fluconazole in sodium chloride injection, usp
02f74fe7-0649-46c5-a06a-0700cd265773
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Fluconazole in Sodium Chloride Injection, USP is indicated for the treatment of: Oropharyngeal and esophageal candidiasis. In open noncomparative studies of relatively small numbers of patients, fluconazole was also effective for the treatment of Candida urinary tract infections, peritonitis, and systemic Candida infections including candidemia, disseminated candidiasis, and pneumonia. Cryptococcal meningitis. Before prescribing fluconazole for AIDS patients with cryptococcal meningitis, please see CLINICAL STUDIES section. Studies comparing fluconazole to amphotericin B in non-HIV infected patients have not been conducted. Prophylaxis: Fluconazole is also indicated to decrease the incidence of candidiasis in patients undergoing bone marrow transplantation who receive cytotoxic chemotherapy and/or radiation therapy. Specimens for fungal culture and other relevant laboratory studies (serology, histopathology) should be obtained prior to therapy to isolate and identify causative organisms. Therapy may be instituted before the results of the cultures and other laboratory studies are known; however, once these results become available, anti-infective therapy should be adjusted accordingly.
Contraindications
Fluconazole is contraindicated in patients who have shown hypersensitivity to fluconazole or to any of its excipients. There is no information regarding cross-hypersensitivity between fluconazole and other azole antifungal agents. Caution should be used in prescribing fluconazole to patients with hypersensitivity to other azoles. Coadministration of other drugs known to prolong the QT interval and which are metabolized via the enzyme CYP3A4 such as erythromycin, pimozide, and quinidine are contraindicated in patients receiving fluconazole. (See CLINICAL PHARMACOLOGY: Drug Interaction Studies and PRECAUTIONS .)
Adverse Reactions
The pattern and incidence of adverse events and laboratory abnormalities recorded during pediatric clinical trials are comparable to those seen in adults. In Phase II/III clinical trials conducted in the United States and in Europe, 577 pediatric patients, ages 1 day to 17 years were treated with fluconazole at doses up to 15 mg/kg/day for up to 1,616 days. Thirteen percent of pediatric patients experienced treatment-related adverse events. The most commonly reported events were vomiting (5%), abdominal pain (3%), nausea (2%), and diarrhea (2%). Treatment was discontinued in 2.3% of patients due to adverse clinical events and in 1.4% of patients due to laboratory test abnormalities. The majority of treatment-related laboratory abnormalities were elevations of transaminases or alkaline phosphatase. Percentage of Patients With Treatment-Related Side Effects Fluconazole (N=577) Comparative Agents (N=451) With any side effect 13.0 9.3 Vomiting 5.4 5.1 Abdominal pain 2.8 1.6 Nausea 2.3 1.6 Diarrhea 2.1 2.2 Clinical Trials Experience in Pediatric Patients Safety in Prophylaxis of Invasive Candida Infections in Premature infants weighing less than 750 grams at birth. In a Phase 3 clinical trial of pediatric patients (premature infants weighing less than 750 grams at birth), the incidence of intestinal perforation in infants receiving fluconazole prophylaxis was higher compared to infants receiving placebo (see PRECAUTIONS: Pediatric Use ). Safety in Pediatric Patients Receiving ECMO A cohort of 20 pediatric patients (1 day to 17 years of age) on ECMO received fluconazole in a prospective, open-label, single-center safety and PK ECMO study. The adverse reaction profile of fluconazole in these patients was similar to that of adult and pediatric non-ECMO patients (See PRECAUTIONS: Pediatric Use ). To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
How Supplied
Fluconazole in Sodium Chloride Injection, USP is supplied as follows: Fluconazole in Sodium Chloride Injection, USP NDC (2 mg per mL) Package Factor 25021-184-82 200 mg per 100 mL in flexible container bag 10 bags per carton 25021-184-87 400 mg per 200 mL in flexible container bag 10 bags per carton Fluconazole in Sodium Chloride Injection, USP for intravenous infusion administration is formulated as iso-osmotic solutions containing 2 mg per mL of fluconazole.
Description
SAGENT ® Rx only
Medication Information
Warnings
(1) Hepatic injury: Fluconazole should be administered with caution to patients with liver dysfunction. Fluconazole has been associated with rare cases of serious hepatic toxicity, including fatalities primarily in patients with serious underlying medical conditions. In cases of fluconazole-associated hepatotoxicity, no obvious relationship to total daily dose, duration of therapy, sex, or age of the patient has been observed. Fluconazole hepatotoxicity has usually, but not always, been reversible on discontinuation of therapy. Patients who develop abnormal liver function tests during fluconazole therapy should be monitored for the development of more severe hepatic injury. Fluconazole should be discontinued if clinical signs and symptoms consistent with liver disease develop that may be attributable to fluconazole.
(2) Anaphylaxis: In rare cases, anaphylaxis has been reported.
(3) Dermatologic: Exfoliative skin disorders during treatment with fluconazole have been reported. Fatal outcomes have been reported in patients with serious underlying diseases. Patients with deep seated fungal infections who develop rashes during treatment with fluconazole should be monitored closely and the drug discontinued if lesions progress. Fluconazole should be discontinued in patients treated for superficial fungal infection who develop a rash that may be attributed to fluconazole.
(4) Potential for fetal harm: There are no adequate and well-controlled clinical trials of fluconazole in pregnant women. Case reports describe a pattern of distinct congenital anomalies in infants exposed in utero to high dose maternal fluconazole (400 to 800 mg/day) during most or all of the first trimester. These reported anomalies are similar to those seen in animal studies. If fluconazole is used during pregnancy or if the patient becomes pregnant while taking the drug, the patient should be informed of the potential hazard to the fetus. Effective contraceptive measures should be considered in women of child-bearing potential who are being treated with fluconazole 400 to 800 mg/day and should continue throughout the treatment period and for approximately 1 week (5 to 6 half-lives) after the final dose. Epidemiological studies suggest a potential risk of spontaneous abortion and congenital abnormalities in infants whose mothers were treated with 150 mg of fluconazole as a single or repeated dose in the first trimester, but these epidemiological studies have limitations and these findings have not been confirmed in controlled clinical trials. (See PRECAUTIONS: Pregnancy .)
Indications and Usage
Fluconazole in Sodium Chloride Injection, USP is indicated for the treatment of:
- Oropharyngeal and esophageal candidiasis. In open noncomparative studies of relatively small numbers of patients, fluconazole was also effective for the treatment of Candida urinary tract infections, peritonitis, and systemic Candida infections including candidemia, disseminated candidiasis, and pneumonia.
- Cryptococcal meningitis. Before prescribing fluconazole for AIDS patients with cryptococcal meningitis, please see CLINICAL STUDIES section. Studies comparing fluconazole to amphotericin B in non-HIV infected patients have not been conducted.
Prophylaxis: Fluconazole is also indicated to decrease the incidence of candidiasis in patients undergoing bone marrow transplantation who receive cytotoxic chemotherapy and/or radiation therapy.
Specimens for fungal culture and other relevant laboratory studies (serology, histopathology) should be obtained prior to therapy to isolate and identify causative organisms. Therapy may be instituted before the results of the cultures and other laboratory studies are known; however, once these results become available, anti-infective therapy should be adjusted accordingly.
Contraindications
Fluconazole is contraindicated in patients who have shown hypersensitivity to fluconazole or to any of its excipients. There is no information regarding cross-hypersensitivity between fluconazole and other azole antifungal agents. Caution should be used in prescribing fluconazole to patients with hypersensitivity to other azoles. Coadministration of other drugs known to prolong the QT interval and which are metabolized via the enzyme CYP3A4 such as erythromycin, pimozide, and quinidine are contraindicated in patients receiving fluconazole. (See CLINICAL PHARMACOLOGY: Drug Interaction Studies and PRECAUTIONS .)
Adverse Reactions
Fluconazole is generally well tolerated.
In some patients, particularly those with serious underlying diseases such as AIDS and cancer, changes in renal and hematological function test results and hepatic abnormalities have been observed during treatment with fluconazole and comparative agents, but the clinical significance and relationship to treatment is uncertain.
How Supplied
Fluconazole in Sodium Chloride Injection, USP is supplied as follows:
| Fluconazole in Sodium Chloride Injection, USP | ||
| NDC | (2 mg per mL) | Package Factor |
| 25021-184-82 | 200 mg per 100 mL in flexible container bag | 10 bags per carton |
| 25021-184-87 | 400 mg per 200 mL in flexible container bag | 10 bags per carton |
Fluconazole in Sodium Chloride Injection, USP for intravenous infusion administration is formulated as iso-osmotic solutions containing 2 mg per mL of fluconazole.
Description
Fluconazole in Sodium Chloride Injection, USP, the first of a new subclass of synthetic triazole antifungal agents, is available as a sterile solution for intravenous use in plastic containers.
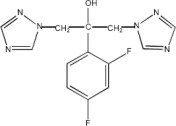
Fluconazole is designated chemically as 2,4-difluoro-α,α1-bis(1H-1,2,4-triazol-1-ylmethyl) benzyl alcohol with an empirical formula of C13H12F2N6O and molecular weight of 306.3. The structural formula is:
Fluconazole is a white crystalline solid which is slightly soluble in water and saline.
Fluconazole in Sodium Chloride Injection, USP is an iso-osmotic, sterile, nonpyrogenic solution of fluconazole in a sodium chloride diluent. Each mL contains 2 mg of fluconazole and 9 mg of sodium chloride. The pH ranges from 4.0 to 8.0. Injection volumes of 100 mL and 200 mL are packaged in plastic containers.
The flexible bag container is fabricated from a specially formulated non-plasticized thermoplastic co-polyolefin. The amount of water that can permeate from the container into the overwrap is insufficient to affect the solution significantly. The suitability of the container material has been confirmed in tests in animals according to USP biological tests for plastic containers.
Section 42229-5
SAGENT®
Rx only
Section 51945-4
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-184-82
100 mL
Fluconazole In Sodium Chloride Injection, USP
200 mg per 100 mL
(2 mg per mL)
Rx only
Iso-Osmotic Sodium Chloride Diluent
For Intravenous Use
Single-Dose Container
General
Some azoles, including fluconazole, have been associated with prolongation of the QT interval on the electrocardiogram. Fluconazole causes QT prolongation via the inhibition of Rectifier Potassium Channel current (Ikr). The QT prolongation caused by other medicinal products (such as amiodarone) may be amplified via the inhibition of cytochrome P450 (CYP) 3A4. (See PRECAUTIONS, Drug Interactions .) During post-marketing surveillance, there have been rare cases of QT prolongation and torsade de pointes in patients taking fluconazole. Most of these reports involved seriously ill patients with multiple confounding risk factors, such as structural heart disease, electrolyte abnormalities, and concomitant medications that may have been contributory. Patients with hypokalemia and advanced cardiac failure are at an increased risk for the occurrence of life-threatening ventricular arrhythmias and torsade de pointes.
Fluconazole should be administered with caution to patients with these potentially proarrhythmic conditions.
Concomitant use of fluconazole and erythromycin has the potential to increase the risk of cardiotoxicity (prolonged QT interval, torsade de pointes) and consequently sudden heart death. This combination should be avoided.
Fluconazole should be administered with caution to patients with renal dysfunction.
Adrenal insufficiency has been reported in patients receiving azoles, including fluconazole. Reversible cases of adrenal insufficiency have been reported in patients receiving fluconazole.
