These Highlights Do Not Include All The Information Needed To Use Stiolto Respimat Safely And Effectively. See Full Prescribing Information For Stiolto Respimat.
01e15aee-40e0-23f3-537f-c96dd63e2cb1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
STIOLTO RESPIMAT is a combination of tiotropium bromide, an anticholinergic and olodaterol, a long-acting beta 2 -adrenergic agonist (LABA) indicated for the long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). ( 1.1 ) Important limitations: STIOLTO RESPIMAT is NOT indicated to treat acute deterioration of COPD. ( 1.1 ) STIOLTO RESPIMAT is NOT indicated to treat asthma. ( 1.1 )
Indications and Usage
STIOLTO RESPIMAT is a combination of tiotropium bromide, an anticholinergic and olodaterol, a long-acting beta 2 -adrenergic agonist (LABA) indicated for the long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). ( 1.1 ) Important limitations: STIOLTO RESPIMAT is NOT indicated to treat acute deterioration of COPD. ( 1.1 ) STIOLTO RESPIMAT is NOT indicated to treat asthma. ( 1.1 )
Dosage and Administration
For oral inhalation only. Two inhalations of STIOLTO RESPIMAT once-daily at the same time of day. ( 2 )
Warnings and Precautions
LABA as monotherapy (without an inhaled corticosteroid) for asthma increases the risk of serious asthma-related events. ( 5.1 ) Do not initiate STIOLTO RESPIMAT in acutely deteriorating COPD patients. ( 5.2 ) Do not use for relief of acute symptoms. Concomitant short-acting beta 2 -agonists can be used as needed for acute relief. ( 5.2 ) Do not exceed the recommended dosage. Excessive use of STIOLTO RESPIMAT, or use in conjunction with other medications containing LABA can result in clinically significant cardiovascular effects and may be fatal. ( 5.3 ) Immediate hypersensitivity reactions: Discontinue STIOLTO RESPIMAT at once and consider alternatives if immediate hypersensitivity reactions, including angioedema, urticaria, rash, bronchospasm, or anaphylaxis, occur. ( 5.4 ) Life-threatening paradoxical bronchospasm can occur. Discontinue STIOLTO RESPIMAT immediately. ( 5.5 ) Use with caution in patients with cardiovascular or convulsive disorders, thyrotoxicosis, or sensitivity to sympathomimetic drugs. ( 5.6 , 5.7 ) Worsening of narrow-angle glaucoma may occur. Use with caution in patients with narrow-angle glaucoma and instruct patients to consult a physician immediately if this occurs. ( 5.8 ) Worsening of urinary retention may occur. Use with caution in patients with prostatic hyperplasia or bladder-neck obstruction and instruct patients to consult a physician immediately if this occurs. ( 5.9 ) Be alert to hypokalemia and hyperglycemia. ( 5.11 )
Contraindications
Use of a LABA, including STIOLTO RESPIMAT, without an inhaled corticosteroid is contraindicated in patients with asthma [see Warnings and Precautions (5.1) ] . STIOLTO RESPIMAT is not indicated for the treatment of asthma. STIOLTO RESPIMAT is contraindicated in patients with a hypersensitivity to tiotropium, ipratropium, olodaterol, or any component of this product [see Warnings and Precautions (5.4) ] . In clinical trials and postmarketing experience with tiotropium, immediate hypersensitivity reactions, including angioedema (including swelling of the lips, tongue, or throat), itching, or rash have been reported. Hypersensitivity reactions were also reported in clinical trials with STIOLTO RESPIMAT.
Adverse Reactions
LABA, such as olodaterol, one of the active components in STIOLTO RESPIMAT, as monotherapy (without an inhaled corticosteroid) for asthma, increase the risk of asthma-related events. STIOLTO RESPIMAT is not indicated for the treatment of asthma [see Warning and Precautions (5.1) ]. The following adverse reactions are described, or described in greater detail, in other sections: Immediate hypersensitivity reactions [see Warnings and Precautions (5.4) ] Paradoxical bronchospasm [see Warnings and Precautions (5.5) ] Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.8) ] Worsening of urinary retention [see Warnings and Precautions (5.9) ]
Drug Interactions
Other adrenergic drugs may potentiate effect. Use with caution. ( 5.3 , 7.1 ) Xanthine derivatives, steroids, diuretics, or non-potassium sparing diuretics may potentiate hypokalemia or ECG changes. Use with caution. ( 7.2 , 7.3 ) MAO inhibitors, tricyclic antidepressants, and drugs that prolong QTc interval may potentiate effect on cardiovascular system. Use with extreme caution. ( 7.4 ) Beta-blockers may decrease effectiveness. Use with caution and only when medically necessary. ( 7.5 ) Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of STIOLTO RESPIMAT with other anticholinergic-containing drugs. ( 7.6 )
Storage and Handling
STIOLTO RESPIMAT Inhalation Spray is supplied in a labeled carton containing one STIOLTO RESPIMAT cartridge and one STIOLTO RESPIMAT inhaler. The STIOLTO RESPIMAT cartridge is provided as an aluminum cylinder with a tamper protection seal on the cap. The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler and should not be interchanged with any other RESPIMAT device delivered product. The STIOLTO RESPIMAT inhaler is a cylindrical shaped plastic inhalation device with a gray colored body and a clear base. The clear base is removed to insert the cartridge. The inhaler contains a dose indicator. The light green-colored cap and the written information on the label of the gray inhaler body indicate that it is labeled for use with the STIOLTO RESPIMAT cartridge. STIOLTO RESPIMAT Inhalation Spray is available as: STIOLTO RESPIMAT Inhalation Spray: 60 metered actuations (NDC 0597-0155-61) STIOLTO RESPIMAT Inhalation Spray: 10 metered actuations (NDC 0597-0155-70) (institutional pack) The STIOLTO RESPIMAT cartridge has a net fill weight of at least 4 grams and when used with the STIOLTO RESPIMAT inhaler, is designed to deliver the labeled number of metered actuations after preparation for use. When the labeled number of actuations has been dispensed from the inhaler, the RESPIMAT locking mechanism will be engaged and no more actuations can be dispensed. After assembly, the STIOLTO RESPIMAT inhaler should be discarded at the latest 3 months after first use or when the locking mechanism is engaged, whichever comes first. Keep out of reach of children. Do not spray into eyes.
How Supplied
STIOLTO RESPIMAT Inhalation Spray is supplied in a labeled carton containing one STIOLTO RESPIMAT cartridge and one STIOLTO RESPIMAT inhaler. The STIOLTO RESPIMAT cartridge is provided as an aluminum cylinder with a tamper protection seal on the cap. The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler and should not be interchanged with any other RESPIMAT device delivered product. The STIOLTO RESPIMAT inhaler is a cylindrical shaped plastic inhalation device with a gray colored body and a clear base. The clear base is removed to insert the cartridge. The inhaler contains a dose indicator. The light green-colored cap and the written information on the label of the gray inhaler body indicate that it is labeled for use with the STIOLTO RESPIMAT cartridge. STIOLTO RESPIMAT Inhalation Spray is available as: STIOLTO RESPIMAT Inhalation Spray: 60 metered actuations (NDC 0597-0155-61) STIOLTO RESPIMAT Inhalation Spray: 10 metered actuations (NDC 0597-0155-70) (institutional pack) The STIOLTO RESPIMAT cartridge has a net fill weight of at least 4 grams and when used with the STIOLTO RESPIMAT inhaler, is designed to deliver the labeled number of metered actuations after preparation for use. When the labeled number of actuations has been dispensed from the inhaler, the RESPIMAT locking mechanism will be engaged and no more actuations can be dispensed. After assembly, the STIOLTO RESPIMAT inhaler should be discarded at the latest 3 months after first use or when the locking mechanism is engaged, whichever comes first. Keep out of reach of children. Do not spray into eyes.
Medication Information
Warnings and Precautions
LABA as monotherapy (without an inhaled corticosteroid) for asthma increases the risk of serious asthma-related events. ( 5.1 ) Do not initiate STIOLTO RESPIMAT in acutely deteriorating COPD patients. ( 5.2 ) Do not use for relief of acute symptoms. Concomitant short-acting beta 2 -agonists can be used as needed for acute relief. ( 5.2 ) Do not exceed the recommended dosage. Excessive use of STIOLTO RESPIMAT, or use in conjunction with other medications containing LABA can result in clinically significant cardiovascular effects and may be fatal. ( 5.3 ) Immediate hypersensitivity reactions: Discontinue STIOLTO RESPIMAT at once and consider alternatives if immediate hypersensitivity reactions, including angioedema, urticaria, rash, bronchospasm, or anaphylaxis, occur. ( 5.4 ) Life-threatening paradoxical bronchospasm can occur. Discontinue STIOLTO RESPIMAT immediately. ( 5.5 ) Use with caution in patients with cardiovascular or convulsive disorders, thyrotoxicosis, or sensitivity to sympathomimetic drugs. ( 5.6 , 5.7 ) Worsening of narrow-angle glaucoma may occur. Use with caution in patients with narrow-angle glaucoma and instruct patients to consult a physician immediately if this occurs. ( 5.8 ) Worsening of urinary retention may occur. Use with caution in patients with prostatic hyperplasia or bladder-neck obstruction and instruct patients to consult a physician immediately if this occurs. ( 5.9 ) Be alert to hypokalemia and hyperglycemia. ( 5.11 )
Indications and Usage
STIOLTO RESPIMAT is a combination of tiotropium bromide, an anticholinergic and olodaterol, a long-acting beta 2 -adrenergic agonist (LABA) indicated for the long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). ( 1.1 ) Important limitations: STIOLTO RESPIMAT is NOT indicated to treat acute deterioration of COPD. ( 1.1 ) STIOLTO RESPIMAT is NOT indicated to treat asthma. ( 1.1 )
Dosage and Administration
For oral inhalation only. Two inhalations of STIOLTO RESPIMAT once-daily at the same time of day. ( 2 )
Contraindications
Use of a LABA, including STIOLTO RESPIMAT, without an inhaled corticosteroid is contraindicated in patients with asthma [see Warnings and Precautions (5.1) ] . STIOLTO RESPIMAT is not indicated for the treatment of asthma. STIOLTO RESPIMAT is contraindicated in patients with a hypersensitivity to tiotropium, ipratropium, olodaterol, or any component of this product [see Warnings and Precautions (5.4) ] . In clinical trials and postmarketing experience with tiotropium, immediate hypersensitivity reactions, including angioedema (including swelling of the lips, tongue, or throat), itching, or rash have been reported. Hypersensitivity reactions were also reported in clinical trials with STIOLTO RESPIMAT.
