].Where possible, consider alternatives, such as azithromycin, which increases rilpivirine concentrations less than other macrolides. |
|
Narcotic Analgesics:
methadone |
↓ R(-) methadone
↓ S(+) methadone |
No dose adjustments are required when initiating coadministration of methadone with EDURANT or EDURANT PED. However, clinical monitoring is recommended as methadone maintenance therapy may need to be adjusted in some patients. |
|
Proton Pump Inhibitors:
e.g., esomeprazole lansoprazole omeprazole pantoprazole rabeprazole |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
In addition to the drugs included in Table 6, the interaction between EDURANT and the following drugs was evaluated in clinical studies and no dose adjustment is needed for either drug [see Clinical Pharmacology (12.3)] : acetaminophen, atorvastatin, chlorzoxazone, cabotegravir, ethinylestradiol, norethindrone, raltegravir, sildenafil, simeprevir and tenofovir disoproxil fumarate. Rilpivirine did not have a clinically significant effect on the pharmacokinetics of digoxin or metformin. No clinically relevant drug-drug interaction is expected when EDURANT or EDURANT PED is coadministered with maraviroc, ribavirin or the NRTIs abacavir, emtricitabine, lamivudine, stavudine and zidovudine.
8.6 Renal Impairment
No dose adjustment of EDURANT or EDURANT PED is required in patients with mild or moderate renal impairment. However, in patients with severe renal impairment or end-stage renal disease, EDURANT or EDURANT PED should be used with caution and with increased monitoring for adverse effects, as rilpivirine concentrations may be increased due to alteration of drug absorption, distribution, and metabolism secondary to renal dysfunction. As rilpivirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)] .
8.7 Hepatic Impairment
No dosage adjustment of EDURANT or EDURANT PED is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. EDURANT and EDURANT PED have not been studied in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)] .
1 Indications and Usage
EDURANT and EDURANT PED are a human immunodeficiency virus type 1 (HIV-1) specific, non-nucleoside reverse transcriptase inhibitor (NNRTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in treatment-naïve patients 2 years of age and older and weighing at least 14 kg with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1).
Limitations of Use:
- More EDURANT treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA ≥50 copies/mL) compared to EDURANT treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1, 14)
EDURANT is indicated in combination with VOCABRIA (cabotegravir), for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine. ( 1.2)
12.1 Mechanism of Action
Rilpivirine is an antiviral drug [see Microbiology (12.4)] .
5.3 Depressive Disorders
The adverse reaction depressive disorders (depressed mood, depression, dysphoria, major depression, mood altered, negative thoughts, suicide attempt, suicidal ideation) has been reported with EDURANT. Patients with severe depressive symptoms should seek immediate medical evaluation to assess the possibility that the symptoms are related to EDURANT or EDURANT PED, and if so, to determine whether the risks of continued therapy outweigh the benefits.
During the Phase 3 trials in adults (N=1368) through 96 weeks, the incidence of depressive disorders (regardless of causality, severity) reported among EDURANT (n=686) or efavirenz (n=682) was 9% and 8%, respectively. Most events were mild or moderate in severity. The incidence of Grade 3 and 4 depressive disorders (regardless of causality) was 1% for both EDURANT and efavirenz. The incidence of discontinuation due to depressive disorders among EDURANT or efavirenz was 1% in each arm. Suicidal ideation was reported in 4 subjects in each arm while suicide attempt was reported in 2 subjects in the EDURANT arm.
During the Phase 2 trial in pediatric subjects 12 to less than 18 years of age (N=36) receiving EDURANT through 48 weeks, the incidence of depressive disorders (regardless of causality, severity) was 19.4% (7/36). Most events were mild or moderate in severity. The incidence of Grade 3 and 4 depressive disorders (regardless of causality) was 5.6% (2/36). None of the subjects discontinued due to depressive disorders. Suicidal ideation and suicide attempt were reported in 1 subject.
5 Warnings and Precautions
- Skin and Hypersensitivity Reactions: Severe skin and hypersensitivity reactions have been reported during postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. Immediately discontinue treatment if hypersensitivity or rash with systemic symptoms or elevations in hepatic serum biochemistries develop and closely monitor clinical status, including hepatic serum biochemistries. ( 5.1)
- Hepatotoxicity: Hepatic adverse events have been reported in patients with underlying liver disease, including hepatitis B or C virus co-infection, or in patients with elevated baseline transaminases. A few cases of hepatotoxicity have occurred in patients with no pre-existing hepatic disease. Monitor liver function tests before and during treatment with EDURANT or EDURANT PED in patients with underlying hepatic disease, such as hepatitis B or C virus co-infection, or marked elevations in transaminase. Also consider monitoring liver functions tests in patients without pre-existing hepatic dysfunction or other risk factors. ( 5.2)
- Depressive Disorders: Severe depressive disorders have been reported. Immediate medical evaluation is recommended for severe depressive disorders. ( 5.3)
- Patients may develop immune reconstitution syndrome. ( 5.5)
2 Dosage and Administration
- One 25 mg EDURANT tablet taken once daily with a meal for patients weighing at least 25 kg. ( 2.2)
- Pediatric patients 2 years of age and older and weighing at least 14 kg to less than 25 kg: Dosage of EDURANT PED is based on body weight. ( 2.3)
- EDURANT PED must be dispersed in drinking water and taken with a meal. ( 2.4)
- Do not substitute EDURANT tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. ( 2.1, 5.6)
- See full prescribing information for dosing information when used in combination with cabotegravir. ( 2.6)
- For pregnant patients who are already on a stable EDURANT regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing more than 25 kg is one 25 mg tablet once daily taken orally with a meal. ( 2.5, 12.3)
- Rifabutin coadministration: Take two 25 mg tablets of EDURANT once daily with a meal for the duration of the rifabutin coadministration. ( 2.7)
3 Dosage Forms and Strengths
6.2 Postmarketing Experience
Adverse reactions have been identified during postmarketing experience in patients receiving a rilpivirine containing regimen. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Renal and Genitourinary Disorders: nephrotic syndrome
Skin and Subcutaneous Tissue Disorders: Severe skin and hypersensitivity reactions including DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms)
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.5 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including EDURANT. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
14.1 Treatment Naïve Adult Subjects
The evidence of efficacy of EDURANT is based on the analyses of 48- and 96-week data from 2 randomized, double-blinded, active controlled, Phase 3 trials TMC278-C209 (ECHO) and TMC278-C215 (THRIVE) in antiretroviral treatment-naïve adults. Antiretroviral treatment-naïve HIV-1 infected subjects enrolled in the Phase 3 trials had a plasma HIV-1 RNA ≥5000 copies/mL and were screened for susceptibility to N(t)RTIs and for absence of specific NNRTI resistance-associated substitutions (RASs). The Phase 3 trials were identical in design, apart from the background regimen (BR). In TMC278-C209, the BR was fixed to the N(t)RTIs, tenofovir disoproxil fumarate plus emtricitabine. In TMC278-C215, the BR consisted of 2 investigator-selected N(t)RTIs: tenofovir disoproxil fumarate plus emtricitabine or zidovudine plus lamivudine or abacavir plus lamivudine. In both trials, randomization was stratified by screening viral load. In TMC278-C215, randomization was also stratified by N(t)RTI BR.
In the pooled analysis for TMC278-C209 and TMC278-C215, demographics and baseline characteristics were balanced between the EDURANT arm and the efavirenz arm. Table 14 displays selected demographic and baseline disease characteristics of the subjects in the EDURANT and efavirenz arms.
| Pooled Data from the Phase 3 TMC278-C209 and TMC278-C215 Trials | ||
|---|---|---|
| EDURANT + BR
N=686 |
Efavirenz + BR
N=682 |
|
| BR=background regimen | ||
| Demographic Characteristics | ||
| Median Age, years (range) | 36 (18–78) | 36 (19–69) |
| Sex | ||
| Male | 76% | 76% |
| Female | 24% | 24% |
| Race | ||
| White | 61% | 60% |
| Black/African American | 24% | 23% |
| Asian | 11% | 14% |
| Other | 2% | 2% |
| Not allowed to ask per local regulations | 1% | 1% |
| Baseline Disease Characteristics | ||
| Median Baseline Plasma HIV-1 RNA (range), log 10copies/mL | 5.0 (2–7) | 5.0 (3–7) |
| Percentage of Patients with Baseline Plasma Viral Load: | ||
| ≤100,000 | 54% | 48% |
| >100,000 to ≤500,000 | 36% | 40% |
| >500,000 | 10% | 12% |
| Median Baseline CD4+ Cell Count (range), cells/mm 3 | 249 (1–888) | 260 (1–1137) |
| Percentage of Subjects with: | ||
| Hepatitis B/C Virus Co-infection | 7% | 10% |
| Percentage of Patients with the Following Background Regimens: | ||
| tenofovir disoproxil fumarate plus emtricitabine | 80% | 80% |
| zidovudine plus lamivudine | 15% | 15% |
| abacavir plus lamivudine | 5% | 5% |
Week 96 efficacy outcomes for subjects treated with EDURANT 25 mg once daily from the pooled analysis are shown in Table 15. The incidence of virologic failure was higher in the EDURANT arm than the efavirenz arm at Week 96. Virologic failures and discontinuations due to adverse events mostly occurred in the first 48 weeks of treatment. Regardless of HIV-1 RNA at the start of therapy, more EDURANT treated subjects with CD4+ cell count less than 200 cells/mm 3 experienced virologic failure compared to EDURANT treated subjects with CD4+ cell count greater than or equal to 200 cells/mm 3.
