These Highlights Do Not Include All The Information Needed To Use Fulvestrant Injection Safely And Effectively. See Full Prescribing Information For Fulvestrant Injection.
015fafb6-23f7-4566-9c23-386eee7b035d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Fulvestrant Injection is an estrogen receptor antagonist indicated for the treatment of: Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy. ( 1 ) HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in postmenopausal women in combination with ribociclib, as initial endocrine based therapy or following disease progression on endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in combination with palbociclib or abemaciclib in women with disease progression after endocrine therapy. ( 1 )
Indications and Usage
Fulvestrant Injection is an estrogen receptor antagonist indicated for the treatment of: Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy. ( 1 ) HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in postmenopausal women in combination with ribociclib, as initial endocrine based therapy or following disease progression on endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in combination with palbociclib or abemaciclib in women with disease progression after endocrine therapy. ( 1 )
Dosage and Administration
Fulvestrant Injection 500 mg should be administered intramuscularly into the buttocks (gluteal area) slowly (1 - 2 minutes per injection) as two 5 mL injections, one in each buttock, on Days 1, 15, 29, and once monthly thereafter. ( 2.1 , 14 ) A dose of 250 mg is recommended in patients with moderate hepatic impairment to be administered intramuscularly into the buttock (gluteal area) slowly (1 - 2 minutes) as one 5 mL injection on Days 1, 15, 29, and once monthly thereafter. ( 2.2 , 5.2 , 8.6 )
Warnings and Precautions
Risk of Bleeding: Use with caution in patients with bleeding diatheses, thrombocytopenia, or anticoagulant use. ( 5.1 ) Increased Exposure in Patients with Hepatic Impairment: Use a 250 mg dose for patients with moderate hepatic impairment. ( 2.2 , 5.2 , 8.6 ) Injection Site Reaction: Use caution while administering Fulvestrant Injection at the dorsogluteal injection site due to the proximity of the underlying sciatic nerve. ( 5.3 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.4 , 8.1 , 8.3 ) Immunoassay Measurement of Serum Estradiol: Fulvestrant Injection can interfere with estradiol measurement by immunoassay, resulting in falsely elevated estradiol levels. ( 5.5 )
Contraindications
Fulvestrant Injection is contraindicated in patients with a known hypersensitivity to the drug or to any of its components. Hypersensitivity reactions, including urticaria and angioedema, have been reported in association with Fulvestrant Injection [see Adverse Reactions ( 6.2 )].
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Risk of Bleeding [see Warnings and Precautions ( 5.1 )] Increased Exposure in Patients with Hepatic Impairment [see Warnings and Precautions ( 5.2 )] Injection Site Reaction [see Warnings and Precautions ( 5.3 )] Embryo-Fetal Toxicity [see Warnings and Precautions ( 5.4 )]
Drug Interactions
There are no known drug-drug interactions. Although, fulvestrant is metabolized by CYP 3A4 in vitro , drug interactions studies with ketoconazole or rifampin did not alter fulvestrant pharmacokinetics. Dose adjustment is not needed in patients co-prescribed CYP 3A4 inhibitors or inducers [see Clinical Pharmacology ( 12.3 )] .
Storage and Handling
Fulvestrant Injection is supplied as two 5 mL clear neutral glass (Type 1) barrels, each containing a sterile, clear, colorless to yellow viscous liquid for intramuscular injection and fitted with a plastic rigid tip cap. It is supplied as follows: NDC Fulvestrant Injection, USP (50 mg per mL) Package Factor 71288- 555 -86 250 mg per 5 mL Single-Dose Prefilled Syringe 2 syringes per carton The 2 x 5 mL single-dose prefilled syringes are presented in a tray with polystyrene plunger rod and 21G x 1-1/2 inch safety needles (SafetyGlide ™ ) for connection to the barrel. Discard each syringe after use. If a patient dose requires only one syringe, unused syringe should be stored as directed below.
How Supplied
Fulvestrant Injection is supplied as two 5 mL clear neutral glass (Type 1) barrels, each containing a sterile, clear, colorless to yellow viscous liquid for intramuscular injection and fitted with a plastic rigid tip cap. It is supplied as follows: NDC Fulvestrant Injection, USP (50 mg per mL) Package Factor 71288- 555 -86 250 mg per 5 mL Single-Dose Prefilled Syringe 2 syringes per carton The 2 x 5 mL single-dose prefilled syringes are presented in a tray with polystyrene plunger rod and 21G x 1-1/2 inch safety needles (SafetyGlide ™ ) for connection to the barrel. Discard each syringe after use. If a patient dose requires only one syringe, unused syringe should be stored as directed below.
Medication Information
Warnings and Precautions
Risk of Bleeding: Use with caution in patients with bleeding diatheses, thrombocytopenia, or anticoagulant use. ( 5.1 ) Increased Exposure in Patients with Hepatic Impairment: Use a 250 mg dose for patients with moderate hepatic impairment. ( 2.2 , 5.2 , 8.6 ) Injection Site Reaction: Use caution while administering Fulvestrant Injection at the dorsogluteal injection site due to the proximity of the underlying sciatic nerve. ( 5.3 ) Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.4 , 8.1 , 8.3 ) Immunoassay Measurement of Serum Estradiol: Fulvestrant Injection can interfere with estradiol measurement by immunoassay, resulting in falsely elevated estradiol levels. ( 5.5 )
Indications and Usage
Fulvestrant Injection is an estrogen receptor antagonist indicated for the treatment of: Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy. ( 1 ) HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in postmenopausal women in combination with ribociclib, as initial endocrine based therapy or following disease progression on endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in combination with palbociclib or abemaciclib in women with disease progression after endocrine therapy. ( 1 )
Dosage and Administration
Fulvestrant Injection 500 mg should be administered intramuscularly into the buttocks (gluteal area) slowly (1 - 2 minutes per injection) as two 5 mL injections, one in each buttock, on Days 1, 15, 29, and once monthly thereafter. ( 2.1 , 14 ) A dose of 250 mg is recommended in patients with moderate hepatic impairment to be administered intramuscularly into the buttock (gluteal area) slowly (1 - 2 minutes) as one 5 mL injection on Days 1, 15, 29, and once monthly thereafter. ( 2.2 , 5.2 , 8.6 )
Contraindications
Fulvestrant Injection is contraindicated in patients with a known hypersensitivity to the drug or to any of its components. Hypersensitivity reactions, including urticaria and angioedema, have been reported in association with Fulvestrant Injection [see Adverse Reactions ( 6.2 )].
Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling: Risk of Bleeding [see Warnings and Precautions ( 5.1 )] Increased Exposure in Patients with Hepatic Impairment [see Warnings and Precautions ( 5.2 )] Injection Site Reaction [see Warnings and Precautions ( 5.3 )] Embryo-Fetal Toxicity [see Warnings and Precautions ( 5.4 )]
Drug Interactions
There are no known drug-drug interactions. Although, fulvestrant is metabolized by CYP 3A4 in vitro , drug interactions studies with ketoconazole or rifampin did not alter fulvestrant pharmacokinetics. Dose adjustment is not needed in patients co-prescribed CYP 3A4 inhibitors or inducers [see Clinical Pharmacology ( 12.3 )] .
