These Highlights Do Not Include All The Information Needed To Use Doxorubicin Hydrochloride Safely And Effectively. See Full Prescribing Information For Doxorubicin Hydrochloride.
00634b2b-4e48-4178-8877-28582af894ad
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
• Cardiomyopathy: Myocardial damage, including acute left ventricular failure, can occur with doxorubicin hydrochloride. The risk of cardiomyopathy is proportional to the cumulative exposure with incidence rates from 1%–20% for cumulative doses ranging from 300 mg/m 2 to 500 mg/m 2 when doxorubicin hydrochloride is administered every 3 weeks. The risk of cardiomyopathy is further increased with concomitant cardiotoxic therapy. Assess left ventricular ejection fraction (LVEF) before and regularly during and after treatment with doxorubicin hydrochloride [see Warnings and Precautions (5.1) ] . • Secondary Malignancies: Secondary acute myelogenous leukemia (AML) and myelodysplastic syndrome (MDS) occur at a higher incidence in patients treated with anthracyclines, including doxorubicin hydrochloride [see Warnings and Precautions (5.2) ] . • Extravasation and Tissue Necrosis: Extravasation of doxorubicin hydrochloride can result in severe local tissue injury and necrosis requiring wide excision of the affected area and skin grafting. Immediately terminate the drug and apply ice to the affected area [see Warnings and Precautions (5.3) ] . • Severe myelosuppression resulting in serious infection, septic shock, requirement for transfusions, hospitalization, and death may occur [see Warnings and Precautions (5.4) ] .
Indications and Usage
Doxorubicin Hydrochloride Injection is an anthracycline topoisomerase inhibitor indicated: • as a component of multi-agent adjuvant chemotherapy for treatment of women with axillary lymph node involvement following resection of primary breast cancer ( 1.1 ) • for the treatment of: acute lymphoblastic leukemia, acute myeloblastic leukemia, Hodgkin lymphoma, Non-Hodgkin lymphoma, metastatic breast cancer, metastatic Wilms' tumor, metastatic neuroblastoma, metastatic soft tissue sarcoma, metastatic bone sarcomas, metastatic ovarian carcinoma, metastatic transitional cell bladder carcinoma, metastatic thyroid carcinoma, metastatic gastric carcinoma, metastatic bronchogenic carcinoma ( 1.2 )
Dosage and Administration
• Single agent : 60 to 75 mg/m 2 given intravenously every 21 days ( 2.2 ) • In combination : 40 to 75 mg/m 2 given intravenously every 21 to 28 days ( 2.2 ) • Discontinue Doxorubicin Hydrochloride Injection in patients who develop signs or symptoms of cardiomyopathy ( 2.3 ) • Reduce dose in patients with hepatic impairment ( 2.4 )
Warnings and Precautions
• Radiation-Induced Toxicity : Can be increased by the administration of Doxorubicin Hydrochloride Injection. Radiation recall can occur in patients who receive Doxorubicin Hydrochloride Injection after prior radiation therapy. ( 5.7 ) • Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and on the use of effective contraception. Advise males with female partners of reproductive potential to use effective contraception. Advise males with pregnant partners to use condoms. ( 5.8 , 8.1 , 8.3 )
Contraindications
Doxorubicin Hydrochloride Injection are contraindicated in patients with: • Severe myocardial insufficiency [see Warnings and Precautions (5.1) ] • Recent (occurring within the past 4–6 weeks) myocardial infarction [see Warnings and Precautions (5.1) ] • Severe persistent drug-induced myelosuppression [see Warnings and Precautions (5.4) ] • Severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin level greater than 5 mg/dL) [see Warnings and Precautions (5.5) ] • Severe hypersensitivity reaction to doxorubicin hydrochloride, including anaphylaxis [see Adverse Reactions (6.2) ]
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling. • Cardiomyopathy and Arrhythmias [see Warnings and Precautions (5.1) ] • Secondary Malignancies [see Warnings and Precautions (5.2) ] • Extravasation and Tissue Necrosis [see Warnings and Precautions (5.3) ] • Severe Myelosuppression [see Warnings and Precautions (5.4) ] • Tumor Lysis Syndrome [see Warnings and Precautions (5.6) ] • Radiation Sensitization and Radiation Recall [see Warnings and Precautions (5.7) ]
Drug Interactions
• Avoid concomitant use of doxorubicin hydrochloride with inhibitors and inducers of CYP3A4, CYP2D6, and/or P-gp ( 7.1 ) • Do not administer doxorubicin hydrochloride in combination with trastuzumab due to increased risk of cardiac dysfunction ( 5.1 , 7.2 )
Medication Information
Warnings and Precautions
• Radiation-Induced Toxicity : Can be increased by the administration of Doxorubicin Hydrochloride Injection. Radiation recall can occur in patients who receive Doxorubicin Hydrochloride Injection after prior radiation therapy. ( 5.7 ) • Embryo-Fetal Toxicity : Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and on the use of effective contraception. Advise males with female partners of reproductive potential to use effective contraception. Advise males with pregnant partners to use condoms. ( 5.8 , 8.1 , 8.3 )
Indications and Usage
Doxorubicin Hydrochloride Injection is an anthracycline topoisomerase inhibitor indicated: • as a component of multi-agent adjuvant chemotherapy for treatment of women with axillary lymph node involvement following resection of primary breast cancer ( 1.1 ) • for the treatment of: acute lymphoblastic leukemia, acute myeloblastic leukemia, Hodgkin lymphoma, Non-Hodgkin lymphoma, metastatic breast cancer, metastatic Wilms' tumor, metastatic neuroblastoma, metastatic soft tissue sarcoma, metastatic bone sarcomas, metastatic ovarian carcinoma, metastatic transitional cell bladder carcinoma, metastatic thyroid carcinoma, metastatic gastric carcinoma, metastatic bronchogenic carcinoma ( 1.2 )
Dosage and Administration
• Single agent : 60 to 75 mg/m 2 given intravenously every 21 days ( 2.2 ) • In combination : 40 to 75 mg/m 2 given intravenously every 21 to 28 days ( 2.2 ) • Discontinue Doxorubicin Hydrochloride Injection in patients who develop signs or symptoms of cardiomyopathy ( 2.3 ) • Reduce dose in patients with hepatic impairment ( 2.4 )
Contraindications
Doxorubicin Hydrochloride Injection are contraindicated in patients with: • Severe myocardial insufficiency [see Warnings and Precautions (5.1) ] • Recent (occurring within the past 4–6 weeks) myocardial infarction [see Warnings and Precautions (5.1) ] • Severe persistent drug-induced myelosuppression [see Warnings and Precautions (5.4) ] • Severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin level greater than 5 mg/dL) [see Warnings and Precautions (5.5) ] • Severe hypersensitivity reaction to doxorubicin hydrochloride, including anaphylaxis [see Adverse Reactions (6.2) ]
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling. • Cardiomyopathy and Arrhythmias [see Warnings and Precautions (5.1) ] • Secondary Malignancies [see Warnings and Precautions (5.2) ] • Extravasation and Tissue Necrosis [see Warnings and Precautions (5.3) ] • Severe Myelosuppression [see Warnings and Precautions (5.4) ] • Tumor Lysis Syndrome [see Warnings and Precautions (5.6) ] • Radiation Sensitization and Radiation Recall [see Warnings and Precautions (5.7) ]
Drug Interactions
• Avoid concomitant use of doxorubicin hydrochloride with inhibitors and inducers of CYP3A4, CYP2D6, and/or P-gp ( 7.1 ) • Do not administer doxorubicin hydrochloride in combination with trastuzumab due to increased risk of cardiac dysfunction ( 5.1 , 7.2 )
Description
• Cardiomyopathy: Myocardial damage, including acute left ventricular failure, can occur with doxorubicin hydrochloride. The risk of cardiomyopathy is proportional to the cumulative exposure with incidence rates from 1%–20% for cumulative doses ranging from 300 mg/m 2 to 500 mg/m 2 when doxorubicin hydrochloride is administered every 3 weeks. The risk of cardiomyopathy is further increased with concomitant cardiotoxic therapy. Assess left ventricular ejection fraction (LVEF) before and regularly during and after treatment with doxorubicin hydrochloride [see Warnings and Precautions (5.1) ] . • Secondary Malignancies: Secondary acute myelogenous leukemia (AML) and myelodysplastic syndrome (MDS) occur at a higher incidence in patients treated with anthracyclines, including doxorubicin hydrochloride [see Warnings and Precautions (5.2) ] . • Extravasation and Tissue Necrosis: Extravasation of doxorubicin hydrochloride can result in severe local tissue injury and necrosis requiring wide excision of the affected area and skin grafting. Immediately terminate the drug and apply ice to the affected area [see Warnings and Precautions (5.3) ] . • Severe myelosuppression resulting in serious infection, septic shock, requirement for transfusions, hospitalization, and death may occur [see Warnings and Precautions (5.4) ] .