When driving vehicles or operating machines, it should be taken into account that occasionally dizziness or seizures may occur.
There have been reports of cases of superinfection with Candida species other than C. albicans, which are often inherently not susceptible to fluconazole (e.g., Candida krusei). Such cases may require alternative antifungal therapy (see CLINICAL PHARMACOLOGY, Microbiology).
To Open
Tear overwrap down side at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. After removing overwrap, check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired.
DO NOT ADD SUPPLEMENTARY MEDICATION.
Preparation for Administration:
- Suspend container from eyelet support.
- Remove plastic protector from outlet port at bottom of container.
- Attach administration set. Refer to complete directions accompanying set.
Human Data
Case reports describe a distinctive and rare pattern of birth defects among infants whose mothers received high-dose (400 to 800 mg/day) fluconazole during most or all of the first trimester of pregnancy. The features seen in these infants include brachycephaly, abnormal facies, abnormal calvarial development, cleft palate, femoral bowing, thin ribs and long bones, arthrogryposis, and congenital heart disease. These effects are similar to those seen in animal studies.
Epidemiological studies suggest a potential risk of spontaneous abortion and congenital abnormalities in infants whose mothers were treated with 150 mg of fluconazole as a single or repeated dose in the first trimester, but these epidemiological studies have limitations and these findings have not been confirmed in controlled clinical trials.
Overdosage
There have been reports of overdose with fluconazole accompanied by hallucination and paranoid behavior.
In the event of overdose, symptomatic treatment (with supportive measures and gastric lavage if clinically indicated) should be instituted.
Fluconazole is largely excreted in urine. A 3-hour hemodialysis session decreases plasma levels by approximately 50%.
In mice and rats receiving very high doses of fluconazole, clinical effects in both species included decreased motility and respiration, ptosis, lacrimation, salivation, urinary incontinence, loss of righting reflex, and cyanosis; death was sometimes preceded by clonic convulsions.
Resistance
A potential for development of resistance to fluconazole is well known. Fungal isolates exhibiting reduced susceptibility to other azoles may also show reduced susceptibility to fluconazole. The frequency of drug resistance development for the various fungi for which this drug is indicated is not known.
Fluconazole resistance may arise from a modification in the quality or quantity of the target enzyme (lanosterol 14-α-demethylase), reduced access to the drug target, or some combination of these mechanisms.
Point mutations in the gene (ERG11) encoding for the target enzyme lead to an altered target with decreased affinity for azoles. Overexpression of ERG11 results in the production of high concentrations of the target enzyme, creating the need for higher intracellular drug concentrations to inhibit all of the enzyme molecules in the cell.
The second major mechanism of drug resistance involves active efflux of fluconazole out of the cell through the activation of two types of multidrug efflux transporters; the major facilitators (encoded by MDR genes) and those of the ATP-binding cassette superfamily (encoded by CDR genes). Upregulation of the MDR gene leads to fluconazole resistance, whereas, upregulation of CDR genes may lead to resistance to multiple azoles.
Resistance in Candida glabrata usually includes upregulation of CDR genes resulting in resistance to multiple azoles. For an isolate where the minimum inhibitory concentration (MIC) is categorized as Intermediate (16 to 32 mcg/mL), the highest fluconazole dose is recommended.
Animal Data
Fluconazole was administered orally to pregnant rabbits during organogenesis in two studies at doses of 5 mg/kg, 10 mg/kg, and 20 mg/kg and at 5 mg/kg, 25 mg/kg, and 75 mg/kg, respectively. Maternal weight gain was impaired at all dose levels (approximately 0.25 to 4 times the 400 mg clinical dose based on body surface area [BSA] comparison), and abortions occurred at 75 mg/kg (approximately 4 times the 400 mg clinical dose based on BSA); no adverse fetal effects were observed.
In several studies in which pregnant rats received fluconazole orally during organogenesis, maternal weight gain was impaired and placental weights were increased at 25 mg/kg. There were no fetal effects at 5 mg/kg or 10 mg/kg; increases in fetal anatomical variants (supernumerary ribs, renal pelvis dilation) and delays in ossification were observed at 25 mg/kg and 50 mg/kg and higher doses. At doses ranging from 80 to 320 mg/kg (approximately 2 to 8 times the 400 mg clinical dose based on BSA), embryolethality in rats was increased and fetal abnormalities included wavy ribs, cleft palate, and abnormal craniofacial ossification. These effects are consistent with the inhibition of estrogen synthesis in rats and may be a result of known effects of lowered estrogen on pregnancy, organogenesis, and parturition.
Geriatric Use
In non-AIDS patients, side effects possibly related to fluconazole treatment were reported in fewer patients aged 65 and older (9%, n =339) than for younger patients (14%, n=2240). However, there was no consistent difference between the older and younger patients with respect to individual side effects. Of the most frequently reported (>1%) side effects, rash, vomiting, and diarrhea occurred in greater proportions of older patients. Similar proportions of older patients (2.4%) and younger patients (1.5%) discontinued fluconazole therapy because of side effects. In post-marketing experience, spontaneous reports of anemia and acute renal failure were more frequent among patients 65 years of age or older than in those between 12 and 65 years of age. Because of the voluntary nature of the reports and the natural increase in the incidence of anemia and renal failure in the elderly, it is however not possible to establish a causal relationship to drug exposure.
Controlled clinical trials of fluconazole did not include sufficient numbers of patients aged 65 and older to evaluate whether they respond differently from younger patients in each indication. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
Fluconazole is primarily cleared by renal excretion as unchanged drug. Because elderly patients are more likely to have decreased renal function, care should be taken to adjust dose based on creatinine clearance. It may be useful to monitor renal function. (See CLINICAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION .)
Multiple Dose
SINCE ORAL ABSORPTION IS RAPID AND ALMOST COMPLETE, THE DAILY DOSE OF FLUCONAZOLE IS THE SAME FOR ORAL AND INTRAVENOUS ADMINISTRATION. In general, a loading dose of twice the daily dose is recommended on the first day of therapy to result in plasma concentrations close to steady-state by the second day of therapy.
The daily dose of fluconazole for the treatment of infection should be based on the infecting organism and the patient's response to therapy. Treatment should be continued until clinical parameters or laboratory tests indicate that active fungal infection has subsided. An inadequate period of treatment may lead to recurrence of active infection. Patients with AIDS and cryptococcal meningitis or recurrent oropharyngeal candidiasis usually require maintenance therapy to prevent relapse.
Pediatric Use
Use in Pediatric Patients for the Treatment of Oropharyngeal Candidiasis
An open-label, randomized, controlled trial has shown fluconazole to be effective in the treatment of oropharyngeal candidiasis in pediatric patients 6 months to 13 years of age. (See CLINICAL STUDIES .)
Use in Pediatric Patients for the Treatment of Candida Esophagitis, Systemic Candida Infections, or Cryptococcal Meningitis
The use of fluconazole in pediatric patients with cryptococcal meningitis, Candida esophagitis, or systemic Candida infections is supported by the efficacy shown for these indications in adults and by the results from several small noncomparative pediatric clinical studies. In addition, pharmacokinetic studies in pediatric patients (See CLINICAL PHARMACOLOGY ) have established a dose proportionality between pediatric patients and adults. (See DOSAGE AND ADMINISTRATION .)
In a noncomparative study of fluconazole administered to pediatric patients (from birth to less than 17 years) with serious systemic fungal infections, most of which were candidemia, the effectiveness of fluconazole was similar to that reported for the treatment of candidemia in adults. Of 17 subjects with culture-confirmed candidemia, 11 of 14 (79%) with baseline symptoms (3 were asymptomatic) had a clinical cure; 13/15 (87%) of evaluable patients had a mycologic cure at the end of treatment but two of these patients relapsed at 10 and 18 days, respectively, following cessation of therapy.
The efficacy of fluconazole for the suppression of cryptococcal meningitis was successful in 4 of 5 pediatric patients (4 years to 10 years of age) treated in a compassionate-use study of fluconazole for the treatment of life-threatening or serious mycosis. There are limited clinical data to support the efficacy of fluconazole for primary treatment of cryptococcal meningitis in pediatric patients.
The safety profile of fluconazole in children has been studied in 577 pediatric patients from 1 day to 17 years of age who received doses ranging from 1 to 15 mg/kg/day for 1 to 1,616 days. (See ADVERSE REACTIONS .)
Use in Pediatric Patients on Extracorporeal Membrane Oxygenation (ECMO)
A prospective, open-label, single-center study was conducted to determine the PK and safety of fluconazole in pediatric patients (ages: from birth to 17 years of age) on ECMO (see CLINICAL PHARMACOLOGY ). A loading dose of 35-mg/kg is recommended in pediatric patients on ECMO due to increased volume of distribution (see DOSAGE AND ADMINISTRATION ).
Use in Prophylaxis of Invasive Candida Infections in Pediatric Patients (premature infants
weighing less than 750 grams at birth)
Safety and effectiveness of fluconazole for the prophylaxis of invasive candidiasis in pediatric patients (premature infants weighing less than 750 grams at birth) have not been established.
A prospective, randomized, double-blind, placebo-controlled, multicenter trial was conducted in premature infants weighing less than 750 grams at birth to evaluate the efficacy and safety of prophylactic fluconazole 6-mg/kg administered twice weekly for 6 weeks versus placebo (NCT00734539). Efficacy was assessed using the endpoint of death or candidiasis by study day 49. The results are summarized in Table 4.
|
*Subjects with missing data are imputed as having candidiasis or died. |
||||
|
**Subjects may be counted more than once as two fluconazole subjects and four placebo subjects diagnosed with candidiasis subsequently died by day 49. |
||||
| Fluconazole (N=188) n (%) | Placebo (N=173) n (%) | P-value | Difference (95% CI) | |
| Death or candidiasis* | 33 (17.6) | 38 (22.0) | 0.2954 | -4.4 (-12.6, 3.8) |
| Components of endpoint** Death Candidiasis Missing |
27 (14.4) 6 (3.2) 2 (1.0) |
25 (14.5) 16 (9.2) 1 (0.5) |
The most common fatal serious adverse reactions in the fluconazole vs placebo arms, respectively, were necrotizing enterocolitis (NEC), 9 (5%) vs 9 (5%); neonatal bacterial sepsis, 6 (3%) vs 7 (4%); and neonatal respiratory failure, 4 (2%) vs 2 (0.6%).
The most common serious adverse reactions (>5%) reported in patients receiving fluconazole prophylaxis are displayed in Table 5.
|
*All serious adverse reactions were assessed and recorded up through 30 days after the final dose of study drug. Serious adverse reactions included both fatal and non-fatal outcomes. |
||
| Adverse Reaction | Fluconazole (N=188) n (%) | Placebo (N=173) n (%) |
| Necrotizing Enterocolitis (NEC) | 27 (14) | 28 (16) |
| Intestinal Perforation (includes ileal/small intestinal perforation) | 13 (7) | 7 (4) |
| Neonatal Respiratory Arrest/ Neonatal Respiratory Failure | 13 (7) | 4 (2) |
| Bacterial Sepsis, Neonatal | 10 (5) | 12 (7) |
Administration
Fluconazole in Sodium Chloride Injection, USP may be administered by intravenous infusion. Fluconazole in Sodium Chloride Injection, USP has been used safely for up to fourteen days of intravenous therapy. The intravenous infusion of fluconazole should be administered at a maximum rate of approximately 200 mg/hour, given as a continuous infusion.
