Drug Facts
014a5f36-0396-498b-901f-9a86df4e11fe
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Chlorhexidine gluconate solution, 0.75%
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Chlorhexidine gluconate solution, 0.75%
Use
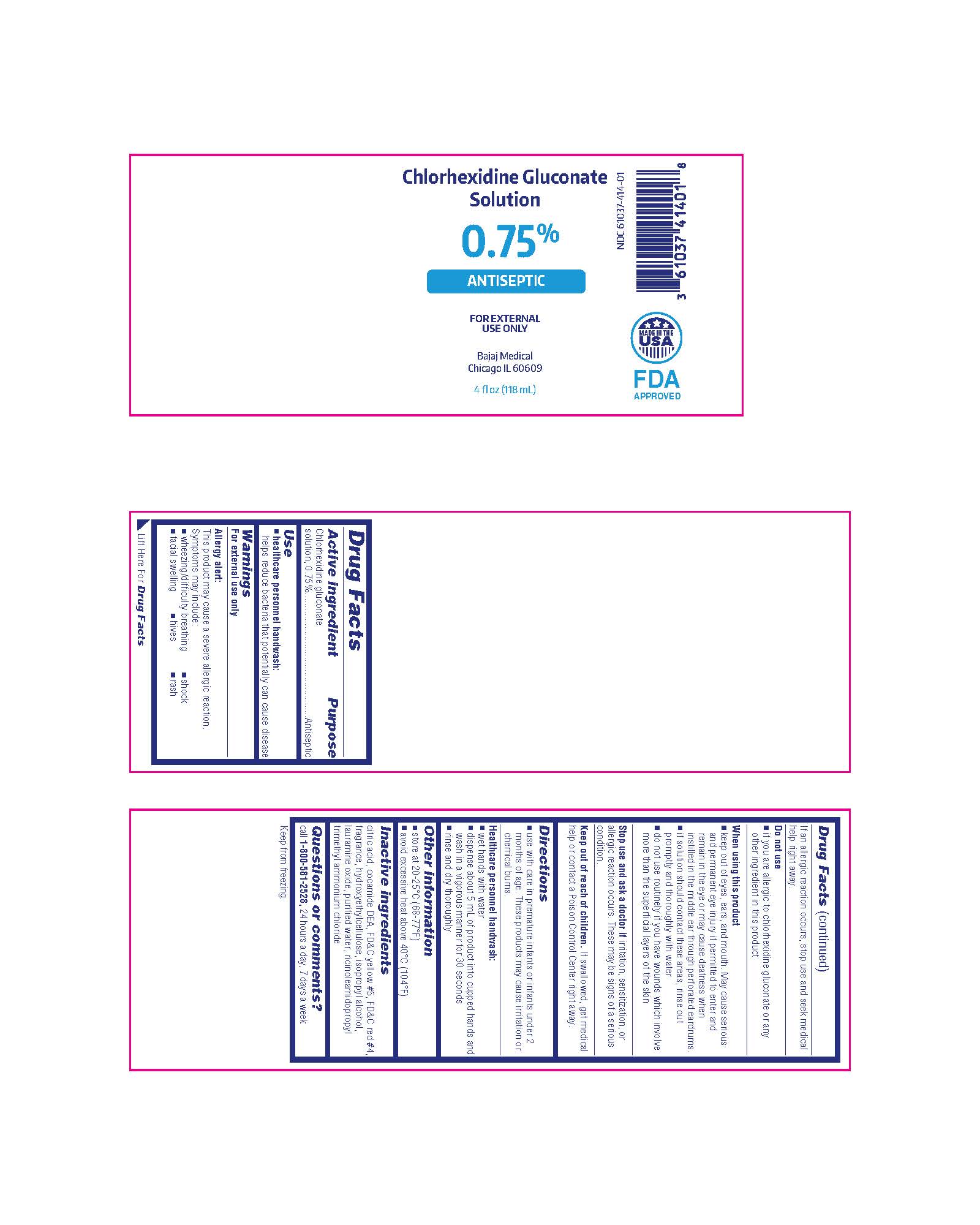
- healthcare personnel handwash: helps reduce bacteria that potentially can cause disease
Section 44425-7
Keep from freezing.
Warnings
For external use only
Allergy alert:
This product may cause a severe allergic reaction.
Symptoms may include:
- wheezing/difficulty breathing
- shock
- facial swelling
- hives
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
Directions
- use with care in premature infants and infants under 2 months of age. These products may cause irritation or chemical burns.