Storage and Handling
Fulvestrant Injection is supplied as two 5 mL clear neutral glass (Type 1) barrels, each containing a sterile, clear, colorless to yellow viscous liquid for intramuscular injection and fitted with a plastic rigid tip cap. It is supplied as follows: NDC Fulvestrant Injection, USP (50 mg per mL) Package Factor 71288- 555 -86 250 mg per 5 mL Single-Dose Prefilled Syringe 2 syringes per carton The 2 x 5 mL single-dose prefilled syringes are presented in a tray with polystyrene plunger rod and 21G x 1-1/2 inch safety needles (SafetyGlide ™ ) for connection to the barrel. Discard each syringe after use. If a patient dose requires only one syringe, unused syringe should be stored as directed below.
How Supplied
Fulvestrant Injection is supplied as two 5 mL clear neutral glass (Type 1) barrels, each containing a sterile, clear, colorless to yellow viscous liquid for intramuscular injection and fitted with a plastic rigid tip cap. It is supplied as follows: NDC Fulvestrant Injection, USP (50 mg per mL) Package Factor 71288- 555 -86 250 mg per 5 mL Single-Dose Prefilled Syringe 2 syringes per carton The 2 x 5 mL single-dose prefilled syringes are presented in a tray with polystyrene plunger rod and 21G x 1-1/2 inch safety needles (SafetyGlide ™ ) for connection to the barrel. Discard each syringe after use. If a patient dose requires only one syringe, unused syringe should be stored as directed below.
Description
Fulvestrant Injection is an estrogen receptor antagonist indicated for the treatment of: Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy. ( 1 ) HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in postmenopausal women in combination with ribociclib, as initial endocrine based therapy or following disease progression on endocrine therapy. ( 1 ) HR-positive, HER2-negative advanced or metastatic breast cancer in combination with palbociclib or abemaciclib in women with disease progression after endocrine therapy. ( 1 )
Section 42229-5
Monotherapy
Fulvestrant Injection is indicated for the treatment of:
- Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy, or
- HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy.
Section 42230-3
|
PATIENT INFORMATION
FULVESTRANT (ful VES trant) INJECTION |
|
|
What is Fulvestrant Injection?
Fulvestrant Injection is a prescription medicine used to treat advanced breast cancer or breast cancer that has spread to other parts of the body (metastatic). Fulvestrant Injection may be used alone, if you have gone through menopause, and your advanced breast cancer is:
|
|
| Fulvestrant Injection may be used in combination with ribociclib, if you have gone through menopause, and your advanced or metastatic breast cancer is HR-positive and HER2-negative, and has not been previously treated with endocrine therapy or has progressed after endocrine therapy. Fulvestrant Injection may be used in combination with palbociclib or abemaciclib if your advanced or metastatic breast cancer is HR-positive and HER2-negative, and has progressed after endocrine therapy. When Fulvestrant Injection is used in combination with palbociclib, abemaciclib, or ribociclib, also read the Patient Information for the prescribed product. It is not known if Fulvestrant Injection is safe and effective in children. It is not known if Fulvestrant Injection is safe and effective in people with severe liver problems. |
|
|
Who should not receive Fulvestrant Injection?
Do not receive Fulvestrant Injection if you have had an allergic reaction to fulvestrant or any of the ingredients in Fulvestrant Injection. See the end of this leaflet for a list of the ingredients in Fulvestrant Injection. Symptoms of an allergic reaction to Fulvestrant Injection may include: • itching or hives • swelling of your face, lips, tongue or throat • trouble breathing |
|
|
What should I tell my healthcare provider before receiving Fulvestrant Injection?
Before receiving Fulvestrant Injection, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Fulvestrant Injection may affect the way other medicines work, and other medicines may affect how Fulvestrant Injection works. Especially tell your healthcare provider if you take a blood thinner medicine. |
|
How will I receive Fulvestrant Injection?
|
|
|
What are the possible side effects of Fulvestrant Injection?
Fulvestrant Injection may cause serious side effects, including:
|
|
|
|
| Fulvestrant Injection may cause fertility problems in males and females. Talk to your healthcare provider if you plan to become pregnant. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects with Fulvestrant Injection. For more information, ask your healthcare provider or pharmacist. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about the safe and effective use of Fulvestrant Injection
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Fulvestrant Injection that is written for health professionals. |
|
|
What are the ingredients in Fulvestrant Injection?
Active ingredient: fulvestrant Inactive ingredients: alcohol, benzyl alcohol, benzyl benzoate, and castor oil. |
This Patient Information has been approved by the U.S. Food and Drug Administration.
Brands listed are the trademarks of their respective owners.
Mfd. for Meitheal Pharmaceuticals
Chicago, IL 60631 (USA)
©2021 Meitheal Pharmaceuticals Inc.
Mfd. by Nanjing King-Friend Biochemical Pharmaceutical Co., Ltd.
Nanjing, China 210061
Revised: February 2021
810038-02
Section 44425-7
Storage
REFRIGERATE, 2° to 8°C (36° to 46°F). TO PROTECT FROM LIGHT, STORE IN THE ORIGINAL CARTON UNTIL TIME OF USE.
Sterile, Nonpyrogenic, PVC-free, DEHP-free.
Section 51945-4
Principal Display Panel – Fulvestrant Injection, 250 mg per 5 mL (50mg per ml) Syringe Label
NDC 71288-555-85
Rx only
Fulvestrant Injection
250 mg per 5 mL
(50 mg per mL)
Date/Site:
Administered by:
For Intramuscular Use Only
PROTECT FROM LIGHT
Both single-dose prefilled syringes must be administered to receive the 500 mg dose.
10 Overdosage
Human experience of overdose with Fulvestrant Injection is limited. There are isolated reports of overdose with Fulvestrant Injection in humans. No adverse reactions were seen in healthy male and female volunteers who received intravenous fulvestrant, which resulted in peak plasma concentrations at the end of the infusion, that were approximately 10 to 15 times those seen after intramuscular injection. The potential toxicity of fulvestrant at these or higher concentrations in cancer patients who may have additional comorbidities is unknown. There is no specific treatment in the event of fulvestrant overdose, and symptoms of overdose are not established. In the event of an overdose, healthcare practitioners should follow general supportive measures and should treat symptomatically.
11 Description
Fulvestrant Injection for intramuscular administration is an estrogen receptor antagonist. The chemical name is 7-alpha-[9-(4,4,5,5,5-penta fluoropentylsulphinyl) nonyl]estra-1,3,5-(10)-triene-3,17-beta-diol. The molecular formula is C32H47F5O3S and its structural formula is:
Fulvestrant is a white powder with a molecular weight of 606.77. The solution for injection is a clear, colorless to yellow, viscous liquid.