Fluconazole in Sodium Chloride Injection, USP in plastic containers are intended only for intravenous administration using sterile equipment.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Do not use if the solution is cloudy or precipitated or if the seal is not intact.
Directions for Intravenous Use of Fluconazole in Plastic Containers
Do not remove unit from overwrap until ready for use. The overwrap is a moisture barrier. The inner bag maintains the sterility of the product.
CAUTION: Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Nursing Mothers
Fluconazole was present in low levels in breast milk following administration of a single 150 mg dose, based on data from a study in 10 breastfeeding women who temporarily or permanently discontinued breastfeeding 5 days to 19 months postpartum. The estimated daily infant dose of fluconazole from breast milk (assuming mean milk consumption of 150 mL/kg/day) based on the mean peak milk concentration (2.61 mcg/mL [range: 1.57 to 3.65 mcg/mL] at 5.2 hours post-dose) was 0.39 mg/kg/day, which is approximately 13% of the recommended pediatric dose for oropharyngeal candidiasis. (Labeled pediatric dose is 6 mg/kg/day on the first day followed by 3 mg/kg/day; estimated infant dose is 13% of 3 mg/kg/day maintenance dose). There are no data on fluconazole levels in milk after repeated use or after high-dose fluconazole. A published survey of 96 breastfeeding women who were treated with fluconazole 150 mg every other day (average of 7.3 capsules [range 1 to 29 capsules]) for lactation-associated candida of the breasts reported no serious adverse reactions in infants. Caution should be exercised when fluconazole is administered to a nursing woman.
Clinical Studies
Cryptococcal meningitis: In a multicenter study comparing fluconazole (200 mg/day) to amphotericin B (0.3 mg/kg/day) for treatment of cryptococcal meningitis in patients with AIDS, a multivariate analysis revealed three pretreatment factors that predicted death during the course of therapy: abnormal mental status, cerebrospinal fluid cryptococcal antigen titer greater than 1:1024, and cerebrospinal fluid white blood cell count of less than 20 cells/mm3. Mortality among high risk patients was 33% and 40% for amphotericin B and fluconazole patients, respectively (p=0.58), with overall deaths 14% (9 of 63 subjects) and 18% (24 of 131 subjects) for the 2 arms of the study (p=0.48). Optimal doses and regimens for patients with acute cryptococcal meningitis and at high risk for treatment failure remain to be determined. (Saag, et al. N Engl J Med 1992; 326:83-9.)
Pediatric Studies
Oropharyngeal candidiasis: An open-label, comparative study of the efficacy and safety of fluconazole (2 to 3 mg/kg/day) and oral nystatin (400,000 I.U. 4 times daily) was conducted in immunocompromised pediatric patients from 6 months to 13 years of age with oropharyngeal candidiasis. Clinical and mycological response rates were higher in pediatric patients treated with fluconazole.
Clinical cure at the end of treatment was reported for 86% of fluconazole-treated patients compared to 46% of nystatin treated patients. Mycologically, 76% of fluconazole treated patients had the infecting organism eradicated compared to 11% for nystatin treated patients.
|
* Subjects without follow-up cultures for any reason were considered nonevaluable for mycological response. |
||
| Fluconazole | Nystatin | |
| Enrolled | 96 | 90 |
| Clinical Cure | 76/88 (86%) | 36/78 (46%) |
| Mycological eradication* | 55/72 (76%) | 6/54 (11%) |
The proportion of patients with clinical relapse 2 weeks after the end of treatment was 14% for subjects receiving fluconazole and 16% for subjects receiving nystatin. At 4 weeks after the end of treatment, the percentages of patients with clinical relapse were 22% for fluconazole and 23% for nystatin.
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
AVOID EXCESSIVE HEAT. PROTECT FROM FREEZING.
Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free.
The container and container closure are not made with natural rubber latex.
Brands listed are the trademarks of their respective owners.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in Switzerland
©2024 Sagent Pharmaceuticals
Revised: March 2024
SAGENT Pharmaceuticals®
Mechanism of Action
Fluconazole is a highly selective inhibitor of fungal cytochrome P450 dependent enzyme lanosterol 14-α-demethylase. This enzyme functions to convert lanosterol to ergosterol. The subsequent loss of normal sterols correlates with the accumulation of 14-α-methyl sterols in fungi and may be responsible for the fungistatic activity of fluconazole. Mammalian cell demethylation is much less sensitive to fluconazole inhibition.
Teratogenic Effects.
Potential for Fetal Harm: Use in pregnancy should be avoided except in patients with severe or potentially life-threatening fungal infections in whom fluconazole may be used if the anticipated benefit outweighs the possible risk to the fetus. A few published case reports describe a pattern of distinct congenital anomalies in infants exposed in utero to high dose maternal fluconazole (400 to 800 mg/day) during most or all of the first trimester. These reported anomalies are similar to those seen in animal studies. Effective contraceptive measures should be considered in women of child-bearing potential who are being treated with fluconazole 400 to 800 mg/day and should continue throughout the treatment period and for approximately 1 week (5 to 6 half-lives) after the final dose. If fluconazole is used during pregnancy, or if the patient becomes pregnant while taking the drug, the patient should be informed of the potential hazard to the fetus. Spontaneous abortions and congenital abnormalities have been suggested as potential risks associated with 150 mg of fluconazole as a single or repeated dose in the first trimester of pregnancy based on retrospective epidemiological studies. There are no adequate and well-controlled studies of fluconazole in pregnant women. (See WARNINGS: Potential for Fetal Harm .)
Antimicrobial Activity
Fluconazole has been shown to be active against most isolates of the following microorganisms both in vitro and in clinical infections.
Candida albicans
Candida glabrata (Many isolates are intermediately susceptible)
Candida parapsilosis
Candida tropicalis
Cryptococcus neoformans
The following in vitro data are available, but their clinical significance is unknown. At least 90% of the following fungi exhibit an in vitro MIC less than or equal to the susceptible breakpoint for fluconazole (https://www.fda.gov/STIC) against isolates of similar genus or organism group. However, the effectiveness of fluconazole in treating clinical infections due to these fungi has not been established in adequate and well-controlled clinical trials.
Candida dubliniensis
Candida guilliermondii
Candida kefyr
Candida lusitaniae
Candida krusei should be considered to be resistant to fluconazole. Resistance in C. krusei appears to be mediated by reduced sensitivity of the target enzyme to inhibition by the agent.
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
Post Marketing Experience
In addition, the following adverse events have occurred during post-marketing experience.
Immunologic: In rare cases, anaphylaxis (including angioedema, face edema and pruritus) has been reported.
Body as a Whole: Asthenia, fatigue, fever, malaise.
Cardiovascular: QT prolongation, torsade de pointes. (See PRECAUTIONS .)
Central Nervous System: Seizures, dizziness.
Hematopoietic and Lymphatic: Leukopenia, including neutropenia and agranulocytosis, thrombocytopenia.
Metabolic: Hypercholesterolemia, hypertriglyceridemia, hypokalemia.
Gastrointestinal: Cholestasis, dry mouth, hepatocellular damage, dyspepsia, vomiting.
Other Senses: Taste perversion.
Musculoskeletal System: myalgia.
Nervous System: Insomnia, paresthesia, somnolence, tremor, vertigo.
Skin and Appendages: Acute generalized exanthematous pustulosis, drug eruption including fixed drug eruption, increased sweating, exfoliative skin disorders including Stevens-Johnson syndrome and toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS) (see WARNINGS ), alopecia.
Pharmacokinetics in Elderly
A pharmacokinetic study was conducted in 22 subjects, 65 years of age or older receiving a single 50 mg oral dose of fluconazole. Ten of these patients were concomitantly receiving diuretics. The Cmax was 1.54 mcg/mL and occurred at 1.3 hours post dose. The mean AUC was 76.4 ± 20.3 mcg•h/mL, and the mean terminal half-life was 46.2 hours. These pharmacokinetic parameter values are higher than analogous values reported for normal young male volunteers. Coadministration of diuretics did not significantly alter the AUC or Cmax. In addition, creatinine clearance (74 mL/min), the percent of drug recovered unchanged in urine (0 to 24 hours, 22%), and the fluconazole renal clearance estimates (0.124 mL/min/kg) for the elderly were generally lower than those of younger volunteers. Thus, the alteration of fluconazole disposition in the elderly appears to be related to reduced renal function characteristic of this group. A plot of each subject's terminal elimination half-life versus creatinine clearance compared to the predicted half-life – creatinine clearance curve derived from normal subjects and subjects with varying degrees of renal insufficiency indicated that 21 of 22 subjects fell within the 95% confidence limit of the predicted half-life – creatinine clearance curves. These results are consistent with the hypothesis that higher values for the pharmacokinetic parameters observed in the elderly subjects compared to normal young male volunteers are due to the decreased kidney function that is expected in the elderly.
Pharmacokinetics and Metabolism
The pharmacokinetic properties (PK) of fluconazole are similar following administration by the intravenous or oral routes. In normal volunteers, the bioavailability of orally administered fluconazole is over 90% compared with intravenous administration. Bioequivalence was established between the 100 mg tablet and both suspension strengths when administered as a single 200 mg dose.
Peak plasma concentrations (Cmax) in fasted normal volunteers occur between 1 and 2 hours with a terminal plasma elimination half-life of approximately 30 hours (range: 20 to 50 hours) after oral administration.
In fasted normal volunteers, administration of a single oral 400 mg dose of fluconazole leads to a mean Cmax of 6.72 mcg/mL (range: 4.12 to 8.08 mcg/mL) and after single or multiple oral doses of 50 to 400 mg, fluconazole plasma concentrations and area under the plasma concentration-time curve (AUC) are dose proportional (Table 1).
|
*AUC0-inf. C max = Peak plasma concentrations, AUC =area under the plasma concentration time curve. |
|||
| Dose regimen |
Cmax
(mcg/mL) |
AUC0-24
(mcg*h/mL) |
Half-life (hours) |
| 50 mg oral (once daily x 7 days) | 2.21 | 37.6 | 26.6 |
| 100 mg oral (once daily x 7 days) | 4.81 | 82.5 | 27.7 |
| 150 mg single oral | 2.70 | 137* | 34.1 |
| 200 mg oral (once daily x 14 days) | 10.12 | 169.5 | 31 |
| 300 mg oral (once daily x 14 days) | 15.98 | 299.4 | 34 |
| 400 mg oral (once daily x 14 days) | 18.89 | 349.9 | 31 |
Steady-state concentrations are reached within 5 to 10 days following oral doses of 50 to 400 mg given once daily. Administration of a loading dose (on Day 1) of twice the usual daily dose results in plasma concentrations close to steady-state by the second day. The apparent volume of distribution of fluconazole approximates that of total body water. Plasma protein binding is low (11 to 12%). Following either single- or multiple oral doses for up to 14 days, fluconazole penetrates into all body fluids studied (see table below). In normal volunteers, saliva concentrations of fluconazole were equal to or slightly greater than plasma concentrations regardless of dose, route, or duration of dosing. In patients with bronchiectasis, sputum concentrations of fluconazole following a single 150 mg oral dose were equal to plasma concentrations at both 4 and 24 hours post dose. In patients with fungal meningitis, fluconazole concentrations in the cerebrospinal fluid (CSF) are approximately 80% of the corresponding plasma concentrations.