Adverse Reactions
LABA, such as olodaterol, one of the active components in STIOLTO RESPIMAT, as monotherapy (without an inhaled corticosteroid) for asthma, increase the risk of asthma-related events. STIOLTO RESPIMAT is not indicated for the treatment of asthma [see Warning and Precautions (5.1) ]. The following adverse reactions are described, or described in greater detail, in other sections: Immediate hypersensitivity reactions [see Warnings and Precautions (5.4) ] Paradoxical bronchospasm [see Warnings and Precautions (5.5) ] Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.8) ] Worsening of urinary retention [see Warnings and Precautions (5.9) ]
Drug Interactions
Other adrenergic drugs may potentiate effect. Use with caution. ( 5.3 , 7.1 ) Xanthine derivatives, steroids, diuretics, or non-potassium sparing diuretics may potentiate hypokalemia or ECG changes. Use with caution. ( 7.2 , 7.3 ) MAO inhibitors, tricyclic antidepressants, and drugs that prolong QTc interval may potentiate effect on cardiovascular system. Use with extreme caution. ( 7.4 ) Beta-blockers may decrease effectiveness. Use with caution and only when medically necessary. ( 7.5 ) Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of STIOLTO RESPIMAT with other anticholinergic-containing drugs. ( 7.6 )
Storage and Handling
STIOLTO RESPIMAT Inhalation Spray is supplied in a labeled carton containing one STIOLTO RESPIMAT cartridge and one STIOLTO RESPIMAT inhaler. The STIOLTO RESPIMAT cartridge is provided as an aluminum cylinder with a tamper protection seal on the cap. The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler and should not be interchanged with any other RESPIMAT device delivered product. The STIOLTO RESPIMAT inhaler is a cylindrical shaped plastic inhalation device with a gray colored body and a clear base. The clear base is removed to insert the cartridge. The inhaler contains a dose indicator. The light green-colored cap and the written information on the label of the gray inhaler body indicate that it is labeled for use with the STIOLTO RESPIMAT cartridge. STIOLTO RESPIMAT Inhalation Spray is available as: STIOLTO RESPIMAT Inhalation Spray: 60 metered actuations (NDC 0597-0155-61) STIOLTO RESPIMAT Inhalation Spray: 10 metered actuations (NDC 0597-0155-70) (institutional pack) The STIOLTO RESPIMAT cartridge has a net fill weight of at least 4 grams and when used with the STIOLTO RESPIMAT inhaler, is designed to deliver the labeled number of metered actuations after preparation for use. When the labeled number of actuations has been dispensed from the inhaler, the RESPIMAT locking mechanism will be engaged and no more actuations can be dispensed. After assembly, the STIOLTO RESPIMAT inhaler should be discarded at the latest 3 months after first use or when the locking mechanism is engaged, whichever comes first. Keep out of reach of children. Do not spray into eyes.
How Supplied
STIOLTO RESPIMAT Inhalation Spray is supplied in a labeled carton containing one STIOLTO RESPIMAT cartridge and one STIOLTO RESPIMAT inhaler. The STIOLTO RESPIMAT cartridge is provided as an aluminum cylinder with a tamper protection seal on the cap. The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler and should not be interchanged with any other RESPIMAT device delivered product. The STIOLTO RESPIMAT inhaler is a cylindrical shaped plastic inhalation device with a gray colored body and a clear base. The clear base is removed to insert the cartridge. The inhaler contains a dose indicator. The light green-colored cap and the written information on the label of the gray inhaler body indicate that it is labeled for use with the STIOLTO RESPIMAT cartridge. STIOLTO RESPIMAT Inhalation Spray is available as: STIOLTO RESPIMAT Inhalation Spray: 60 metered actuations (NDC 0597-0155-61) STIOLTO RESPIMAT Inhalation Spray: 10 metered actuations (NDC 0597-0155-70) (institutional pack) The STIOLTO RESPIMAT cartridge has a net fill weight of at least 4 grams and when used with the STIOLTO RESPIMAT inhaler, is designed to deliver the labeled number of metered actuations after preparation for use. When the labeled number of actuations has been dispensed from the inhaler, the RESPIMAT locking mechanism will be engaged and no more actuations can be dispensed. After assembly, the STIOLTO RESPIMAT inhaler should be discarded at the latest 3 months after first use or when the locking mechanism is engaged, whichever comes first. Keep out of reach of children. Do not spray into eyes.
Description
STIOLTO RESPIMAT is a combination of tiotropium bromide, an anticholinergic and olodaterol, a long-acting beta 2 -adrenergic agonist (LABA) indicated for the long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). ( 1.1 ) Important limitations: STIOLTO RESPIMAT is NOT indicated to treat acute deterioration of COPD. ( 1.1 ) STIOLTO RESPIMAT is NOT indicated to treat asthma. ( 1.1 )
Section 42229-5
Important Limitations of Use
- STIOLTO RESPIMAT is not indicated to treat acute deteriorations of COPD [see Warnings and Precautions (5.2)].
- STIOLTO RESPIMAT is not indicated to treat asthma. The safety and effectiveness of STIOLTO RESPIMAT in asthma have not been established.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 1/2025 | |
|
PATIENT INFORMATION STIOLTO® RESPIMAT® (sti-OL-to– RES peh mat) (tiotropium bromide and olodaterol inhalation spray), for oral inhalation use |
||
What is STIOLTO RESPIMAT?
|
||
Do not use STIOLTO RESPIMAT if you:
|
||
Before you use STIOLTO RESPIMAT, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
|
||
|
How should I use STIOLTO RESPIMAT?
Read the step-by-step instructions for using STIOLTO RESPIMAT at the end of this Patient Information leaflet.
|
||
|
What are the possible side effects with STIOLTO RESPIMAT?
STIOLTO RESPIMAT can cause serious side effects, including:
|
||
|
|
|
These are not all the side effects of STIOLTO RESPIMAT. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store STIOLTO RESPIMAT?
|
||
|
General information about the safe and effective use of STIOLTO RESPIMAT
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use STIOLTO RESPIMAT for a condition for which it was not prescribed. Do not give STIOLTO RESPIMAT to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about STIOLTO RESPIMAT that is written for health professionals. |
||
|
Active ingredients: tiotropium bromide and olodaterol Inactive ingredients: benzalkonium chloride, edetate disodium, hydrochloric acid, and water for injection Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc. Ridgefield, CT 06877 USA Licensed from: Boehringer Ingelheim International GmbH SPIRIVA®, HANDIHALER®, STIOLTO ® and RESPIMAT® are registered trademarks of and are used under license from Boehringer Ingelheim International GmbH Copyright © 2025 Boehringer Ingelheim International GmbH ALL RIGHTS RESERVED COL10537BA092025 For more information about STIOLTO RESPIMAT, including current prescribing information, a video demonstration or a Quick Start Guide on how to use STIOLTO RESPIMAT, go to www.STIOLTO.com, scan the code, or call 1-800-542-6257.
|
Section 44425-7
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Avoid freezing.
Section 59845-8
Instructions for Use
STIOLTO® RESPIMAT® (sti-OL-to- RES peh mat)
(tiotropium bromide and olodaterol inhalation spray), for oral inhalation use
For Oral Inhalation Only
Do not spray STIOLTO RESPIMAT into your eyes.
Read these Instructions for Use before you start using STIOLTO RESPIMAT and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your doctor about your medical condition or your treatment.
You will need to use this inhaler 1 time each day, at the same time each day. Each time you use it take 2 puffs.
Do not turn the clear base before inserting the cartridge.
How to store your STIOLTO RESPIMAT inhaler
- Store STIOLTO RESPIMAT at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not freeze your STIOLTO RESPIMAT cartridge and inhaler.
- If STIOLTO RESPIMAT has not been used for more than 3 days, release 1 puff towards the ground.
- If STIOLTO RESPIMAT has not been used for more than 21 days, repeat steps 4 to 6 under the "Prepare for first use" until a mist is visible. Then repeat steps 4 to 6 three more times.
- Keep your STIOLTO RESPIMAT cartridge, inhaler, and all medicines out of the reach of children.
How to care for your STIOLTO RESPIMAT inhaler
Clean the mouthpiece, including the metal part inside the mouthpiece, with a damp cloth or tissue only, at least 1 time each week. Any minor discoloration in the mouthpiece does not affect your STIOLTO RESPIMAT inhaler.
When to get a new STIOLTO RESPIMAT inhaler
- The scale on your inhaler will show the number of puffs you have, if used as indicated (2 puffs 1 time each day).
- The dose indicator will show you approximately how much medicine is left.
- When the dose indicator enters the red area of the scale, it will show you approximately how many puffs are left before you need a refill or new prescription.
- When the dose indicator reaches the end of the red scale, your STIOLTO RESPIMAT is empty and automatically locks. At this point, the clear base cannot be turned any further.
- Three months after insertion of cartridge, throw away the STIOLTO RESPIMAT even if it has not been used, or when the inhaler is locked, or when it expires, whichever comes first.
Prepare for first use
1. Remove clear base
|
|
2. Insert cartridge
|
|
3. Replace clear base
|
|
4. Turn
|
|
5. Open
|
|
6. Press
|
Daily use (T O P)
T
urn
|
|
O
pen
|
|
P
ress
|
Answers to Common Questions
It is difficult to insert the cartridge deep enough:
Did you accidentally turn the clear base before inserting the cartridge? Open the cap, press the dose-release button, then insert the cartridge.
Did you insert the cartridge with the wide end first? Insert the cartridge with the narrow end first.
I cannot press the dose-release button:
Did you turn the clear base? If not, turn the clear base in a continuous movement until it clicks (half a turn).
Is the dose indicator on the STIOLTO RESPIMAT pointing to 0 (zero)? The STIOLTO RESPIMAT inhaler is locked after the labeled number of puffs have been used. Prepare and use your new STIOLTO RESPIMAT inhaler.
I cannot turn the clear base:
Did you turn the clear base already? If the clear base has already been turned, follow steps "Open" and "Press" under "Daily use" to get your medicine.