Section 42229-5
Cardiomyopathy
Discontinue Doxorubicin Hydrochloride Injection in patients who develop signs or symptoms of cardiomyopathy [see Warnings and Precautions (5.1)].
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 7/2024 | ||
|
Patient Information
|
||
|
What is the most important information I should know about Doxorubicin?
|
||
|
|
|
|
Your healthcare provider will do tests to check the strength of your heart muscle before, during, and after your treatment with Doxorubicin. |
||
|
||
|
What is Doxorubicin?
|
||
|
Do not receive Doxorubicin if:
|
||
|
Before you receive Doxorubicin, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
How will I receive Doxorubicin?
|
||
|
What are the possible side effects of Doxorubicin?
The most common side effects of Doxorubicin include:
Other side effects:
Doxorubicin may cause fertility problems in males. This could affect your ability to father a child. Talk to your healthcare provider if this is a concern for you.
Doxorubicin may cause fertility problems in females. Your periods (menstrual cycle) may completely stop when you receive Doxorubicin. Your periods may or may not return following treatment. Early menopause has also happened. Talk to your healthcare provider if this is a concern for you. |
||
|
General information about the safe and effective use of Doxorubicin.
|
||
|
What are the ingredients in Doxorubicin?
Pfizer Labs Division of Pfizer Inc. New York, NY 10001
LAB-0703-5.0 For more information, call 1-800-438-1985 or visit www.pfizer.com |
Section 44425-7
Storage
Store all vials at 2°C to 8°C (36°F to 46°F). Protect from light.
Storage of Doxorubicin Hydrochloride Injection under refrigerated conditions can result in the formation of a gelled product. Place gelled product at room temperature [15°C to 30°C (59°F to 86°F)] for 2 to 4 hours to return the product to a slightly viscous, mobile solution.
10 Overdosage
Few cases of overdose have been described.
A 58-year-old man with acute lymphoblastic leukemia received 10-fold overdose of doxorubicin hydrochloride (300 mg/m2) in one day. He was treated with charcoal filtration, hemopoietic growth factor (G-CSF), proton pump inhibitor and antimicrobial prophylaxis. The patient suffered sinus tachycardia, grade 4 neutropenia and thrombocytopenia for 11 days, severe mucositis and sepsis. The patient recovered completely 26 days after the overdose.
A 17-year-old girl with osteogenic sarcoma received 150 mg of doxorubicin hydrochloride daily for 2 days (intended dose was 50 mg per day for 3 days). The patient developed severe mucositis on days 4–7 after the overdose and chills and pyrexia on day 7. The patient was treated with antibiotics and platelets and recovered 18 days after overdose.
15 References
-
1."Hazardous Drugs". OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description
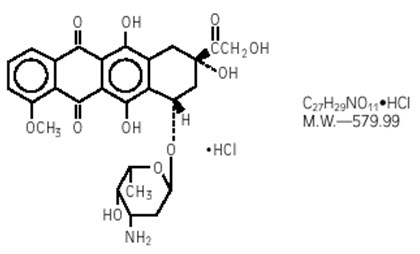
Doxorubicin hydrochloride is an anthracycline topoisomerase inhibitor isolated from cultures of Streptomyces peucetius var. caesius. The chemical name of doxorubicin hydrochloride is 5,12-Naphthacenedione, 10-[(3-amino-2,3,6-trideoxy-α-L-lyxo-hexopyranosyl)oxy]-7,8,9,10-tetrahydro-6,8,11-trihydroxy-8-(hydroxylacetyl)-1-methoxy-, hydrochloride (8S-cis)-. The chemical structure of doxorubicin hydrochloride is:
Doxorubicin Hydrochloride Injection, for intravenous use is a clear red, sterile, isotonic aqueous solution provided in vials containing 10 mg/5 mL doxorubicin hydrochloride (equivalent to 9.37 mg of doxorubicin free base), 20 mg/10 mL doxorubicin hydrochloride (equivalent to 18.74 mg of doxorubicin free base), 50 mg/25 mL doxorubicin hydrochloride (equivalent to 46.86 mg of doxorubicin free base), 150 mg/75 mL doxorubicin hydrochloride (140.58 mg of doxorubicin free base), or 200 mg/100 mL doxorubicin hydrochloride (equivalent to 187.4 mg of doxorubicin free base). The drug product has demonstrated inherent antimicrobial activity suitable for a multiple dose presentation. Each milliliter of solution contains 2 mg of doxorubicin hydrochloride and 9 mg of sodium chloride. The pH of the solution is adjusted to 3.0 with hydrochloric acid, USP.
1.2 Other Cancers
Doxorubicin Hydrochloride Injection is indicated for the treatment of
-
•acute lymphoblastic leukemia
-
•acute myeloblastic leukemia
-
•Hodgkin lymphoma
-
•non-Hodgkin lymphoma (NHL)
-
•metastatic breast cancer
-
•metastatic Wilms' tumor
-
•metastatic neuroblastoma
-
•metastatic soft tissue sarcoma
-
•metastatic bone sarcoma
-
•metastatic ovarian carcinoma
-
•metastatic transitional cell bladder carcinoma
-
•metastatic thyroid carcinoma
-
•metastatic gastric carcinoma
-
•metastatic bronchogenic carcinoma
8.4 Pediatric Use
Based on postmarketing reports, pediatric patients treated with doxorubicin hydrochloride are at risk for developing late cardiovascular dysfunction. Risk factors include young age at treatment (especially < 5 years), high cumulative doses and receipt of combined modality therapy. Long-term periodic cardiovascular monitoring is recommended for all pediatric patients who have received doxorubicin hydrochloride. Doxorubicin hydrochloride, as a component of intensive chemotherapy regimens administered to pediatric patients, may contribute to prepubertal growth failure and may also contribute to gonadal impairment, which is usually temporary.