A single oral 150 mg dose of fluconazole administered to 27 patients penetrated into vaginal tissue, resulting in tissue: plasma ratios ranging from 0.94 to 1.14 over the first 48 hours following dosing.
A single oral 150 mg dose of fluconazole administered to 14 patients penetrated into vaginal fluid, resulting in fluid: plasma ratios ranging from 0.36 to 0.71 over the first 72 hours following dosing.
|
* Relative to concurrent concentrations in plasma in subjects with normal renal function. |
|
|
† Independent of degree of meningeal inflammation. |
|
|
Tissue or Fluid |
Ratio of Fluconazole
Tissue (Fluid)/Plasma Concentration* |
| Cerebrospinal fluid† Saliva Sputum Blister fluid Urine Normal skin Nails Blister skin Vaginal tissue Vaginal fluid |
0.5 to 0.9 1 1 1 10 10 1 2 1 0.4 to 0.7 |
Mean body clearance in adults is reported to be 0.23 (17%) mL/min/kg. In normal volunteers, fluconazole is cleared primarily by renal excretion, with approximately 80% of the administered dose appearing in the urine as unchanged drug. About 11% of the dose is excreted in the urine as metabolites.
The pharmacokinetics of fluconazole are markedly affected by reduction in renal function. There is an inverse relationship between the elimination half-life and creatinine clearance. The dose of fluconazole may need to be reduced in patients with impaired renal function. (See DOSAGE AND ADMINISTRATION .) A 3-hour hemodialysis session decreases plasma concentrations by approximately 50%.
In normal volunteers, fluconazole administration (doses ranging from 200 mg to 400 mg once daily for up to 14 days) was associated with small and inconsistent effects on testosterone concentrations, endogenous corticosteroid concentrations, and the adrenocorticotropic hormone (ACTH)-stimulated cortisol response.
Dosing in Pediatric Patients On Ecmo
The recommended dosage of fluconazole in pediatric patients 3 months and older on ECMO is 35-mg/kg on the first day (not to exceed 800 mg) followed by 12-mg/kg once daily (not to exceed 400 mg).
For patients from birth to 3 months postnatal age, and gestational age less than 30 weeks, a loading dose of 35- mg/kg on the first day followed by 9-mg/kg once daily is recommended.
For patients from birth to 3 months postnatal age and gestational age 30 weeks and above, a loading dose of 35-mg/kg on the first day followed by 12-mg/kg once daily is recommended.
Cryptococcal meningitis: For the treatment of acute cryptococcal meningitis, the recommended dosage is 12 mg/kg on the first day, followed by 6 mg/kg once daily. A dosage of 12 mg/kg once daily may be used, based on medical judgment of the patient's response to therapy. The recommended duration of treatment for initial therapy of cryptococcal meningitis is 10 to 12 weeks after the cerebrospinal fluid becomes culture negative. For suppression of relapse of cryptococcal meningitis in pediatric patients with AIDS, the recommended dose of fluconazole is 6 mg/kg once daily.
In Patients Receiving Multiple Doses
Sixteen percent of over 4000 patients treated with fluconazole in clinical trials of 7 days or more experienced adverse events. Treatment was discontinued in 1.5% of patients due to adverse clinical events and in 1.3% of patients due to laboratory test abnormalities.
Clinical adverse events were reported more frequently in HIV infected patients (21%) than in non-HIV infected patients (13%); however, the patterns in HIV infected and non-HIV infected patients were similar. The proportions of patients discontinuing therapy due to clinical adverse events were similar in the two groups (1.5%).
The following treatment-related clinical adverse events occurred at an incidence of 1% or greater in 4048 patients receiving fluconazole for 7 or more days in clinical trials: nausea 3.7%, headache 1.9%, skin rash 1.8%, vomiting 1.7%, abdominal pain 1.7%, and diarrhea 1.5%.
Pharmacokinetics in Pediatric Patients
In pediatrics patients from 2 days to 15 years of age, the following pharmacokinetic data have been reported following the administration of fluconazole:
|
*Data for Clearance, Cmax, AUC and Vdss are presented as arithmetic mean (CV%) and Half-life as arithmetic mean only. *AUC0-inf; ^AUC0-24; Abbreviations :Cmax=Peak plasma concentrations, AUC =area under the plasma concentration time curve; and Vdss=volume of distribution at steady state. |
||||||
|
There are limited data available in patients 61 days to less than 9 months of age. |
||||||
|
Age Studied |
Dose (mg/kg) |
Clearance (mL/min/kg) |
Half-life (Hours) |
C
max
(mcg/mL) |
AUC
(mcg*h/mL) |
Vdss (L/kg) |
|
2 to 60 days |
Intravenous 25 mg/kg on day one followed by Intravenous 12 mg/kg once daily |
0.29 (35%) N=8 |
54.2 |
23.4(29%) N=8 |
439 (25%) |
1.13 (31%) |
| 9 months to 13 years | Single-Oral 2 mg/kg | 0.40 (38%) N=14 | 25.0 | 2.9 (22%) N=16 | 94.7 (34%)* N=14 | N/A |
| 9 months to 13 years | Single-Oral 8 mg/kg | 0.51 (60%) N=15 | 19.5 | 9.8 (20%) N=15 | 362.5 (58%)*=14 | N/A |
| 5 to 15 years |
Multiple Intravenous 2 mg/kg |
0.49 (40%) N=4 |
17.4 | 5.5 (25%) N=5 |
67.4 (26%)^ N=4 |
0.722 (36%) N=4 |
| 5 to 15 years |
Multiple Intravenous 4 mg/kg |
0.59 (64%) N=5 | 15.2 | 11.4 (44%) N=6 | 139.1 (46%)^ N=5 |
0.729 (33%) N=5 |
| 5 to 15 years |
Multiple Intravenous 8 mg/kg |
0.66 (31%) N=7 | 17.6 | 14.1 (22%) N=8 | 196.7(25%)^ N=7 |
1.069 (37%) N=7 |
In pediatric patients (premature newborns; gestational age 26 to 29 weeks and postnatal age from birth to 1 (day), the mean (%cv) clearance within 36 hours of birth was 0.180 (35%, N=7) mL/min/kg, which increased with time to a mean of 0.218 (31%, N=9) mL/min/kg 6 days later and 0.333 (56%, N=4) mL/min/kg 12 days later. Similarly, the half-life was 73.6 hours, which decreased with time to a mean of 53.2 hours 6 days later and 46.6 hours 12 days later.
In a study of 13 pediatric patients (preterm and term infants with median gestational age (GA) of 37 weeks, GA range 24 to 39 weeks; median postnatal age [PNA] 19 days, PNA range 5 to 262 days) 12 infants received a 25 mg/kg loading dose, and 9/12 (75%) achieved an AUC0-24 of >400-mg*h/L in the first 24 hours. A population pharmacokinetic model using data from 55 pediatric patients (GA 23 to 40 weeks, PNA 1 to 88 days) found that a loading dose of 25 mg/kg is necessary to reach target AUC >400-mg*h/L within 24 hours of initiating therapy in pediatric patients younger than 3 months of age. A maintenance dose of 9 mg/kg daily should be used in pediatric patients born at GA less than 30 weeks and 12 mg/kg daily in pediatric patients with GA equal or greater than 30 weeks. (See DOSAGE AND ADMINISTRATION .)
A population PK model using data from 21 pediatric patients ages from birth to 17 years supported with extracorporeal membrane oxygenation (ECMO), and 19 pediatric non-ECMO patients ages from birth to 2 years found that clearance was related to serum creatinine while a higher volume of distribution was related to presence of ECMO support. The median volume of distribution was 1.3 L/kg in pediatric patients on ECMO and 0.9 L/kg in those not on ECMO. Simulations suggested that a loading dose of 35 mg/kg is needed to achieve the target AUC0-24 >400 mg*h/L within the first 24 hours in pediatric patients on ECMO. (See DOSAGE AND ADMINISTRATION.)
Adverse Reactions in Pediatric Patients
The pattern and incidence of adverse events and laboratory abnormalities recorded during pediatric clinical trials are comparable to those seen in adults.
In Phase II/III clinical trials conducted in the United States and in Europe, 577 pediatric patients, ages 1 day to 17 years were treated with fluconazole at doses up to 15 mg/kg/day for up to 1,616 days. Thirteen percent of pediatric patients experienced treatment-related adverse events. The most commonly reported events were vomiting (5%), abdominal pain (3%), nausea (2%), and diarrhea (2%). Treatment was discontinued in 2.3% of patients due to adverse clinical events and in 1.4% of patients due to laboratory test abnormalities. The majority of treatment-related laboratory abnormalities were elevations of transaminases or alkaline phosphatase.
| Percentage of Patients With Treatment-Related Side Effects | ||
|
Fluconazole
(N=577) |
Comparative Agents
(N=451) |
|
| With any side effect | 13.0 | 9.3 |
| Vomiting | 5.4 | 5.1 |
| Abdominal pain | 2.8 | 1.6 |
| Nausea | 2.3 | 1.6 |
| Diarrhea | 2.1 | 2.2 |
Clinical Trials Experience in Pediatric Patients
Safety in Prophylaxis of Invasive Candida Infections in Premature infants weighing less than 750 grams at birth.
In a Phase 3 clinical trial of pediatric patients (premature infants weighing less than 750 grams at birth), the incidence of intestinal perforation in infants receiving fluconazole prophylaxis was higher compared to infants receiving placebo (see PRECAUTIONS: Pediatric Use).
Safety in Pediatric Patients Receiving ECMO
A cohort of 20 pediatric patients (1 day to 17 years of age) on ECMO received fluconazole in a prospective, open-label, single-center safety and PK ECMO study. The adverse reaction profile of fluconazole in these patients was similar to that of adult and pediatric non-ECMO patients (See PRECAUTIONS: Pediatric Use).
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Dosage in Patients With Impaired Renal Function
Fluconazole is cleared primarily by renal excretion as unchanged drug. In patients with impaired renal function who will receive multiple doses of fluconazole, an initial loading dose of 50 mg to 400 mg should be given. After the loading dose, the daily dose (according to indication) should be based on the following summary:
| Creatinine Clearance (mL/min) | Recommended Dose (%) |
| >50 | 100 |
| ≤50 (no dialysis) | 50 |
| Hemodialysis | 100% after each hemodialysis |
Patients on hemodialysis should receive 100% of the recommended dose after each hemodialysis; on non-dialysis days, patients should receive a reduced dose according to their creatinine clearance.
These are suggested dose adjustments based on pharmacokinetics following administration of multiple doses. Further adjustment may be needed depending upon clinical condition.
When serum creatinine is the only measure of renal function available, the following formula (based on sex, weight, and age of the patient) should be used to estimate the creatinine clearance in adults:
| Males: | Weight (kg) × (140 - age) |
| 72 × serum creatinine (mg/100 mL) |
Females: 0.85 × above value
Although the pharmacokinetics of fluconazole has not been studied in pediatric patients with renal insufficiency, dosage reduction in pediatric patientswith renal insufficiency should parallel that recommended for adults. The following formula may be used to estimate creatinine clearance in pediatric patients:
| K × | linear length or height (cm) |
| serum creatinine (mg/100 mL) |
(Where K=0.55 for pediatric patients older than 1 year and 0.45 for infants.)