Is the dose indicator on the STIOLTO RESPIMAT pointing to 0 (zero)? The STIOLTO RESPIMAT inhaler is locked after the labeled number of puffs have been used. Prepare and use your new STIOLTO RESPIMAT inhaler.
The dose indicator on the STIOLTO RESPIMAT reaches 0 (zero) too soon:
Did you use STIOLTO RESPIMAT as indicated (2 puffs 1 time each day)? STIOLTO RESPIMAT will deliver the labeled number of puffs if used at 2 puffs 1 time each day.
Did you turn the clear base before you inserted the cartridge? The dose indicator counts each turn of the clear base regardless whether a cartridge has been inserted or not.
Did you spray in the air often to check whether the STIOLTO RESPIMAT is working? After you have prepared STIOLTO RESPIMAT, no test-spraying is required if used daily.
Did you insert the cartridge into a used STIOLTO RESPIMAT? Always insert a new cartridge into a NEW STIOLTO RESPIMAT.
My STIOLTO RESPIMAT sprays automatically:
Was the cap open when you turned the clear base? Close the cap, then turn the clear base.
Did you press the dose-release button when turning the clear base? Close the cap, so the dose-release button is covered, then turn the clear base.
Did you stop when turning the clear base before it clicked? Turn the clear base in a continuous movement until it clicks (half a turn).
My STIOLTO RESPIMAT does not spray:
Did you insert a cartridge? If not, insert a cartridge.
Did you repeat Turn, Open, Press (TOP) less than 3 times after inserting the cartridge? Repeat Turn, Open, Press (TOP) 3 times after inserting the cartridge as shown in steps 4 to 6 under "Prepare for first use".
Is the dose indicator on the STIOLTO RESPIMAT pointing to 0 (zero)? You have used up all your medicine and the inhaler is locked.
For more information about STIOLTO RESPIMAT, including current prescribing information, a video demonstration or a Quick Start Guide on how to use STIOLTO RESPIMAT, go to www.STIOLTO.com, scan the code, or call 1-800-542-6257.
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT 06877 USA
Licensed from:
Boehringer Ingelheim International GmbH
SPIRIVA®, HANDIHALER®, STIOLTO® and RESPIMAT® are registered trademarks of and are used under license from Boehringer Ingelheim International GmbH
Copyright © 2025 Boehringer Ingelheim International GmbH
ALL RIGHTS RESERVED
Revised: January 2025
COL10537BA092025
10 Overdosage
STIOLTO RESPIMAT contains both tiotropium bromide and olodaterol; therefore, the risks associated with overdosage for the individual components described below apply to STIOLTO RESPIMAT.
11 Description
STIOLTO RESPIMAT is a combination of tiotropium, an anticholinergic, and olodaterol, a long-acting beta2-adrenergic agonist (LABA).
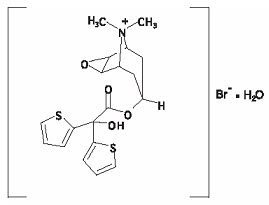
The drug substance tiotropium bromide monohydrate is chemically described as (1α, 2ß, 4ß, 5α, 7ß)-7-[(Hydroxydi-2-thienylacetyl)oxy]-9,9-dimethyl-3-oxa-9-azoniatricyclo[3.3.1.02,4] nonane bromide monohydrate. It is a synthetic, non-chiral, quaternary ammonium compound. Tiotropium bromide is a white or yellowish white powder. It is sparingly soluble in water and soluble in methanol.
The structural formula is:
Tiotropium bromide (monohydrate) has a molecular mass of 490.4 and a molecular formula of C19H22NO4S2Br ∙ H2O.
The drug substance olodaterol hydrochloride is chemically described as 2H-1,4-Benzoxazin-3H(4H)-one, 6-hydroxy-8-[(1R)-1-hydroxy-2-[[2-(4-methoxyphenyl)-1,1-dimethylethyl]-amino]ethyl]-, monohydrochloride. Olodaterol hydrochloride is a white to off-white powder that is sparingly-slightly soluble in water and slightly soluble in ethanol. The molecular weight is 422.9 g/mole (salt): 386.5 g/mole (base), and the molecular formula is C21H26N2O5 × HCl as a hydrochloride. The conversion factor from salt to free base is 1.094.
The structural formula is:
The drug product, STIOLTO RESPIMAT, is composed of a sterile aqueous solution of tiotropium bromide and olodaterol hydrochloride filled into a 4.5 mL plastic container crimped into an aluminum cylinder (STIOLTO RESPIMAT cartridge) for use with the STIOLTO RESPIMAT inhaler.
Excipients include, benzalkonium chloride, edetate disodium, hydrochloric acid, and water for injection.
The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler. The STIOLTO RESPIMAT inhaler is a hand held, pocket sized oral inhalation device that uses mechanical energy to generate a slow-moving aerosol cloud of medication from a metered volume of the drug solution. The STIOLTO RESPIMAT inhaler has a light green-colored cap.
When used with the STIOLTO RESPIMAT inhaler each cartridge, containing 4 grams of sterile aqueous solution, delivers the labeled number of metered actuations after preparation for use. Each dose (one dose equals two actuations) from the STIOLTO RESPIMAT inhaler delivers 5 mcg tiotropium (equivalent to 6.247 mcg tiotropium bromide monohydrate) and 5 mcg olodaterol (equivalent to 5.473 mcg olodaterol hydrochloride) in 22.1 mcL from the mouthpiece. As with all inhaled drugs, the actual amount of drug delivered to the lung may depend on patient factors, such as the coordination between the actuation of the inhaler and inspiration through the delivery system. The duration of inspiration should be at least as long as the spray duration (1.5 seconds).
7.5 Beta Blockers
Beta-adrenergic receptor antagonists (beta-blockers) and the olodaterol component of STIOLTO RESPIMAT may interfere with the effect of each other when administered concurrently. Beta-blockers not only block the therapeutic effects of beta-agonists, but may produce severe bronchospasm in COPD patients. Therefore, patients with COPD should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-blockers in patients with COPD. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
8.4 Pediatric Use
COPD does not normally occur in children. The safety and effectiveness of STIOLTO RESPIMAT in the pediatric population has not been established.
8.5 Geriatric Use
Based on available data, no adjustment of STIOLTO RESPIMAT dosage in geriatric patients is warranted [see Clinical Pharmacology (12.3)].
Of the 1,029 patients who received STIOLTO RESPIMAT at the recommended dose once daily in the clinical studies from the pooled 1-year database, 525 (51.0%) were <65 years of age, 407 (39.6%) were 65 to <75, 96 (9.3%) were 75 to <85, and 1 (0.1%) was ≥85.
No overall differences in effectiveness were observed, and in the 1-year pooled data, the adverse drug reaction profiles were similar in the older population compared to the patient population overall.
14 Clinical Studies
The safety and efficacy of STIOLTO RESPIMAT were evaluated in a clinical development program that included three dose ranging trials, two active-controlled trials, three active- and placebo-controlled trials, and one placebo-controlled trial. The efficacy of STIOLTO RESPIMAT is based primarily on two 4-week dose-ranging trials in 592 COPD patients and two confirmatory active-controlled 52-week trials (Trials 1 and 2) in 5,162 COPD patients.
4 Contraindications
Use of a LABA, including STIOLTO RESPIMAT, without an inhaled corticosteroid is contraindicated in patients with asthma [see Warnings and Precautions (5.1)]. STIOLTO RESPIMAT is not indicated for the treatment of asthma.
STIOLTO RESPIMAT is contraindicated in patients with a hypersensitivity to tiotropium, ipratropium, olodaterol, or any component of this product [see Warnings and Precautions (5.4)].
In clinical trials and postmarketing experience with tiotropium, immediate hypersensitivity reactions, including angioedema (including swelling of the lips, tongue, or throat), itching, or rash have been reported. Hypersensitivity reactions were also reported in clinical trials with STIOLTO RESPIMAT.
6 Adverse Reactions
LABA, such as olodaterol, one of the active components in STIOLTO RESPIMAT, as monotherapy (without an inhaled corticosteroid) for asthma, increase the risk of asthma-related events. STIOLTO RESPIMAT is not indicated for the treatment of asthma [see Warning and Precautions (5.1)].
The following adverse reactions are described, or described in greater detail, in other sections:
- Immediate hypersensitivity reactions [see Warnings and Precautions (5.4)]
- Paradoxical bronchospasm [see Warnings and Precautions (5.5)]
- Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.8)]
- Worsening of urinary retention [see Warnings and Precautions (5.9)]
7 Drug Interactions
- Other adrenergic drugs may potentiate effect. Use with caution. (5.3, 7.1)
- Xanthine derivatives, steroids, diuretics, or non-potassium sparing diuretics may potentiate hypokalemia or ECG changes. Use with caution. (7.2, 7.3)
- MAO inhibitors, tricyclic antidepressants, and drugs that prolong QTc interval may potentiate effect on cardiovascular system. Use with extreme caution. (7.4)
- Beta-blockers may decrease effectiveness. Use with caution and only when medically necessary. (7.5)
- Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of STIOLTO RESPIMAT with other anticholinergic-containing drugs. (7.6)
7.1 Adrenergic Drugs
If additional adrenergic drugs are to be administered by any route, they should be used with caution because the sympathetic effects of olodaterol, one component of STIOLTO RESPIMAT, may be potentiated [see Warnings and Precautions (5.3, 5.6, 5.10, 5.11)].
7.6 Anticholinergics
There is potential for an additive interaction with concomitantly used anticholinergic medications. Therefore, avoid co-administration of STIOLTO RESPIMAT with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic adverse effects [see Warnings and Precautions (5.8, 5.9) and Adverse Reactions (6)].
8.7 Renal Impairment
No dose adjustment is required for patients with renal impairment. However, patients with moderate to severe renal impairment (creatinine clearance of <60 mL/min) treated with STIOLTO RESPIMAT should be monitored closely for anticholinergic side effects [see Dosage and Administration (2), Warnings and Precautions (5.10), and Clinical Pharmacology (12.3)].
5.10 Renal Impairment
Because tiotropium is a predominantly renally excreted drug, patients with moderate to severe renal impairment (creatinine clearance of <60 mL/min) treated with STIOLTO RESPIMAT should be monitored closely for anticholinergic side effects [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
2.1 Recommended Dosage
The recommended dosage of STIOLTO RESPIMAT is two inhalations once-daily at the same time of the day. Do not use STIOLTO RESPIMAT more than two inhalations every 24 hours.
