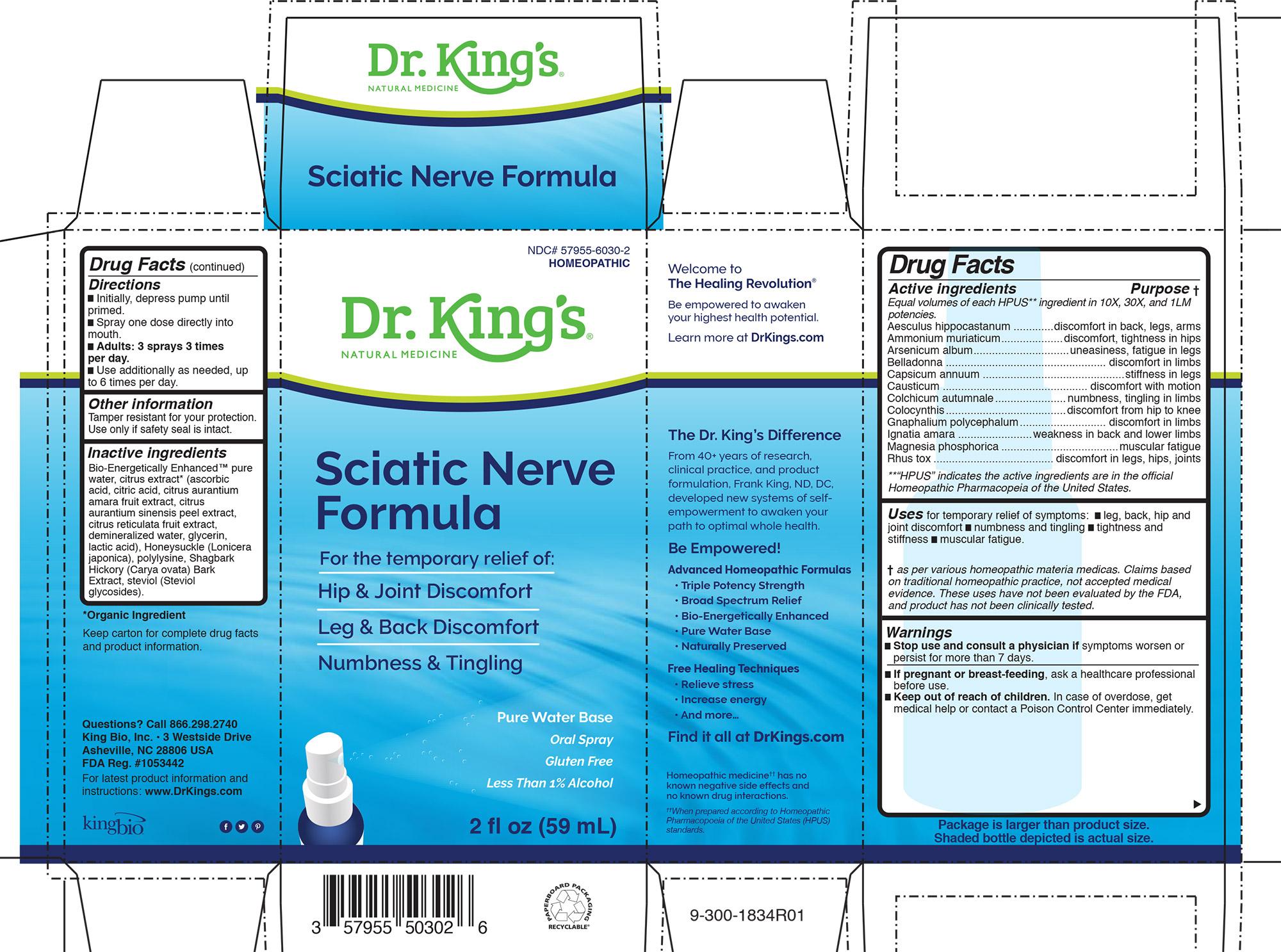
Sciatic Nerve Formula™
00734376-119f-4d32-a94b-417a52ea5612
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts ____________________________________________________________________________________________________________ Equal volumes of each ingredient in 10X, 30X, and LM1 potencies. HPUS active ingredients: Aesculus hippocastanum, Ammonium muriaticum, Arsenicum album, Belladonna, Capsicum annuum, Causticum, Colchicum autumnale, Colocynthis, Gnaphalium polycephalum, Ignatia amara, Magnesia phosphoria, Rhus toxicodendron .
Medication Information
Warnings and Precautions
Warnings
█ Stop use and consult a physician if symptoms worsen or
persist for more than 7 days
Indications and Usage
Uses for temporary relief of symptoms: █ leg, back, hip and
joint discomfort █ numbness and tingling █ tightness and
stiffness █ muscular fatigue.
Dosage and Administration
Directions:
- Initially, depress pump until primed.
- Spray one dose directly into mouth.
- Adults: 3 sprays 3 times per day.
- Use additionally as needed, up to 6 times per day.
Description
Drug Facts ____________________________________________________________________________________________________________ Equal volumes of each ingredient in 10X, 30X, and LM1 potencies. HPUS active ingredients: Aesculus hippocastanum, Ammonium muriaticum, Arsenicum album, Belladonna, Capsicum annuum, Causticum, Colchicum autumnale, Colocynthis, Gnaphalium polycephalum, Ignatia amara, Magnesia phosphoria, Rhus toxicodendron .