| EDURANT + BR
N=686 |
Efavirenz + BR
N=682 |
|
|---|---|---|
| N=total number of subjects per treatment group; BR=background regimen. | ||
| Note: Analysis was based on the last observed viral load data within the Week 96 window (Week 90–103), respectively. | ||
|
HIV-1 RNA <50 copies/mL
CI=Predicted difference (95% CI) of response rate is -0.2 (-4.7; 4.3) at Week 96.
|
76% | 77% |
|
HIV-1 RNA ≥50 copies/mL
Includes subjects who had ≥50 copies/mL in the Week 96 window, subjects who discontinued early due to lack or loss of efficacy, subjects who discontinued for reasons other than an adverse event, death or lack or loss of efficacy and at the time of discontinuation had a viral value of ≥50 copies/mL, and subjects who had a switch in background regimen that was not permitted by the protocol.
|
16% | 10% |
|
No virologic data at Week 96 window
Reasons |
||
| Discontinued study due to adverse event or death
Includes subjects who discontinued due to an adverse event or death if this resulted in no on-treatment virologic data in the Week 96 window.
|
4% | 8% |
| Discontinued study for other reasons and last available HIV-1 RNA <50 copies/mL (or missing)
Includes subjects who discontinued for reasons other than an adverse event, death or lack or loss of efficacy, e.g., withdrew consent, loss to follow-up, etc.
|
4% | 5% |
| Missing data during window but on study | <1% | <1% |
| HIV-1 RNA <50 copies/mL by Baseline HIV-1 RNA (copies/mL) | ||
| ≤100,000 | 82% | 78% |
| >100,000 | 70% | 75% |
| HIV-1 RNA ≥50 copies/mL by Baseline HIV-1 RNA (copies/mL) | ||
| ≤100,000 | 9% | 8% |
| >100,000 | 24% | 11% |
| HIV-1 RNA <50 copies/mL by CD4+ cell count (cells/mm 3) | ||
| <200 | 68% | 74% |
| ≥200 | 81% | 77% |
| HIV-1 RNA ≥50 copies/mL by CD4+ cell count (cells/mm 3) | ||
| <200 | 27% | 10% |
| ≥200 | 10% | 9% |
At Week 96, the mean CD4+ cell count increase from baseline was 228 cells/mm 3 for EDURANT-treated subjects and 219 cells/mm 3 for efavirenz-treated subjects in the pooled analysis of the TMC278-C209 and TMC278-C215 trials.
Study TMC278-C204 was a randomized, active-controlled, Phase 2b trial in antiretroviral treatment-naïve HIV-1-infected adult subjects consisting of 2 parts: an initial 96 weeks, partially-blinded dose-finding part [EDURANT doses blinded] followed by a long-term, open-label part. After Week 96, subjects randomized to one of the 3 doses of EDURANT were switched to EDURANT 25 mg once daily. Subjects in the control arm received efavirenz 600 mg once daily in addition to a BR in both parts of the study. The BR consisted of 2 investigator-selected N(t)RTIs: zidovudine plus lamivudine or tenofovir disoproxil fumarate plus emtricitabine.
Study TMC278-C204 enrolled 368 HIV-1-infected treatment-naïve adult subjects who had a plasma HIV-1 RNA ≥5000 copies/mL, previously received ≤2 weeks of treatment with an N(t)RTI or protease inhibitor, had no prior use of NNRTIs, and were screened for susceptibility to N(t)RTI and for absence of specific NNRTI RASs.
At 96 weeks, the proportion of subjects with <50 HIV-1 RNA copies/mL receiving EDURANT 25 mg (N=93) compared to subjects receiving efavirenz (N=89) was 76% and 71%, respectively. The mean increase from baseline in CD4+ counts was 146 cells/mm 3in subjects receiving EDURANT 25 mg and 160 cells/mm 3in subjects receiving efavirenz.
At 240 weeks, 60% (56/93) of subjects who originally received 25 mg once daily achieved HIV RNA <50 copies/mL compared to 57% (51/89) of subjects in the control group.
2.1 Overview of Different Dosage Forms
EDURANT is available in two dosage forms:
- EDURANT 25 mg film-coated tablets for adults and pediatric patients weighing at least 25 kg.
- EDURANT PED 2.5 mg tablets for oral suspension should only be given to pediatric patients weighing at least 14 kg to less than 25 kg [see Dosage and Administration (2.3)] .
Do not substitute EDURANT tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. A difference in bioavailability between 1 × 25 mg film-coated tablet and 10 × 2.5 mg tablets for oral suspension was observed; therefore, they are NOT substitutable [see Warnings and Precautions (5.6)and Clinical Pharmacology (12.3)].
Take EDURANT and EDURANT PED once daily with a meal in combination with other antiretrovirals [see Clinical Pharmacology (12.3)].
2.5 Recommended Dosage During Pregnancy
For pregnant patients who are already on a stable EDURANT regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing at least 25 kg is one 25 mg tablet once daily taken orally with a meal. Refer to Table 1 for dosing recommendations for pediatric patients [see Dosage and Administration (2.2, 2.3)]. Lower exposures of rilpivirine were observed during pregnancy, therefore viral load should be monitored closely [see Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
5.1 Skin and Hypersensitivity Reactions
Severe skin and hypersensitivity reactions have been reported during the postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. While some skin reactions were accompanied by constitutional symptoms such as fever, other skin reactions were associated with organ dysfunctions, including elevations in hepatic serum biochemistries. During the Phase 3 clinical trials, treatment-related rashes with at least Grade 2 severity were reported in 3% of subjects receiving EDURANT. No Grade 4 rash was reported. Overall, most rashes were Grade 1 or 2 and occurred in the first four to six weeks of therapy [see Adverse Reactions (6.1and 6.2)] . Discontinue EDURANT or EDURANT PED immediately if signs or symptoms of severe skin or hypersensitivity reactions develop, including but not limited to, severe rash or rash accompanied by fever, blisters, mucosal involvement, conjunctivitis, facial edema, angioedema, hepatitis or eosinophilia. Clinical status including laboratory parameters should be monitored and appropriate therapy should be initiated.
5.6 Different Formulations Are Not Substitutable
EDURANT and EDURANT PED have differing pharmacokinetic profiles and are not substitutable on a milligram-per-milligram basis. A difference in bioavailability between 1 × 25 mg film-coated tablet and 10 × 2.5 mg tablets for oral suspension was observed; therefore, they are not substitutable [see Clinical Pharmacology (12.3)]. When a pediatric patient weighs 25 kg or greater, they must switch from EDURANT PED tablets for oral suspension to one 25 mg EDURANT tablet daily [see Dosage and Administration (2.3)]. Incorrect dosing of a given formulation may result in underdosing and loss of therapeutic effect and possible development of resistance or possible clinically significant adverse reactions from greater exposure to rilpivirine.
1.1 Treatment of Hiv 1 in Treatment Naïve Patients
EDURANT and EDURANT PED, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-naïve patients 2 years of age and older and weighing at least 14 kg with plasma HIV-1 RNA less than or equal to 100,000 copies/mL at the start of therapy.
Principal Display Panel 25 Mg Tablet Bottle Label
30 Tablets
NDC59676-278-01
EDURANT
®
(rilpivirine) tablets
25 mg
Each tablet contains 27.5 mg of rilpivirine
hydrochloride, which is equivalent to 25 mg
of rilpivirine.
ALERT: Find out about
medicines that should NOT be
taken with EDURANT
®from
your healthcare provider.
Rx only
2.7 Recommended Dosage With Rifabutin Coadministration
If EDURANT is coadministered with rifabutin, the EDURANT dose should be increased to 50 mg (two 25 mg tablets) once daily, taken with a meal. When rifabutin coadministration is stopped, the EDURANT dose should be decreased to 25 mg once daily, taken with a meal [see Drug Interactions (7)and Clinical Pharmacology (12.3)].
Note that use of CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) with rifabutin is contraindicated. Refer to CABENUVA labeling for additional detail.