Each injection contains as inactive ingredients: 10% w/v (12% v/v) Alcohol, USP, 10% w/v Benzyl Alcohol, NF, and 15% w/v Benzyl Benzoate, USP, as co-solvents, and made up to 100% w/v with Castor Oil, USP as a co-solvent and release rate modifier.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. A multi-center, single-arm, open-label, study of fulvestrant was conducted in 30 girls with McCune-Albright Syndrome (MAS) associated with Progressive Precocious Puberty (PPP). The median age at informed consent was 6 years old (range: 1 to 8).
The first 10 patients initially received fulvestrant 2 mg/kg. Based on PK data from the first 6 patients, all 10 patients receiving 2 mg/kg were escalated to a dose of 4 mg/kg and all other patients received 4 mg/kg from study entry.
Baseline measurements for vaginal bleeding days, bone age, growth velocity, and Tanner staging for at least 6 months prior to study entry were provided retrospectively by the parent, guardian or local consultant. All measurements during the study period were collected prospectively. Patients' baseline characteristics included the following: a mean ± SD chronological age of 5.9 ± 1.8 years; a mean rate of bone age advancement (change in bone age in years divided by change in chronological age in years) of 2.0 ± 1.03; and a mean growth velocity z-score of 2.4 ± 3.26.
Twenty-nine of 30 patients completed the 12-month study period. The following results were observed: 35% (95% CI: 16%, 57%) of the 23 patients with baseline vaginal bleeding experienced a complete cessation of vaginal bleeding on-treatment (month 0 to 12); a reduction in the rate of bone age advancement during the 12-month study period compared to baseline (mean change=-0.9 [95% CI: -1.4, -0.4]); and a reduction in mean growth velocity Z-score on-treatment compared to baseline (mean change=-1.1 [95% CI: -2.7, 0.4]). There were no clinically meaningful changes in median Tanner stage (breast or pubic), mean uterine volume, or mean ovarian volume, or predicted adult height (PAH) on-treatment compared to baseline. The effect of Fulvestrant Injection on bone mineral density in children has not been studied and is not known.
Eight patients (27%) experienced adverse reactions that were considered possibly related to Fulvestrant Injection. These included injection site reactions (inflammation, pain, hematoma, pruritus, rash), abdominal pain, contusion, tachycardia, hot flash, extremity pain, and vomiting. Nine (30%) patients reported an SAE, none of which were considered related to Fulvestrant Injection. No patients discontinued study treatment due to an AE and no patients died.
8.5 Geriatric Use
For Fulvestrant Injection 250 mg, when tumor response was considered by age, objective responses were seen in 22% and 24% of patients under 65 years of age and in 11% and 16% of patients 65 years of age and older, who were treated with Fulvestrant Injection in Study 0021 and Study 0020, respectively.
14 Clinical Studies
The efficacy of Fulvestrant Injection 500 mg versus Fulvestrant Injection 250 mg was compared in CONFIRM. The efficacy of Fulvestrant Injection 250 mg was compared to 1 mg anastrozole in Studies 0020 and 0021. The efficacy of Fulvestrant Injection 500 mg was compared to 1 mg anastrozole in FALCON. The efficacy of Fulvestrant Injection 500 mg in combination with palbociclib 125 mg was compared to Fulvestrant Injection 500 mg plus placebo in PALOMA-3. The efficacy of Fulvestrant Injection 500 mg in combination with abemaciclib 150 mg was compared to Fulvestrant Injection 500 mg plus placebo in MONARCH 2. The efficacy of Fulvestrant Injection 500 mg in combination with ribociclib 600 mg was compared to Fulvestrant Injection 500 mg plus placebo in MONALEESA-3.
4 Contraindications
Fulvestrant Injection is contraindicated in patients with a known hypersensitivity to the drug or to any of its components. Hypersensitivity reactions, including urticaria and angioedema, have been reported in association with Fulvestrant Injection [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following adverse reactions are discussed in more detail in other sections of the labeling:
7 Drug Interactions
There are no known drug-drug interactions. Although, fulvestrant is metabolized by CYP 3A4 in vitro, drug interactions studies with ketoconazole or rifampin did not alter fulvestrant pharmacokinetics. Dose adjustment is not needed in patients co-prescribed CYP 3A4 inhibitors or inducers [see Clinical Pharmacology (12.3)].
5.1 Risk of Bleeding
Because Fulvestrant Injection is administered intramuscularly, it should be used with caution in patients with bleeding diatheses, thrombocytopenia, or anticoagulant use.
8.7 Renal Impairment
Negligible amounts of fulvestrant are eliminated in urine; therefore, a study in patients with renal impairment was not conducted. In the advanced breast cancer trials, fulvestrant concentrations in women with estimated creatinine clearance as low as 30 mL/min were similar to women with normal creatinine.
12.2 Pharmacodynamics
In a clinical study in postmenopausal women with primary breast cancer treated with single doses of Fulvestrant Injection 15-22 days prior to surgery, there was evidence of increasing down-regulation of ER with increasing dose. This was associated with a dose-related decrease in the expression of the progesterone receptor, an estrogen-regulated protein. These effects on the ER pathway were also associated with a decrease in Ki67 labeling index, a marker of cell proliferation.
8.6 Hepatic Impairment
Fulvestrant Injection is metabolized primarily in the liver.
The pharmacokinetics of fulvestrant were evaluated after a single dose of 100 mg in subjects with mild and moderate hepatic impairment and normal hepatic function (n=7 subjects/group), using a shorter-acting intramuscular injection formulation. Subjects with mild hepatic impairment (Child-Pugh class A) had comparable mean AUC and clearance values to those with normal hepatic function. In subjects with moderate hepatic impairment (Child-Pugh class B), the average AUC of fulvestrant increased by 70% compared to patients with normal hepatic function. AUC was positively correlated with total bilirubin concentration (p=0.012). Fulvestrant Injection has not been studied in patients with severe hepatic impairment (Child-Pugh class C).
A dose of Fulvestrant Injection 250 mg is recommended in patients with moderate hepatic impairment (Child-Pugh class B) [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)].
1 Indications and Usage
Fulvestrant Injection is an estrogen receptor antagonist indicated for the treatment of:
- Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy. (1)
- HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy. (1)
- HR-positive, HER2-negative advanced or metastatic breast cancer in postmenopausal women in combination with ribociclib, as initial endocrine based therapy or following disease progression on endocrine therapy. (1)
- HR-positive, HER2-negative advanced or metastatic breast cancer in combination with palbociclib or abemaciclib in women with disease progression after endocrine therapy. (1)
12.1 Mechanism of Action
Many breast cancers have estrogen receptors (ER) and the growth of these tumors can be stimulated by estrogen. Fulvestrant is an estrogen receptor antagonist that binds to the estrogen receptor in a competitive manner with affinity comparable to that of estradiol and downregulates the ER protein in human breast cancer cells.