8.6 Hepatic Impairment
No dose adjustment is needed in patients with mild and moderate hepatic impairment. A study in subjects with severe hepatic impairment was not performed [see Clinical Pharmacology (12.3)].
1 Indications and Usage
STIOLTO RESPIMAT is a combination of tiotropium bromide, an anticholinergic and olodaterol, a long-acting beta2-adrenergic agonist (LABA) indicated for the long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). (1.1)
Important limitations:
5.7 Coexisting Conditions
Olodaterol, like other sympathomimetic amines, should be used with caution in patients with convulsive disorders or thyrotoxicosis, in patients with known or suspected prolongation of the QT interval, and in patients who are unusually responsive to sympathomimetic amines. Doses of the related beta2-agonist albuterol, when administered intravenously, have been reported to aggravate pre-existing diabetes mellitus and ketoacidosis.
5 Warnings and Precautions
- LABA as monotherapy (without an inhaled corticosteroid) for asthma increases the risk of serious asthma-related events. (5.1)
- Do not initiate STIOLTO RESPIMAT in acutely deteriorating COPD patients. (5.2)
- Do not use for relief of acute symptoms. Concomitant short-acting beta2-agonists can be used as needed for acute relief. (5.2)
- Do not exceed the recommended dosage. Excessive use of STIOLTO RESPIMAT, or use in conjunction with other medications containing LABA can result in clinically significant cardiovascular effects and may be fatal. (5.3)
- Immediate hypersensitivity reactions: Discontinue STIOLTO RESPIMAT at once and consider alternatives if immediate hypersensitivity reactions, including angioedema, urticaria, rash, bronchospasm, or anaphylaxis, occur. (5.4)
- Life-threatening paradoxical bronchospasm can occur. Discontinue STIOLTO RESPIMAT immediately. (5.5)
- Use with caution in patients with cardiovascular or convulsive disorders, thyrotoxicosis, or sensitivity to sympathomimetic drugs. (5.6, 5.7)
- Worsening of narrow-angle glaucoma may occur. Use with caution in patients with narrow-angle glaucoma and instruct patients to consult a physician immediately if this occurs. (5.8)
- Worsening of urinary retention may occur. Use with caution in patients with prostatic hyperplasia or bladder-neck obstruction and instruct patients to consult a physician immediately if this occurs. (5.9)
- Be alert to hypokalemia and hyperglycemia. (5.11)
5.6 Cardiovascular Effects
Olodaterol, like other beta2-agonists, can produce a clinically significant cardiovascular effect in some patients as measured by increases in pulse rate, systolic or diastolic blood pressure, and/or symptoms. If such effects occur, STIOLTO RESPIMAT may need to be discontinued. In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the T wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Long acting beta2-adrenergic agonists should be administered with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, hypertrophic obstructive cardiomyopathy, and hypertension.
2 Dosage and Administration
- For oral inhalation only.
- Two inhalations of STIOLTO RESPIMAT once-daily at the same time of day. (2)
3 Dosage Forms and Strengths
Inhalation spray: Each actuation from the mouthpiece delivers 2.5 mcg tiotropium (equivalent to 3.124 mcg tiotropium bromide monohydrate), and 2.5 mcg olodaterol (equivalent to 2.736 mcg olodaterol hydrochloride). Two actuations equal one dose. (3)
5.5 Paradoxical Bronchospasm
As with other inhaled medicines, STIOLTO RESPIMAT may cause paradoxical bronchospasm that may be life-threatening. If paradoxical bronchospasm occurs, STIOLTO RESPIMAT should be stopped immediately and alternative therapy instituted.
8 Use in Specific Populations
2.2 Administration Information
For oral inhalation only.
Prior to first use, the STIOLTO RESPIMAT cartridge is inserted into the STIOLTO RESPIMAT inhaler and the unit is primed. When using the unit for the first time, patients are to actuate the inhaler toward the ground until an aerosol cloud is visible and then repeat the process three more times. The unit is then considered primed and ready for use. If not used for more than 3 days, patients are to actuate the inhaler once to prepare the inhaler for use. If not used for more than 21 days, patients are to actuate the inhaler until an aerosol cloud is visible and then repeat the process three more times to prepare the inhaler for use [see Patient Counseling Information (17)].
No dosage adjustment is required for geriatric, hepatically-impaired, or renally-impaired patients. However, patients with moderate to severe renal impairment given STIOLTO RESPIMAT should be monitored closely for anticholinergic effects [see Warnings and Precautions (5.10), Use in Specific Populations (8.5, 8.6, 8.7), and Clinical Pharmacology (12.3)].
1.1 Maintenance Treatment of Copd
STIOLTO RESPIMAT is a combination of tiotropium bromide and olodaterol indicated for long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.11 Hypokalemia and Hyperglycemia
Beta-adrenergic agonists may produce significant hypokalemia in some patients, which has the potential to produce adverse cardiovascular effects [see Clinical Pharmacology (12.2)]. The decrease in serum potassium is usually transient, not requiring supplementation. Inhalation of high doses of beta2-adrenergic agonists may produce increases in plasma glucose.
In patients with severe COPD, hypokalemia may be potentiated by hypoxia and concomitant treatment [see Drug Interactions (7.2)], which may increase the susceptibility for cardiac arrhythmias.
Clinically notable decreases in serum potassium or changes in blood glucose were infrequent during clinical studies with long-term administration of olodaterol with the rates similar to those for placebo controls. Olodaterol has not been investigated in patients whose diabetes mellitus is not well controlled.
5.9 Worsening of Urinary Retention
STIOLTO RESPIMAT should be used with caution in patients with urinary retention. Prescribers and patients should be alert for signs and symptoms of prostatic hyperplasia or bladder-neck obstruction (e.g., difficulty passing urine, painful urination), especially in patients with prostatic hyperplasia or bladder neck obstruction. Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
7.3 Non Potassium Sparing Diuretics
The ECG changes and/or hypokalemia that may result from the administration of non-potassium sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dosage of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the co-administration of STIOLTO RESPIMAT with non-potassium sparing diuretics.
16 How Supplied/storage and Handling
STIOLTO RESPIMAT Inhalation Spray is supplied in a labeled carton containing one STIOLTO RESPIMAT cartridge and one STIOLTO RESPIMAT inhaler.
The STIOLTO RESPIMAT cartridge is provided as an aluminum cylinder with a tamper protection seal on the cap. The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler and should not be interchanged with any other RESPIMAT device delivered product.
The STIOLTO RESPIMAT inhaler is a cylindrical shaped plastic inhalation device with a gray colored body and a clear base. The clear base is removed to insert the cartridge. The inhaler contains a dose indicator. The light green-colored cap and the written information on the label of the gray inhaler body indicate that it is labeled for use with the STIOLTO RESPIMAT cartridge.
STIOLTO RESPIMAT Inhalation Spray is available as:
- STIOLTO RESPIMAT Inhalation Spray: 60 metered actuations (NDC 0597-0155-61)
- STIOLTO RESPIMAT Inhalation Spray: 10 metered actuations (NDC 0597-0155-70) (institutional pack)
The STIOLTO RESPIMAT cartridge has a net fill weight of at least 4 grams and when used with the STIOLTO RESPIMAT inhaler, is designed to deliver the labeled number of metered actuations after preparation for use.
When the labeled number of actuations has been dispensed from the inhaler, the RESPIMAT locking mechanism will be engaged and no more actuations can be dispensed.
After assembly, the STIOLTO RESPIMAT inhaler should be discarded at the latest 3 months after first use or when the locking mechanism is engaged, whichever comes first.
Keep out of reach of children. Do not spray into eyes.
5.8 Worsening of Narrow Angle Glaucoma
STIOLTO RESPIMAT should be used with caution in patients with narrow-angle glaucoma. Prescribers and patients should be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
5.4 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions, including urticaria, angioedema (including swelling of the lips, tongue or throat), rash, bronchospasm, anaphylaxis, or itching may occur after administration of STIOLTO RESPIMAT. If such a reaction occurs, therapy with STIOLTO RESPIMAT should be stopped at once and alternative treatments should be considered. Given the similar structural formula of atropine to tiotropium, patients with a history of hypersensitivity reactions to atropine or its derivatives should be closely monitored for similar hypersensitivity reactions to STIOLTO RESPIMAT.
5.2 Deterioration of Disease and Acute Episodes
STIOLTO RESPIMAT should not be initiated in patients with acutely deteriorating COPD, which may be a life-threatening condition. STIOLTO RESPIMAT has not been studied in patients with acutely deteriorating COPD. The use of STIOLTO RESPIMAT in this setting is inappropriate.
STIOLTO RESPIMAT should not be used for the relief of acute symptoms, i.e., as rescue therapy for the treatment of acute episodes of bronchospasm. STIOLTO RESPIMAT has not been studied in the relief of acute symptoms and extra doses should not be used for that purpose. Acute symptoms should be treated with an inhaled short-acting beta2-agonist.
When beginning STIOLTO RESPIMAT, patients who have been taking inhaled, short-acting beta2-agonists on a regular basis (e.g., four times a day) should be instructed to discontinue the regular use of these drugs and use them only for symptomatic relief of acute respiratory symptoms. When prescribing STIOLTO RESPIMAT, the healthcare provider should also prescribe an inhaled, short-acting beta2-agonist and instruct the patient on how it should be used. Increasing inhaled beta2-agonist use is a signal of deteriorating disease for which prompt medical attention is indicated.
COPD may deteriorate acutely over a period of hours or chronically over several days or longer. If STIOLTO RESPIMAT no longer controls symptoms of bronchoconstriction, or the patient's inhaled, short-acting beta2-agonist becomes less effective or the patient needs more inhalation of short-acting beta2-agonist than usual, these may be markers of deterioration of disease. In this setting, a re-evaluation of the patient and the COPD treatment regimen should be undertaken at once. Increasing the daily dosage of STIOLTO RESPIMAT beyond the recommended dosage is not appropriate in this situation.