1.2 Treatment of Hiv 1 in Combination With Cabotegravir
EDURANT is indicated in combination with VOCABRIA (cabotegravir) for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable antiretroviral regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine, for use as [see Dosage and Administration (2.6)] :
- oral lead-in to assess the tolerability of rilpivirine prior to administration of rilpivirine extended-release injectable suspension, a component of CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension).
- oral therapy for patients who will miss planned injection dosing with CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension).
2.2 Recommended Dosage in Treatment Naïve Adult Patients
The recommended dosage of EDURANT in adult patients is one 25 mg tablet taken orally once daily with a meal [see Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
Principal Display Panel 2.5 Mg Tablet Blister Pack Carton
NDC 59676-280-90
Rx only
EDURANT ® PED
(rilpivirine)
tablets for oral suspension
2.5 mg
90 tablets
9 blister cards, 10 tablets each
Attention: EDURANT PED is NOT a substitute for EDURANT
DISPERSE IN DRINKING WATER
Do not crush, chew, or swallow whole, see Instructions for Use
2.4 Preparation and Administration Instructions for Edurant Ped Only
Advise patients or caregivers of patients taking EDURANT PED to refer to the Instructions for Use to properly prepare and take the medication.
EDURANT PED must be dispersed in drinking water and taken immediately with a meal. If not taken immediately, then the oral suspension should be discarded, and a new dose of medicine should be prepared. The patient should not chew or swallow EDURANT PED whole. The following instructions should be followed:
- Place the tablets for oral suspension in a cup, add 5 mL (1 teaspoon) of drinking water at room temperature. Do not crush the tablets.
- Swirl the cup carefully for 1–2 minutes to disperse the tablets. The oral suspension will start to look cloudy.
- Take all the prepared oral suspension immediately or to aid in administration, the oral suspension can be further diluted with 5 mL (1 teaspoon) of drinking water, milk, orange juice or applesauce. Swirl and take all the medicine immediately. A spoon can be used if needed.
- Make sure the entire dose is taken and no medicine is left in the cup. If required, add another 5 mL (1 teaspoon) of drinking water (or alternative beverage or soft food), swirl and drink immediately.
14.4 Treatment Naïve Pediatric Subjects (2 to Less Than 12 Years of Age)
The pharmacokinetics, safety, tolerability and efficacy of EDURANT and EDURANT PED weight-adjusted doses 25, 15 and 12.5 mg once daily in combination with an investigator-selected BR containing two NRTIs, was evaluated in trial TMC278-C213 Cohort 2, a single-arm, open-label Phase 2 trial in antiretroviral treatment-naïve HIV-1 infected pediatric subjects 6 to less than 12 years of age and weighing at least 17 kg. The Week 48 analysis included 18 subjects, 17 (94%) subjects completed the 48-week treatment period, and 1 (6%) subject discontinued the study early due to reaching a virologic endpoint. The 18 subjects had a median age of 9 years (range 6 to 11 years) and the median weight at baseline was 25 kg (range 17 to 51 kg). 89% were Black and 39% were female. The median baseline plasma viral load was 55,400 (range 567–149,000) copies/mL, and the median absolute baseline CD4+ cell count was 432.5 (range 12–2,068) cells/µL.
The number of subjects with HIV-1 RNA <50 copies/mL at Week 48 was 13/18 (72%), while 3/18 (17%) subjects had HIV-1 RNA ≥50 copies/mL at Week 48 [see Microbiology (12.4)] . Two out of 18 (11%) participants in the 15 mg once daily (20 to ≤25 kg) dose-weight group had missing viral load data at Week 48 but remained on study. The viral load for these 2 subjects was <50 copies/mL, post-Week 48. The mean increase (SE) in CD4+ from baseline was 215.9 (62.42) cells/µL at Week 48.
The safety and efficacy of EDURANT and EDURANT PED in treatment naïve pediatric subjects 2 to less than 6 years of age is supported by evidence from adequate and well-controlled studies of EDURANT in adults with additional population pharmacokinetic data from adults and pediatric subjects 6 years and older [see Use in Specific Populations (8.4)and Clinical Pharmacology (12.3)] .
14.2 Virologically Suppressed Adults Treated in Combination With Cabotegravir
The use of EDURANT in combination with VOCABRIA (cabotegravir) as an oral lead-in and in patients who miss planned injections with CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) was evaluated in two Phase 3 randomized, multicenter, active-controlled, parallel-arm, open-label, non-inferiority trials (Trial 201584: FLAIR [NCT02938520], Trial 201585: ATLAS [NCT2951052]), and one Phase 3b randomized, multicenter, parallel-group, open-label, non-inferiority trial (Trial 207966: ATLAS-2M [NCT03299049]) in subjects who were virologically suppressed (HIV-1 RNA <50 copies/mL). See full prescribing information for VOCABRIA and CABENUVA for additional information.
14.3 Treatment Naïve Pediatric Subjects (≥12 to Less Than 18 Years of Age)
The pharmacokinetics, safety, tolerability and efficacy of EDURANT 25 mg once daily, in combination with an investigator-selected background regimen (BR) containing two NRTIs, was evaluated in trial TMC278-C213 Cohort 1, a single-arm, open-label Phase 2 trial in antiretroviral treatment-naïve HIV-1 infected pediatric subjects 12 to less than 18 years of age and weighing at least 32 kg. Thirty six (36) subjects were enrolled in the trial to complete at least 48 weeks of treatment. The 36 subjects had a median age of 14.5 years (range: 12 to 17 years), and were 56% female, 89% Black and 11% Asian.
In the efficacy analysis, most subjects (75%; 28/36) had baseline HIV RNA <100,000 copies/mL. For these 28 subjects the median baseline plasma HIV-1 RNA was 44,250 (range: 2,060–92,600 copies/mL) and the median baseline CD4+ cell count was 445.5 cells/mm 3(range: 123 to 983 cells/mm 3).
Among the subjects who had baseline HIV RNA ≤100,000, the proportion with HIV-1 RNA <50 copies/mL at Week 48 was 79% (22/28), versus 50% (4/8) in those with >100,000 copies/mL. The proportion of virologic failures among subjects with a baseline viral load ≤100,000 copies/mL was 21% (6/28), versus 38% (3/8) in those with >100,000 copies/mL. At Week 48, the mean increase in CD4+ cell count from baseline was 201.2 cells/mm 3.
5.4 Risk of Adverse Reactions Or Loss of Virologic Response Due to Drug Interactions
The concomitant use of EDURANT or EDURANT PED and other drugs may result in potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.7), Contraindications (4), and Drug Interactions (7)] :
- Loss of therapeutic effect of EDURANT or EDURANT PED and possible development of resistance.
In healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in EDURANT) have been shown to prolong the QTc interval of the electrocardiogram. Consider alternatives to EDURANT or EDURANT PED when coadministered with a drug that is known to have a risk of torsade de pointes [see Drug Interactions (7) and Clinical Pharmacology (12.2)] .
See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during EDURANT or EDURANT PED therapy and review concomitant medications during therapy.
2.3 Recommended Dosage in Treatment Naïve Pediatric Patients 2 Years of Age and Older and Weighing At Least 14 Kg
The recommended dosage of EDURANT and EDURANT PED in pediatric patients 2 years of age and older and weighing at least 14 kg is based on body weight (see Table 1). Both EDURANT and EDURANT PED should be taken orally once daily with a meal [see Use in Specific Populations (8.4)and Clinical Pharmacology (12.3)] .
| Body Weight (kg) | EDURANT 25 mg Tablets | EDURANT PED 2.5 mg Tablets for Oral Suspension | Total Daily Dose |
|---|---|---|---|
| 14 kg to less than 20 kg | Not recommended | 5 tablets once daily | 12.5 mg EDURANT PED once daily |
| 20 kg to less than 25 kg | Not recommended | 6 tablets once daily | 15 mg EDURANT PED once daily |
| Greater than or equal to 25 kg | 1 tablet once daily | Not recommended | 25 mg EDURANT once daily |
2.6 Recommended Dosage in Combination With Cabotegravir in Adults and Adolescents 12 Years of Age and Older and Weighing At Least 35 Kg
Consult the prescribing information for CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) before initiating EDURANT to ensure therapy with CABENUVA is appropriate.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
- More EDURANT treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA ≥50 copies/mL) compared to EDURANT treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL [see Clinical Studies (14.1)] .
Section 42230-3 (42230-3)
| PATIENT INFORMATION | ||||
|---|---|---|---|---|
| EDURANT
® (ee dur ant)
(rilpivirine) tablets, for oral use |
EDURANT
® PED (ee dur ant ped)
(rilpivirine) tablets, for oral suspension |
|||
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 11/2024 | |||
What are EDURANT and EDURANT PED?