In vitro studies demonstrated that fulvestrant is a reversible inhibitor of the growth of tamoxifen-resistant, as well as estrogen-sensitive human breast cancer (MCF-7) cell lines. In in vivo tumor studies, fulvestrant delayed the establishment of tumors from xenografts of human breast cancer MCF-7 cells in nude mice. Fulvestrant inhibited the growth of established MCF-7 xenografts and of tamoxifen-resistant breast tumor xenografts.
Fulvestrant showed no agonist-type effects in in vivo uterotrophic assays in immature or ovariectomized mice and rats. In in vivo studies in immature rats and ovariectomized monkeys, fulvestrant blocked the uterotrophic action of estradiol. In postmenopausal women, the absence of changes in plasma concentrations of FSH and LH in response to fulvestrant treatment (250 mg monthly) suggests no peripheral steroidal effects.
5.4 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, Fulvestrant Injection can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of fulvestrant to pregnant rats and rabbits during organogenesis resulted in embryo-fetal toxicity at daily doses that are significantly less than the maximum recommended human dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with Fulvestrant Injection and for one year after the last dose [see Use in Specific Populations (8.1), (8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
- Risk of Bleeding: Use with caution in patients with bleeding diatheses, thrombocytopenia, or anticoagulant use. (5.1)
- Increased Exposure in Patients with Hepatic Impairment: Use a 250 mg dose for patients with moderate hepatic impairment. (2.2, 5.2, 8.6)
- Injection Site Reaction: Use caution while administering Fulvestrant Injection at the dorsogluteal injection site due to the proximity of the underlying sciatic nerve. (5.3)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.4, 8.1, 8.3)
- Immunoassay Measurement of Serum Estradiol: Fulvestrant Injection can interfere with estradiol measurement by immunoassay, resulting in falsely elevated estradiol levels. (5.5)
2 Dosage and Administration
- Fulvestrant Injection 500 mg should be administered intramuscularly into the buttocks (gluteal area) slowly (1 - 2 minutes per injection) as two 5 mL injections, one in each buttock, on Days 1, 15, 29, and once monthly thereafter. (2.1, 14)
- A dose of 250 mg is recommended in patients with moderate hepatic impairment to be administered intramuscularly into the buttock (gluteal area) slowly (1 - 2 minutes) as one 5 mL injection on Days 1, 15, 29, and once monthly thereafter. (2.2, 5.2, 8.6)
5.3 Injection Site Reaction
Injection site related events including sciatica, neuralgia, neuropathic pain, and peripheral neuropathy have been reported with Fulvestrant Injection. Caution should be taken while administering Fulvestrant Injection at the dorsogluteal injection site due to the proximity of the underlying sciatic nerve [see Dosage and Administration (2.3) and Adverse Reactions (6.1)].
2.3 Administration Technique
Administer the injection according to the local guidelines for performing large volume intramuscular injections.
NOTE: Due to the proximity of the underlying sciatic nerve, caution should be taken if administering Fulvestrant Injection at the dorsogluteal injection site [see Warnings and Precautions (5.3) and Adverse Reactions (6.1)].
The proper method of administration of Fulvestrant Injection for intramuscular use is described in the following instructions.
For each single-dose prefilled syringe:
- Remove glass syringe barrel from tray and check that it is not damaged.
- Remove perforated patient record label from syringe.
- Inspect drug product in glass syringe for any visible particulate matter or discoloration prior to use. Discard if particulate matter or discoloration is present.
- Peel open the safety needle (SafetyGlide™) outer packaging.
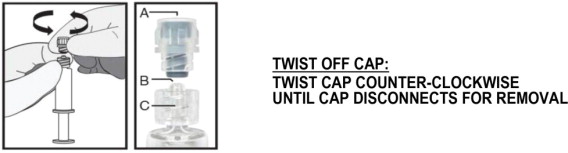
- Hold the syringe upright on the ribbed part (C). With the other hand, take hold of the cap (A) and carefully TWIST THE CAP in a counter-clockwise direction until the cap disconnects for removal (see Figure 1).
Figure 1
- Pull the cap (A) off in a straight upward direction. DO NOT TOUCH THE STERILE SYRINGE TIP (Luer-Lok) (B) (see Figure 2).
Figure 2
- Attach the safety needle to the syringe tip (Luer-Lok). Twist needle until firmly seated (see Figure 3). Confirm that the needle is locked to the Luer connector before moving or tilting the syringe out of the vertical plane to avoid spillage of syringe contents.
Figure 3
For Administration:
- Pull shield straight off needle to avoid damaging needle point.
- Remove needle sheath.
- Expel excess gas from the syringe (a small gas bubble may remain).
- Administer intramuscularly slowly (1-2 minutes/injection) into the buttock (gluteal area). For user convenience, the needle ‘bevel up’ position is orientated to the lever arm, as shown in Figure 4.
Figure 4
- After injection, immediately activate the lever arm to deploy the needle shielding by applying a single-finger stroke to the activation assisted lever arm to push the lever arm completely forward. Listen for a click. Confirm that the needle shielding has completely covered the needle (see Figure 5). NOTE: Activate away from self and others.
Figure 5
- Discard the empty syringe into an approved sharps collector in accordance with applicable regulations and institutional policy.
- Repeat steps 1 through 13 for second syringe.
How To Use Fulvestrant Injection
For the 2 x 5 mL syringe package, the contents of both syringes must be injected to receive the 500 mg recommended dose.
SAFETYGLIDE ™ INSTRUCTIONS FROM BECTON DICKINSON
SafetyGlide™ is a trademark of Becton Dickinson and Company.
Important Administration Information
To help avoid HIV (AIDS), HBV (Hepatitis), and other infectious diseases due to accidental needlesticks, contaminated needles should not be recapped or removed, unless there is no alternative or that such action is required by a specific medical procedure. Hands must remain behind the needle at all times during use and disposal.
Do not autoclave SafetyGlide™ Needle before use.
Becton Dickinson guarantees the contents of their unopened or undamaged packages to be sterile, non-toxic and non-pyrogenic.
3 Dosage Forms and Strengths
Fulvestrant Injection, an injection for intramuscular administration, is supplied as 5-mL single-dose prefilled syringes containing 250 mg per 5 mL fulvestrant.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Fulvestrant Injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
For Fulvestrant Injection 250 mg, other adverse reactions reported as drug-related and seen infrequently (<1%) include thromboembolic phenomena, myalgia, vertigo, leukopenia, and hypersensitivity reactions including angioedema and urticaria.
Vaginal bleeding has been reported infrequently (<1%), mainly in patients during the first 6 weeks after changing from existing hormonal therapy to treatment with Fulvestrant Injection. If bleeding persists, further evaluation should be considered.
Elevation of bilirubin, elevation of gamma GT, hepatitis, and liver failure have been reported infrequently (<1%).