There are no recommended dose adjustments based on age. Doxorubicin clearance was increased in patients aged 2 years to 20 years as compared to adults, while doxorubicin clearance was similar in infants less than 2 years as compared to adults [see Clinical Pharmacology (12.3)].
8.5 Geriatric Use
Clinical experience in patients who were 65 years of age and older who received doxorubicin hydrochloride-based chemotherapy regimens for metastatic breast cancer showed no overall differences in safety and effectiveness compared with younger patients.
4 Contraindications
Doxorubicin Hydrochloride Injection are contraindicated in patients with:
-
•Severe myocardial insufficiency [see Warnings and Precautions (5.1)]
-
•Recent (occurring within the past 4–6 weeks) myocardial infarction [see Warnings and Precautions (5.1)]
-
•Severe persistent drug-induced myelosuppression [see Warnings and Precautions (5.4)]
-
•Severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin level greater than 5 mg/dL) [see Warnings and Precautions (5.5)]
-
•Severe hypersensitivity reaction to doxorubicin hydrochloride, including anaphylaxis [see Adverse Reactions (6.2)]
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling.
-
•Cardiomyopathy and Arrhythmias [see Warnings and Precautions (5.1)]
-
•Secondary Malignancies [see Warnings and Precautions (5.2)]
-
•Extravasation and Tissue Necrosis [see Warnings and Precautions (5.3)]
-
•Severe Myelosuppression [see Warnings and Precautions (5.4)]
-
•Tumor Lysis Syndrome [see Warnings and Precautions (5.6)]
-
•Radiation Sensitization and Radiation Recall [see Warnings and Precautions (5.7)]
7 Drug Interactions
12.3 Pharmacokinetics
Pharmacokinetic studies conducted in patients with various types of tumors have shown that doxorubicin follows multiphasic disposition after intravenous injection. In four patients, doxorubicin demonstrated dose-independent pharmacokinetics across a dose range of 30 mg/m2 to 70 mg/m2.
8.6 Hepatic Impairment
The clearance of doxorubicin was reduced in patients with elevated serum total bilirubin levels.
Doxorubicin Hydrochloride Injection is contraindicated in patients with severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin levels greater than 5 mg/dL) [see Contraindications (4)]. Reduce the dose of Doxorubicin Hydrochloride Injection in patients with serum total bilirubin levels greater than 1.2 mg/dL [See Dosage and Administration (2.4), Warnings and Precautions (5.5)].
1 Indications and Usage
Doxorubicin Hydrochloride Injection is an anthracycline topoisomerase inhibitor indicated:
-
•as a component of multi-agent adjuvant chemotherapy for treatment of women with axillary lymph node involvement following resection of primary breast cancer (1.1)
-
•for the treatment of: acute lymphoblastic leukemia, acute myeloblastic leukemia, Hodgkin lymphoma, Non-Hodgkin lymphoma, metastatic breast cancer, metastatic Wilms' tumor, metastatic neuroblastoma, metastatic soft tissue sarcoma, metastatic bone sarcomas, metastatic ovarian carcinoma, metastatic transitional cell bladder carcinoma, metastatic thyroid carcinoma, metastatic gastric carcinoma, metastatic bronchogenic carcinoma (1.2)
12.1 Mechanism of Action
The cytotoxic effect of doxorubicin hydrochloride on malignant cells and its toxic effects on various organs are thought to be related to nucleotide base intercalation and cell membrane lipid binding activities of doxorubicin. Intercalation inhibits nucleotide replication and action of DNA and RNA polymerases. The interaction of doxorubicin with topoisomerase II to form DNA-cleavable complexes appears to be an important mechanism of doxorubicin hydrochloride cytocidal activity.
5.6 Tumor Lysis Syndrome
Doxorubicin hydrochloride can induce tumor lysis syndrome in patients with rapidly growing tumors. Evaluate blood uric acid levels, potassium, calcium, phosphate, and creatinine after initial treatment. Hydration, urine alkalinization, and prophylaxis with allopurinol to prevent hyperuricemia may minimize potential complications of tumor lysis syndrome.
5.8 Embryo Fetal Toxicity
Based on findings in animals and its mechanism of action, Doxorubicin Hydrochloride Injection can cause fetal harm when administered to a pregnant woman; avoid the use of Doxorubicin Hydrochloride Injection during the 1st trimester. Available human data do not establish the presence or absence of major birth defects and miscarriage related to the use of doxorubicin hydrochloride during the 2nd and 3rd trimesters. Doxorubicin hydrochloride was teratogenic and embryotoxic in rats and rabbits at doses lower than the recommended human dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with Doxorubicin Hydrochloride Injection and for 6 months after treatment. Advise males with female partners of reproductive potential to use effective contraception during treatment with Doxorubicin Hydrochloride Injection and for 3 months after treatment. Advise males with pregnant partners to use condoms during treatment with Doxorubicin Hydrochloride Injection and for at least 10 days after the final dose [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
1.1 Adjuvant Breast Cancer
Doxorubicin Hydrochloride Injection is indicated as a component of multi-agent adjuvant chemotherapy for treatment of women with axillary lymph node involvement following resection of primary breast cancer.
5 Warnings and Precautions
-
•Radiation-Induced Toxicity: Can be increased by the administration of Doxorubicin Hydrochloride Injection. Radiation recall can occur in patients who receive Doxorubicin Hydrochloride Injection after prior radiation therapy. (5.7)
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and on the use of effective contraception. Advise males with female partners of reproductive potential to use effective contraception. Advise males with pregnant partners to use condoms. (5.8, 8.1, 8.3)
5.2 Secondary Malignancies
The risk of developing secondary acute myelogenous leukemia (AML) and myelodysplastic syndrome (MDS) is increased following treatment with doxorubicin hydrochloride. Cumulative incidences ranged from 0.2% at five years to 1.5% at 10 years in two separate trials involving the adjuvant treatment of women with breast cancer. These leukemias generally occur within 1 to 3 years of treatment.
14.1 Adjuvant Breast Cancer
The efficacy of doxorubicin hydrochloride-containing regimens for the post-operative, adjuvant treatment of surgically resected breast cancer was evaluated in a meta-analysis conducted by the Early Breast Cancer Trialists Collaborative Group (EBCTCG). The EBCTCG meta-analyses compared cyclophosphamide, methotrexate, and fluorouracil (CMF) to no chemotherapy (19 trials including 7523 patients) and doxorubicin hydrochloride-containing regimens with CMF as an active control (6 trials including 3510 patients). Data from the meta-analysis of trials comparing CMF to no therapy were used to establish the historical treatment effect size for CMF regimens. The major efficacy outcome measures were disease-free survival (DFS) and overall survival (OS).
Of the 3510 women (2157 received doxorubicin hydrochloride-containing regimens and 1353 received CMF treatment) with early breast cancer involving axillary lymph nodes included in the six trials from the meta-analyses, approximately 70% were premenopausal and 30% were postmenopausal.