Do Not Use
- if you are allergic to chlorhexidine gluconate or any other ingredient in this product
Active Ingredient
Chlorhexidine gluconate solution, 0.75%
Other Information
- store at 20-25°C (68-77°F)
- avoid excessive heat above 40°C (104°F)
Inactive Ingredients
citric acid, cocamide DEA, FD&C yellow #5, FD&C red #4, fragrance, hydroxyethylcellulose, isopropyl alcohol, lauramine oxide, purified water, ricinoleamidopropyl trimethyl ammonium chloride
Questions Or Comments?
call 1-800-581-2528, 24 hours a day, 7 days a week
When Using This Product
- keep out of eyes, ears, and mouth. May cause serious and permanent eye injury if permitted to enter and remain in the eye or may cause deafness when instilled in the middle ear through perforated eardrums.
- if solution should contact these areas, rinse out promptly and thoroughly with water
- do not use routinely if you have wounds which involve more than the superficial layers of the skin
Stop Use and Ask A Doctor If
irritation, sensitization, or allergic reaction occurs. These may be signs of a serious condition.
Healthcare Personnel Handwash:
- wet hands with water
- dispense about 5 mL of product into cupped hands and wash in a vigorous manner for 30 seconds
- rinse and dry thoroughly
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
4 fl oz (118 mL)
NDC 61037-414-01
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
8 fl oz (236 mL)
NDC 61037-414-02
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
16 fl oz (473 mL)
NDC 61037-414-05
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
32 fl oz (946 mL)
NDC 61037-414-06
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
128 fl oz (3785 mL)
NDC 61037-414-07
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
8 fl oz (236 mL)
NDC 61037-414-03
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
8 fl oz (236 mL)
NDC 61037-414-04
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
40.6 fl oz (1.2 L)
NDC 61037-414-08
MADE IN THE USA
FDA APPROVED
Structured Label Content
Use
- healthcare personnel handwash: helps reduce bacteria that potentially can cause disease
Section 44425-7 (44425-7)
Keep from freezing.
Purpose
Antiseptic
Warnings
For external use only
Allergy alert:
This product may cause a severe allergic reaction.
Symptoms may include:
- wheezing/difficulty breathing
- shock
- facial swelling
- hives
- rash
If an allergic reaction occurs, stop use and seek medical help right away.
Directions
- use with care in premature infants and infants under 2 months of age. These products may cause irritation or chemical burns.
Do Not Use (Do not use)
- if you are allergic to chlorhexidine gluconate or any other ingredient in this product
Active Ingredient (Active ingredient)
Chlorhexidine gluconate solution, 0.75%
Other Information (Other information)
- store at 20-25°C (68-77°F)
- avoid excessive heat above 40°C (104°F)
Inactive Ingredients (Inactive ingredients)
citric acid, cocamide DEA, FD&C yellow #5, FD&C red #4, fragrance, hydroxyethylcellulose, isopropyl alcohol, lauramine oxide, purified water, ricinoleamidopropyl trimethyl ammonium chloride
Questions Or Comments? (Questions or comments?)
call 1-800-581-2528, 24 hours a day, 7 days a week
When Using This Product (When using this product)
- keep out of eyes, ears, and mouth. May cause serious and permanent eye injury if permitted to enter and remain in the eye or may cause deafness when instilled in the middle ear through perforated eardrums.
- if solution should contact these areas, rinse out promptly and thoroughly with water
- do not use routinely if you have wounds which involve more than the superficial layers of the skin
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
irritation, sensitization, or allergic reaction occurs. These may be signs of a serious condition.
Healthcare Personnel Handwash: (Healthcare personnel handwash:)
- wet hands with water
- dispense about 5 mL of product into cupped hands and wash in a vigorous manner for 30 seconds
- rinse and dry thoroughly
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel (Package/Label Principal Display Panel)
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
4 fl oz (118 mL)
NDC 61037-414-01
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
8 fl oz (236 mL)
NDC 61037-414-02
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
16 fl oz (473 mL)
NDC 61037-414-05
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
32 fl oz (946 mL)
NDC 61037-414-06
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
128 fl oz (3785 mL)
NDC 61037-414-07
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
8 fl oz (236 mL)
NDC 61037-414-03
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
8 fl oz (236 mL)
NDC 61037-414-04
MADE IN THE USA
FDA APPROVED
Chlorhexidine Gluconate Solution
0.75%
ANTISEPTIC
FOR EXTERNAL USE ONLY
Bajaj Medical
Chicago IL 60609
40.6 fl oz (1.2 L)
NDC 61037-414-08
MADE IN THE USA
FDA APPROVED
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:26.975025 · Updated: 2026-03-14T23:00:52.351984