Principal Display Panel 2.5 Mcg/2.5 Mcg Cartridge Carton
NDC 0597-0155-61
Stiolto® Respimat®
(tiotropium bromide and olodaterol
inhalation spray)
2.5 mcg/2.5 mcg per actuation*
FOR ORAL INHALATION ONLY
ATTENTION PHARMACIST:
Dispense with Instructions for Use
Rx only
4 Grams
60 Metered Inhalations
(Two inhalations equal one dose)
Boehringer
Ingelheim
7.7 Inhibitors of Cytochrome P450 and P Gp Efflux Transporter
In a drug interaction study using the strong dual CYP and P-gp inhibitor ketoconazole, a 1.7-fold increase of olodaterol maximum plasma concentrations and AUC was observed [see Pharmacokinetics (12.3)]. Olodaterol was evaluated in clinical trials for up to one year at doses up to twice the recommended therapeutic dosage. No dosage adjustment of STIOLTO RESPIMAT is necessary.
7.2 Sympathomimetics, Xanthine Derivatives, Steroids, Or Diuretics
Tiotropium has been used concomitantly with short-acting and long-acting sympathomimetic (beta-agonists) bronchodilators, methylxanthines, and oral and inhaled steroids, without increases in adverse reactions. Concomitant treatment with xanthine derivatives, steroids, or diuretics may potentiate any hypokalemic effect of olodaterol [see Warnings and Precautions (5.11)].
6.1 Clinical Trials Experience in Chronic Obstructive Pulmonary Disease
Because clinical trials are conducted under widely varying conditions, the incidence of adverse reactions observed in the clinical trials of a drug cannot be directly compared to the incidences in the clinical trials of another drug and may not reflect the incidences observed in practice.
The clinical program for STIOLTO RESPIMAT included 7,151 subjects with COPD in two 52-week active-controlled trials, one 12-week placebo-controlled trial, three 6-week placebo-controlled cross-over trials, and four additional trials of shorter duration. A total of 1,988 subjects received at least 1 dose of STIOLTO RESPIMAT. Adverse reactions observed in the ≤12-week trials were consistent with those observed in the 52-week trials, which formed the primary safety database.
The primary safety database consisted of pooled data from the two 52-week double-blind, active-controlled, parallel group confirmatory clinical trials (Trials 1 and 2). These trials included 5,162 adult COPD patients (72.9% males and 27.1% females) 40 years of age and older. Of these patients, 1,029 were treated with STIOLTO RESPIMAT once daily. The STIOLTO RESPIMAT group was composed of mostly Caucasians (71.1%) with a mean age of 63.8 years and a mean percent predicted FEV1 at baseline of 43.2%. In these two trials, tiotropium 5 mcg and olodaterol 5 mcg were included as active control arms and no placebo was used.
In these two clinical trials, 74% of patients exposed to STIOLTO RESPIMAT reported an adverse reaction compared to 76.6% and 73.3% in the olodaterol 5 mcg and tiotropium 5 mcg groups, respectively. The proportion of patients who discontinued due to an adverse reaction was 7.4% for STIOLTO RESPIMAT treated patients compared to 9.9% and 9.0% for olodaterol 5 mcg and tiotropium 5 mcg treated patients. The adverse reaction most commonly leading to discontinuation was worsening COPD.
The most common serious adverse reactions were COPD exacerbation and pneumonia.
Table 1 shows all adverse drug reactions that occurred with an incidence of >3% in the STIOLTO RESPIMAT treatment group and a higher incidence rate than the active comparator groups listed.
| Treatment | STIOLTO RESPIMAT (once daily) |
Tiotropium (5 mcg once daily) |
Olodaterol (5 mcg once daily) |
|---|---|---|---|
| Body system (adverse drug reaction) | n=1,029 n (%) |
n=1,033 n (%) |
n=1,038 n (%) |
| Infections and infestations | |||
| Nasopharyngitis | 128 (12.4) | 121 (11.7) | 131 (12.6) |
| Respiratory, thoracic, and mediastinal disorders | |||
| Cough | 40 (3.9) | 45 (4.4) | 31 (3.0) |
| Musculoskeletal and connective tissue disorders | |||
| Back Pain | 37 (3.6) | 19 (1.8) | 35 (3.4) |
Other adverse drug reactions in patients receiving STIOLTO RESPIMAT that occurred in ≤3% of patients in clinical studies are listed below:
Metabolism and nutrition disorders: dehydration
Nervous system disorders: dizziness, insomnia
Eye disorders: glaucoma, intraocular pressure increased, vision blurred
Cardiac/vascular disorders: atrial fibrillation, palpitations, supraventricular tachycardia, tachycardia, hypertension
Respiratory, thoracic, and mediastinal disorders: epistaxis, pharyngitis, dysphonia, bronchospasm, laryngitis, sinusitis
Gastrointestinal disorders: dry mouth, constipation, oropharyngeal candidiasis, dysphagia, gastroesophageal reflux disease, gingivitis, glossitis, stomatitis, intestinal obstruction including ileus paralytic
Skin and subcutaneous disorders: rash, pruritus, angioneurotic edema, urticaria, skin infection, and skin ulcer, dry skin, hypersensitivity (including immediate reactions)
Musculoskeletal and connective tissue disorders: arthralgia, joint swelling
Renal and urinary disorders: urinary retention, dysuria, and urinary tract infection
5.1 Serious Asthma Related Events – Hospitalizations, Intubations, Death
- The safety and efficacy of STIOLTO RESPIMAT in patients with asthma have not been established. STIOLTO RESPIMAT is not indicated for the treatment of asthma [see Contraindications (4)].
- Use of long-acting beta2-adrenergic agonists (LABA) as monotherapy [without inhaled corticosteroids (ICS)] for asthma is associated with an increased risk of asthma-related death. Available data from controlled clinical trials also suggest that use of LABA as monotherapy increases the risk of asthma-related hospitalization in pediatric and adolescent patients. These findings are considered a class effect of LABA monotherapy. When LABA are used in fixed-dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared with ICS alone.
- A 28-week, placebo-controlled US study comparing the safety of another LABA (salmeterol) with placebo, each added to usual asthma therapy, showed an increase in asthma-related deaths in patients receiving salmeterol (13/13,176 in patients treated with salmeterol vs. 3/13,179 in patients treated with placebo; RR 4.37, 95% CI 1.25, 15.34). The increased risk of asthma-related death is considered a class effect of LABA, including olodaterol, one of the active ingredients in STIOLTO RESPIMAT.
- No study adequate to determine whether the rate of asthma-related death is increased in patients treated with STIOLTO RESPIMAT has been conducted.
- Available data do not suggest an increased risk of death with use of LABA in patients with COPD.
5.3 Excessive Use of Stiolto Respimat and Use With Other Long Acting Beta2
As with other inhaled drugs containing beta2-adrenergic agents, STIOLTO RESPIMAT should not be used more often than recommended, at higher doses than recommended, or in conjunction with other medications containing long-acting beta2-agonists, as an overdose may result. Clinically significant cardiovascular effects and fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs.
7.4 Monoamine Oxidase Inhibitors, Tricyclic Antidepressants, Qtc Prolonging Drugs
STIOLTO RESPIMAT, as with other drugs containing beta2-agonists, should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants or other drugs known to prolong the QTc interval because the action of adrenergic agonists on the cardiovascular system may be potentiated by these agents. Drugs that are known to prolong the QTc interval may be associated with an increased risk of ventricular arrhythmias.
Structured Label Content
Section 42229-5 (42229-5)
Important Limitations of Use
- STIOLTO RESPIMAT is not indicated to treat acute deteriorations of COPD [see Warnings and Precautions (5.2)].
- STIOLTO RESPIMAT is not indicated to treat asthma. The safety and effectiveness of STIOLTO RESPIMAT in asthma have not been established.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 1/2025 | |
|
PATIENT INFORMATION STIOLTO® RESPIMAT® (sti-OL-to– RES peh mat) (tiotropium bromide and olodaterol inhalation spray), for oral inhalation use |
||
What is STIOLTO RESPIMAT?
|
||
Do not use STIOLTO RESPIMAT if you:
|
||
Before you use STIOLTO RESPIMAT, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take:
|
||
|
How should I use STIOLTO RESPIMAT?
Read the step-by-step instructions for using STIOLTO RESPIMAT at the end of this Patient Information leaflet.
|
||
|
What are the possible side effects with STIOLTO RESPIMAT?
STIOLTO RESPIMAT can cause serious side effects, including:
|
||
|
|
|
These are not all the side effects of STIOLTO RESPIMAT. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store STIOLTO RESPIMAT?
|
||
|
General information about the safe and effective use of STIOLTO RESPIMAT
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use STIOLTO RESPIMAT for a condition for which it was not prescribed. Do not give STIOLTO RESPIMAT to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about STIOLTO RESPIMAT that is written for health professionals. |
||
|
Active ingredients: tiotropium bromide and olodaterol Inactive ingredients: benzalkonium chloride, edetate disodium, hydrochloric acid, and water for injection Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc. Ridgefield, CT 06877 USA Licensed from: Boehringer Ingelheim International GmbH SPIRIVA®, HANDIHALER®, STIOLTO ® and RESPIMAT® are registered trademarks of and are used under license from Boehringer Ingelheim International GmbH Copyright © 2025 Boehringer Ingelheim International GmbH ALL RIGHTS RESERVED COL10537BA092025 For more information about STIOLTO RESPIMAT, including current prescribing information, a video demonstration or a Quick Start Guide on how to use STIOLTO RESPIMAT, go to www.STIOLTO.com, scan the code, or call 1-800-542-6257.
|
Section 44425-7 (44425-7)
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Avoid freezing.
Section 59845-8 (59845-8)
Instructions for Use
STIOLTO® RESPIMAT® (sti-OL-to- RES peh mat)
(tiotropium bromide and olodaterol inhalation spray), for oral inhalation use
For Oral Inhalation Only
Do not spray STIOLTO RESPIMAT into your eyes.
Read these Instructions for Use before you start using STIOLTO RESPIMAT and each time you get a refill. There may be new information. This leaflet does not take the place of talking to your doctor about your medical condition or your treatment.
You will need to use this inhaler 1 time each day, at the same time each day. Each time you use it take 2 puffs.
Do not turn the clear base before inserting the cartridge.
How to store your STIOLTO RESPIMAT inhaler
- Store STIOLTO RESPIMAT at room temperature between 68°F to 77°F (20°C to 25°C).
- Do not freeze your STIOLTO RESPIMAT cartridge and inhaler.
- If STIOLTO RESPIMAT has not been used for more than 3 days, release 1 puff towards the ground.