At the time of the meta-analysis, 1745 first recurrences and 1348 deaths had occurred. The analyses demonstrated that doxorubicin hydrochloride-containing regimens retained at least 75% of the historical CMF adjuvant effect on DFS with a hazard ratio (HR) of 0.91 (95% CI: 0.82, 1.01) and on OS with a HR of 0.91 (95% CI: 0.81, 1.03). Efficacy results are provided in Table 3 and Figures 1 and 2.
|
Study
(starting year) |
Regimens | No. of Cycles | No. of Patients |
Doxorubicin Hydrochloride‑Containing Regimens vs. CMF
HR Hazard ratio of less than 1 indicates that the treatment with doxorubicin hydrochloride-containing regimens is associated with lower risk of disease recurrences or death compared to the treatment with CMF.
(95% CI)
|
|
|---|---|---|---|---|---|
| DFS | OS | ||||
| Abbreviations: DFS = disease free survival; OS = overall survival; AC = doxorubicin hydrochloride, cyclophosphamide; AVbCMF = doxorubicin hydrochloride, vinblastine, cyclophosphamide, methotrexate, fluorouracil; CMF = cyclophosphamide, methotrexate, fluorouracil; CMFVA = cyclophosphamide, methotrexate, fluorouracil, vincristine, doxorubicin hydrochloride; FAC = fluorouracil, doxorubicin hydrochloride, cyclophosphamide; FACV = fluorouracil, doxorubicin hydrochloride, cyclophosphamide, vincristine; HR = hazard ratio; CI = confidence interval | |||||
|
|
|||||
|
NSABP B-15 |
AC |
4 |
1562 Includes pooled data from patients who received either AC alone for 4 cycles, or who were treated with AC for 4 cycles followed by 3 cycles of CMF.
|
0.93 (0.82, 1.06) |
0.97 (0.83, 1.12) |
|
|
|||||
|
CMF |
6 |
776 |
|||
|
|
|||||
|
SECSG 2 |
FAC |
6 |
260 |
0.86 (0.66, 1.13) |
0.93 (0.69, 1.26) |
|
|
|||||
|
CMF |
6 |
268 |
|||
|
|
|||||
|
ONCOFRANCE |
FACV |
12 |
138 |
0.71 (0.49, 1.03) |
0.65 (0.44, 0.96) |
|
|
|||||
|
CMF |
12 |
113 |
|||
|
|
|||||
|
SE Sweden BCG A |
AC |
6 |
21 |
0.59 (0.22, 1.61) |
0.53 (0.21, 1.37) |
|
|
|||||
|
CMF |
6 |
22 |
|||
|
|
|||||
|
NSABC Israel Br0283 |
AVbCMF Patients received alternating cycles of AVb and CMF.
|
4 |
55 |
0.91 (0.53, 1.57) |
0.88 (0.47, 1.63) |
|
|
|||||
|
CMF |
6 |
50 |
|||
|
|
|||||
|
Austrian BCSG 3 |
CMFVA |
6 |
121 |
||
|
1.07 (0.73, 1.55) |
0.93 (0.64, 1.35) |
||||
|
CMF |
8 |
124 |
|||
|
Combined Studies |
Doxorubicin Hydrochloride-Containing Regimen |
2157 |
|||
|
0.91 (0.82, 1.01) |
0.91 (0.81, 1.03) |
||||
|
CMF |
1353 |
|
Figure 1. Meta-analysis of Disease-Free Survival |
|
|
|
Figure 2. Meta-analysis of Overall Survival |
2 Dosage and Administration
-
•Single agent: 60 to 75 mg/m2 given intravenously every 21 days (2.2)
-
•In combination: 40 to 75 mg/m2 given intravenously every 21 to 28 days (2.2)
-
•Discontinue Doxorubicin Hydrochloride Injection in patients who develop signs or symptoms of cardiomyopathy (2.3)
-
•Reduce dose in patients with hepatic impairment (2.4)
5.4 Severe Myelosuppression
Doxorubicin hydrochloride can cause myelosuppression. In Study 1, the incidence of severe myelosuppression was: grade 4 leukopenia (0.3%), grade 3 leukopenia (3%), and grade 4 thrombocytopenia (0.1%). A dose-dependent, reversible neutropenia is the predominant manifestation of myelosuppression from doxorubicin hydrochloride. When doxorubicin hydrochloride is administered every 21 days, the neutrophil count reaches its nadir 10 to 14 days after administration with recovery usually occurring by day 21.
Obtain complete blood counts prior to each treatment and carefully monitor patients during treatment for possible clinical complications due to myelosuppression. Delay next dose of Doxorubicin Hydrochloride Injection if severe myelosuppression has not improved. Consider dose reduction for patients with prolonged myelosuppression based on severity of reaction.
3 Dosage Forms and Strengths
Doxorubicin Hydrochloride Injection:
-
•10 mg/5 mL, 20 mg/10 mL, and 50 mg/25 mL (2 mg/mL) clear red solution in a single-dose vial.
-
•200 mg/100 mL (2 mg/mL) clear red solution in a multiple-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of Doxorubicin Hydrochloride Injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac – Cardiogenic shock
Cutaneous – Skin and nail hyperpigmentation, oncolysis, rash, itching, photosensitivity, urticaria, acral erythema, palmar plantar erythrodysesthesia
Gastrointestinal – Nausea, mucositis, stomatitis, necrotizing colitis, typhlitis, gastric erosions, gastrointestinal tract bleeding, hematochezia, esophagitis, anorexia, abdominal pain, dehydration, diarrhea, hyperpigmentation of the oral mucosa
Hypersensitivity – Anaphylaxis
Laboratory Abnormalities – Increased ALT, increased AST
Neurological – Peripheral sensory and motor neuropathy, seizures, coma
Ocular – Conjunctivitis, keratitis, lacrimation
Vascular – Phlebosclerosis, phlebitis/thrombophlebitis, hot flashes, thromboembolism
Other – Malaise/asthenia, fever, chills, weight gain
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.5 Preparation and Administration
Doxorubicin Hydrochloride Injection is a hazardous drug. Follow applicable special handling and disposal procedures.1
7.2 Concomitant Use of Trastuzumab
Concomitant use of trastuzumab and doxorubicin hydrochloride results in an increased risk of cardiac dysfunction. Avoid concomitant administration of Doxorubicin Hydrochloride Injection and trastuzumab [see Warnings and Precautions (5.1)].
Patients receiving doxorubicin after stopping treatment with trastuzumab may also be at an increased risk of developing cardiotoxicity. Trastuzumab may persist in the circulation for up to 7 months. Therefore, avoid anthracycline-based therapy for up to 7 months after stopping trastuzumab when possible. If anthracyclines are used before this time, carefully monitor cardiac function.
7.3 Concomitant Use of Dexrazoxane
Do not administer dexrazoxane as a cardioprotectant at the initiation of doxorubicin hydrochloride-containing chemotherapy regimens. In a randomized trial in women with metastatic breast cancer, initiation of dexrazoxane with doxorubicin hydrochloride-based chemotherapy resulted in a significantly lower tumor response rate (48% vs. 63%; p = 0.007) and shorter time to progression compared to doxorubicin hydrochloride-based chemotherapy alone.