8 Use in Specific Populations
- Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other trials and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
Fulvestrant Injection is supplied as two 5 mL clear neutral glass (Type 1) barrels, each containing a sterile, clear, colorless to yellow viscous liquid for intramuscular injection and fitted with a plastic rigid tip cap. It is supplied as follows:
| NDC | Fulvestrant Injection, USP (50 mg per mL) | Package Factor |
| 71288-555-86 | 250 mg per 5 mL Single-Dose Prefilled Syringe | 2 syringes per carton |
The 2 x 5 mL single-dose prefilled syringes are presented in a tray with polystyrene plunger rod and 21G x 1-1/2 inch safety needles (SafetyGlide™) for connection to the barrel.
Discard each syringe after use. If a patient dose requires only one syringe, unused syringe should be stored as directed below.
5.5 Immunoassay Measurement of Serum Estradiol
Due to structural similarity of fulvestrant and estradiol, Fulvestrant Injection can interfere with estradiol measurement by immunoassay, resulting in falsely elevated estradiol levels.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenesis studies were conducted in rats and mice. Positive findings were observed in both species. Rats were treated at intramuscular doses of 15 mg/kg/30 days, 10 mg/rat/30 days and 10 mg/rat/15 days.
These doses correspond to 0.9-, 1.5-, and 3-fold (in females) and 0.8-, 0.8-, and 2-fold (in males) the systemic exposure [AUC0-30 days] achieved in women receiving the recommended dose of 500 mg/month. An increased incidence of benign ovarian granulosa cell tumors and testicular Leydig cell tumors was evident, in females dosed at 10 mg/rat/15 days and males dosed at 15 mg/rat/30 days, respectively. Mice were treated at oral doses of 0, 20, 150 and 500 mg/kg/day. These doses correspond to 0-, 0.8-, 8.4- and 18-fold (in females) and 0.8-, 7.1- and 11.9-fold (in males), the systemic exposure (AUC0-30 days) achieved in women receiving the recommended dose of 500 mg/month. There was an increased incidence of sex cord stromal tumors (both benign and malignant) in the ovary of mice at doses of 150 and 500 mg/kg/day. Induction of such tumors is consistent with the pharmacology-related endocrine feedback alterations in gonadotropin levels caused by an antiestrogen.
Fulvestrant was not mutagenic or clastogenic in multiple in vitro tests with and without the addition of a mammalian liver metabolic activation factor (bacterial mutation assay in strains of Salmonella typhimurium and Escherichia coli, in vitro cytogenetics study in human lymphocytes, mammalian cell mutation assay in mouse lymphoma cells and in vivo micronucleus test in rat).
In female rats, fulvestrant administered at doses ≥0.01 mg/kg/day (0.6% the human recommended dose based on body surface area [BSA in mg/m2]), for 2 weeks prior to and for 1 week following mating, caused a reduction in fertility and embryonic survival. No adverse effects on female fertility and embryonic survival were evident in female animals dosed at 0.001 mg/kg/day (0.06% the human dose based on BSA in mg/m2). Restoration of female fertility to values similar to controls was evident following a 29-day withdrawal period after dosing at 2 mg/kg/day (equivalent to the human dose based on BSA in mg/m2). The effects of fulvestrant on the fertility of female rats appear to be consistent with its antiestrogenic activity. The potential effects of fulvestrant on the fertility of male animals were not studied, but in a 6-month toxicology study, male rats treated with intramuscular doses of 15 mg/kg/30 days, 10 mg/rat/30 days, or 10 mg/rat/15 days fulvestrant showed a loss of spermatozoa from the seminiferous tubules, seminiferous tubular atrophy, and degenerative changes in the epididymides. Changes in the testes and epididymides had not recovered 20 weeks after cessation of dosing. These fulvestrant doses correspond to 1.3-, 1.2-, and 3.5-fold the systemic exposure [AUC0-30 days] achieved in women receiving the recommended dose of 500 mg/month.
5.2 Increased Exposure in Patients With Hepatic Impairment
The safety and pharmacokinetics of Fulvestrant Injection were evaluated in a study in seven subjects with moderate hepatic impairment (Child-Pugh class B) and seven subjects with normal hepatic function. Exposure was increased in patients with moderate hepatic impairment, therefore a dose of 250 mg is recommended [see Dosage and Administration (2.2)].
Fulvestrant Injection has not been studied in patients with severe hepatic impairment (Child-Pugh class C) [see Use in Specific Populations (8.6)].
Structured Label Content
Section 42229-5 (42229-5)
Monotherapy
Fulvestrant Injection is indicated for the treatment of:
- Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy, or
- HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy.
Section 42230-3 (42230-3)
|
PATIENT INFORMATION
FULVESTRANT (ful VES trant) INJECTION |
|
|
What is Fulvestrant Injection?
Fulvestrant Injection is a prescription medicine used to treat advanced breast cancer or breast cancer that has spread to other parts of the body (metastatic). Fulvestrant Injection may be used alone, if you have gone through menopause, and your advanced breast cancer is:
|
|
| Fulvestrant Injection may be used in combination with ribociclib, if you have gone through menopause, and your advanced or metastatic breast cancer is HR-positive and HER2-negative, and has not been previously treated with endocrine therapy or has progressed after endocrine therapy. Fulvestrant Injection may be used in combination with palbociclib or abemaciclib if your advanced or metastatic breast cancer is HR-positive and HER2-negative, and has progressed after endocrine therapy. When Fulvestrant Injection is used in combination with palbociclib, abemaciclib, or ribociclib, also read the Patient Information for the prescribed product. It is not known if Fulvestrant Injection is safe and effective in children. It is not known if Fulvestrant Injection is safe and effective in people with severe liver problems. |
|
|
Who should not receive Fulvestrant Injection?
Do not receive Fulvestrant Injection if you have had an allergic reaction to fulvestrant or any of the ingredients in Fulvestrant Injection. See the end of this leaflet for a list of the ingredients in Fulvestrant Injection. Symptoms of an allergic reaction to Fulvestrant Injection may include: • itching or hives • swelling of your face, lips, tongue or throat • trouble breathing |
|
|
What should I tell my healthcare provider before receiving Fulvestrant Injection?
Before receiving Fulvestrant Injection, tell your healthcare provider about all of your medical conditions, including if you:
|
|
|
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Fulvestrant Injection may affect the way other medicines work, and other medicines may affect how Fulvestrant Injection works. Especially tell your healthcare provider if you take a blood thinner medicine. |
|
How will I receive Fulvestrant Injection?
|
|
|
What are the possible side effects of Fulvestrant Injection?
Fulvestrant Injection may cause serious side effects, including:
|
|
|
|
| Fulvestrant Injection may cause fertility problems in males and females. Talk to your healthcare provider if you plan to become pregnant. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects with Fulvestrant Injection. For more information, ask your healthcare provider or pharmacist. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
General information about the safe and effective use of Fulvestrant Injection
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about Fulvestrant Injection that is written for health professionals. |
|
|
What are the ingredients in Fulvestrant Injection?