If you take EDURANT in combination with oral VOCABRIA (cabotegravir), you should also read the Patient Information that comes with oral VOCABRIA (cabotegravir). It is not known if EDURANT or EDURANT PED is safe and effective in children less than 2 years of age or who weigh less than 31 Ibs (14 kg). |
||||
| Do not take EDURANT or EDURANT PED if you are taking any of the following medicines: | ||||
|
|
|||
Before taking EDURANT or EDURANT PED, tell your healthcare provider about all your medical conditions, including if you:
|
||||
|
How should I take EDURANT or EDURANT PED?
See the " Instructions for Use" for detailed instructions on how to prepare and give a dose of EDURANT PED tablets for oral suspension.
|
||||
|
What are the possible side effects of EDURANT and EDURANT PED?
EDURANT and EDURANT PED can cause serious side effects including:
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
|
The most common side effects of EDURANT or EDURANT PED include depression, headache, trouble sleeping (insomnia) and rash.
These are not all the possible side effects of EDURANT or EDURANT PED. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store EDURANT or EDURANT PED?
|
||||
|
General information about the safe and effective use of EDURANT and EDURANT PED.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use EDURANT or EDURANT PED for a condition for which it was not prescribed. Do not give EDURANT or EDURANT PED to other people even if they have the same condition you have. It may harm them. You can ask your healthcare provider or pharmacist for information about EDURANT or EDURANT PED that is written for health professionals. |
||||
|
What are the ingredients in EDURANT and EDURANT PED?
Active ingredient: rilpivirine. Inactive ingredients: EDURANT 25 mg tablets: croscarmellose sodium, lactose monohydrate, magnesium stearate, polysorbate 20, povidone K30 and silicified microcrystalline cellulose. The tablet coating contains hypromellose 2910 6 mPa.s, lactose monohydrate, PEG 3000, titanium dioxide and triacetin. EDURANT PED 2.5 mg tablets for oral suspension: croscarmellose sodium, lactose monohydrate, mannitol, microcrystalline cellulose, povidone K30, polysorbate 20, sodium lauryl sulfate and sodium stearyl fumarate. Manufactured for: Janssen Products, LP, Horsham PA 19044, USA For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2011, 2024 For more information go to www.EDURANT.com or call 1-800-526-7736 |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
EDURANT Tablets
EDURANT ® (rilpivirine) 25 mg tablets are supplied as white to off-white, film-coated, round, biconvex, 6.4 mm tablets. Each tablet contains 27.5 mg of rilpivirine hydrochloride, which is equivalent to 25 mg of rilpivirine. Each tablet is debossed with "TMC" on one side and "25" on the other side.
EDURANT 25 mg tablets are packaged in bottles in the following configuration: 25 mg tablets-bottles of 30 (NDC 59676-278-01).
Store EDURANT tablets in the original bottle in order to protect from light. Store EDURANT tablets at 20° to 25°C (68° to 77°F); with excursions permitted to 15° to 30°C (59° to 86°F) [see USP controlled room temperature].
Section 59845-8 (59845-8)
|
INSTRUCTIONS FOR USE
EDURANT ® PED ( ee dur ant ped) (rilpivirine) tablets, for oral suspension |
|
|---|---|
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: 11/2024 |
| This Instructions for Use contains information on how to prepare and give EDURANT PED. Read this Instructions for Use before your child starts taking EDURANT PED for the first time and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your child's medical condition or treatment.
Important information you need to know before giving EDURANT PED:
|
|
Supplies needed to prepare and give EDURANT PED:
|
|
| Step 1: Prepare EDURANT PED | |
|
|
|
| Step 2: Place the tablets in a small cup | |
|
|
|
|
|
|
| If you spill any medicine, clean up the spill. Throw away the rest of the prepared medicine and make a new dose.
You must give the prepared medicine right away. If you do not give the prepared medicine right away, throw away the mixture and prepare a new dose of medicine. |
|
| Step 3: Give EDURANT PED | |
|
|
Step 4: Clean the dosing items
|
|
How should I store EDURANT PED?
|
|
| Manufactured for: Janssen Products, LP, Horsham PA 19044, USA
For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2011, 2024 For more information go to www.EDURANT.com or call 1-800-526-7736 |
10 Overdosage (10 OVERDOSAGE)
There is no specific antidote for overdose with EDURANT or EDURANT PED. Human experience of overdose with EDURANT or EDURANT PED is limited. Treatment of overdose with EDURANT or EDURANT PED consists of general supportive measures including monitoring of vital signs and ECG (QT interval) as well as observation of the clinical status of the patient. It is advisable to contact a poison control center to obtain the latest recommendations for the management of an overdose. Since rilpivirine is highly bound to plasma protein, dialysis is unlikely to result in significant removal of the active substance.
11 Description (11 DESCRIPTION)
EDURANT ® (rilpivirine) is a non-nucleoside reverse transcriptase inhibitor (NNRTI) of human immunodeficiency virus type 1 (HIV-1).
The chemical name for rilpivirine hydrochloride is 4-[[4-[[4-[(E)-2-cyanoethenyl]-2,6-dimethylphenyl]amino]-2-pyrimidinyl]amino]benzonitrile monohydrochloride. Its molecular formula is C 22H 18N 6∙ HCl and its molecular weight is 402.88. Rilpivirine hydrochloride has the following structural formula:
Rilpivirine hydrochloride is a white to almost white powder. Rilpivirine hydrochloride is practically insoluble in water over a wide pH range.
EDURANT 25 mg tablets are available as a white to off-white, film-coated, round, biconvex, 6.4 mm tablet for oral administration. Each tablet contains 27.5 mg of rilpivirine hydrochloride, which is equivalent to 25 mg of rilpivirine. Each EDURANT 25 mg tablet also contains the inactive ingredients croscarmellose sodium, lactose monohydrate, magnesium stearate, polysorbate 20, povidone K30 and silicified microcrystalline cellulose. The tablet coating contains hypromellose 2910 6 mPa.s, lactose monohydrate, PEG 3000, titanium dioxide and triacetin.
EDURANT PED 2.5 mg tablets for oral suspension are available as white to almost white, round 6.5 mm tablet, debossed with "TMC" on one side and "PED" on the other side. Each tablet for oral suspension contains 2.75 mg of rilpivirine hydrochloride equivalent to 2.5 mg rilpivirine. Each tablet for oral suspension also contains the inactive ingredients croscarmellose sodium, lactose monohydrate, mannitol, microcrystalline cellulose, polysorbate 20, povidone K30, sodium lauryl sulfate and sodium stearyl fumarate.
8.4 Pediatric Use
The safety and effectiveness of EDURANT and EDURANT PED has been established for the treatment of HIV-1 infection in treatment-naïve pediatric patients 2 years of age and older and weighing at least 14 kg. Use of EDURANT or EDURANT PED in this population is supported by three trials: TMC278-C213, TMC278HTX2002 and MOCHA.
8.5 Geriatric Use
Clinical studies of EDURANT did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, caution should be exercised in the administration and monitoring of EDURANT in elderly patients reflecting the greater frequency of decreased renal and hepatic function, and of concomitant disease or other drug therapy.
5.2 Hepatotoxicity
Hepatic adverse events have been reported in patients receiving a rilpivirine-containing regimen. Patients with underlying hepatitis B or C virus infection, or marked elevations in transaminases prior to treatment may be at increased risk for worsening or development of transaminase elevations with use of EDURANT or EDURANT PED. A few cases of hepatic toxicity have been reported in adult patients receiving a rilpivirine-containing regimen who had no pre-existing hepatic disease or other identifiable risk factors. Appropriate laboratory testing prior to initiating therapy and monitoring for hepatotoxicity during therapy with EDURANT or EDURANT PED is recommended in patients with underlying hepatic disease such as hepatitis B or C virus infection, or in patients with marked elevations in transaminases prior to treatment initiation. Liver enzyme monitoring should also be considered for patients without pre-existing hepatic dysfunction or other risk factors.