5.3 Extravasation and Tissue Necrosis
Extravasation of doxorubicin hydrochloride can cause severe local tissue injury manifesting as blistering, ulceration, and necrosis requiring wide excision of the affected area and skin grafting. Extravasation should be considered if a patient experiences a burning or stinging sensation or shows other evidence indicating peri-venous infiltration or extravasation; however, extravasation may be present in patients who do not experience a stinging or burning sensation or when blood return is present on aspiration of the infusion needle.
When given via a peripheral venous line, infuse Doxorubicin Hydrochloride Injection over 10 minutes or less to minimize the risk of thrombosis or perivenous extravasation.
If extravasation is suspected, immediately discontinue the intravenous injection or continuous intravenous infusion [see Dosage and Administration (2.5)]. Apply ice to the site intermittently for 15 minutes, 4 times a day for 3 days. In adults, if appropriate, administer dexrazoxane at the site of extravasation as soon as possible and within the first 6 hours after extravasation.
7.4 Concomitant Use of 6 Mercaptopurine
Doxorubicin hydrochloride may potentiate 6-mercaptopurine-induced hepatotoxicity. In 11 patients with refractory leukemia treated with 6-mercaptopurine (500 mg/m2 intravenously daily for 5 days per cycle every 2–3 weeks) and doxorubicin hydrochloride (50 mg/m2 intravenous once per cycle every 2–3 weeks) alone or with vincristine and prednisone, all developed hepatic dysfunction manifested by increased total serum bilirubin, alkaline phosphatase and aspartate aminotransferase.
2.2 Recommended Dosage for Other Cancers
-
•The recommended dosage of Doxorubicin Hydrochloride Injection when used as a single agent is 60 mg/m2 to 75 mg/m2 intravenously every 21 days.
-
•The recommended dosage of Doxorubicin Hydrochloride Injection, when administered in combination with other chemotherapy drugs, is 40 mg/m2 to 75 mg/m2 intravenously every 21 to 28 days.
-
•Consider use of the lower Doxorubicin Hydrochloride Injection dose in the recommended dosage range or longer intervals between cycles for heavily pretreated patients, elderly patients, or obese patients.
-
•Cumulative doses above 550 mg/m2 are associated with an increased risk of cardiomyopathy [see Warnings and Precautions (5.1)].
5.5 Use in Patients With Hepatic Impairment
The clearance of doxorubicin is decreased in patients with elevated serum bilirubin with an increased risk of toxicity [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)]. Doxorubicin Hydrochloride Injection is contraindicated in patients with severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin level greater than 5 mg/dL) [see Contraindications (4)]. Reduce the dose of Doxorubicin Hydrochloride Injection in patients with serum bilirubin levels of 1.2 to 5 mg/dL [see Dosage and Administration (2.4)]. Obtain liver tests including ALT, AST, alkaline phosphatase, and bilirubin prior to and during therapy.
2.4 Dosage Modifications for Hepatic Impairment
Doxorubicin Hydrochloride Injection is contraindicated in patients with severe hepatic impairment (Child-Pugh Class C or serum bilirubin greater than 5 mg/dL) [see Contraindications (4)].
Dosage modifications for Doxorubicin Hydrochloride Injection in patients with elevated serum total bilirubin concentrations [see Warnings and Precautions (5.5), Use in Specific Populations (8.6)] are provided in Table 1.
| Serum Total Bilirubin Concentration | Dosage Modification |
|---|---|
|
1.2–3 mg/dL |
50% |
|
3.1–5 mg/dL |
75% |
|
Greater than 5 mg/dL |
Do not initiate Doxorubicin Hydrochloride Injection; |
Principal Display Panel 10 Mg/5 Ml Vial Label
NDC 0069-4004-05
Single-dose
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
10 mg/5 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
Principal Display Panel 10 Mg/5 Ml Vial Carton
NDC 0069-4004-05
Rx only
Single-dose: Discard unused portion
DOXOrubicin
Hydrochloride
Injection
10 mg/5 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 20 Mg/10 Ml Vial Label
NDC 0069-4015-10
Single-dose
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
Principal Display Panel 50 Mg/25 Ml Vial Label
NDC 0069-4026-25
Single-dose
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
50 mg/25 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
2.1 Recommended Dosage for Adjuvant Breast Cancer
The recommended dosage of Doxorubicin Hydrochloride Injection is 60 mg/m2 administered as an intravenous bolus on day 1 of each 21-day treatment cycle, in combination with cyclophosphamide, for a total of four cycles.
Principal Display Panel 20 Mg/10 Ml Vial Carton
NDC 0069-4015-10
Rx only
Single-dose: Discard unused portion
DOXOrubicin
Hydrochloride
Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 50 Mg/25 Ml Vial Carton
NDC 0069-4026-25
Rx only
Single-dose: Discard unused portion
DOXOrubicin
Hydrochloride
Injection
50 mg/25 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 200 Mg/100 Ml Vial Label
NDC 0069-4037-01
Multiple-dose Vial
Rx only
DOXOrubicin
Hydrochloride Injection
200 mg/100 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 200 Mg/100 Ml Vial Carton
NDC 0069-4037-01
Rx only
Multiple-dose Vial
DOXOrubicin
Hydrochloride
Injection
200 mg/100 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel – 10 Mg/5 Ml Vial Onco Tain®
NDC 0069-4205-05
Single-dose vial
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
10 mg/5 mL
(2 mg/mL)
For Intravenous Use Only
Principal Display Panel – 20 Mg/10 Ml Vial Onco Tain®
NDC 0069-0255-10
Single-dose vial
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
Warning: Hazardous Drug
Principal Display Panel – 50 Mg/25 Ml Vial Onco Tain®
NDC 0069-3358-25
Single-dose vial
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
50 mg/25 mL
(2 mg/mL)
For Intravenous Use Only
Warning: Hazardous Drug
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Doxorubicin hydrochloride treatment can increase the risk of secondary malignancies based on postmarketing reports [see Warnings and Precautions (5.2)]. Doxorubicin hydrochloride was mutagenic in the in vitro Ames assay, and clastogenic in multiple in vitro assays (CHO cell, V79 hamster cell, human lymphoblast, and SCE assays) and the in vivo mouse micronucleus assay.
Doxorubicin hydrochloride decreased fertility in female rats at the doses of 0.05 and 0.2 mg/kg/day (approximately 0.005 and 0.02 times the recommended human dose, based on body surface area).
A single intravenous dose of 0.1 mg/kg doxorubicin hydrochloride (approximately 0.01 times the recommended human dose based on body surface area) was toxic to male reproductive organs in animal studies, producing testicular atrophy, diffuse degeneration of the seminiferous tubules, and oligospermia/hypospermia in rats. Doxorubicin hydrochloride induces DNA damage in rabbit spermatozoa and dominant lethal mutations in mice.
