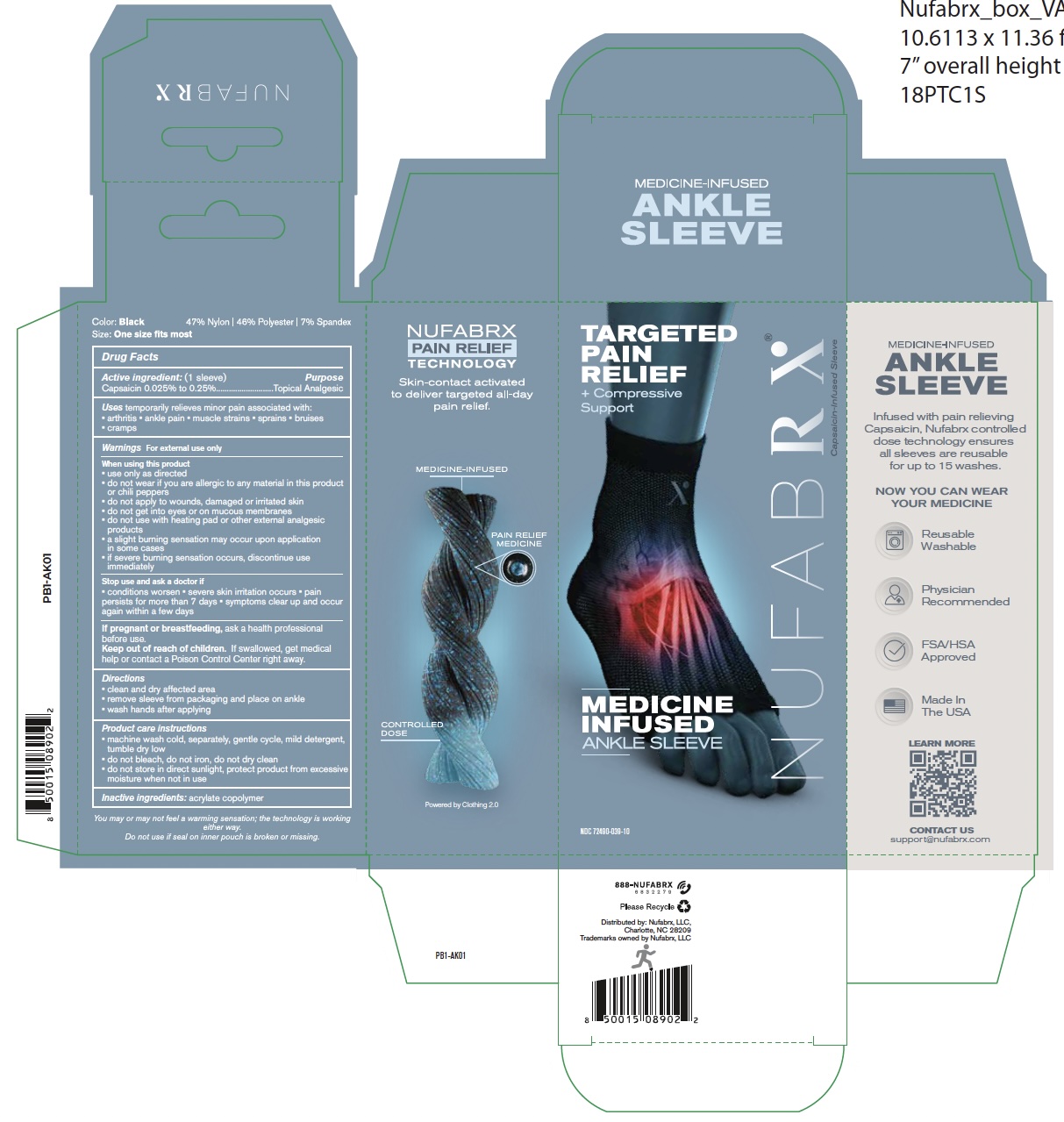
72490-039 Nufabrx Ankle Sleeve
013d60c7-a613-7cdf-e063-6294a90ad584
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Capsaicin 0.25% to 0.25%
Purpose
Topical Analgesic
Medication Information
Purpose
Topical Analgesic
Description
Capsaicin 0.25% to 0.25%
Uses
Temporary relieves minor pain associated with:
- arthritis
- ankle pain
- muscle strains
- sprains
- bruises
- cramps
Label
Warnings
For external use only.
Directions
- clean and dry affected area
- remove sleeve from packaging and place on ankle
- wash hands after applying
Active Ingredient
Capsaicin 0.25% to 0.25%
Inactive Ingredient
acrylate copolymer
When Using This Product
- use only as directed
- do not wear if you are allergic to any material in this product or chili peppers
- do not apply to wounds, damaged or irritated skin
- do not get into eyes or on mucous membranes
- do not use with heating pad or other external analgesic products
- a slight burning sensation may occur upon application in some cases
- if severe burning sensation occurs, discontinue use immediately
Product Care Instructions
- machine wash cold, separately, gentle cycle, mild detergent, tumble dry low
- do not bleach, do not iron, do not dry clean
- do not store in direct sublight, protect product from excessive moisture when not in use
Keep Out of Reach Children
If swallowed, get medical help or contact a Poison Control Center right away.
Stop Use and Ask A Doctor If
- conditions worsen
- severe skin irritation occurs
- pain persists for more than 7 days
- symptoms clear up and occur again within a few days
If Pregnant Or Breast Feeding
Ask a health professional before use.
Structured Label Content
Uses
Temporary relieves minor pain associated with:
- arthritis
- ankle pain
- muscle strains
- sprains
- bruises
- cramps
Label
Purpose
Topical Analgesic
Warnings
For external use only.
Directions
- clean and dry affected area
- remove sleeve from packaging and place on ankle
- wash hands after applying
Active Ingredient (Active ingredient)
Capsaicin 0.25% to 0.25%
Inactive Ingredient (Inactive ingredient)
acrylate copolymer
When Using This Product (When using this product)
- use only as directed
- do not wear if you are allergic to any material in this product or chili peppers
- do not apply to wounds, damaged or irritated skin
- do not get into eyes or on mucous membranes
- do not use with heating pad or other external analgesic products
- a slight burning sensation may occur upon application in some cases
- if severe burning sensation occurs, discontinue use immediately
Product Care Instructions (Product care instructions)
- machine wash cold, separately, gentle cycle, mild detergent, tumble dry low
- do not bleach, do not iron, do not dry clean
- do not store in direct sublight, protect product from excessive moisture when not in use
Keep Out of Reach Children (Keep out of reach children)
If swallowed, get medical help or contact a Poison Control Center right away.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- conditions worsen
- severe skin irritation occurs
- pain persists for more than 7 days
- symptoms clear up and occur again within a few days
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
Ask a health professional before use.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:53.086232 · Updated: 2026-03-14T23:11:49.333849