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Fluconazole showed no evidence of carcinogenic potential in mice and rats treated orally for 24 months at doses of 2.5 mg/kg/day, 5 mg/kg/day, or 10 mg/kg/day (approximately 2 to 7 times the recommended human dose). Male rats treated with 5 mg/kg/day and 10 mg/kg/day had an increased incidence of hepatocellular adenomas.
Fluconazole, with or without metabolic activation, was negative in tests for mutagenicity in four strains of S. typhimurium, and in the mouse lymphoma L5178Y system. Cytogenetic studies in vivo (murine bone marrow cells, following oral administration of fluconazole) and in vitro (human lymphocytes exposed to fluconazole at 1000 mcg/mL) showed no evidence of chromosomal mutations.
Fluconazole did not affect the fertility of male or female rats treated orally with daily doses of 5 mg/kg, 10 mg/kg, or 20 mg/kg or with parenteral doses of 5 mg/kg, 25 mg/kg, or 75 mg/kg, although the onset of parturition was slightly delayed at 20 mg/kg PO. In an intravenous perinatal study in rats at 5 mg/kg, 20 mg/kg, and 40 mg/kg, dystocia and prolongation of parturition were observed in a few dams at 20 mg/kg (approximately 5 to 15 times the recommended human dose) and 40 mg/kg, but not at 5 mg/kg. The disturbances in parturition were reflected by a slight increase in the number of still born pups and decrease of neonatal survival at these dose levels. The effects on parturition in rats are consistent with the species specific estrogen-lowering property produced by high doses of fluconazole. Such a hormone change has not been observed in women treated with fluconazole. (See CLINICAL PHARMACOLOGY .)
Structured Label Content
Warnings (WARNINGS)
(1) Hepatic injury: Fluconazole should be administered with caution to patients with liver dysfunction. Fluconazole has been associated with rare cases of serious hepatic toxicity, including fatalities primarily in patients with serious underlying medical conditions. In cases of fluconazole-associated hepatotoxicity, no obvious relationship to total daily dose, duration of therapy, sex, or age of the patient has been observed. Fluconazole hepatotoxicity has usually, but not always, been reversible on discontinuation of therapy. Patients who develop abnormal liver function tests during fluconazole therapy should be monitored for the development of more severe hepatic injury. Fluconazole should be discontinued if clinical signs and symptoms consistent with liver disease develop that may be attributable to fluconazole.
(2) Anaphylaxis: In rare cases, anaphylaxis has been reported.
(3) Dermatologic: Exfoliative skin disorders during treatment with fluconazole have been reported. Fatal outcomes have been reported in patients with serious underlying diseases. Patients with deep seated fungal infections who develop rashes during treatment with fluconazole should be monitored closely and the drug discontinued if lesions progress. Fluconazole should be discontinued in patients treated for superficial fungal infection who develop a rash that may be attributed to fluconazole.
(4) Potential for fetal harm: There are no adequate and well-controlled clinical trials of fluconazole in pregnant women. Case reports describe a pattern of distinct congenital anomalies in infants exposed in utero to high dose maternal fluconazole (400 to 800 mg/day) during most or all of the first trimester. These reported anomalies are similar to those seen in animal studies. If fluconazole is used during pregnancy or if the patient becomes pregnant while taking the drug, the patient should be informed of the potential hazard to the fetus. Effective contraceptive measures should be considered in women of child-bearing potential who are being treated with fluconazole 400 to 800 mg/day and should continue throughout the treatment period and for approximately 1 week (5 to 6 half-lives) after the final dose. Epidemiological studies suggest a potential risk of spontaneous abortion and congenital abnormalities in infants whose mothers were treated with 150 mg of fluconazole as a single or repeated dose in the first trimester, but these epidemiological studies have limitations and these findings have not been confirmed in controlled clinical trials. (See PRECAUTIONS: Pregnancy .)
Indications and Usage (INDICATIONS AND USAGE)
Fluconazole in Sodium Chloride Injection, USP is indicated for the treatment of:
- Oropharyngeal and esophageal candidiasis. In open noncomparative studies of relatively small numbers of patients, fluconazole was also effective for the treatment of Candida urinary tract infections, peritonitis, and systemic Candida infections including candidemia, disseminated candidiasis, and pneumonia.
- Cryptococcal meningitis. Before prescribing fluconazole for AIDS patients with cryptococcal meningitis, please see CLINICAL STUDIES section. Studies comparing fluconazole to amphotericin B in non-HIV infected patients have not been conducted.
Prophylaxis: Fluconazole is also indicated to decrease the incidence of candidiasis in patients undergoing bone marrow transplantation who receive cytotoxic chemotherapy and/or radiation therapy.
Specimens for fungal culture and other relevant laboratory studies (serology, histopathology) should be obtained prior to therapy to isolate and identify causative organisms. Therapy may be instituted before the results of the cultures and other laboratory studies are known; however, once these results become available, anti-infective therapy should be adjusted accordingly.
Contraindications (CONTRAINDICATIONS)
Fluconazole is contraindicated in patients who have shown hypersensitivity to fluconazole or to any of its excipients. There is no information regarding cross-hypersensitivity between fluconazole and other azole antifungal agents. Caution should be used in prescribing fluconazole to patients with hypersensitivity to other azoles. Coadministration of other drugs known to prolong the QT interval and which are metabolized via the enzyme CYP3A4 such as erythromycin, pimozide, and quinidine are contraindicated in patients receiving fluconazole. (See CLINICAL PHARMACOLOGY: Drug Interaction Studies and PRECAUTIONS .)
Adverse Reactions (ADVERSE REACTIONS)
Fluconazole is generally well tolerated.
In some patients, particularly those with serious underlying diseases such as AIDS and cancer, changes in renal and hematological function test results and hepatic abnormalities have been observed during treatment with fluconazole and comparative agents, but the clinical significance and relationship to treatment is uncertain.
How Supplied (HOW SUPPLIED)
Fluconazole in Sodium Chloride Injection, USP is supplied as follows:
| Fluconazole in Sodium Chloride Injection, USP | ||
| NDC | (2 mg per mL) | Package Factor |
| 25021-184-82 | 200 mg per 100 mL in flexible container bag | 10 bags per carton |
| 25021-184-87 | 400 mg per 200 mL in flexible container bag | 10 bags per carton |
Fluconazole in Sodium Chloride Injection, USP for intravenous infusion administration is formulated as iso-osmotic solutions containing 2 mg per mL of fluconazole.
Description (DESCRIPTION)
Fluconazole in Sodium Chloride Injection, USP, the first of a new subclass of synthetic triazole antifungal agents, is available as a sterile solution for intravenous use in plastic containers.
Fluconazole is designated chemically as 2,4-difluoro-α,α1-bis(1H-1,2,4-triazol-1-ylmethyl) benzyl alcohol with an empirical formula of C13H12F2N6O and molecular weight of 306.3. The structural formula is:
Fluconazole is a white crystalline solid which is slightly soluble in water and saline.
Fluconazole in Sodium Chloride Injection, USP is an iso-osmotic, sterile, nonpyrogenic solution of fluconazole in a sodium chloride diluent. Each mL contains 2 mg of fluconazole and 9 mg of sodium chloride. The pH ranges from 4.0 to 8.0. Injection volumes of 100 mL and 200 mL are packaged in plastic containers.
The flexible bag container is fabricated from a specially formulated non-plasticized thermoplastic co-polyolefin. The amount of water that can permeate from the container into the overwrap is insufficient to affect the solution significantly. The suitability of the container material has been confirmed in tests in animals according to USP biological tests for plastic containers.
Section 42229-5 (42229-5)
SAGENT®
Rx only
Section 51945-4 (51945-4)
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bag Label
NDC 25021-184-82
100 mL
Fluconazole In Sodium Chloride Injection, USP
200 mg per 100 mL
(2 mg per mL)
Rx only
Iso-Osmotic Sodium Chloride Diluent
For Intravenous Use
Single-Dose Container
General
Some azoles, including fluconazole, have been associated with prolongation of the QT interval on the electrocardiogram. Fluconazole causes QT prolongation via the inhibition of Rectifier Potassium Channel current (Ikr). The QT prolongation caused by other medicinal products (such as amiodarone) may be amplified via the inhibition of cytochrome P450 (CYP) 3A4. (See PRECAUTIONS, Drug Interactions .) During post-marketing surveillance, there have been rare cases of QT prolongation and torsade de pointes in patients taking fluconazole. Most of these reports involved seriously ill patients with multiple confounding risk factors, such as structural heart disease, electrolyte abnormalities, and concomitant medications that may have been contributory. Patients with hypokalemia and advanced cardiac failure are at an increased risk for the occurrence of life-threatening ventricular arrhythmias and torsade de pointes.
Fluconazole should be administered with caution to patients with these potentially proarrhythmic conditions.
Concomitant use of fluconazole and erythromycin has the potential to increase the risk of cardiotoxicity (prolonged QT interval, torsade de pointes) and consequently sudden heart death. This combination should be avoided.
Fluconazole should be administered with caution to patients with renal dysfunction.
Adrenal insufficiency has been reported in patients receiving azoles, including fluconazole. Reversible cases of adrenal insufficiency have been reported in patients receiving fluconazole.
When driving vehicles or operating machines, it should be taken into account that occasionally dizziness or seizures may occur.
There have been reports of cases of superinfection with Candida species other than C. albicans, which are often inherently not susceptible to fluconazole (e.g., Candida krusei). Such cases may require alternative antifungal therapy (see CLINICAL PHARMACOLOGY, Microbiology).
To Open
Tear overwrap down side at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. After removing overwrap, check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired.
DO NOT ADD SUPPLEMENTARY MEDICATION.
Preparation for Administration:
- Suspend container from eyelet support.
- Remove plastic protector from outlet port at bottom of container.
- Attach administration set. Refer to complete directions accompanying set.
Human Data
Case reports describe a distinctive and rare pattern of birth defects among infants whose mothers received high-dose (400 to 800 mg/day) fluconazole during most or all of the first trimester of pregnancy. The features seen in these infants include brachycephaly, abnormal facies, abnormal calvarial development, cleft palate, femoral bowing, thin ribs and long bones, arthrogryposis, and congenital heart disease. These effects are similar to those seen in animal studies.
Epidemiological studies suggest a potential risk of spontaneous abortion and congenital abnormalities in infants whose mothers were treated with 150 mg of fluconazole as a single or repeated dose in the first trimester, but these epidemiological studies have limitations and these findings have not been confirmed in controlled clinical trials.
Overdosage (OVERDOSAGE)
There have been reports of overdose with fluconazole accompanied by hallucination and paranoid behavior.
In the event of overdose, symptomatic treatment (with supportive measures and gastric lavage if clinically indicated) should be instituted.
Fluconazole is largely excreted in urine. A 3-hour hemodialysis session decreases plasma levels by approximately 50%.
In mice and rats receiving very high doses of fluconazole, clinical effects in both species included decreased motility and respiration, ptosis, lacrimation, salivation, urinary incontinence, loss of righting reflex, and cyanosis; death was sometimes preceded by clonic convulsions.
Resistance
A potential for development of resistance to fluconazole is well known. Fungal isolates exhibiting reduced susceptibility to other azoles may also show reduced susceptibility to fluconazole. The frequency of drug resistance development for the various fungi for which this drug is indicated is not known.