Principal Display Panel – 200 Mg/100 Ml Vial Onco Tain®
NDC 0069-1442-04
Multiple-dose Vial
Rx only
DOXOrubicin
Hydrochloride Injection
200 mg/100 mL
(2 mg/mL)
For Intravenous Use Only
Warning: Hazardous Drug
5.7 Potentiation of Radiation Toxicity and Radiation Recall
Doxorubicin hydrochloride can increase radiation-induced toxicity to the myocardium, mucosa, skin, and liver. Radiation recall, including but not limited to cutaneous and pulmonary toxicity, can occur in patients who receive doxorubicin hydrochloride after prior radiation therapy.
Warning: Cardiomyopathy, Secondary Malignancies, Extravasation and Tissue Necrosis, and Severe Myelosuppression
-
•Cardiomyopathy: Myocardial damage, including acute left ventricular failure, can occur with doxorubicin hydrochloride. The risk of cardiomyopathy is proportional to the cumulative exposure with incidence rates from 1%–20% for cumulative doses ranging from 300 mg/m2 to 500 mg/m2 when doxorubicin hydrochloride is administered every 3 weeks. The risk of cardiomyopathy is further increased with concomitant cardiotoxic therapy. Assess left ventricular ejection fraction (LVEF) before and regularly during and after treatment with doxorubicin hydrochloride [see Warnings and Precautions (5.1)].
-
•Secondary Malignancies: Secondary acute myelogenous leukemia (AML) and myelodysplastic syndrome (MDS) occur at a higher incidence in patients treated with anthracyclines, including doxorubicin hydrochloride [see Warnings and Precautions (5.2)].
-
•Extravasation and Tissue Necrosis: Extravasation of doxorubicin hydrochloride can result in severe local tissue injury and necrosis requiring wide excision of the affected area and skin grafting. Immediately terminate the drug and apply ice to the affected area [see Warnings and Precautions (5.3)].
-
•Severe myelosuppression resulting in serious infection, septic shock, requirement for transfusions, hospitalization, and death may occur [see Warnings and Precautions (5.4)].
Structured Label Content
Section 42229-5 (42229-5)
Cardiomyopathy
Discontinue Doxorubicin Hydrochloride Injection in patients who develop signs or symptoms of cardiomyopathy [see Warnings and Precautions (5.1)].
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 7/2024 | ||
|
Patient Information
|
||
|
What is the most important information I should know about Doxorubicin?
|
||
|
|
|
|
Your healthcare provider will do tests to check the strength of your heart muscle before, during, and after your treatment with Doxorubicin. |
||
|
||
|
What is Doxorubicin?
|
||
|
Do not receive Doxorubicin if:
|
||
|
Before you receive Doxorubicin, tell your healthcare provider about all of your medical conditions, including if you:
|
||
|
How will I receive Doxorubicin?
|
||
|
What are the possible side effects of Doxorubicin?
The most common side effects of Doxorubicin include:
Other side effects:
Doxorubicin may cause fertility problems in males. This could affect your ability to father a child. Talk to your healthcare provider if this is a concern for you.
Doxorubicin may cause fertility problems in females. Your periods (menstrual cycle) may completely stop when you receive Doxorubicin. Your periods may or may not return following treatment. Early menopause has also happened. Talk to your healthcare provider if this is a concern for you. |
||
|
General information about the safe and effective use of Doxorubicin.
|
||
|
What are the ingredients in Doxorubicin?
Pfizer Labs Division of Pfizer Inc. New York, NY 10001
LAB-0703-5.0 For more information, call 1-800-438-1985 or visit www.pfizer.com |
Section 44425-7 (44425-7)
Storage
Store all vials at 2°C to 8°C (36°F to 46°F). Protect from light.
Storage of Doxorubicin Hydrochloride Injection under refrigerated conditions can result in the formation of a gelled product. Place gelled product at room temperature [15°C to 30°C (59°F to 86°F)] for 2 to 4 hours to return the product to a slightly viscous, mobile solution.
10 Overdosage (10 OVERDOSAGE)
Few cases of overdose have been described.
A 58-year-old man with acute lymphoblastic leukemia received 10-fold overdose of doxorubicin hydrochloride (300 mg/m2) in one day. He was treated with charcoal filtration, hemopoietic growth factor (G-CSF), proton pump inhibitor and antimicrobial prophylaxis. The patient suffered sinus tachycardia, grade 4 neutropenia and thrombocytopenia for 11 days, severe mucositis and sepsis. The patient recovered completely 26 days after the overdose.
A 17-year-old girl with osteogenic sarcoma received 150 mg of doxorubicin hydrochloride daily for 2 days (intended dose was 50 mg per day for 3 days). The patient developed severe mucositis on days 4–7 after the overdose and chills and pyrexia on day 7. The patient was treated with antibiotics and platelets and recovered 18 days after overdose.
15 References (15 REFERENCES)
-
1."Hazardous Drugs". OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
Doxorubicin hydrochloride is an anthracycline topoisomerase inhibitor isolated from cultures of Streptomyces peucetius var. caesius. The chemical name of doxorubicin hydrochloride is 5,12-Naphthacenedione, 10-[(3-amino-2,3,6-trideoxy-α-L-lyxo-hexopyranosyl)oxy]-7,8,9,10-tetrahydro-6,8,11-trihydroxy-8-(hydroxylacetyl)-1-methoxy-, hydrochloride (8S-cis)-. The chemical structure of doxorubicin hydrochloride is:
Doxorubicin Hydrochloride Injection, for intravenous use is a clear red, sterile, isotonic aqueous solution provided in vials containing 10 mg/5 mL doxorubicin hydrochloride (equivalent to 9.37 mg of doxorubicin free base), 20 mg/10 mL doxorubicin hydrochloride (equivalent to 18.74 mg of doxorubicin free base), 50 mg/25 mL doxorubicin hydrochloride (equivalent to 46.86 mg of doxorubicin free base), 150 mg/75 mL doxorubicin hydrochloride (140.58 mg of doxorubicin free base), or 200 mg/100 mL doxorubicin hydrochloride (equivalent to 187.4 mg of doxorubicin free base). The drug product has demonstrated inherent antimicrobial activity suitable for a multiple dose presentation. Each milliliter of solution contains 2 mg of doxorubicin hydrochloride and 9 mg of sodium chloride. The pH of the solution is adjusted to 3.0 with hydrochloric acid, USP.
1.2 Other Cancers
Doxorubicin Hydrochloride Injection is indicated for the treatment of
-
•acute lymphoblastic leukemia
-
•acute myeloblastic leukemia
-
•Hodgkin lymphoma
-
•non-Hodgkin lymphoma (NHL)
-
•metastatic breast cancer
-
•metastatic Wilms' tumor
-
•metastatic neuroblastoma
-
•metastatic soft tissue sarcoma
-
•metastatic bone sarcoma
-
•metastatic ovarian carcinoma
-
•metastatic transitional cell bladder carcinoma
-
•metastatic thyroid carcinoma
-
•metastatic gastric carcinoma
-
•metastatic bronchogenic carcinoma
8.4 Pediatric Use
Based on postmarketing reports, pediatric patients treated with doxorubicin hydrochloride are at risk for developing late cardiovascular dysfunction. Risk factors include young age at treatment (especially < 5 years), high cumulative doses and receipt of combined modality therapy. Long-term periodic cardiovascular monitoring is recommended for all pediatric patients who have received doxorubicin hydrochloride. Doxorubicin hydrochloride, as a component of intensive chemotherapy regimens administered to pediatric patients, may contribute to prepubertal growth failure and may also contribute to gonadal impairment, which is usually temporary.