Section 44425-7
Other information
Tamper resistant for your protection.
Use only if safety seal is intact.
Section 50565-1
Keep out of reach of children. In case of overdose, get
medical help or contact a Poison Control Center immediately.
Section 51727-6
Inactive Ingredients
Bio-Energetically Enhanced™ pure
water, citrus extract* (ascorbic
acid, citric acid, citrus aurantium
amara fruit extract, citrus
aurantium sinensis peel extract,
citrus reticulata fruit extract,
demineralized water, glycerin,
lactic acid), Honeysuckle (Lonicera
japonica), polylysine, Shagbark
Hickory (Carya ovata) Bark
Extract, steviol (Steviol
glycosides).
Section 51945-4
Section 53413-1
Questions? Call 866.298.2740
King Bio, Inc. • 3 Westside Drive
Asheville, NC 28806 USA
FDA Reg. #1053442
For latest product information and
instructions: www.DrKings.com
Section 53414-9
If pregnant or breast-feeding, ask a healthcare professional
before use.
Section 55105-1
Drug Facts
________________________________________________
Active ingredients Purpose
Equal volumes of each HPUS** ingredient in 10X, 30X, and 1LM
potencies.
Aesculus hippocastanum .............discomfort in back, legs, arms
Ammonium muriaticum....................discomfort, tightness in hips
Arsenicum album...............................uneasiness, fatigue in legs
Belladonna .................................................... discomfort in limbs
Capsicum annuum ..............................................stiffness in legs
Causticum ............................................... discomfort with motion
Colchicum autumnale.......................numbness, tingling in limbs
Colocynthis.......................................discomfort from hip to knee
Gnaphalium polycephalum............................ discomfort in limbs
Ignatia amara ........................weakness in back and lower limbs
Magnesia phosphorica ......................................muscular fatigue
Rhus tox ....................................... discomfort in legs, hips, joints
**“HPUS” indicates the active ingredients are in the official
Homeopathic Pharmacopeia of the United States.
Section 55106-9
Drug Facts
____________________________________________________________________________________________________________
Equal volumes of each ingredient in 10X, 30X, and LM1 potencies.
HPUS active ingredients: Aesculus hippocastanum, Ammonium muriaticum, Arsenicum album, Belladonna, Capsicum annuum, Causticum, Colchicum autumnale, Colocynthis, Gnaphalium polycephalum, Ignatia amara, Magnesia phosphoria, Rhus toxicodendron .
Structured Label Content
Indications and Usage (34067-9)
Uses for temporary relief of symptoms: █ leg, back, hip and
joint discomfort █ numbness and tingling █ tightness and
stiffness █ muscular fatigue.
Dosage and Administration (34068-7)
Directions:
- Initially, depress pump until primed.
- Spray one dose directly into mouth.
- Adults: 3 sprays 3 times per day.
- Use additionally as needed, up to 6 times per day.
Warnings and Precautions (34071-1)
Warnings
█ Stop use and consult a physician if symptoms worsen or
persist for more than 7 days
Section 44425-7 (44425-7)
Other information
Tamper resistant for your protection.
Use only if safety seal is intact.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get
medical help or contact a Poison Control Center immediately.
Section 51727-6 (51727-6)
Inactive Ingredients
Bio-Energetically Enhanced™ pure
water, citrus extract* (ascorbic
acid, citric acid, citrus aurantium
amara fruit extract, citrus
aurantium sinensis peel extract,
citrus reticulata fruit extract,
demineralized water, glycerin,
lactic acid), Honeysuckle (Lonicera
japonica), polylysine, Shagbark
Hickory (Carya ovata) Bark
Extract, steviol (Steviol
glycosides).
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions? Call 866.298.2740
King Bio, Inc. • 3 Westside Drive
Asheville, NC 28806 USA
FDA Reg. #1053442
For latest product information and
instructions: www.DrKings.com
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a healthcare professional
before use.
Section 55105-1 (55105-1)
Drug Facts
________________________________________________
Active ingredients Purpose
Equal volumes of each HPUS** ingredient in 10X, 30X, and 1LM
potencies.
Aesculus hippocastanum .............discomfort in back, legs, arms
Ammonium muriaticum....................discomfort, tightness in hips
Arsenicum album...............................uneasiness, fatigue in legs
Belladonna .................................................... discomfort in limbs
Capsicum annuum ..............................................stiffness in legs
Causticum ............................................... discomfort with motion
Colchicum autumnale.......................numbness, tingling in limbs
Colocynthis.......................................discomfort from hip to knee
Gnaphalium polycephalum............................ discomfort in limbs
Ignatia amara ........................weakness in back and lower limbs
Magnesia phosphorica ......................................muscular fatigue
Rhus tox ....................................... discomfort in legs, hips, joints
**“HPUS” indicates the active ingredients are in the official
Homeopathic Pharmacopeia of the United States.
Section 55106-9 (55106-9)
Drug Facts
____________________________________________________________________________________________________________
Equal volumes of each ingredient in 10X, 30X, and LM1 potencies.
HPUS active ingredients: Aesculus hippocastanum, Ammonium muriaticum, Arsenicum album, Belladonna, Capsicum annuum, Causticum, Colchicum autumnale, Colocynthis, Gnaphalium polycephalum, Ignatia amara, Magnesia phosphoria, Rhus toxicodendron .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:55.669960 · Updated: 2026-03-14T23:14:57.671574