Fluconazole resistance may arise from a modification in the quality or quantity of the target enzyme (lanosterol 14-α-demethylase), reduced access to the drug target, or some combination of these mechanisms.
Point mutations in the gene (ERG11) encoding for the target enzyme lead to an altered target with decreased affinity for azoles. Overexpression of ERG11 results in the production of high concentrations of the target enzyme, creating the need for higher intracellular drug concentrations to inhibit all of the enzyme molecules in the cell.
The second major mechanism of drug resistance involves active efflux of fluconazole out of the cell through the activation of two types of multidrug efflux transporters; the major facilitators (encoded by MDR genes) and those of the ATP-binding cassette superfamily (encoded by CDR genes). Upregulation of the MDR gene leads to fluconazole resistance, whereas, upregulation of CDR genes may lead to resistance to multiple azoles.
Resistance in Candida glabrata usually includes upregulation of CDR genes resulting in resistance to multiple azoles. For an isolate where the minimum inhibitory concentration (MIC) is categorized as Intermediate (16 to 32 mcg/mL), the highest fluconazole dose is recommended.
Animal Data
Fluconazole was administered orally to pregnant rabbits during organogenesis in two studies at doses of 5 mg/kg, 10 mg/kg, and 20 mg/kg and at 5 mg/kg, 25 mg/kg, and 75 mg/kg, respectively. Maternal weight gain was impaired at all dose levels (approximately 0.25 to 4 times the 400 mg clinical dose based on body surface area [BSA] comparison), and abortions occurred at 75 mg/kg (approximately 4 times the 400 mg clinical dose based on BSA); no adverse fetal effects were observed.
In several studies in which pregnant rats received fluconazole orally during organogenesis, maternal weight gain was impaired and placental weights were increased at 25 mg/kg. There were no fetal effects at 5 mg/kg or 10 mg/kg; increases in fetal anatomical variants (supernumerary ribs, renal pelvis dilation) and delays in ossification were observed at 25 mg/kg and 50 mg/kg and higher doses. At doses ranging from 80 to 320 mg/kg (approximately 2 to 8 times the 400 mg clinical dose based on BSA), embryolethality in rats was increased and fetal abnormalities included wavy ribs, cleft palate, and abnormal craniofacial ossification. These effects are consistent with the inhibition of estrogen synthesis in rats and may be a result of known effects of lowered estrogen on pregnancy, organogenesis, and parturition.
Geriatric Use
In non-AIDS patients, side effects possibly related to fluconazole treatment were reported in fewer patients aged 65 and older (9%, n =339) than for younger patients (14%, n=2240). However, there was no consistent difference between the older and younger patients with respect to individual side effects. Of the most frequently reported (>1%) side effects, rash, vomiting, and diarrhea occurred in greater proportions of older patients. Similar proportions of older patients (2.4%) and younger patients (1.5%) discontinued fluconazole therapy because of side effects. In post-marketing experience, spontaneous reports of anemia and acute renal failure were more frequent among patients 65 years of age or older than in those between 12 and 65 years of age. Because of the voluntary nature of the reports and the natural increase in the incidence of anemia and renal failure in the elderly, it is however not possible to establish a causal relationship to drug exposure.
Controlled clinical trials of fluconazole did not include sufficient numbers of patients aged 65 and older to evaluate whether they respond differently from younger patients in each indication. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
Fluconazole is primarily cleared by renal excretion as unchanged drug. Because elderly patients are more likely to have decreased renal function, care should be taken to adjust dose based on creatinine clearance. It may be useful to monitor renal function. (See CLINICAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION .)
Multiple Dose
SINCE ORAL ABSORPTION IS RAPID AND ALMOST COMPLETE, THE DAILY DOSE OF FLUCONAZOLE IS THE SAME FOR ORAL AND INTRAVENOUS ADMINISTRATION. In general, a loading dose of twice the daily dose is recommended on the first day of therapy to result in plasma concentrations close to steady-state by the second day of therapy.
The daily dose of fluconazole for the treatment of infection should be based on the infecting organism and the patient's response to therapy. Treatment should be continued until clinical parameters or laboratory tests indicate that active fungal infection has subsided. An inadequate period of treatment may lead to recurrence of active infection. Patients with AIDS and cryptococcal meningitis or recurrent oropharyngeal candidiasis usually require maintenance therapy to prevent relapse.
Pediatric Use
Use in Pediatric Patients for the Treatment of Oropharyngeal Candidiasis
An open-label, randomized, controlled trial has shown fluconazole to be effective in the treatment of oropharyngeal candidiasis in pediatric patients 6 months to 13 years of age. (See CLINICAL STUDIES .)
Use in Pediatric Patients for the Treatment of Candida Esophagitis, Systemic Candida Infections, or Cryptococcal Meningitis
The use of fluconazole in pediatric patients with cryptococcal meningitis, Candida esophagitis, or systemic Candida infections is supported by the efficacy shown for these indications in adults and by the results from several small noncomparative pediatric clinical studies. In addition, pharmacokinetic studies in pediatric patients (See CLINICAL PHARMACOLOGY ) have established a dose proportionality between pediatric patients and adults. (See DOSAGE AND ADMINISTRATION .)
In a noncomparative study of fluconazole administered to pediatric patients (from birth to less than 17 years) with serious systemic fungal infections, most of which were candidemia, the effectiveness of fluconazole was similar to that reported for the treatment of candidemia in adults. Of 17 subjects with culture-confirmed candidemia, 11 of 14 (79%) with baseline symptoms (3 were asymptomatic) had a clinical cure; 13/15 (87%) of evaluable patients had a mycologic cure at the end of treatment but two of these patients relapsed at 10 and 18 days, respectively, following cessation of therapy.
The efficacy of fluconazole for the suppression of cryptococcal meningitis was successful in 4 of 5 pediatric patients (4 years to 10 years of age) treated in a compassionate-use study of fluconazole for the treatment of life-threatening or serious mycosis. There are limited clinical data to support the efficacy of fluconazole for primary treatment of cryptococcal meningitis in pediatric patients.
The safety profile of fluconazole in children has been studied in 577 pediatric patients from 1 day to 17 years of age who received doses ranging from 1 to 15 mg/kg/day for 1 to 1,616 days. (See ADVERSE REACTIONS .)
Use in Pediatric Patients on Extracorporeal Membrane Oxygenation (ECMO)
A prospective, open-label, single-center study was conducted to determine the PK and safety of fluconazole in pediatric patients (ages: from birth to 17 years of age) on ECMO (see CLINICAL PHARMACOLOGY ). A loading dose of 35-mg/kg is recommended in pediatric patients on ECMO due to increased volume of distribution (see DOSAGE AND ADMINISTRATION ).
Use in Prophylaxis of Invasive Candida Infections in Pediatric Patients (premature infants
weighing less than 750 grams at birth)
Safety and effectiveness of fluconazole for the prophylaxis of invasive candidiasis in pediatric patients (premature infants weighing less than 750 grams at birth) have not been established.
A prospective, randomized, double-blind, placebo-controlled, multicenter trial was conducted in premature infants weighing less than 750 grams at birth to evaluate the efficacy and safety of prophylactic fluconazole 6-mg/kg administered twice weekly for 6 weeks versus placebo (NCT00734539). Efficacy was assessed using the endpoint of death or candidiasis by study day 49. The results are summarized in Table 4.
|
*Subjects with missing data are imputed as having candidiasis or died. |
||||
|
**Subjects may be counted more than once as two fluconazole subjects and four placebo subjects diagnosed with candidiasis subsequently died by day 49. |
||||
| Fluconazole (N=188) n (%) | Placebo (N=173) n (%) | P-value | Difference (95% CI) | |
| Death or candidiasis* | 33 (17.6) | 38 (22.0) | 0.2954 | -4.4 (-12.6, 3.8) |
| Components of endpoint** Death Candidiasis Missing |
27 (14.4) 6 (3.2) 2 (1.0) |
25 (14.5) 16 (9.2) 1 (0.5) |
The most common fatal serious adverse reactions in the fluconazole vs placebo arms, respectively, were necrotizing enterocolitis (NEC), 9 (5%) vs 9 (5%); neonatal bacterial sepsis, 6 (3%) vs 7 (4%); and neonatal respiratory failure, 4 (2%) vs 2 (0.6%).
The most common serious adverse reactions (>5%) reported in patients receiving fluconazole prophylaxis are displayed in Table 5.
|
*All serious adverse reactions were assessed and recorded up through 30 days after the final dose of study drug. Serious adverse reactions included both fatal and non-fatal outcomes. |
||
| Adverse Reaction | Fluconazole (N=188) n (%) | Placebo (N=173) n (%) |
| Necrotizing Enterocolitis (NEC) | 27 (14) | 28 (16) |
| Intestinal Perforation (includes ileal/small intestinal perforation) | 13 (7) | 7 (4) |
| Neonatal Respiratory Arrest/ Neonatal Respiratory Failure | 13 (7) | 4 (2) |
| Bacterial Sepsis, Neonatal | 10 (5) | 12 (7) |
Administration
Fluconazole in Sodium Chloride Injection, USP may be administered by intravenous infusion. Fluconazole in Sodium Chloride Injection, USP has been used safely for up to fourteen days of intravenous therapy. The intravenous infusion of fluconazole should be administered at a maximum rate of approximately 200 mg/hour, given as a continuous infusion.
Fluconazole in Sodium Chloride Injection, USP in plastic containers are intended only for intravenous administration using sterile equipment.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
Do not use if the solution is cloudy or precipitated or if the seal is not intact.
Directions for Intravenous Use of Fluconazole in Plastic Containers
Do not remove unit from overwrap until ready for use. The overwrap is a moisture barrier. The inner bag maintains the sterility of the product.
CAUTION: Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Nursing Mothers
Fluconazole was present in low levels in breast milk following administration of a single 150 mg dose, based on data from a study in 10 breastfeeding women who temporarily or permanently discontinued breastfeeding 5 days to 19 months postpartum. The estimated daily infant dose of fluconazole from breast milk (assuming mean milk consumption of 150 mL/kg/day) based on the mean peak milk concentration (2.61 mcg/mL [range: 1.57 to 3.65 mcg/mL] at 5.2 hours post-dose) was 0.39 mg/kg/day, which is approximately 13% of the recommended pediatric dose for oropharyngeal candidiasis. (Labeled pediatric dose is 6 mg/kg/day on the first day followed by 3 mg/kg/day; estimated infant dose is 13% of 3 mg/kg/day maintenance dose). There are no data on fluconazole levels in milk after repeated use or after high-dose fluconazole. A published survey of 96 breastfeeding women who were treated with fluconazole 150 mg every other day (average of 7.3 capsules [range 1 to 29 capsules]) for lactation-associated candida of the breasts reported no serious adverse reactions in infants. Caution should be exercised when fluconazole is administered to a nursing woman.
Clinical Studies (CLINICAL STUDIES)
Cryptococcal meningitis: In a multicenter study comparing fluconazole (200 mg/day) to amphotericin B (0.3 mg/kg/day) for treatment of cryptococcal meningitis in patients with AIDS, a multivariate analysis revealed three pretreatment factors that predicted death during the course of therapy: abnormal mental status, cerebrospinal fluid cryptococcal antigen titer greater than 1:1024, and cerebrospinal fluid white blood cell count of less than 20 cells/mm3. Mortality among high risk patients was 33% and 40% for amphotericin B and fluconazole patients, respectively (p=0.58), with overall deaths 14% (9 of 63 subjects) and 18% (24 of 131 subjects) for the 2 arms of the study (p=0.48). Optimal doses and regimens for patients with acute cryptococcal meningitis and at high risk for treatment failure remain to be determined. (Saag, et al. N Engl J Med 1992; 326:83-9.)