- If STIOLTO RESPIMAT has not been used for more than 21 days, repeat steps 4 to 6 under the "Prepare for first use" until a mist is visible. Then repeat steps 4 to 6 three more times.
- Keep your STIOLTO RESPIMAT cartridge, inhaler, and all medicines out of the reach of children.
How to care for your STIOLTO RESPIMAT inhaler
Clean the mouthpiece, including the metal part inside the mouthpiece, with a damp cloth or tissue only, at least 1 time each week. Any minor discoloration in the mouthpiece does not affect your STIOLTO RESPIMAT inhaler.
When to get a new STIOLTO RESPIMAT inhaler
- The scale on your inhaler will show the number of puffs you have, if used as indicated (2 puffs 1 time each day).
- The dose indicator will show you approximately how much medicine is left.
- When the dose indicator enters the red area of the scale, it will show you approximately how many puffs are left before you need a refill or new prescription.
- When the dose indicator reaches the end of the red scale, your STIOLTO RESPIMAT is empty and automatically locks. At this point, the clear base cannot be turned any further.
- Three months after insertion of cartridge, throw away the STIOLTO RESPIMAT even if it has not been used, or when the inhaler is locked, or when it expires, whichever comes first.
Prepare for first use
1. Remove clear base
|
|
2. Insert cartridge
|
|
3. Replace clear base
|
|
4. Turn
|
|
5. Open
|
|
6. Press
|
Daily use (T O P)
T
urn
|
|
O
pen
|
|
P
ress
|
Answers to Common Questions
It is difficult to insert the cartridge deep enough:
Did you accidentally turn the clear base before inserting the cartridge? Open the cap, press the dose-release button, then insert the cartridge.
Did you insert the cartridge with the wide end first? Insert the cartridge with the narrow end first.
I cannot press the dose-release button:
Did you turn the clear base? If not, turn the clear base in a continuous movement until it clicks (half a turn).
Is the dose indicator on the STIOLTO RESPIMAT pointing to 0 (zero)? The STIOLTO RESPIMAT inhaler is locked after the labeled number of puffs have been used. Prepare and use your new STIOLTO RESPIMAT inhaler.
I cannot turn the clear base:
Did you turn the clear base already? If the clear base has already been turned, follow steps "Open" and "Press" under "Daily use" to get your medicine.
Is the dose indicator on the STIOLTO RESPIMAT pointing to 0 (zero)? The STIOLTO RESPIMAT inhaler is locked after the labeled number of puffs have been used. Prepare and use your new STIOLTO RESPIMAT inhaler.
The dose indicator on the STIOLTO RESPIMAT reaches 0 (zero) too soon:
Did you use STIOLTO RESPIMAT as indicated (2 puffs 1 time each day)? STIOLTO RESPIMAT will deliver the labeled number of puffs if used at 2 puffs 1 time each day.
Did you turn the clear base before you inserted the cartridge? The dose indicator counts each turn of the clear base regardless whether a cartridge has been inserted or not.
Did you spray in the air often to check whether the STIOLTO RESPIMAT is working? After you have prepared STIOLTO RESPIMAT, no test-spraying is required if used daily.
Did you insert the cartridge into a used STIOLTO RESPIMAT? Always insert a new cartridge into a NEW STIOLTO RESPIMAT.
My STIOLTO RESPIMAT sprays automatically:
Was the cap open when you turned the clear base? Close the cap, then turn the clear base.
Did you press the dose-release button when turning the clear base? Close the cap, so the dose-release button is covered, then turn the clear base.
Did you stop when turning the clear base before it clicked? Turn the clear base in a continuous movement until it clicks (half a turn).
My STIOLTO RESPIMAT does not spray:
Did you insert a cartridge? If not, insert a cartridge.
Did you repeat Turn, Open, Press (TOP) less than 3 times after inserting the cartridge? Repeat Turn, Open, Press (TOP) 3 times after inserting the cartridge as shown in steps 4 to 6 under "Prepare for first use".
Is the dose indicator on the STIOLTO RESPIMAT pointing to 0 (zero)? You have used up all your medicine and the inhaler is locked.
For more information about STIOLTO RESPIMAT, including current prescribing information, a video demonstration or a Quick Start Guide on how to use STIOLTO RESPIMAT, go to www.STIOLTO.com, scan the code, or call 1-800-542-6257.
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT 06877 USA
Licensed from:
Boehringer Ingelheim International GmbH
SPIRIVA®, HANDIHALER®, STIOLTO® and RESPIMAT® are registered trademarks of and are used under license from Boehringer Ingelheim International GmbH
Copyright © 2025 Boehringer Ingelheim International GmbH
ALL RIGHTS RESERVED
Revised: January 2025
COL10537BA092025
10 Overdosage (10 OVERDOSAGE)
STIOLTO RESPIMAT contains both tiotropium bromide and olodaterol; therefore, the risks associated with overdosage for the individual components described below apply to STIOLTO RESPIMAT.
11 Description (11 DESCRIPTION)
STIOLTO RESPIMAT is a combination of tiotropium, an anticholinergic, and olodaterol, a long-acting beta2-adrenergic agonist (LABA).
The drug substance tiotropium bromide monohydrate is chemically described as (1α, 2ß, 4ß, 5α, 7ß)-7-[(Hydroxydi-2-thienylacetyl)oxy]-9,9-dimethyl-3-oxa-9-azoniatricyclo[3.3.1.02,4] nonane bromide monohydrate. It is a synthetic, non-chiral, quaternary ammonium compound. Tiotropium bromide is a white or yellowish white powder. It is sparingly soluble in water and soluble in methanol.
The structural formula is:
Tiotropium bromide (monohydrate) has a molecular mass of 490.4 and a molecular formula of C19H22NO4S2Br ∙ H2O.
The drug substance olodaterol hydrochloride is chemically described as 2H-1,4-Benzoxazin-3H(4H)-one, 6-hydroxy-8-[(1R)-1-hydroxy-2-[[2-(4-methoxyphenyl)-1,1-dimethylethyl]-amino]ethyl]-, monohydrochloride. Olodaterol hydrochloride is a white to off-white powder that is sparingly-slightly soluble in water and slightly soluble in ethanol. The molecular weight is 422.9 g/mole (salt): 386.5 g/mole (base), and the molecular formula is C21H26N2O5 × HCl as a hydrochloride. The conversion factor from salt to free base is 1.094.
The structural formula is:
The drug product, STIOLTO RESPIMAT, is composed of a sterile aqueous solution of tiotropium bromide and olodaterol hydrochloride filled into a 4.5 mL plastic container crimped into an aluminum cylinder (STIOLTO RESPIMAT cartridge) for use with the STIOLTO RESPIMAT inhaler.
Excipients include, benzalkonium chloride, edetate disodium, hydrochloric acid, and water for injection.
The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler. The STIOLTO RESPIMAT inhaler is a hand held, pocket sized oral inhalation device that uses mechanical energy to generate a slow-moving aerosol cloud of medication from a metered volume of the drug solution. The STIOLTO RESPIMAT inhaler has a light green-colored cap.
When used with the STIOLTO RESPIMAT inhaler each cartridge, containing 4 grams of sterile aqueous solution, delivers the labeled number of metered actuations after preparation for use. Each dose (one dose equals two actuations) from the STIOLTO RESPIMAT inhaler delivers 5 mcg tiotropium (equivalent to 6.247 mcg tiotropium bromide monohydrate) and 5 mcg olodaterol (equivalent to 5.473 mcg olodaterol hydrochloride) in 22.1 mcL from the mouthpiece. As with all inhaled drugs, the actual amount of drug delivered to the lung may depend on patient factors, such as the coordination between the actuation of the inhaler and inspiration through the delivery system. The duration of inspiration should be at least as long as the spray duration (1.5 seconds).
7.5 Beta Blockers (7.5 Beta-Blockers)
Beta-adrenergic receptor antagonists (beta-blockers) and the olodaterol component of STIOLTO RESPIMAT may interfere with the effect of each other when administered concurrently. Beta-blockers not only block the therapeutic effects of beta-agonists, but may produce severe bronchospasm in COPD patients. Therefore, patients with COPD should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-blockers in patients with COPD. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
8.4 Pediatric Use
COPD does not normally occur in children. The safety and effectiveness of STIOLTO RESPIMAT in the pediatric population has not been established.
8.5 Geriatric Use
Based on available data, no adjustment of STIOLTO RESPIMAT dosage in geriatric patients is warranted [see Clinical Pharmacology (12.3)].
Of the 1,029 patients who received STIOLTO RESPIMAT at the recommended dose once daily in the clinical studies from the pooled 1-year database, 525 (51.0%) were <65 years of age, 407 (39.6%) were 65 to <75, 96 (9.3%) were 75 to <85, and 1 (0.1%) was ≥85.
No overall differences in effectiveness were observed, and in the 1-year pooled data, the adverse drug reaction profiles were similar in the older population compared to the patient population overall.
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of STIOLTO RESPIMAT were evaluated in a clinical development program that included three dose ranging trials, two active-controlled trials, three active- and placebo-controlled trials, and one placebo-controlled trial. The efficacy of STIOLTO RESPIMAT is based primarily on two 4-week dose-ranging trials in 592 COPD patients and two confirmatory active-controlled 52-week trials (Trials 1 and 2) in 5,162 COPD patients.
4 Contraindications (4 CONTRAINDICATIONS)
Use of a LABA, including STIOLTO RESPIMAT, without an inhaled corticosteroid is contraindicated in patients with asthma [see Warnings and Precautions (5.1)]. STIOLTO RESPIMAT is not indicated for the treatment of asthma.
STIOLTO RESPIMAT is contraindicated in patients with a hypersensitivity to tiotropium, ipratropium, olodaterol, or any component of this product [see Warnings and Precautions (5.4)].
In clinical trials and postmarketing experience with tiotropium, immediate hypersensitivity reactions, including angioedema (including swelling of the lips, tongue, or throat), itching, or rash have been reported. Hypersensitivity reactions were also reported in clinical trials with STIOLTO RESPIMAT.
6 Adverse Reactions (6 ADVERSE REACTIONS)
LABA, such as olodaterol, one of the active components in STIOLTO RESPIMAT, as monotherapy (without an inhaled corticosteroid) for asthma, increase the risk of asthma-related events. STIOLTO RESPIMAT is not indicated for the treatment of asthma [see Warning and Precautions (5.1)].