4 Contraindications (4 CONTRAINDICATIONS)
EDURANT and EDURANT PED are contraindicated for coadministration with the drugs in Table 2 for which significant decreases in rilpivirine plasma concentrations may occur due to CYP3A enzyme induction or gastric pH increase, which may result in loss of virologic response and possible resistance to EDURANT or EDURANT PED or to the class of NNRTIs [see Drug Interactions (7)and Clinical Pharmacology (12.3)] .
| Drug Class | Contraindicated Drugs in Class | Clinical Comment |
|---|---|---|
| Anticonvulsants | Carbamazepine
Oxcarbazepine Phenobarbital Phenytoin |
Potential for significant decreases in rilpivirine plasma concentrations due to CYP3A enzyme induction, which may result in loss of virologic response. |
| Antimycobacterials | Rifampin
Rifapentine |
|
| Glucocorticoid
(systemic) |
Dexamethasone
(more than a single-dose treatment) |
|
| Herbal Products | St. John's wort
( Hypericum perforatum) |
|
| Proton Pump Inhibitors | e.g., Esomeprazole
Lansoprazole Omeprazole Pantoprazole Rabeprazole |
Potential for significant decreases in rilpivirine plasma concentrations due to gastric pH increase, which may result in loss of virologic response. |
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed below and in other sections of the labeling:
- Skin and Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Depressive Disorders [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Rilpivirine is primarily metabolized by cytochrome P450 (CYP)3A, and drugs that induce or inhibit CYP3A may thus affect the clearance of rilpivirine. Coadministration of EDURANT or EDURANT PED and drugs that induce CYP3A may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance to rilpivirine or to the class of NNRTIs. Coadministration of EDURANT or EDURANT PED and drugs that inhibit CYP3A may result in increased plasma concentrations of rilpivirine. Coadministration of EDURANT or EDURANT PED with drugs that increase gastric pH may result in decreased plasma concentrations of rilpivirine and loss of virologic response and possible resistance to rilpivirine or to the class of NNRTIs.
EDURANT or EDURANT PED at the recommended doses are not likely to have a clinically relevant effect on the exposure of drugs metabolized by CYP enzymes.
Table 6 shows the established and other potentially significant drug interactions based on which alterations in dose or regimen of EDURANT or EDURANT PED and/or coadministered drug may be recommended. Drugs that are not recommended for coadministration with EDURANT or EDURANT PED are also included in Table 6 [see Dosage and Administration (2), Contraindications (4), and Clinical Pharmacology (12.3)].
| Concomitant Drug Class:
Drug Name |
Effect on Concentration of Rilpivirine or Concomitant Drug | Clinical Comment |
|---|---|---|
| ↑=increase, ↓=decrease, ↔=no change | ||
|
Antacids:
antacids (e.g., aluminum or magnesium hydroxide, calcium carbonate) |
↔ rilpivirine
(antacids taken at least 2 hours before or at least 4 hours after rilpivirine) |
The combination of EDURANT or EDURANT PED and antacids should be used with caution as coadministration may cause significant decreases in rilpivirine plasma concentrations (increase in gastric pH). Antacids should only be administered either at least 2 hours before or at least 4 hours after EDURANT or EDURANT PED. |
| ↓ rilpivirine
(concomitant intake) |
||
|
Anticonvulsants:
carbamazepine oxcarbazepine phenobarbital phenytoin |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
|
Antimycobacterials:
rifampin rifapentine |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
|
Antimycobacterials:
rifabutin The interaction between EDURANT and the drug was evaluated in a clinical study. All other drug-drug interactions shown are predicted.
|
↓ rilpivirine | Concomitant use of EDURANT with rifabutin may cause a decrease in the plasma concentrations of rilpivirine (induction of CYP3A enzymes). Throughout coadministration of EDURANT with rifabutin, the EDURANT dose should be increased from 25 mg once daily to 50 mg once daily. When rifabutin coadministration is stopped, the EDURANT dose should be decreased to 25 mg once daily. |
|
Azole Antifungal Agents:
fluconazole itraconazole ketoconazole This interaction study has been performed with a dose higher than the recommended dose for EDURANT assessing the maximal effect on the coadministered drug. The dosing recommendation is applicable to the recommended doses of EDURANT once daily.
posaconazole voriconazole |
↑ rilpivirine
↓ ketoconazole |
Concomitant use of EDURANT or EDURANT PED with azole antifungal agents may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No rilpivirine dose adjustment is required when EDURANT or EDURANT PED is coadministered with azole antifungal agents. Clinically monitor for breakthrough fungal infections when azole antifungals are coadministered with EDURANT or EDURANT PED. |
|
Glucocorticoid (systemic):
dexamethasone (more than a single-dose treatment) |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
|
H
2-Receptor Antagonists:
cimetidine famotidine nizatidine ranitidine |
↔ rilpivirine
(famotidine taken 12 hours before rilpivirine or 4 hours after rilpivirine) |
The combination of EDURANT or EDURANT PED and H 2-receptor antagonists should be used with caution as coadministration may cause significant decreases in rilpivirine plasma concentrations (increase in gastric pH). H 2-receptor antagonists should only be administered at least 12 hours before or at least 4 hours after EDURANT or EDURANT PED. |
| ↓ rilpivirine
(famotidine taken 2 hours before rilpivirine) |
||
|
Herbal Products:
St. John's wort ( Hypericum perforatum) |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
| HIV-Antiviral Agents: Non-nucleoside Reverse Transcriptase Inhibitors (NNRTIs) | ||
| NNRTI
(delavirdine) |
↑ rilpivirine
↔ delavirdine |
It is not recommended to coadminister EDURANT or EDURANT PED with delavirdine and other NNRTIs. |
| Other NNRTIs
(efavirenz, etravirine, nevirapine) |
↓ rilpivirine
↔ other NNRTIs |
|
| HIV-Antiviral Agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) | ||
| didanosine | ↔ rilpivirine
↔ didanosine |
No dose adjustment is required when EDURANT or EDURANT PED is coadministered with didanosine. Didanosine is to be administered on an empty stomach and at least two hours before or at least four hours after EDURANT or EDURANT PED (which should be administered with a meal). |
| HIV-Antiviral Agents: Protease Inhibitors (PIs)-Boosted (i.e., with coadministration of low-dose ritonavir) or Unboosted (i.e., without coadministration of low-dose ritonavir) | ||
| darunavir/ritonavir | ↑ rilpivirine
↔ boosted darunavir |
Concomitant use of EDURANT or EDURANT PED with darunavir/ritonavir may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No dose adjustment is required when EDURANT or EDURANT PED is coadministered with darunavir/ritonavir. |
| Lopinavir/ritonavir | ↑ rilpivirine
↔ boosted lopinavir |
Concomitant use of EDURANT or EDURANT PED with lopinavir/ritonavir may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). No dose adjustment is required when EDURANT or EDURANT PED is coadministered with lopinavir/ritonavir. |
| Other boosted PIs
(atazanavir/ritonavir, fosamprenavir/ritonavir, saquinavir/ritonavir, tipranavir/ritonavir) |
↑ rilpivirine
↔ boosted PI |
Concomitant use of EDURANT or EDURANT PED with boosted PIs may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). EDURANT or EDURANT PED is not expected to affect the plasma concentrations of coadministered PIs. |
| Unboosted PIs (atazanavir, fosamprenavir, indinavir, nelfinavir) | ↑ rilpivirine
↔ unboosted PI |
Concomitant use of EDURANT or EDURANT PED with unboosted PIs may cause an increase in the plasma concentrations of rilpivirine (inhibition of CYP3A enzymes). EDURANT or EDURANT PED is not expected to affect the plasma concentrations of coadministered PIs. |
|
Macrolide or ketolide antibiotics:
azithromycin clarithromycin erythromycin |
↑ rilpivirine
↔ azithromycin ↔ clarithromycin ↔ erythromycin |
Macrolides are expected to increase concentrations of rilpivirine and are associated with a risk of Torsade de Pointes [Warnings and Precautions (5.4)].Where possible, consider alternatives, such as azithromycin, which increases rilpivirine concentrations less than other macrolides. |
|
Narcotic Analgesics:
methadone |
↓ R(-) methadone
↓ S(+) methadone |
No dose adjustments are required when initiating coadministration of methadone with EDURANT or EDURANT PED. However, clinical monitoring is recommended as methadone maintenance therapy may need to be adjusted in some patients. |
|
Proton Pump Inhibitors:
e.g., esomeprazole lansoprazole omeprazole pantoprazole rabeprazole |
↓ rilpivirine | Coadministration is contraindicated with EDURANT or EDURANT PED [see Contraindications (4)] . |
In addition to the drugs included in Table 6, the interaction between EDURANT and the following drugs was evaluated in clinical studies and no dose adjustment is needed for either drug [see Clinical Pharmacology (12.3)] : acetaminophen, atorvastatin, chlorzoxazone, cabotegravir, ethinylestradiol, norethindrone, raltegravir, sildenafil, simeprevir and tenofovir disoproxil fumarate. Rilpivirine did not have a clinically significant effect on the pharmacokinetics of digoxin or metformin. No clinically relevant drug-drug interaction is expected when EDURANT or EDURANT PED is coadministered with maraviroc, ribavirin or the NRTIs abacavir, emtricitabine, lamivudine, stavudine and zidovudine.
8.6 Renal Impairment
No dose adjustment of EDURANT or EDURANT PED is required in patients with mild or moderate renal impairment. However, in patients with severe renal impairment or end-stage renal disease, EDURANT or EDURANT PED should be used with caution and with increased monitoring for adverse effects, as rilpivirine concentrations may be increased due to alteration of drug absorption, distribution, and metabolism secondary to renal dysfunction. As rilpivirine is highly bound to plasma proteins, it is unlikely that it will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)] .