Pediatric Studies
Oropharyngeal candidiasis: An open-label, comparative study of the efficacy and safety of fluconazole (2 to 3 mg/kg/day) and oral nystatin (400,000 I.U. 4 times daily) was conducted in immunocompromised pediatric patients from 6 months to 13 years of age with oropharyngeal candidiasis. Clinical and mycological response rates were higher in pediatric patients treated with fluconazole.
Clinical cure at the end of treatment was reported for 86% of fluconazole-treated patients compared to 46% of nystatin treated patients. Mycologically, 76% of fluconazole treated patients had the infecting organism eradicated compared to 11% for nystatin treated patients.
|
* Subjects without follow-up cultures for any reason were considered nonevaluable for mycological response. |
||
| Fluconazole | Nystatin | |
| Enrolled | 96 | 90 |
| Clinical Cure | 76/88 (86%) | 36/78 (46%) |
| Mycological eradication* | 55/72 (76%) | 6/54 (11%) |
The proportion of patients with clinical relapse 2 weeks after the end of treatment was 14% for subjects receiving fluconazole and 16% for subjects receiving nystatin. At 4 weeks after the end of treatment, the percentages of patients with clinical relapse were 22% for fluconazole and 23% for nystatin.
Storage Conditions
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
AVOID EXCESSIVE HEAT. PROTECT FROM FREEZING.
Sterile, Nonpyrogenic, Preservative-free, PVC-free, DEHP-free.
The container and container closure are not made with natural rubber latex.
Brands listed are the trademarks of their respective owners.
SAGENT®
Mfd. for SAGENT Pharmaceuticals
Schaumburg, IL 60195 (USA)
Made in Switzerland
©2024 Sagent Pharmaceuticals
Revised: March 2024
SAGENT Pharmaceuticals®
Mechanism of Action
Fluconazole is a highly selective inhibitor of fungal cytochrome P450 dependent enzyme lanosterol 14-α-demethylase. This enzyme functions to convert lanosterol to ergosterol. The subsequent loss of normal sterols correlates with the accumulation of 14-α-methyl sterols in fungi and may be responsible for the fungistatic activity of fluconazole. Mammalian cell demethylation is much less sensitive to fluconazole inhibition.
Teratogenic Effects.
Potential for Fetal Harm: Use in pregnancy should be avoided except in patients with severe or potentially life-threatening fungal infections in whom fluconazole may be used if the anticipated benefit outweighs the possible risk to the fetus. A few published case reports describe a pattern of distinct congenital anomalies in infants exposed in utero to high dose maternal fluconazole (400 to 800 mg/day) during most or all of the first trimester. These reported anomalies are similar to those seen in animal studies. Effective contraceptive measures should be considered in women of child-bearing potential who are being treated with fluconazole 400 to 800 mg/day and should continue throughout the treatment period and for approximately 1 week (5 to 6 half-lives) after the final dose. If fluconazole is used during pregnancy, or if the patient becomes pregnant while taking the drug, the patient should be informed of the potential hazard to the fetus. Spontaneous abortions and congenital abnormalities have been suggested as potential risks associated with 150 mg of fluconazole as a single or repeated dose in the first trimester of pregnancy based on retrospective epidemiological studies. There are no adequate and well-controlled studies of fluconazole in pregnant women. (See WARNINGS: Potential for Fetal Harm .)
Antimicrobial Activity
Fluconazole has been shown to be active against most isolates of the following microorganisms both in vitro and in clinical infections.
Candida albicans
Candida glabrata (Many isolates are intermediately susceptible)
Candida parapsilosis
Candida tropicalis
Cryptococcus neoformans
The following in vitro data are available, but their clinical significance is unknown. At least 90% of the following fungi exhibit an in vitro MIC less than or equal to the susceptible breakpoint for fluconazole (https://www.fda.gov/STIC) against isolates of similar genus or organism group. However, the effectiveness of fluconazole in treating clinical infections due to these fungi has not been established in adequate and well-controlled clinical trials.
Candida dubliniensis
Candida guilliermondii
Candida kefyr
Candida lusitaniae
Candida krusei should be considered to be resistant to fluconazole. Resistance in C. krusei appears to be mediated by reduced sensitivity of the target enzyme to inhibition by the agent.
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
Post Marketing Experience (Post-Marketing Experience)
In addition, the following adverse events have occurred during post-marketing experience.
Immunologic: In rare cases, anaphylaxis (including angioedema, face edema and pruritus) has been reported.
Body as a Whole: Asthenia, fatigue, fever, malaise.
Cardiovascular: QT prolongation, torsade de pointes. (See PRECAUTIONS .)
Central Nervous System: Seizures, dizziness.
Hematopoietic and Lymphatic: Leukopenia, including neutropenia and agranulocytosis, thrombocytopenia.
Metabolic: Hypercholesterolemia, hypertriglyceridemia, hypokalemia.
Gastrointestinal: Cholestasis, dry mouth, hepatocellular damage, dyspepsia, vomiting.
Other Senses: Taste perversion.
Musculoskeletal System: myalgia.
Nervous System: Insomnia, paresthesia, somnolence, tremor, vertigo.
Skin and Appendages: Acute generalized exanthematous pustulosis, drug eruption including fixed drug eruption, increased sweating, exfoliative skin disorders including Stevens-Johnson syndrome and toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS) (see WARNINGS ), alopecia.
Pharmacokinetics in Elderly
A pharmacokinetic study was conducted in 22 subjects, 65 years of age or older receiving a single 50 mg oral dose of fluconazole. Ten of these patients were concomitantly receiving diuretics. The Cmax was 1.54 mcg/mL and occurred at 1.3 hours post dose. The mean AUC was 76.4 ± 20.3 mcg•h/mL, and the mean terminal half-life was 46.2 hours. These pharmacokinetic parameter values are higher than analogous values reported for normal young male volunteers. Coadministration of diuretics did not significantly alter the AUC or Cmax. In addition, creatinine clearance (74 mL/min), the percent of drug recovered unchanged in urine (0 to 24 hours, 22%), and the fluconazole renal clearance estimates (0.124 mL/min/kg) for the elderly were generally lower than those of younger volunteers. Thus, the alteration of fluconazole disposition in the elderly appears to be related to reduced renal function characteristic of this group. A plot of each subject's terminal elimination half-life versus creatinine clearance compared to the predicted half-life – creatinine clearance curve derived from normal subjects and subjects with varying degrees of renal insufficiency indicated that 21 of 22 subjects fell within the 95% confidence limit of the predicted half-life – creatinine clearance curves. These results are consistent with the hypothesis that higher values for the pharmacokinetic parameters observed in the elderly subjects compared to normal young male volunteers are due to the decreased kidney function that is expected in the elderly.
Pharmacokinetics and Metabolism
The pharmacokinetic properties (PK) of fluconazole are similar following administration by the intravenous or oral routes. In normal volunteers, the bioavailability of orally administered fluconazole is over 90% compared with intravenous administration. Bioequivalence was established between the 100 mg tablet and both suspension strengths when administered as a single 200 mg dose.
Peak plasma concentrations (Cmax) in fasted normal volunteers occur between 1 and 2 hours with a terminal plasma elimination half-life of approximately 30 hours (range: 20 to 50 hours) after oral administration.
In fasted normal volunteers, administration of a single oral 400 mg dose of fluconazole leads to a mean Cmax of 6.72 mcg/mL (range: 4.12 to 8.08 mcg/mL) and after single or multiple oral doses of 50 to 400 mg, fluconazole plasma concentrations and area under the plasma concentration-time curve (AUC) are dose proportional (Table 1).
|
*AUC0-inf. C max = Peak plasma concentrations, AUC =area under the plasma concentration time curve. |
|||
| Dose regimen |
Cmax
(mcg/mL) |
AUC0-24
(mcg*h/mL) |
Half-life (hours) |
| 50 mg oral (once daily x 7 days) | 2.21 | 37.6 | 26.6 |
| 100 mg oral (once daily x 7 days) | 4.81 | 82.5 | 27.7 |
| 150 mg single oral | 2.70 | 137* | 34.1 |
| 200 mg oral (once daily x 14 days) | 10.12 | 169.5 | 31 |
| 300 mg oral (once daily x 14 days) | 15.98 | 299.4 | 34 |
| 400 mg oral (once daily x 14 days) | 18.89 | 349.9 | 31 |
Steady-state concentrations are reached within 5 to 10 days following oral doses of 50 to 400 mg given once daily. Administration of a loading dose (on Day 1) of twice the usual daily dose results in plasma concentrations close to steady-state by the second day. The apparent volume of distribution of fluconazole approximates that of total body water. Plasma protein binding is low (11 to 12%). Following either single- or multiple oral doses for up to 14 days, fluconazole penetrates into all body fluids studied (see table below). In normal volunteers, saliva concentrations of fluconazole were equal to or slightly greater than plasma concentrations regardless of dose, route, or duration of dosing. In patients with bronchiectasis, sputum concentrations of fluconazole following a single 150 mg oral dose were equal to plasma concentrations at both 4 and 24 hours post dose. In patients with fungal meningitis, fluconazole concentrations in the cerebrospinal fluid (CSF) are approximately 80% of the corresponding plasma concentrations.
A single oral 150 mg dose of fluconazole administered to 27 patients penetrated into vaginal tissue, resulting in tissue: plasma ratios ranging from 0.94 to 1.14 over the first 48 hours following dosing.
A single oral 150 mg dose of fluconazole administered to 14 patients penetrated into vaginal fluid, resulting in fluid: plasma ratios ranging from 0.36 to 0.71 over the first 72 hours following dosing.
|
* Relative to concurrent concentrations in plasma in subjects with normal renal function. |
|
|
† Independent of degree of meningeal inflammation. |
|
|
Tissue or Fluid |
Ratio of Fluconazole
Tissue (Fluid)/Plasma Concentration* |
| Cerebrospinal fluid† Saliva Sputum Blister fluid Urine Normal skin Nails Blister skin Vaginal tissue Vaginal fluid |
0.5 to 0.9 1 1 1 10 10 1 2 1 0.4 to 0.7 |
Mean body clearance in adults is reported to be 0.23 (17%) mL/min/kg. In normal volunteers, fluconazole is cleared primarily by renal excretion, with approximately 80% of the administered dose appearing in the urine as unchanged drug. About 11% of the dose is excreted in the urine as metabolites.
The pharmacokinetics of fluconazole are markedly affected by reduction in renal function. There is an inverse relationship between the elimination half-life and creatinine clearance. The dose of fluconazole may need to be reduced in patients with impaired renal function. (See DOSAGE AND ADMINISTRATION .) A 3-hour hemodialysis session decreases plasma concentrations by approximately 50%.
In normal volunteers, fluconazole administration (doses ranging from 200 mg to 400 mg once daily for up to 14 days) was associated with small and inconsistent effects on testosterone concentrations, endogenous corticosteroid concentrations, and the adrenocorticotropic hormone (ACTH)-stimulated cortisol response.