The following adverse reactions are described, or described in greater detail, in other sections:
- Immediate hypersensitivity reactions [see Warnings and Precautions (5.4)]
- Paradoxical bronchospasm [see Warnings and Precautions (5.5)]
- Worsening of narrow-angle glaucoma [see Warnings and Precautions (5.8)]
- Worsening of urinary retention [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Other adrenergic drugs may potentiate effect. Use with caution. (5.3, 7.1)
- Xanthine derivatives, steroids, diuretics, or non-potassium sparing diuretics may potentiate hypokalemia or ECG changes. Use with caution. (7.2, 7.3)
- MAO inhibitors, tricyclic antidepressants, and drugs that prolong QTc interval may potentiate effect on cardiovascular system. Use with extreme caution. (7.4)
- Beta-blockers may decrease effectiveness. Use with caution and only when medically necessary. (7.5)
- Anticholinergics: May interact additively with concomitantly used anticholinergic medications. Avoid administration of STIOLTO RESPIMAT with other anticholinergic-containing drugs. (7.6)
7.1 Adrenergic Drugs
If additional adrenergic drugs are to be administered by any route, they should be used with caution because the sympathetic effects of olodaterol, one component of STIOLTO RESPIMAT, may be potentiated [see Warnings and Precautions (5.3, 5.6, 5.10, 5.11)].
7.6 Anticholinergics
There is potential for an additive interaction with concomitantly used anticholinergic medications. Therefore, avoid co-administration of STIOLTO RESPIMAT with other anticholinergic-containing drugs as this may lead to an increase in anticholinergic adverse effects [see Warnings and Precautions (5.8, 5.9) and Adverse Reactions (6)].
8.7 Renal Impairment
No dose adjustment is required for patients with renal impairment. However, patients with moderate to severe renal impairment (creatinine clearance of <60 mL/min) treated with STIOLTO RESPIMAT should be monitored closely for anticholinergic side effects [see Dosage and Administration (2), Warnings and Precautions (5.10), and Clinical Pharmacology (12.3)].
5.10 Renal Impairment
Because tiotropium is a predominantly renally excreted drug, patients with moderate to severe renal impairment (creatinine clearance of <60 mL/min) treated with STIOLTO RESPIMAT should be monitored closely for anticholinergic side effects [see Use in Specific Populations (8.7) and Clinical Pharmacology (12.3)].
2.1 Recommended Dosage
The recommended dosage of STIOLTO RESPIMAT is two inhalations once-daily at the same time of the day. Do not use STIOLTO RESPIMAT more than two inhalations every 24 hours.
8.6 Hepatic Impairment
No dose adjustment is needed in patients with mild and moderate hepatic impairment. A study in subjects with severe hepatic impairment was not performed [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
STIOLTO RESPIMAT is a combination of tiotropium bromide, an anticholinergic and olodaterol, a long-acting beta2-adrenergic agonist (LABA) indicated for the long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). (1.1)
Important limitations:
5.7 Coexisting Conditions
Olodaterol, like other sympathomimetic amines, should be used with caution in patients with convulsive disorders or thyrotoxicosis, in patients with known or suspected prolongation of the QT interval, and in patients who are unusually responsive to sympathomimetic amines. Doses of the related beta2-agonist albuterol, when administered intravenously, have been reported to aggravate pre-existing diabetes mellitus and ketoacidosis.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- LABA as monotherapy (without an inhaled corticosteroid) for asthma increases the risk of serious asthma-related events. (5.1)
- Do not initiate STIOLTO RESPIMAT in acutely deteriorating COPD patients. (5.2)
- Do not use for relief of acute symptoms. Concomitant short-acting beta2-agonists can be used as needed for acute relief. (5.2)
- Do not exceed the recommended dosage. Excessive use of STIOLTO RESPIMAT, or use in conjunction with other medications containing LABA can result in clinically significant cardiovascular effects and may be fatal. (5.3)
- Immediate hypersensitivity reactions: Discontinue STIOLTO RESPIMAT at once and consider alternatives if immediate hypersensitivity reactions, including angioedema, urticaria, rash, bronchospasm, or anaphylaxis, occur. (5.4)
- Life-threatening paradoxical bronchospasm can occur. Discontinue STIOLTO RESPIMAT immediately. (5.5)
- Use with caution in patients with cardiovascular or convulsive disorders, thyrotoxicosis, or sensitivity to sympathomimetic drugs. (5.6, 5.7)
- Worsening of narrow-angle glaucoma may occur. Use with caution in patients with narrow-angle glaucoma and instruct patients to consult a physician immediately if this occurs. (5.8)
- Worsening of urinary retention may occur. Use with caution in patients with prostatic hyperplasia or bladder-neck obstruction and instruct patients to consult a physician immediately if this occurs. (5.9)
- Be alert to hypokalemia and hyperglycemia. (5.11)
5.6 Cardiovascular Effects
Olodaterol, like other beta2-agonists, can produce a clinically significant cardiovascular effect in some patients as measured by increases in pulse rate, systolic or diastolic blood pressure, and/or symptoms. If such effects occur, STIOLTO RESPIMAT may need to be discontinued. In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the T wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Long acting beta2-adrenergic agonists should be administered with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, hypertrophic obstructive cardiomyopathy, and hypertension.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For oral inhalation only.
- Two inhalations of STIOLTO RESPIMAT once-daily at the same time of day. (2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Inhalation spray: Each actuation from the mouthpiece delivers 2.5 mcg tiotropium (equivalent to 3.124 mcg tiotropium bromide monohydrate), and 2.5 mcg olodaterol (equivalent to 2.736 mcg olodaterol hydrochloride). Two actuations equal one dose. (3)
5.5 Paradoxical Bronchospasm
As with other inhaled medicines, STIOLTO RESPIMAT may cause paradoxical bronchospasm that may be life-threatening. If paradoxical bronchospasm occurs, STIOLTO RESPIMAT should be stopped immediately and alternative therapy instituted.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
2.2 Administration Information
For oral inhalation only.
Prior to first use, the STIOLTO RESPIMAT cartridge is inserted into the STIOLTO RESPIMAT inhaler and the unit is primed. When using the unit for the first time, patients are to actuate the inhaler toward the ground until an aerosol cloud is visible and then repeat the process three more times. The unit is then considered primed and ready for use. If not used for more than 3 days, patients are to actuate the inhaler once to prepare the inhaler for use. If not used for more than 21 days, patients are to actuate the inhaler until an aerosol cloud is visible and then repeat the process three more times to prepare the inhaler for use [see Patient Counseling Information (17)].
No dosage adjustment is required for geriatric, hepatically-impaired, or renally-impaired patients. However, patients with moderate to severe renal impairment given STIOLTO RESPIMAT should be monitored closely for anticholinergic effects [see Warnings and Precautions (5.10), Use in Specific Populations (8.5, 8.6, 8.7), and Clinical Pharmacology (12.3)].
1.1 Maintenance Treatment of Copd (1.1 Maintenance Treatment of COPD)
STIOLTO RESPIMAT is a combination of tiotropium bromide and olodaterol indicated for long-term, once-daily maintenance treatment of patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.11 Hypokalemia and Hyperglycemia
Beta-adrenergic agonists may produce significant hypokalemia in some patients, which has the potential to produce adverse cardiovascular effects [see Clinical Pharmacology (12.2)]. The decrease in serum potassium is usually transient, not requiring supplementation. Inhalation of high doses of beta2-adrenergic agonists may produce increases in plasma glucose.
In patients with severe COPD, hypokalemia may be potentiated by hypoxia and concomitant treatment [see Drug Interactions (7.2)], which may increase the susceptibility for cardiac arrhythmias.
Clinically notable decreases in serum potassium or changes in blood glucose were infrequent during clinical studies with long-term administration of olodaterol with the rates similar to those for placebo controls. Olodaterol has not been investigated in patients whose diabetes mellitus is not well controlled.
5.9 Worsening of Urinary Retention
STIOLTO RESPIMAT should be used with caution in patients with urinary retention. Prescribers and patients should be alert for signs and symptoms of prostatic hyperplasia or bladder-neck obstruction (e.g., difficulty passing urine, painful urination), especially in patients with prostatic hyperplasia or bladder neck obstruction. Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
7.3 Non Potassium Sparing Diuretics (7.3 Non-Potassium Sparing Diuretics)
The ECG changes and/or hypokalemia that may result from the administration of non-potassium sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dosage of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the co-administration of STIOLTO RESPIMAT with non-potassium sparing diuretics.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
STIOLTO RESPIMAT Inhalation Spray is supplied in a labeled carton containing one STIOLTO RESPIMAT cartridge and one STIOLTO RESPIMAT inhaler.
The STIOLTO RESPIMAT cartridge is provided as an aluminum cylinder with a tamper protection seal on the cap. The STIOLTO RESPIMAT cartridge is only intended for use with the STIOLTO RESPIMAT inhaler and should not be interchanged with any other RESPIMAT device delivered product.
The STIOLTO RESPIMAT inhaler is a cylindrical shaped plastic inhalation device with a gray colored body and a clear base. The clear base is removed to insert the cartridge. The inhaler contains a dose indicator. The light green-colored cap and the written information on the label of the gray inhaler body indicate that it is labeled for use with the STIOLTO RESPIMAT cartridge.
STIOLTO RESPIMAT Inhalation Spray is available as:
- STIOLTO RESPIMAT Inhalation Spray: 60 metered actuations (NDC 0597-0155-61)
- STIOLTO RESPIMAT Inhalation Spray: 10 metered actuations (NDC 0597-0155-70) (institutional pack)
The STIOLTO RESPIMAT cartridge has a net fill weight of at least 4 grams and when used with the STIOLTO RESPIMAT inhaler, is designed to deliver the labeled number of metered actuations after preparation for use.
When the labeled number of actuations has been dispensed from the inhaler, the RESPIMAT locking mechanism will be engaged and no more actuations can be dispensed.
After assembly, the STIOLTO RESPIMAT inhaler should be discarded at the latest 3 months after first use or when the locking mechanism is engaged, whichever comes first.
Keep out of reach of children. Do not spray into eyes.
5.8 Worsening of Narrow Angle Glaucoma (5.8 Worsening of Narrow-Angle Glaucoma)
STIOLTO RESPIMAT should be used with caution in patients with narrow-angle glaucoma. Prescribers and patients should be alert for signs and symptoms of acute narrow-angle glaucoma (e.g., eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema). Instruct patients to consult a physician immediately should any of these signs or symptoms develop.