8.7 Hepatic Impairment
No dosage adjustment of EDURANT or EDURANT PED is required in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment. EDURANT and EDURANT PED have not been studied in patients with severe hepatic impairment (Child-Pugh Class C) [see Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
EDURANT and EDURANT PED are a human immunodeficiency virus type 1 (HIV-1) specific, non-nucleoside reverse transcriptase inhibitor (NNRTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in treatment-naïve patients 2 years of age and older and weighing at least 14 kg with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1).
Limitations of Use:
- More EDURANT treated subjects with HIV-1 RNA greater than 100,000 copies/mL at the start of therapy experienced virologic failure (HIV-1 RNA ≥50 copies/mL) compared to EDURANT treated subjects with HIV-1 RNA less than or equal to 100,000 copies/mL. ( 1.1, 14)
EDURANT is indicated in combination with VOCABRIA (cabotegravir), for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine. ( 1.2)
12.1 Mechanism of Action
Rilpivirine is an antiviral drug [see Microbiology (12.4)] .
5.3 Depressive Disorders
The adverse reaction depressive disorders (depressed mood, depression, dysphoria, major depression, mood altered, negative thoughts, suicide attempt, suicidal ideation) has been reported with EDURANT. Patients with severe depressive symptoms should seek immediate medical evaluation to assess the possibility that the symptoms are related to EDURANT or EDURANT PED, and if so, to determine whether the risks of continued therapy outweigh the benefits.
During the Phase 3 trials in adults (N=1368) through 96 weeks, the incidence of depressive disorders (regardless of causality, severity) reported among EDURANT (n=686) or efavirenz (n=682) was 9% and 8%, respectively. Most events were mild or moderate in severity. The incidence of Grade 3 and 4 depressive disorders (regardless of causality) was 1% for both EDURANT and efavirenz. The incidence of discontinuation due to depressive disorders among EDURANT or efavirenz was 1% in each arm. Suicidal ideation was reported in 4 subjects in each arm while suicide attempt was reported in 2 subjects in the EDURANT arm.
During the Phase 2 trial in pediatric subjects 12 to less than 18 years of age (N=36) receiving EDURANT through 48 weeks, the incidence of depressive disorders (regardless of causality, severity) was 19.4% (7/36). Most events were mild or moderate in severity. The incidence of Grade 3 and 4 depressive disorders (regardless of causality) was 5.6% (2/36). None of the subjects discontinued due to depressive disorders. Suicidal ideation and suicide attempt were reported in 1 subject.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Skin and Hypersensitivity Reactions: Severe skin and hypersensitivity reactions have been reported during postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. Immediately discontinue treatment if hypersensitivity or rash with systemic symptoms or elevations in hepatic serum biochemistries develop and closely monitor clinical status, including hepatic serum biochemistries. ( 5.1)
- Hepatotoxicity: Hepatic adverse events have been reported in patients with underlying liver disease, including hepatitis B or C virus co-infection, or in patients with elevated baseline transaminases. A few cases of hepatotoxicity have occurred in patients with no pre-existing hepatic disease. Monitor liver function tests before and during treatment with EDURANT or EDURANT PED in patients with underlying hepatic disease, such as hepatitis B or C virus co-infection, or marked elevations in transaminase. Also consider monitoring liver functions tests in patients without pre-existing hepatic dysfunction or other risk factors. ( 5.2)
- Depressive Disorders: Severe depressive disorders have been reported. Immediate medical evaluation is recommended for severe depressive disorders. ( 5.3)
- Patients may develop immune reconstitution syndrome. ( 5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- One 25 mg EDURANT tablet taken once daily with a meal for patients weighing at least 25 kg. ( 2.2)
- Pediatric patients 2 years of age and older and weighing at least 14 kg to less than 25 kg: Dosage of EDURANT PED is based on body weight. ( 2.3)
- EDURANT PED must be dispersed in drinking water and taken with a meal. ( 2.4)
- Do not substitute EDURANT tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. ( 2.1, 5.6)
- See full prescribing information for dosing information when used in combination with cabotegravir. ( 2.6)
- For pregnant patients who are already on a stable EDURANT regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing more than 25 kg is one 25 mg tablet once daily taken orally with a meal. ( 2.5, 12.3)
- Rifabutin coadministration: Take two 25 mg tablets of EDURANT once daily with a meal for the duration of the rifabutin coadministration. ( 2.7)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
6.2 Postmarketing Experience
Adverse reactions have been identified during postmarketing experience in patients receiving a rilpivirine containing regimen. Because these reactions are reported voluntarily from a population of unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Renal and Genitourinary Disorders: nephrotic syndrome
Skin and Subcutaneous Tissue Disorders: Severe skin and hypersensitivity reactions including DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.5 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including EDURANT. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
14.1 Treatment Naïve Adult Subjects (14.1 Treatment-Naïve Adult Subjects)
The evidence of efficacy of EDURANT is based on the analyses of 48- and 96-week data from 2 randomized, double-blinded, active controlled, Phase 3 trials TMC278-C209 (ECHO) and TMC278-C215 (THRIVE) in antiretroviral treatment-naïve adults. Antiretroviral treatment-naïve HIV-1 infected subjects enrolled in the Phase 3 trials had a plasma HIV-1 RNA ≥5000 copies/mL and were screened for susceptibility to N(t)RTIs and for absence of specific NNRTI resistance-associated substitutions (RASs). The Phase 3 trials were identical in design, apart from the background regimen (BR). In TMC278-C209, the BR was fixed to the N(t)RTIs, tenofovir disoproxil fumarate plus emtricitabine. In TMC278-C215, the BR consisted of 2 investigator-selected N(t)RTIs: tenofovir disoproxil fumarate plus emtricitabine or zidovudine plus lamivudine or abacavir plus lamivudine. In both trials, randomization was stratified by screening viral load. In TMC278-C215, randomization was also stratified by N(t)RTI BR.
In the pooled analysis for TMC278-C209 and TMC278-C215, demographics and baseline characteristics were balanced between the EDURANT arm and the efavirenz arm. Table 14 displays selected demographic and baseline disease characteristics of the subjects in the EDURANT and efavirenz arms.
| Pooled Data from the Phase 3 TMC278-C209 and TMC278-C215 Trials | ||
|---|---|---|
| EDURANT + BR
N=686 |
Efavirenz + BR
N=682 |
|
| BR=background regimen | ||
| Demographic Characteristics | ||
| Median Age, years (range) | 36 (18–78) | 36 (19–69) |
| Sex | ||
| Male | 76% | 76% |
| Female | 24% | 24% |
| Race | ||
| White | 61% | 60% |
| Black/African American | 24% | 23% |
| Asian | 11% | 14% |
| Other | 2% | 2% |
| Not allowed to ask per local regulations | 1% | 1% |
| Baseline Disease Characteristics | ||
| Median Baseline Plasma HIV-1 RNA (range), log 10copies/mL | 5.0 (2–7) | 5.0 (3–7) |
| Percentage of Patients with Baseline Plasma Viral Load: | ||
| ≤100,000 | 54% | 48% |
| >100,000 to ≤500,000 | 36% | 40% |
| >500,000 | 10% | 12% |
| Median Baseline CD4+ Cell Count (range), cells/mm 3 | 249 (1–888) | 260 (1–1137) |
| Percentage of Subjects with: | ||
| Hepatitis B/C Virus Co-infection | 7% | 10% |
| Percentage of Patients with the Following Background Regimens: | ||
| tenofovir disoproxil fumarate plus emtricitabine | 80% | 80% |
| zidovudine plus lamivudine | 15% | 15% |
| abacavir plus lamivudine | 5% | 5% |
Week 96 efficacy outcomes for subjects treated with EDURANT 25 mg once daily from the pooled analysis are shown in Table 15. The incidence of virologic failure was higher in the EDURANT arm than the efavirenz arm at Week 96. Virologic failures and discontinuations due to adverse events mostly occurred in the first 48 weeks of treatment. Regardless of HIV-1 RNA at the start of therapy, more EDURANT treated subjects with CD4+ cell count less than 200 cells/mm 3 experienced virologic failure compared to EDURANT treated subjects with CD4+ cell count greater than or equal to 200 cells/mm 3.
| EDURANT + BR
N=686 |
Efavirenz + BR
N=682 |
|
|---|---|---|
| N=total number of subjects per treatment group; BR=background regimen. | ||
| Note: Analysis was based on the last observed viral load data within the Week 96 window (Week 90–103), respectively. | ||
|
HIV-1 RNA <50 copies/mL
CI=Predicted difference (95% CI) of response rate is -0.2 (-4.7; 4.3) at Week 96.