Dosing in Pediatric Patients On Ecmo (Dosing in Pediatric Patients on ECMO)
The recommended dosage of fluconazole in pediatric patients 3 months and older on ECMO is 35-mg/kg on the first day (not to exceed 800 mg) followed by 12-mg/kg once daily (not to exceed 400 mg).
For patients from birth to 3 months postnatal age, and gestational age less than 30 weeks, a loading dose of 35- mg/kg on the first day followed by 9-mg/kg once daily is recommended.
For patients from birth to 3 months postnatal age and gestational age 30 weeks and above, a loading dose of 35-mg/kg on the first day followed by 12-mg/kg once daily is recommended.
Cryptococcal meningitis: For the treatment of acute cryptococcal meningitis, the recommended dosage is 12 mg/kg on the first day, followed by 6 mg/kg once daily. A dosage of 12 mg/kg once daily may be used, based on medical judgment of the patient's response to therapy. The recommended duration of treatment for initial therapy of cryptococcal meningitis is 10 to 12 weeks after the cerebrospinal fluid becomes culture negative. For suppression of relapse of cryptococcal meningitis in pediatric patients with AIDS, the recommended dose of fluconazole is 6 mg/kg once daily.
In Patients Receiving Multiple Doses
Sixteen percent of over 4000 patients treated with fluconazole in clinical trials of 7 days or more experienced adverse events. Treatment was discontinued in 1.5% of patients due to adverse clinical events and in 1.3% of patients due to laboratory test abnormalities.
Clinical adverse events were reported more frequently in HIV infected patients (21%) than in non-HIV infected patients (13%); however, the patterns in HIV infected and non-HIV infected patients were similar. The proportions of patients discontinuing therapy due to clinical adverse events were similar in the two groups (1.5%).
The following treatment-related clinical adverse events occurred at an incidence of 1% or greater in 4048 patients receiving fluconazole for 7 or more days in clinical trials: nausea 3.7%, headache 1.9%, skin rash 1.8%, vomiting 1.7%, abdominal pain 1.7%, and diarrhea 1.5%.
Pharmacokinetics in Pediatric Patients
In pediatrics patients from 2 days to 15 years of age, the following pharmacokinetic data have been reported following the administration of fluconazole:
|
*Data for Clearance, Cmax, AUC and Vdss are presented as arithmetic mean (CV%) and Half-life as arithmetic mean only. *AUC0-inf; ^AUC0-24; Abbreviations :Cmax=Peak plasma concentrations, AUC =area under the plasma concentration time curve; and Vdss=volume of distribution at steady state. |
||||||
|
There are limited data available in patients 61 days to less than 9 months of age. |
||||||
|
Age Studied |
Dose (mg/kg) |
Clearance (mL/min/kg) |
Half-life (Hours) |
C
max
(mcg/mL) |
AUC
(mcg*h/mL) |
Vdss (L/kg) |
|
2 to 60 days |
Intravenous 25 mg/kg on day one followed by Intravenous 12 mg/kg once daily |
0.29 (35%) N=8 |
54.2 |
23.4(29%) N=8 |
439 (25%) |
1.13 (31%) |
| 9 months to 13 years | Single-Oral 2 mg/kg | 0.40 (38%) N=14 | 25.0 | 2.9 (22%) N=16 | 94.7 (34%)* N=14 | N/A |
| 9 months to 13 years | Single-Oral 8 mg/kg | 0.51 (60%) N=15 | 19.5 | 9.8 (20%) N=15 | 362.5 (58%)*=14 | N/A |
| 5 to 15 years |
Multiple Intravenous 2 mg/kg |
0.49 (40%) N=4 |
17.4 | 5.5 (25%) N=5 |
67.4 (26%)^ N=4 |
0.722 (36%) N=4 |
| 5 to 15 years |
Multiple Intravenous 4 mg/kg |
0.59 (64%) N=5 | 15.2 | 11.4 (44%) N=6 | 139.1 (46%)^ N=5 |
0.729 (33%) N=5 |
| 5 to 15 years |
Multiple Intravenous 8 mg/kg |
0.66 (31%) N=7 | 17.6 | 14.1 (22%) N=8 | 196.7(25%)^ N=7 |
1.069 (37%) N=7 |
In pediatric patients (premature newborns; gestational age 26 to 29 weeks and postnatal age from birth to 1 (day), the mean (%cv) clearance within 36 hours of birth was 0.180 (35%, N=7) mL/min/kg, which increased with time to a mean of 0.218 (31%, N=9) mL/min/kg 6 days later and 0.333 (56%, N=4) mL/min/kg 12 days later. Similarly, the half-life was 73.6 hours, which decreased with time to a mean of 53.2 hours 6 days later and 46.6 hours 12 days later.
In a study of 13 pediatric patients (preterm and term infants with median gestational age (GA) of 37 weeks, GA range 24 to 39 weeks; median postnatal age [PNA] 19 days, PNA range 5 to 262 days) 12 infants received a 25 mg/kg loading dose, and 9/12 (75%) achieved an AUC0-24 of >400-mg*h/L in the first 24 hours. A population pharmacokinetic model using data from 55 pediatric patients (GA 23 to 40 weeks, PNA 1 to 88 days) found that a loading dose of 25 mg/kg is necessary to reach target AUC >400-mg*h/L within 24 hours of initiating therapy in pediatric patients younger than 3 months of age. A maintenance dose of 9 mg/kg daily should be used in pediatric patients born at GA less than 30 weeks and 12 mg/kg daily in pediatric patients with GA equal or greater than 30 weeks. (See DOSAGE AND ADMINISTRATION .)
A population PK model using data from 21 pediatric patients ages from birth to 17 years supported with extracorporeal membrane oxygenation (ECMO), and 19 pediatric non-ECMO patients ages from birth to 2 years found that clearance was related to serum creatinine while a higher volume of distribution was related to presence of ECMO support. The median volume of distribution was 1.3 L/kg in pediatric patients on ECMO and 0.9 L/kg in those not on ECMO. Simulations suggested that a loading dose of 35 mg/kg is needed to achieve the target AUC0-24 >400 mg*h/L within the first 24 hours in pediatric patients on ECMO. (See DOSAGE AND ADMINISTRATION.)
Adverse Reactions in Pediatric Patients
The pattern and incidence of adverse events and laboratory abnormalities recorded during pediatric clinical trials are comparable to those seen in adults.
In Phase II/III clinical trials conducted in the United States and in Europe, 577 pediatric patients, ages 1 day to 17 years were treated with fluconazole at doses up to 15 mg/kg/day for up to 1,616 days. Thirteen percent of pediatric patients experienced treatment-related adverse events. The most commonly reported events were vomiting (5%), abdominal pain (3%), nausea (2%), and diarrhea (2%). Treatment was discontinued in 2.3% of patients due to adverse clinical events and in 1.4% of patients due to laboratory test abnormalities. The majority of treatment-related laboratory abnormalities were elevations of transaminases or alkaline phosphatase.
| Percentage of Patients With Treatment-Related Side Effects | ||
|
Fluconazole
(N=577) |
Comparative Agents
(N=451) |
|
| With any side effect | 13.0 | 9.3 |
| Vomiting | 5.4 | 5.1 |
| Abdominal pain | 2.8 | 1.6 |
| Nausea | 2.3 | 1.6 |
| Diarrhea | 2.1 | 2.2 |
Clinical Trials Experience in Pediatric Patients
Safety in Prophylaxis of Invasive Candida Infections in Premature infants weighing less than 750 grams at birth.
In a Phase 3 clinical trial of pediatric patients (premature infants weighing less than 750 grams at birth), the incidence of intestinal perforation in infants receiving fluconazole prophylaxis was higher compared to infants receiving placebo (see PRECAUTIONS: Pediatric Use).
Safety in Pediatric Patients Receiving ECMO
A cohort of 20 pediatric patients (1 day to 17 years of age) on ECMO received fluconazole in a prospective, open-label, single-center safety and PK ECMO study. The adverse reaction profile of fluconazole in these patients was similar to that of adult and pediatric non-ECMO patients (See PRECAUTIONS: Pediatric Use).
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Dosage in Patients With Impaired Renal Function (Dosage In Patients With Impaired Renal Function)
Fluconazole is cleared primarily by renal excretion as unchanged drug. In patients with impaired renal function who will receive multiple doses of fluconazole, an initial loading dose of 50 mg to 400 mg should be given. After the loading dose, the daily dose (according to indication) should be based on the following summary:
| Creatinine Clearance (mL/min) | Recommended Dose (%) |
| >50 | 100 |
| ≤50 (no dialysis) | 50 |
| Hemodialysis | 100% after each hemodialysis |
Patients on hemodialysis should receive 100% of the recommended dose after each hemodialysis; on non-dialysis days, patients should receive a reduced dose according to their creatinine clearance.
These are suggested dose adjustments based on pharmacokinetics following administration of multiple doses. Further adjustment may be needed depending upon clinical condition.
When serum creatinine is the only measure of renal function available, the following formula (based on sex, weight, and age of the patient) should be used to estimate the creatinine clearance in adults:
| Males: | Weight (kg) × (140 - age) |
| 72 × serum creatinine (mg/100 mL) |
Females: 0.85 × above value
Although the pharmacokinetics of fluconazole has not been studied in pediatric patients with renal insufficiency, dosage reduction in pediatric patientswith renal insufficiency should parallel that recommended for adults. The following formula may be used to estimate creatinine clearance in pediatric patients:
| K × | linear length or height (cm) |
| serum creatinine (mg/100 mL) |
(Where K=0.55 for pediatric patients older than 1 year and 0.45 for infants.)
Carcinogenesis, Mutagenesis, and Impairment of Fertility
Fluconazole showed no evidence of carcinogenic potential in mice and rats treated orally for 24 months at doses of 2.5 mg/kg/day, 5 mg/kg/day, or 10 mg/kg/day (approximately 2 to 7 times the recommended human dose). Male rats treated with 5 mg/kg/day and 10 mg/kg/day had an increased incidence of hepatocellular adenomas.
Fluconazole, with or without metabolic activation, was negative in tests for mutagenicity in four strains of S. typhimurium, and in the mouse lymphoma L5178Y system. Cytogenetic studies in vivo (murine bone marrow cells, following oral administration of fluconazole) and in vitro (human lymphocytes exposed to fluconazole at 1000 mcg/mL) showed no evidence of chromosomal mutations.
Fluconazole did not affect the fertility of male or female rats treated orally with daily doses of 5 mg/kg, 10 mg/kg, or 20 mg/kg or with parenteral doses of 5 mg/kg, 25 mg/kg, or 75 mg/kg, although the onset of parturition was slightly delayed at 20 mg/kg PO. In an intravenous perinatal study in rats at 5 mg/kg, 20 mg/kg, and 40 mg/kg, dystocia and prolongation of parturition were observed in a few dams at 20 mg/kg (approximately 5 to 15 times the recommended human dose) and 40 mg/kg, but not at 5 mg/kg. The disturbances in parturition were reflected by a slight increase in the number of still born pups and decrease of neonatal survival at these dose levels. The effects on parturition in rats are consistent with the species specific estrogen-lowering property produced by high doses of fluconazole. Such a hormone change has not been observed in women treated with fluconazole. (See CLINICAL PHARMACOLOGY .)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:34.774341 · Updated: 2026-03-14T22:16:20.175496