Active ingredient: fulvestrant Inactive ingredients: alcohol, benzyl alcohol, benzyl benzoate, and castor oil. |
This Patient Information has been approved by the U.S. Food and Drug Administration.
Brands listed are the trademarks of their respective owners.
Mfd. for Meitheal Pharmaceuticals
Chicago, IL 60631 (USA)
©2021 Meitheal Pharmaceuticals Inc.
Mfd. by Nanjing King-Friend Biochemical Pharmaceutical Co., Ltd.
Nanjing, China 210061
Revised: February 2021
810038-02
Section 44425-7 (44425-7)
Storage
REFRIGERATE, 2° to 8°C (36° to 46°F). TO PROTECT FROM LIGHT, STORE IN THE ORIGINAL CARTON UNTIL TIME OF USE.
Sterile, Nonpyrogenic, PVC-free, DEHP-free.
Section 51945-4 (51945-4)
Principal Display Panel – Fulvestrant Injection, 250 mg per 5 mL (50mg per ml) Syringe Label
NDC 71288-555-85
Rx only
Fulvestrant Injection
250 mg per 5 mL
(50 mg per mL)
Date/Site:
Administered by:
For Intramuscular Use Only
PROTECT FROM LIGHT
Both single-dose prefilled syringes must be administered to receive the 500 mg dose.
10 Overdosage (10 OVERDOSAGE)
Human experience of overdose with Fulvestrant Injection is limited. There are isolated reports of overdose with Fulvestrant Injection in humans. No adverse reactions were seen in healthy male and female volunteers who received intravenous fulvestrant, which resulted in peak plasma concentrations at the end of the infusion, that were approximately 10 to 15 times those seen after intramuscular injection. The potential toxicity of fulvestrant at these or higher concentrations in cancer patients who may have additional comorbidities is unknown. There is no specific treatment in the event of fulvestrant overdose, and symptoms of overdose are not established. In the event of an overdose, healthcare practitioners should follow general supportive measures and should treat symptomatically.
11 Description (11 DESCRIPTION)
Fulvestrant Injection for intramuscular administration is an estrogen receptor antagonist. The chemical name is 7-alpha-[9-(4,4,5,5,5-penta fluoropentylsulphinyl) nonyl]estra-1,3,5-(10)-triene-3,17-beta-diol. The molecular formula is C32H47F5O3S and its structural formula is:
Fulvestrant is a white powder with a molecular weight of 606.77. The solution for injection is a clear, colorless to yellow, viscous liquid.
Each injection contains as inactive ingredients: 10% w/v (12% v/v) Alcohol, USP, 10% w/v Benzyl Alcohol, NF, and 15% w/v Benzyl Benzoate, USP, as co-solvents, and made up to 100% w/v with Castor Oil, USP as a co-solvent and release rate modifier.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. A multi-center, single-arm, open-label, study of fulvestrant was conducted in 30 girls with McCune-Albright Syndrome (MAS) associated with Progressive Precocious Puberty (PPP). The median age at informed consent was 6 years old (range: 1 to 8).
The first 10 patients initially received fulvestrant 2 mg/kg. Based on PK data from the first 6 patients, all 10 patients receiving 2 mg/kg were escalated to a dose of 4 mg/kg and all other patients received 4 mg/kg from study entry.
Baseline measurements for vaginal bleeding days, bone age, growth velocity, and Tanner staging for at least 6 months prior to study entry were provided retrospectively by the parent, guardian or local consultant. All measurements during the study period were collected prospectively. Patients' baseline characteristics included the following: a mean ± SD chronological age of 5.9 ± 1.8 years; a mean rate of bone age advancement (change in bone age in years divided by change in chronological age in years) of 2.0 ± 1.03; and a mean growth velocity z-score of 2.4 ± 3.26.
Twenty-nine of 30 patients completed the 12-month study period. The following results were observed: 35% (95% CI: 16%, 57%) of the 23 patients with baseline vaginal bleeding experienced a complete cessation of vaginal bleeding on-treatment (month 0 to 12); a reduction in the rate of bone age advancement during the 12-month study period compared to baseline (mean change=-0.9 [95% CI: -1.4, -0.4]); and a reduction in mean growth velocity Z-score on-treatment compared to baseline (mean change=-1.1 [95% CI: -2.7, 0.4]). There were no clinically meaningful changes in median Tanner stage (breast or pubic), mean uterine volume, or mean ovarian volume, or predicted adult height (PAH) on-treatment compared to baseline. The effect of Fulvestrant Injection on bone mineral density in children has not been studied and is not known.
Eight patients (27%) experienced adverse reactions that were considered possibly related to Fulvestrant Injection. These included injection site reactions (inflammation, pain, hematoma, pruritus, rash), abdominal pain, contusion, tachycardia, hot flash, extremity pain, and vomiting. Nine (30%) patients reported an SAE, none of which were considered related to Fulvestrant Injection. No patients discontinued study treatment due to an AE and no patients died.
8.5 Geriatric Use
For Fulvestrant Injection 250 mg, when tumor response was considered by age, objective responses were seen in 22% and 24% of patients under 65 years of age and in 11% and 16% of patients 65 years of age and older, who were treated with Fulvestrant Injection in Study 0021 and Study 0020, respectively.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of Fulvestrant Injection 500 mg versus Fulvestrant Injection 250 mg was compared in CONFIRM. The efficacy of Fulvestrant Injection 250 mg was compared to 1 mg anastrozole in Studies 0020 and 0021. The efficacy of Fulvestrant Injection 500 mg was compared to 1 mg anastrozole in FALCON. The efficacy of Fulvestrant Injection 500 mg in combination with palbociclib 125 mg was compared to Fulvestrant Injection 500 mg plus placebo in PALOMA-3. The efficacy of Fulvestrant Injection 500 mg in combination with abemaciclib 150 mg was compared to Fulvestrant Injection 500 mg plus placebo in MONARCH 2. The efficacy of Fulvestrant Injection 500 mg in combination with ribociclib 600 mg was compared to Fulvestrant Injection 500 mg plus placebo in MONALEESA-3.
4 Contraindications (4 CONTRAINDICATIONS)
Fulvestrant Injection is contraindicated in patients with a known hypersensitivity to the drug or to any of its components. Hypersensitivity reactions, including urticaria and angioedema, have been reported in association with Fulvestrant Injection [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in more detail in other sections of the labeling:
7 Drug Interactions (7 DRUG INTERACTIONS)
There are no known drug-drug interactions. Although, fulvestrant is metabolized by CYP 3A4 in vitro, drug interactions studies with ketoconazole or rifampin did not alter fulvestrant pharmacokinetics. Dose adjustment is not needed in patients co-prescribed CYP 3A4 inhibitors or inducers [see Clinical Pharmacology (12.3)].