There are no recommended dose adjustments based on age. Doxorubicin clearance was increased in patients aged 2 years to 20 years as compared to adults, while doxorubicin clearance was similar in infants less than 2 years as compared to adults [see Clinical Pharmacology (12.3)].
8.5 Geriatric Use
Clinical experience in patients who were 65 years of age and older who received doxorubicin hydrochloride-based chemotherapy regimens for metastatic breast cancer showed no overall differences in safety and effectiveness compared with younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
Doxorubicin Hydrochloride Injection are contraindicated in patients with:
-
•Severe myocardial insufficiency [see Warnings and Precautions (5.1)]
-
•Recent (occurring within the past 4–6 weeks) myocardial infarction [see Warnings and Precautions (5.1)]
-
•Severe persistent drug-induced myelosuppression [see Warnings and Precautions (5.4)]
-
•Severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin level greater than 5 mg/dL) [see Warnings and Precautions (5.5)]
-
•Severe hypersensitivity reaction to doxorubicin hydrochloride, including anaphylaxis [see Adverse Reactions (6.2)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling.
-
•Cardiomyopathy and Arrhythmias [see Warnings and Precautions (5.1)]
-
•Secondary Malignancies [see Warnings and Precautions (5.2)]
-
•Extravasation and Tissue Necrosis [see Warnings and Precautions (5.3)]
-
•Severe Myelosuppression [see Warnings and Precautions (5.4)]
-
•Tumor Lysis Syndrome [see Warnings and Precautions (5.6)]
-
•Radiation Sensitization and Radiation Recall [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
12.3 Pharmacokinetics
Pharmacokinetic studies conducted in patients with various types of tumors have shown that doxorubicin follows multiphasic disposition after intravenous injection. In four patients, doxorubicin demonstrated dose-independent pharmacokinetics across a dose range of 30 mg/m2 to 70 mg/m2.
8.6 Hepatic Impairment
The clearance of doxorubicin was reduced in patients with elevated serum total bilirubin levels.
Doxorubicin Hydrochloride Injection is contraindicated in patients with severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin levels greater than 5 mg/dL) [see Contraindications (4)]. Reduce the dose of Doxorubicin Hydrochloride Injection in patients with serum total bilirubin levels greater than 1.2 mg/dL [See Dosage and Administration (2.4), Warnings and Precautions (5.5)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Doxorubicin Hydrochloride Injection is an anthracycline topoisomerase inhibitor indicated:
-
•as a component of multi-agent adjuvant chemotherapy for treatment of women with axillary lymph node involvement following resection of primary breast cancer (1.1)
-
•for the treatment of: acute lymphoblastic leukemia, acute myeloblastic leukemia, Hodgkin lymphoma, Non-Hodgkin lymphoma, metastatic breast cancer, metastatic Wilms' tumor, metastatic neuroblastoma, metastatic soft tissue sarcoma, metastatic bone sarcomas, metastatic ovarian carcinoma, metastatic transitional cell bladder carcinoma, metastatic thyroid carcinoma, metastatic gastric carcinoma, metastatic bronchogenic carcinoma (1.2)
12.1 Mechanism of Action
The cytotoxic effect of doxorubicin hydrochloride on malignant cells and its toxic effects on various organs are thought to be related to nucleotide base intercalation and cell membrane lipid binding activities of doxorubicin. Intercalation inhibits nucleotide replication and action of DNA and RNA polymerases. The interaction of doxorubicin with topoisomerase II to form DNA-cleavable complexes appears to be an important mechanism of doxorubicin hydrochloride cytocidal activity.
5.6 Tumor Lysis Syndrome
Doxorubicin hydrochloride can induce tumor lysis syndrome in patients with rapidly growing tumors. Evaluate blood uric acid levels, potassium, calcium, phosphate, and creatinine after initial treatment. Hydration, urine alkalinization, and prophylaxis with allopurinol to prevent hyperuricemia may minimize potential complications of tumor lysis syndrome.
5.8 Embryo Fetal Toxicity (5.8 Embryo-Fetal Toxicity)
Based on findings in animals and its mechanism of action, Doxorubicin Hydrochloride Injection can cause fetal harm when administered to a pregnant woman; avoid the use of Doxorubicin Hydrochloride Injection during the 1st trimester. Available human data do not establish the presence or absence of major birth defects and miscarriage related to the use of doxorubicin hydrochloride during the 2nd and 3rd trimesters. Doxorubicin hydrochloride was teratogenic and embryotoxic in rats and rabbits at doses lower than the recommended human dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with Doxorubicin Hydrochloride Injection and for 6 months after treatment. Advise males with female partners of reproductive potential to use effective contraception during treatment with Doxorubicin Hydrochloride Injection and for 3 months after treatment. Advise males with pregnant partners to use condoms during treatment with Doxorubicin Hydrochloride Injection and for at least 10 days after the final dose [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
1.1 Adjuvant Breast Cancer
Doxorubicin Hydrochloride Injection is indicated as a component of multi-agent adjuvant chemotherapy for treatment of women with axillary lymph node involvement following resection of primary breast cancer.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Radiation-Induced Toxicity: Can be increased by the administration of Doxorubicin Hydrochloride Injection. Radiation recall can occur in patients who receive Doxorubicin Hydrochloride Injection after prior radiation therapy. (5.7)
-
•Embryo-Fetal Toxicity: Can cause fetal harm. Advise females of reproductive potential of the potential risk to a fetus and on the use of effective contraception. Advise males with female partners of reproductive potential to use effective contraception. Advise males with pregnant partners to use condoms. (5.8, 8.1, 8.3)
5.2 Secondary Malignancies
The risk of developing secondary acute myelogenous leukemia (AML) and myelodysplastic syndrome (MDS) is increased following treatment with doxorubicin hydrochloride. Cumulative incidences ranged from 0.2% at five years to 1.5% at 10 years in two separate trials involving the adjuvant treatment of women with breast cancer. These leukemias generally occur within 1 to 3 years of treatment.
14.1 Adjuvant Breast Cancer
The efficacy of doxorubicin hydrochloride-containing regimens for the post-operative, adjuvant treatment of surgically resected breast cancer was evaluated in a meta-analysis conducted by the Early Breast Cancer Trialists Collaborative Group (EBCTCG). The EBCTCG meta-analyses compared cyclophosphamide, methotrexate, and fluorouracil (CMF) to no chemotherapy (19 trials including 7523 patients) and doxorubicin hydrochloride-containing regimens with CMF as an active control (6 trials including 3510 patients). Data from the meta-analysis of trials comparing CMF to no therapy were used to establish the historical treatment effect size for CMF regimens. The major efficacy outcome measures were disease-free survival (DFS) and overall survival (OS).
Of the 3510 women (2157 received doxorubicin hydrochloride-containing regimens and 1353 received CMF treatment) with early breast cancer involving axillary lymph nodes included in the six trials from the meta-analyses, approximately 70% were premenopausal and 30% were postmenopausal.