|
76% | 77% |
|
HIV-1 RNA ≥50 copies/mL
Includes subjects who had ≥50 copies/mL in the Week 96 window, subjects who discontinued early due to lack or loss of efficacy, subjects who discontinued for reasons other than an adverse event, death or lack or loss of efficacy and at the time of discontinuation had a viral value of ≥50 copies/mL, and subjects who had a switch in background regimen that was not permitted by the protocol.
|
16% | 10% |
|
No virologic data at Week 96 window
Reasons |
||
| Discontinued study due to adverse event or death
Includes subjects who discontinued due to an adverse event or death if this resulted in no on-treatment virologic data in the Week 96 window.
|
4% | 8% |
| Discontinued study for other reasons and last available HIV-1 RNA <50 copies/mL (or missing)
Includes subjects who discontinued for reasons other than an adverse event, death or lack or loss of efficacy, e.g., withdrew consent, loss to follow-up, etc.
|
4% | 5% |
| Missing data during window but on study | <1% | <1% |
| HIV-1 RNA <50 copies/mL by Baseline HIV-1 RNA (copies/mL) | ||
| ≤100,000 | 82% | 78% |
| >100,000 | 70% | 75% |
| HIV-1 RNA ≥50 copies/mL by Baseline HIV-1 RNA (copies/mL) | ||
| ≤100,000 | 9% | 8% |
| >100,000 | 24% | 11% |
| HIV-1 RNA <50 copies/mL by CD4+ cell count (cells/mm 3) | ||
| <200 | 68% | 74% |
| ≥200 | 81% | 77% |
| HIV-1 RNA ≥50 copies/mL by CD4+ cell count (cells/mm 3) | ||
| <200 | 27% | 10% |
| ≥200 | 10% | 9% |
At Week 96, the mean CD4+ cell count increase from baseline was 228 cells/mm 3 for EDURANT-treated subjects and 219 cells/mm 3 for efavirenz-treated subjects in the pooled analysis of the TMC278-C209 and TMC278-C215 trials.
Study TMC278-C204 was a randomized, active-controlled, Phase 2b trial in antiretroviral treatment-naïve HIV-1-infected adult subjects consisting of 2 parts: an initial 96 weeks, partially-blinded dose-finding part [EDURANT doses blinded] followed by a long-term, open-label part. After Week 96, subjects randomized to one of the 3 doses of EDURANT were switched to EDURANT 25 mg once daily. Subjects in the control arm received efavirenz 600 mg once daily in addition to a BR in both parts of the study. The BR consisted of 2 investigator-selected N(t)RTIs: zidovudine plus lamivudine or tenofovir disoproxil fumarate plus emtricitabine.
Study TMC278-C204 enrolled 368 HIV-1-infected treatment-naïve adult subjects who had a plasma HIV-1 RNA ≥5000 copies/mL, previously received ≤2 weeks of treatment with an N(t)RTI or protease inhibitor, had no prior use of NNRTIs, and were screened for susceptibility to N(t)RTI and for absence of specific NNRTI RASs.
At 96 weeks, the proportion of subjects with <50 HIV-1 RNA copies/mL receiving EDURANT 25 mg (N=93) compared to subjects receiving efavirenz (N=89) was 76% and 71%, respectively. The mean increase from baseline in CD4+ counts was 146 cells/mm 3in subjects receiving EDURANT 25 mg and 160 cells/mm 3in subjects receiving efavirenz.
At 240 weeks, 60% (56/93) of subjects who originally received 25 mg once daily achieved HIV RNA <50 copies/mL compared to 57% (51/89) of subjects in the control group.
2.1 Overview of Different Dosage Forms
EDURANT is available in two dosage forms:
- EDURANT 25 mg film-coated tablets for adults and pediatric patients weighing at least 25 kg.
- EDURANT PED 2.5 mg tablets for oral suspension should only be given to pediatric patients weighing at least 14 kg to less than 25 kg [see Dosage and Administration (2.3)] .
Do not substitute EDURANT tablets and EDURANT PED tablets for oral suspension on a milligram-per-milligram basis due to differing pharmacokinetic profiles. A difference in bioavailability between 1 × 25 mg film-coated tablet and 10 × 2.5 mg tablets for oral suspension was observed; therefore, they are NOT substitutable [see Warnings and Precautions (5.6)and Clinical Pharmacology (12.3)].
Take EDURANT and EDURANT PED once daily with a meal in combination with other antiretrovirals [see Clinical Pharmacology (12.3)].
2.5 Recommended Dosage During Pregnancy
For pregnant patients who are already on a stable EDURANT regimen prior to pregnancy and who are virologically suppressed (HIV-1 RNA less than 50 copies per mL) the recommended dosage in adults and pediatric patients weighing at least 25 kg is one 25 mg tablet once daily taken orally with a meal. Refer to Table 1 for dosing recommendations for pediatric patients [see Dosage and Administration (2.2, 2.3)]. Lower exposures of rilpivirine were observed during pregnancy, therefore viral load should be monitored closely [see Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
5.1 Skin and Hypersensitivity Reactions
Severe skin and hypersensitivity reactions have been reported during the postmarketing experience, including cases of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), with rilpivirine-containing regimens. While some skin reactions were accompanied by constitutional symptoms such as fever, other skin reactions were associated with organ dysfunctions, including elevations in hepatic serum biochemistries. During the Phase 3 clinical trials, treatment-related rashes with at least Grade 2 severity were reported in 3% of subjects receiving EDURANT. No Grade 4 rash was reported. Overall, most rashes were Grade 1 or 2 and occurred in the first four to six weeks of therapy [see Adverse Reactions (6.1and 6.2)] . Discontinue EDURANT or EDURANT PED immediately if signs or symptoms of severe skin or hypersensitivity reactions develop, including but not limited to, severe rash or rash accompanied by fever, blisters, mucosal involvement, conjunctivitis, facial edema, angioedema, hepatitis or eosinophilia. Clinical status including laboratory parameters should be monitored and appropriate therapy should be initiated.
5.6 Different Formulations Are Not Substitutable
EDURANT and EDURANT PED have differing pharmacokinetic profiles and are not substitutable on a milligram-per-milligram basis. A difference in bioavailability between 1 × 25 mg film-coated tablet and 10 × 2.5 mg tablets for oral suspension was observed; therefore, they are not substitutable [see Clinical Pharmacology (12.3)]. When a pediatric patient weighs 25 kg or greater, they must switch from EDURANT PED tablets for oral suspension to one 25 mg EDURANT tablet daily [see Dosage and Administration (2.3)]. Incorrect dosing of a given formulation may result in underdosing and loss of therapeutic effect and possible development of resistance or possible clinically significant adverse reactions from greater exposure to rilpivirine.
1.1 Treatment of Hiv 1 in Treatment Naïve Patients (1.1 Treatment of HIV-1 in Treatment-Naïve Patients)
EDURANT and EDURANT PED, in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in antiretroviral treatment-naïve patients 2 years of age and older and weighing at least 14 kg with plasma HIV-1 RNA less than or equal to 100,000 copies/mL at the start of therapy.
Principal Display Panel 25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label)
30 Tablets
NDC59676-278-01
EDURANT
®
(rilpivirine) tablets
25 mg
Each tablet contains 27.5 mg of rilpivirine
hydrochloride, which is equivalent to 25 mg
of rilpivirine.
ALERT: Find out about
medicines that should NOT be
taken with EDURANT
®from
your healthcare provider.
Rx only
2.7 Recommended Dosage With Rifabutin Coadministration (2.7 Recommended Dosage with Rifabutin Coadministration)
If EDURANT is coadministered with rifabutin, the EDURANT dose should be increased to 50 mg (two 25 mg tablets) once daily, taken with a meal. When rifabutin coadministration is stopped, the EDURANT dose should be decreased to 25 mg once daily, taken with a meal [see Drug Interactions (7)and Clinical Pharmacology (12.3)].
Note that use of CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) with rifabutin is contraindicated. Refer to CABENUVA labeling for additional detail.
1.2 Treatment of Hiv 1 in Combination With Cabotegravir (1.2 Treatment of HIV-1 in Combination with Cabotegravir)
EDURANT is indicated in combination with VOCABRIA (cabotegravir) for short-term treatment of HIV-1 infection in adults and adolescents 12 years and older and weighing at least 35 kg who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable antiretroviral regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine, for use as [see Dosage and Administration (2.6)] :
- oral lead-in to assess the tolerability of rilpivirine prior to administration of rilpivirine extended-release injectable suspension, a component of CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension).
- oral therapy for patients who will miss planned injection dosing with CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension).