5.1 Risk of Bleeding
Because Fulvestrant Injection is administered intramuscularly, it should be used with caution in patients with bleeding diatheses, thrombocytopenia, or anticoagulant use.
8.7 Renal Impairment
Negligible amounts of fulvestrant are eliminated in urine; therefore, a study in patients with renal impairment was not conducted. In the advanced breast cancer trials, fulvestrant concentrations in women with estimated creatinine clearance as low as 30 mL/min were similar to women with normal creatinine.
12.2 Pharmacodynamics
In a clinical study in postmenopausal women with primary breast cancer treated with single doses of Fulvestrant Injection 15-22 days prior to surgery, there was evidence of increasing down-regulation of ER with increasing dose. This was associated with a dose-related decrease in the expression of the progesterone receptor, an estrogen-regulated protein. These effects on the ER pathway were also associated with a decrease in Ki67 labeling index, a marker of cell proliferation.
8.6 Hepatic Impairment
Fulvestrant Injection is metabolized primarily in the liver.
The pharmacokinetics of fulvestrant were evaluated after a single dose of 100 mg in subjects with mild and moderate hepatic impairment and normal hepatic function (n=7 subjects/group), using a shorter-acting intramuscular injection formulation. Subjects with mild hepatic impairment (Child-Pugh class A) had comparable mean AUC and clearance values to those with normal hepatic function. In subjects with moderate hepatic impairment (Child-Pugh class B), the average AUC of fulvestrant increased by 70% compared to patients with normal hepatic function. AUC was positively correlated with total bilirubin concentration (p=0.012). Fulvestrant Injection has not been studied in patients with severe hepatic impairment (Child-Pugh class C).
A dose of Fulvestrant Injection 250 mg is recommended in patients with moderate hepatic impairment (Child-Pugh class B) [see Dosage and Administration (2.2) and Warnings and Precautions (5.2)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Fulvestrant Injection is an estrogen receptor antagonist indicated for the treatment of:
- Hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced breast cancer in postmenopausal women not previously treated with endocrine therapy. (1)
- HR-positive advanced breast cancer in postmenopausal women with disease progression following endocrine therapy. (1)
- HR-positive, HER2-negative advanced or metastatic breast cancer in postmenopausal women in combination with ribociclib, as initial endocrine based therapy or following disease progression on endocrine therapy. (1)
- HR-positive, HER2-negative advanced or metastatic breast cancer in combination with palbociclib or abemaciclib in women with disease progression after endocrine therapy. (1)
12.1 Mechanism of Action
Many breast cancers have estrogen receptors (ER) and the growth of these tumors can be stimulated by estrogen. Fulvestrant is an estrogen receptor antagonist that binds to the estrogen receptor in a competitive manner with affinity comparable to that of estradiol and downregulates the ER protein in human breast cancer cells.
In vitro studies demonstrated that fulvestrant is a reversible inhibitor of the growth of tamoxifen-resistant, as well as estrogen-sensitive human breast cancer (MCF-7) cell lines. In in vivo tumor studies, fulvestrant delayed the establishment of tumors from xenografts of human breast cancer MCF-7 cells in nude mice. Fulvestrant inhibited the growth of established MCF-7 xenografts and of tamoxifen-resistant breast tumor xenografts.
Fulvestrant showed no agonist-type effects in in vivo uterotrophic assays in immature or ovariectomized mice and rats. In in vivo studies in immature rats and ovariectomized monkeys, fulvestrant blocked the uterotrophic action of estradiol. In postmenopausal women, the absence of changes in plasma concentrations of FSH and LH in response to fulvestrant treatment (250 mg monthly) suggests no peripheral steroidal effects.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, Fulvestrant Injection can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of fulvestrant to pregnant rats and rabbits during organogenesis resulted in embryo-fetal toxicity at daily doses that are significantly less than the maximum recommended human dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with Fulvestrant Injection and for one year after the last dose [see Use in Specific Populations (8.1), (8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risk of Bleeding: Use with caution in patients with bleeding diatheses, thrombocytopenia, or anticoagulant use. (5.1)
- Increased Exposure in Patients with Hepatic Impairment: Use a 250 mg dose for patients with moderate hepatic impairment. (2.2, 5.2, 8.6)
- Injection Site Reaction: Use caution while administering Fulvestrant Injection at the dorsogluteal injection site due to the proximity of the underlying sciatic nerve. (5.3)
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.4, 8.1, 8.3)
- Immunoassay Measurement of Serum Estradiol: Fulvestrant Injection can interfere with estradiol measurement by immunoassay, resulting in falsely elevated estradiol levels. (5.5)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Fulvestrant Injection 500 mg should be administered intramuscularly into the buttocks (gluteal area) slowly (1 - 2 minutes per injection) as two 5 mL injections, one in each buttock, on Days 1, 15, 29, and once monthly thereafter. (2.1, 14)
- A dose of 250 mg is recommended in patients with moderate hepatic impairment to be administered intramuscularly into the buttock (gluteal area) slowly (1 - 2 minutes) as one 5 mL injection on Days 1, 15, 29, and once monthly thereafter. (2.2, 5.2, 8.6)
5.3 Injection Site Reaction
Injection site related events including sciatica, neuralgia, neuropathic pain, and peripheral neuropathy have been reported with Fulvestrant Injection. Caution should be taken while administering Fulvestrant Injection at the dorsogluteal injection site due to the proximity of the underlying sciatic nerve [see Dosage and Administration (2.3) and Adverse Reactions (6.1)].
2.3 Administration Technique
Administer the injection according to the local guidelines for performing large volume intramuscular injections.
NOTE: Due to the proximity of the underlying sciatic nerve, caution should be taken if administering Fulvestrant Injection at the dorsogluteal injection site [see Warnings and Precautions (5.3) and Adverse Reactions (6.1)].
The proper method of administration of Fulvestrant Injection for intramuscular use is described in the following instructions.
For each single-dose prefilled syringe:
- Remove glass syringe barrel from tray and check that it is not damaged.
- Remove perforated patient record label from syringe.
- Inspect drug product in glass syringe for any visible particulate matter or discoloration prior to use. Discard if particulate matter or discoloration is present.
- Peel open the safety needle (SafetyGlide™) outer packaging.
- Hold the syringe upright on the ribbed part (C). With the other hand, take hold of the cap (A) and carefully TWIST THE CAP in a counter-clockwise direction until the cap disconnects for removal (see Figure 1).
Figure 1
- Pull the cap (A) off in a straight upward direction. DO NOT TOUCH THE STERILE SYRINGE TIP (Luer-Lok) (B) (see Figure 2).
Figure 2
- Attach the safety needle to the syringe tip (Luer-Lok). Twist needle until firmly seated (see Figure 3). Confirm that the needle is locked to the Luer connector before moving or tilting the syringe out of the vertical plane to avoid spillage of syringe contents.