5.4 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions, including urticaria, angioedema (including swelling of the lips, tongue or throat), rash, bronchospasm, anaphylaxis, or itching may occur after administration of STIOLTO RESPIMAT. If such a reaction occurs, therapy with STIOLTO RESPIMAT should be stopped at once and alternative treatments should be considered. Given the similar structural formula of atropine to tiotropium, patients with a history of hypersensitivity reactions to atropine or its derivatives should be closely monitored for similar hypersensitivity reactions to STIOLTO RESPIMAT.
5.2 Deterioration of Disease and Acute Episodes
STIOLTO RESPIMAT should not be initiated in patients with acutely deteriorating COPD, which may be a life-threatening condition. STIOLTO RESPIMAT has not been studied in patients with acutely deteriorating COPD. The use of STIOLTO RESPIMAT in this setting is inappropriate.
STIOLTO RESPIMAT should not be used for the relief of acute symptoms, i.e., as rescue therapy for the treatment of acute episodes of bronchospasm. STIOLTO RESPIMAT has not been studied in the relief of acute symptoms and extra doses should not be used for that purpose. Acute symptoms should be treated with an inhaled short-acting beta2-agonist.
When beginning STIOLTO RESPIMAT, patients who have been taking inhaled, short-acting beta2-agonists on a regular basis (e.g., four times a day) should be instructed to discontinue the regular use of these drugs and use them only for symptomatic relief of acute respiratory symptoms. When prescribing STIOLTO RESPIMAT, the healthcare provider should also prescribe an inhaled, short-acting beta2-agonist and instruct the patient on how it should be used. Increasing inhaled beta2-agonist use is a signal of deteriorating disease for which prompt medical attention is indicated.
COPD may deteriorate acutely over a period of hours or chronically over several days or longer. If STIOLTO RESPIMAT no longer controls symptoms of bronchoconstriction, or the patient's inhaled, short-acting beta2-agonist becomes less effective or the patient needs more inhalation of short-acting beta2-agonist than usual, these may be markers of deterioration of disease. In this setting, a re-evaluation of the patient and the COPD treatment regimen should be undertaken at once. Increasing the daily dosage of STIOLTO RESPIMAT beyond the recommended dosage is not appropriate in this situation.
Principal Display Panel 2.5 Mcg/2.5 Mcg Cartridge Carton (PRINCIPAL DISPLAY PANEL - 2.5 mcg/2.5 mcg Cartridge Carton)
NDC 0597-0155-61
Stiolto® Respimat®
(tiotropium bromide and olodaterol
inhalation spray)
2.5 mcg/2.5 mcg per actuation*
FOR ORAL INHALATION ONLY
ATTENTION PHARMACIST:
Dispense with Instructions for Use
Rx only
4 Grams
60 Metered Inhalations
(Two inhalations equal one dose)
Boehringer
Ingelheim
7.7 Inhibitors of Cytochrome P450 and P Gp Efflux Transporter (7.7 Inhibitors of Cytochrome P450 and P-gp Efflux Transporter)
In a drug interaction study using the strong dual CYP and P-gp inhibitor ketoconazole, a 1.7-fold increase of olodaterol maximum plasma concentrations and AUC was observed [see Pharmacokinetics (12.3)]. Olodaterol was evaluated in clinical trials for up to one year at doses up to twice the recommended therapeutic dosage. No dosage adjustment of STIOLTO RESPIMAT is necessary.
7.2 Sympathomimetics, Xanthine Derivatives, Steroids, Or Diuretics (7.2 Sympathomimetics, Xanthine Derivatives, Steroids, or Diuretics)
Tiotropium has been used concomitantly with short-acting and long-acting sympathomimetic (beta-agonists) bronchodilators, methylxanthines, and oral and inhaled steroids, without increases in adverse reactions. Concomitant treatment with xanthine derivatives, steroids, or diuretics may potentiate any hypokalemic effect of olodaterol [see Warnings and Precautions (5.11)].
6.1 Clinical Trials Experience in Chronic Obstructive Pulmonary Disease
Because clinical trials are conducted under widely varying conditions, the incidence of adverse reactions observed in the clinical trials of a drug cannot be directly compared to the incidences in the clinical trials of another drug and may not reflect the incidences observed in practice.
The clinical program for STIOLTO RESPIMAT included 7,151 subjects with COPD in two 52-week active-controlled trials, one 12-week placebo-controlled trial, three 6-week placebo-controlled cross-over trials, and four additional trials of shorter duration. A total of 1,988 subjects received at least 1 dose of STIOLTO RESPIMAT. Adverse reactions observed in the ≤12-week trials were consistent with those observed in the 52-week trials, which formed the primary safety database.
The primary safety database consisted of pooled data from the two 52-week double-blind, active-controlled, parallel group confirmatory clinical trials (Trials 1 and 2). These trials included 5,162 adult COPD patients (72.9% males and 27.1% females) 40 years of age and older. Of these patients, 1,029 were treated with STIOLTO RESPIMAT once daily. The STIOLTO RESPIMAT group was composed of mostly Caucasians (71.1%) with a mean age of 63.8 years and a mean percent predicted FEV1 at baseline of 43.2%. In these two trials, tiotropium 5 mcg and olodaterol 5 mcg were included as active control arms and no placebo was used.
In these two clinical trials, 74% of patients exposed to STIOLTO RESPIMAT reported an adverse reaction compared to 76.6% and 73.3% in the olodaterol 5 mcg and tiotropium 5 mcg groups, respectively. The proportion of patients who discontinued due to an adverse reaction was 7.4% for STIOLTO RESPIMAT treated patients compared to 9.9% and 9.0% for olodaterol 5 mcg and tiotropium 5 mcg treated patients. The adverse reaction most commonly leading to discontinuation was worsening COPD.
The most common serious adverse reactions were COPD exacerbation and pneumonia.
Table 1 shows all adverse drug reactions that occurred with an incidence of >3% in the STIOLTO RESPIMAT treatment group and a higher incidence rate than the active comparator groups listed.
| Treatment | STIOLTO RESPIMAT (once daily) |
Tiotropium (5 mcg once daily) |
Olodaterol (5 mcg once daily) |
|---|---|---|---|
| Body system (adverse drug reaction) | n=1,029 n (%) |
n=1,033 n (%) |
n=1,038 n (%) |
| Infections and infestations | |||
| Nasopharyngitis | 128 (12.4) | 121 (11.7) | 131 (12.6) |
| Respiratory, thoracic, and mediastinal disorders | |||
| Cough | 40 (3.9) | 45 (4.4) | 31 (3.0) |
| Musculoskeletal and connective tissue disorders | |||
| Back Pain | 37 (3.6) | 19 (1.8) | 35 (3.4) |
Other adverse drug reactions in patients receiving STIOLTO RESPIMAT that occurred in ≤3% of patients in clinical studies are listed below:
Metabolism and nutrition disorders: dehydration
Nervous system disorders: dizziness, insomnia
Eye disorders: glaucoma, intraocular pressure increased, vision blurred
Cardiac/vascular disorders: atrial fibrillation, palpitations, supraventricular tachycardia, tachycardia, hypertension
Respiratory, thoracic, and mediastinal disorders: epistaxis, pharyngitis, dysphonia, bronchospasm, laryngitis, sinusitis
Gastrointestinal disorders: dry mouth, constipation, oropharyngeal candidiasis, dysphagia, gastroesophageal reflux disease, gingivitis, glossitis, stomatitis, intestinal obstruction including ileus paralytic
Skin and subcutaneous disorders: rash, pruritus, angioneurotic edema, urticaria, skin infection, and skin ulcer, dry skin, hypersensitivity (including immediate reactions)
Musculoskeletal and connective tissue disorders: arthralgia, joint swelling
Renal and urinary disorders: urinary retention, dysuria, and urinary tract infection
5.1 Serious Asthma Related Events – Hospitalizations, Intubations, Death (5.1 Serious Asthma-Related Events – Hospitalizations, Intubations, Death)
- The safety and efficacy of STIOLTO RESPIMAT in patients with asthma have not been established. STIOLTO RESPIMAT is not indicated for the treatment of asthma [see Contraindications (4)].
- Use of long-acting beta2-adrenergic agonists (LABA) as monotherapy [without inhaled corticosteroids (ICS)] for asthma is associated with an increased risk of asthma-related death. Available data from controlled clinical trials also suggest that use of LABA as monotherapy increases the risk of asthma-related hospitalization in pediatric and adolescent patients. These findings are considered a class effect of LABA monotherapy. When LABA are used in fixed-dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared with ICS alone.
- A 28-week, placebo-controlled US study comparing the safety of another LABA (salmeterol) with placebo, each added to usual asthma therapy, showed an increase in asthma-related deaths in patients receiving salmeterol (13/13,176 in patients treated with salmeterol vs. 3/13,179 in patients treated with placebo; RR 4.37, 95% CI 1.25, 15.34). The increased risk of asthma-related death is considered a class effect of LABA, including olodaterol, one of the active ingredients in STIOLTO RESPIMAT.
- No study adequate to determine whether the rate of asthma-related death is increased in patients treated with STIOLTO RESPIMAT has been conducted.
- Available data do not suggest an increased risk of death with use of LABA in patients with COPD.
5.3 Excessive Use of Stiolto Respimat and Use With Other Long Acting Beta2 (5.3 Excessive Use of STIOLTO RESPIMAT and Use With Other Long-Acting Beta2)
As with other inhaled drugs containing beta2-adrenergic agents, STIOLTO RESPIMAT should not be used more often than recommended, at higher doses than recommended, or in conjunction with other medications containing long-acting beta2-agonists, as an overdose may result. Clinically significant cardiovascular effects and fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs.
7.4 Monoamine Oxidase Inhibitors, Tricyclic Antidepressants, Qtc Prolonging Drugs (7.4 Monoamine Oxidase Inhibitors, Tricyclic Antidepressants, QTc Prolonging Drugs)
STIOLTO RESPIMAT, as with other drugs containing beta2-agonists, should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants or other drugs known to prolong the QTc interval because the action of adrenergic agonists on the cardiovascular system may be potentiated by these agents. Drugs that are known to prolong the QTc interval may be associated with an increased risk of ventricular arrhythmias.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:11.409150 · Updated: 2026-03-14T22:29:13.416831