2.2 Recommended Dosage in Treatment Naïve Adult Patients (2.2 Recommended Dosage in Treatment-Naïve Adult Patients)
The recommended dosage of EDURANT in adult patients is one 25 mg tablet taken orally once daily with a meal [see Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
Principal Display Panel 2.5 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 2.5 mg Tablet Blister Pack Carton)
NDC 59676-280-90
Rx only
EDURANT ® PED
(rilpivirine)
tablets for oral suspension
2.5 mg
90 tablets
9 blister cards, 10 tablets each
Attention: EDURANT PED is NOT a substitute for EDURANT
DISPERSE IN DRINKING WATER
Do not crush, chew, or swallow whole, see Instructions for Use
2.4 Preparation and Administration Instructions for Edurant Ped Only (2.4 Preparation and Administration Instructions for EDURANT PED Only)
Advise patients or caregivers of patients taking EDURANT PED to refer to the Instructions for Use to properly prepare and take the medication.
EDURANT PED must be dispersed in drinking water and taken immediately with a meal. If not taken immediately, then the oral suspension should be discarded, and a new dose of medicine should be prepared. The patient should not chew or swallow EDURANT PED whole. The following instructions should be followed:
- Place the tablets for oral suspension in a cup, add 5 mL (1 teaspoon) of drinking water at room temperature. Do not crush the tablets.
- Swirl the cup carefully for 1–2 minutes to disperse the tablets. The oral suspension will start to look cloudy.
- Take all the prepared oral suspension immediately or to aid in administration, the oral suspension can be further diluted with 5 mL (1 teaspoon) of drinking water, milk, orange juice or applesauce. Swirl and take all the medicine immediately. A spoon can be used if needed.
- Make sure the entire dose is taken and no medicine is left in the cup. If required, add another 5 mL (1 teaspoon) of drinking water (or alternative beverage or soft food), swirl and drink immediately.
14.4 Treatment Naïve Pediatric Subjects (2 to Less Than 12 Years of Age) (14.4 Treatment-Naïve Pediatric Subjects (2 to less than 12 Years of Age))
The pharmacokinetics, safety, tolerability and efficacy of EDURANT and EDURANT PED weight-adjusted doses 25, 15 and 12.5 mg once daily in combination with an investigator-selected BR containing two NRTIs, was evaluated in trial TMC278-C213 Cohort 2, a single-arm, open-label Phase 2 trial in antiretroviral treatment-naïve HIV-1 infected pediatric subjects 6 to less than 12 years of age and weighing at least 17 kg. The Week 48 analysis included 18 subjects, 17 (94%) subjects completed the 48-week treatment period, and 1 (6%) subject discontinued the study early due to reaching a virologic endpoint. The 18 subjects had a median age of 9 years (range 6 to 11 years) and the median weight at baseline was 25 kg (range 17 to 51 kg). 89% were Black and 39% were female. The median baseline plasma viral load was 55,400 (range 567–149,000) copies/mL, and the median absolute baseline CD4+ cell count was 432.5 (range 12–2,068) cells/µL.
The number of subjects with HIV-1 RNA <50 copies/mL at Week 48 was 13/18 (72%), while 3/18 (17%) subjects had HIV-1 RNA ≥50 copies/mL at Week 48 [see Microbiology (12.4)] . Two out of 18 (11%) participants in the 15 mg once daily (20 to ≤25 kg) dose-weight group had missing viral load data at Week 48 but remained on study. The viral load for these 2 subjects was <50 copies/mL, post-Week 48. The mean increase (SE) in CD4+ from baseline was 215.9 (62.42) cells/µL at Week 48.
The safety and efficacy of EDURANT and EDURANT PED in treatment naïve pediatric subjects 2 to less than 6 years of age is supported by evidence from adequate and well-controlled studies of EDURANT in adults with additional population pharmacokinetic data from adults and pediatric subjects 6 years and older [see Use in Specific Populations (8.4)and Clinical Pharmacology (12.3)] .
14.2 Virologically Suppressed Adults Treated in Combination With Cabotegravir (14.2 Virologically-Suppressed Adults Treated in Combination with Cabotegravir)
The use of EDURANT in combination with VOCABRIA (cabotegravir) as an oral lead-in and in patients who miss planned injections with CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) was evaluated in two Phase 3 randomized, multicenter, active-controlled, parallel-arm, open-label, non-inferiority trials (Trial 201584: FLAIR [NCT02938520], Trial 201585: ATLAS [NCT2951052]), and one Phase 3b randomized, multicenter, parallel-group, open-label, non-inferiority trial (Trial 207966: ATLAS-2M [NCT03299049]) in subjects who were virologically suppressed (HIV-1 RNA <50 copies/mL). See full prescribing information for VOCABRIA and CABENUVA for additional information.
14.3 Treatment Naïve Pediatric Subjects (≥12 to Less Than 18 Years of Age) (14.3 Treatment-Naïve Pediatric Subjects (≥12 to less than 18 Years of Age))
The pharmacokinetics, safety, tolerability and efficacy of EDURANT 25 mg once daily, in combination with an investigator-selected background regimen (BR) containing two NRTIs, was evaluated in trial TMC278-C213 Cohort 1, a single-arm, open-label Phase 2 trial in antiretroviral treatment-naïve HIV-1 infected pediatric subjects 12 to less than 18 years of age and weighing at least 32 kg. Thirty six (36) subjects were enrolled in the trial to complete at least 48 weeks of treatment. The 36 subjects had a median age of 14.5 years (range: 12 to 17 years), and were 56% female, 89% Black and 11% Asian.
In the efficacy analysis, most subjects (75%; 28/36) had baseline HIV RNA <100,000 copies/mL. For these 28 subjects the median baseline plasma HIV-1 RNA was 44,250 (range: 2,060–92,600 copies/mL) and the median baseline CD4+ cell count was 445.5 cells/mm 3(range: 123 to 983 cells/mm 3).
Among the subjects who had baseline HIV RNA ≤100,000, the proportion with HIV-1 RNA <50 copies/mL at Week 48 was 79% (22/28), versus 50% (4/8) in those with >100,000 copies/mL. The proportion of virologic failures among subjects with a baseline viral load ≤100,000 copies/mL was 21% (6/28), versus 38% (3/8) in those with >100,000 copies/mL. At Week 48, the mean increase in CD4+ cell count from baseline was 201.2 cells/mm 3.
5.4 Risk of Adverse Reactions Or Loss of Virologic Response Due to Drug Interactions (5.4 Risk of Adverse Reactions or Loss of Virologic Response Due to Drug Interactions)
The concomitant use of EDURANT or EDURANT PED and other drugs may result in potentially significant drug interactions, some of which may lead to [see Dosage and Administration (2.7), Contraindications (4), and Drug Interactions (7)] :
- Loss of therapeutic effect of EDURANT or EDURANT PED and possible development of resistance.
In healthy subjects, 75 mg once daily and 300 mg once daily (3 times and 12 times the dose in EDURANT) have been shown to prolong the QTc interval of the electrocardiogram. Consider alternatives to EDURANT or EDURANT PED when coadministered with a drug that is known to have a risk of torsade de pointes [see Drug Interactions (7) and Clinical Pharmacology (12.2)] .
See Table 6 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during EDURANT or EDURANT PED therapy and review concomitant medications during therapy.
2.3 Recommended Dosage in Treatment Naïve Pediatric Patients 2 Years of Age and Older and Weighing At Least 14 Kg (2.3 Recommended Dosage in Treatment-Naïve Pediatric Patients 2 Years of Age and Older and Weighing at least 14 kg)
The recommended dosage of EDURANT and EDURANT PED in pediatric patients 2 years of age and older and weighing at least 14 kg is based on body weight (see Table 1). Both EDURANT and EDURANT PED should be taken orally once daily with a meal [see Use in Specific Populations (8.4)and Clinical Pharmacology (12.3)] .
| Body Weight (kg) | EDURANT 25 mg Tablets | EDURANT PED 2.5 mg Tablets for Oral Suspension | Total Daily Dose |
|---|---|---|---|
| 14 kg to less than 20 kg | Not recommended | 5 tablets once daily | 12.5 mg EDURANT PED once daily |
| 20 kg to less than 25 kg | Not recommended | 6 tablets once daily | 15 mg EDURANT PED once daily |
| Greater than or equal to 25 kg | 1 tablet once daily | Not recommended | 25 mg EDURANT once daily |
2.6 Recommended Dosage in Combination With Cabotegravir in Adults and Adolescents 12 Years of Age and Older and Weighing At Least 35 Kg (2.6 Recommended Dosage in Combination with Cabotegravir in Adults and Adolescents 12 Years of Age and Older and Weighing at least 35 kg)
Consult the prescribing information for CABENUVA (cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension) before initiating EDURANT to ensure therapy with CABENUVA is appropriate.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:09.202049 · Updated: 2026-03-14T22:32:10.275892