Figure 3
For Administration:
- Pull shield straight off needle to avoid damaging needle point.
- Remove needle sheath.
- Expel excess gas from the syringe (a small gas bubble may remain).
- Administer intramuscularly slowly (1-2 minutes/injection) into the buttock (gluteal area). For user convenience, the needle ‘bevel up’ position is orientated to the lever arm, as shown in Figure 4.
Figure 4
- After injection, immediately activate the lever arm to deploy the needle shielding by applying a single-finger stroke to the activation assisted lever arm to push the lever arm completely forward. Listen for a click. Confirm that the needle shielding has completely covered the needle (see Figure 5). NOTE: Activate away from self and others.
Figure 5
- Discard the empty syringe into an approved sharps collector in accordance with applicable regulations and institutional policy.
- Repeat steps 1 through 13 for second syringe.
How To Use Fulvestrant Injection
For the 2 x 5 mL syringe package, the contents of both syringes must be injected to receive the 500 mg recommended dose.
SAFETYGLIDE ™ INSTRUCTIONS FROM BECTON DICKINSON
SafetyGlide™ is a trademark of Becton Dickinson and Company.
Important Administration Information
To help avoid HIV (AIDS), HBV (Hepatitis), and other infectious diseases due to accidental needlesticks, contaminated needles should not be recapped or removed, unless there is no alternative or that such action is required by a specific medical procedure. Hands must remain behind the needle at all times during use and disposal.
Do not autoclave SafetyGlide™ Needle before use.
Becton Dickinson guarantees the contents of their unopened or undamaged packages to be sterile, non-toxic and non-pyrogenic.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Fulvestrant Injection, an injection for intramuscular administration, is supplied as 5-mL single-dose prefilled syringes containing 250 mg per 5 mL fulvestrant.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Fulvestrant Injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
For Fulvestrant Injection 250 mg, other adverse reactions reported as drug-related and seen infrequently (<1%) include thromboembolic phenomena, myalgia, vertigo, leukopenia, and hypersensitivity reactions including angioedema and urticaria.
Vaginal bleeding has been reported infrequently (<1%), mainly in patients during the first 6 weeks after changing from existing hormonal therapy to treatment with Fulvestrant Injection. If bleeding persists, further evaluation should be considered.
Elevation of bilirubin, elevation of gamma GT, hepatitis, and liver failure have been reported infrequently (<1%).
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other trials and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Fulvestrant Injection is supplied as two 5 mL clear neutral glass (Type 1) barrels, each containing a sterile, clear, colorless to yellow viscous liquid for intramuscular injection and fitted with a plastic rigid tip cap. It is supplied as follows:
| NDC | Fulvestrant Injection, USP (50 mg per mL) | Package Factor |
| 71288-555-86 | 250 mg per 5 mL Single-Dose Prefilled Syringe | 2 syringes per carton |
The 2 x 5 mL single-dose prefilled syringes are presented in a tray with polystyrene plunger rod and 21G x 1-1/2 inch safety needles (SafetyGlide™) for connection to the barrel.
Discard each syringe after use. If a patient dose requires only one syringe, unused syringe should be stored as directed below.
5.5 Immunoassay Measurement of Serum Estradiol
Due to structural similarity of fulvestrant and estradiol, Fulvestrant Injection can interfere with estradiol measurement by immunoassay, resulting in falsely elevated estradiol levels.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Two-year carcinogenesis studies were conducted in rats and mice. Positive findings were observed in both species. Rats were treated at intramuscular doses of 15 mg/kg/30 days, 10 mg/rat/30 days and 10 mg/rat/15 days.
These doses correspond to 0.9-, 1.5-, and 3-fold (in females) and 0.8-, 0.8-, and 2-fold (in males) the systemic exposure [AUC0-30 days] achieved in women receiving the recommended dose of 500 mg/month. An increased incidence of benign ovarian granulosa cell tumors and testicular Leydig cell tumors was evident, in females dosed at 10 mg/rat/15 days and males dosed at 15 mg/rat/30 days, respectively. Mice were treated at oral doses of 0, 20, 150 and 500 mg/kg/day. These doses correspond to 0-, 0.8-, 8.4- and 18-fold (in females) and 0.8-, 7.1- and 11.9-fold (in males), the systemic exposure (AUC0-30 days) achieved in women receiving the recommended dose of 500 mg/month. There was an increased incidence of sex cord stromal tumors (both benign and malignant) in the ovary of mice at doses of 150 and 500 mg/kg/day. Induction of such tumors is consistent with the pharmacology-related endocrine feedback alterations in gonadotropin levels caused by an antiestrogen.
Fulvestrant was not mutagenic or clastogenic in multiple in vitro tests with and without the addition of a mammalian liver metabolic activation factor (bacterial mutation assay in strains of Salmonella typhimurium and Escherichia coli, in vitro cytogenetics study in human lymphocytes, mammalian cell mutation assay in mouse lymphoma cells and in vivo micronucleus test in rat).
In female rats, fulvestrant administered at doses ≥0.01 mg/kg/day (0.6% the human recommended dose based on body surface area [BSA in mg/m2]), for 2 weeks prior to and for 1 week following mating, caused a reduction in fertility and embryonic survival. No adverse effects on female fertility and embryonic survival were evident in female animals dosed at 0.001 mg/kg/day (0.06% the human dose based on BSA in mg/m2). Restoration of female fertility to values similar to controls was evident following a 29-day withdrawal period after dosing at 2 mg/kg/day (equivalent to the human dose based on BSA in mg/m2). The effects of fulvestrant on the fertility of female rats appear to be consistent with its antiestrogenic activity. The potential effects of fulvestrant on the fertility of male animals were not studied, but in a 6-month toxicology study, male rats treated with intramuscular doses of 15 mg/kg/30 days, 10 mg/rat/30 days, or 10 mg/rat/15 days fulvestrant showed a loss of spermatozoa from the seminiferous tubules, seminiferous tubular atrophy, and degenerative changes in the epididymides. Changes in the testes and epididymides had not recovered 20 weeks after cessation of dosing. These fulvestrant doses correspond to 1.3-, 1.2-, and 3.5-fold the systemic exposure [AUC0-30 days] achieved in women receiving the recommended dose of 500 mg/month.
5.2 Increased Exposure in Patients With Hepatic Impairment (5.2 Increased Exposure in Patients with Hepatic Impairment)
The safety and pharmacokinetics of Fulvestrant Injection were evaluated in a study in seven subjects with moderate hepatic impairment (Child-Pugh class B) and seven subjects with normal hepatic function. Exposure was increased in patients with moderate hepatic impairment, therefore a dose of 250 mg is recommended [see Dosage and Administration (2.2)].
Fulvestrant Injection has not been studied in patients with severe hepatic impairment (Child-Pugh class C) [see Use in Specific Populations (8.6)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:39.864559 · Updated: 2026-03-14T22:06:28.970908