At the time of the meta-analysis, 1745 first recurrences and 1348 deaths had occurred. The analyses demonstrated that doxorubicin hydrochloride-containing regimens retained at least 75% of the historical CMF adjuvant effect on DFS with a hazard ratio (HR) of 0.91 (95% CI: 0.82, 1.01) and on OS with a HR of 0.91 (95% CI: 0.81, 1.03). Efficacy results are provided in Table 3 and Figures 1 and 2.
|
Study
(starting year) |
Regimens | No. of Cycles | No. of Patients |
Doxorubicin Hydrochloride‑Containing Regimens vs. CMF
HR Hazard ratio of less than 1 indicates that the treatment with doxorubicin hydrochloride-containing regimens is associated with lower risk of disease recurrences or death compared to the treatment with CMF.
(95% CI)
|
|
|---|---|---|---|---|---|
| DFS | OS | ||||
| Abbreviations: DFS = disease free survival; OS = overall survival; AC = doxorubicin hydrochloride, cyclophosphamide; AVbCMF = doxorubicin hydrochloride, vinblastine, cyclophosphamide, methotrexate, fluorouracil; CMF = cyclophosphamide, methotrexate, fluorouracil; CMFVA = cyclophosphamide, methotrexate, fluorouracil, vincristine, doxorubicin hydrochloride; FAC = fluorouracil, doxorubicin hydrochloride, cyclophosphamide; FACV = fluorouracil, doxorubicin hydrochloride, cyclophosphamide, vincristine; HR = hazard ratio; CI = confidence interval | |||||
|
|
|||||
|
NSABP B-15 |
AC |
4 |
1562 Includes pooled data from patients who received either AC alone for 4 cycles, or who were treated with AC for 4 cycles followed by 3 cycles of CMF.
|
0.93 (0.82, 1.06) |
0.97 (0.83, 1.12) |
|
|
|||||
|
CMF |
6 |
776 |
|||
|
|
|||||
|
SECSG 2 |
FAC |
6 |
260 |
0.86 (0.66, 1.13) |
0.93 (0.69, 1.26) |
|
|
|||||
|
CMF |
6 |
268 |
|||
|
|
|||||
|
ONCOFRANCE |
FACV |
12 |
138 |
0.71 (0.49, 1.03) |
0.65 (0.44, 0.96) |
|
|
|||||
|
CMF |
12 |
113 |
|||
|
|
|||||
|
SE Sweden BCG A |
AC |
6 |
21 |
0.59 (0.22, 1.61) |
0.53 (0.21, 1.37) |
|
|
|||||
|
CMF |
6 |
22 |
|||
|
|
|||||
|
NSABC Israel Br0283 |
AVbCMF Patients received alternating cycles of AVb and CMF.
|
4 |
55 |
0.91 (0.53, 1.57) |
0.88 (0.47, 1.63) |
|
|
|||||
|
CMF |
6 |
50 |
|||
|
|
|||||
|
Austrian BCSG 3 |
CMFVA |
6 |
121 |
||
|
1.07 (0.73, 1.55) |
0.93 (0.64, 1.35) |
||||
|
CMF |
8 |
124 |
|||
|
Combined Studies |
Doxorubicin Hydrochloride-Containing Regimen |
2157 |
|||
|
0.91 (0.82, 1.01) |
0.91 (0.81, 1.03) |
||||
|
CMF |
1353 |
|
Figure 1. Meta-analysis of Disease-Free Survival |
|
|
|
Figure 2. Meta-analysis of Overall Survival |
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Single agent: 60 to 75 mg/m2 given intravenously every 21 days (2.2)
-
•In combination: 40 to 75 mg/m2 given intravenously every 21 to 28 days (2.2)
-
•Discontinue Doxorubicin Hydrochloride Injection in patients who develop signs or symptoms of cardiomyopathy (2.3)
-
•Reduce dose in patients with hepatic impairment (2.4)
5.4 Severe Myelosuppression
Doxorubicin hydrochloride can cause myelosuppression. In Study 1, the incidence of severe myelosuppression was: grade 4 leukopenia (0.3%), grade 3 leukopenia (3%), and grade 4 thrombocytopenia (0.1%). A dose-dependent, reversible neutropenia is the predominant manifestation of myelosuppression from doxorubicin hydrochloride. When doxorubicin hydrochloride is administered every 21 days, the neutrophil count reaches its nadir 10 to 14 days after administration with recovery usually occurring by day 21.
Obtain complete blood counts prior to each treatment and carefully monitor patients during treatment for possible clinical complications due to myelosuppression. Delay next dose of Doxorubicin Hydrochloride Injection if severe myelosuppression has not improved. Consider dose reduction for patients with prolonged myelosuppression based on severity of reaction.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Doxorubicin Hydrochloride Injection:
-
•10 mg/5 mL, 20 mg/10 mL, and 50 mg/25 mL (2 mg/mL) clear red solution in a single-dose vial.
-
•200 mg/100 mL (2 mg/mL) clear red solution in a multiple-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of Doxorubicin Hydrochloride Injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac – Cardiogenic shock
Cutaneous – Skin and nail hyperpigmentation, oncolysis, rash, itching, photosensitivity, urticaria, acral erythema, palmar plantar erythrodysesthesia
Gastrointestinal – Nausea, mucositis, stomatitis, necrotizing colitis, typhlitis, gastric erosions, gastrointestinal tract bleeding, hematochezia, esophagitis, anorexia, abdominal pain, dehydration, diarrhea, hyperpigmentation of the oral mucosa
Hypersensitivity – Anaphylaxis
Laboratory Abnormalities – Increased ALT, increased AST
Neurological – Peripheral sensory and motor neuropathy, seizures, coma
Ocular – Conjunctivitis, keratitis, lacrimation
Vascular – Phlebosclerosis, phlebitis/thrombophlebitis, hot flashes, thromboembolism
Other – Malaise/asthenia, fever, chills, weight gain
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.5 Preparation and Administration
Doxorubicin Hydrochloride Injection is a hazardous drug. Follow applicable special handling and disposal procedures.1
7.2 Concomitant Use of Trastuzumab
Concomitant use of trastuzumab and doxorubicin hydrochloride results in an increased risk of cardiac dysfunction. Avoid concomitant administration of Doxorubicin Hydrochloride Injection and trastuzumab [see Warnings and Precautions (5.1)].
Patients receiving doxorubicin after stopping treatment with trastuzumab may also be at an increased risk of developing cardiotoxicity. Trastuzumab may persist in the circulation for up to 7 months. Therefore, avoid anthracycline-based therapy for up to 7 months after stopping trastuzumab when possible. If anthracyclines are used before this time, carefully monitor cardiac function.
7.3 Concomitant Use of Dexrazoxane
Do not administer dexrazoxane as a cardioprotectant at the initiation of doxorubicin hydrochloride-containing chemotherapy regimens. In a randomized trial in women with metastatic breast cancer, initiation of dexrazoxane with doxorubicin hydrochloride-based chemotherapy resulted in a significantly lower tumor response rate (48% vs. 63%; p = 0.007) and shorter time to progression compared to doxorubicin hydrochloride-based chemotherapy alone.
5.3 Extravasation and Tissue Necrosis
Extravasation of doxorubicin hydrochloride can cause severe local tissue injury manifesting as blistering, ulceration, and necrosis requiring wide excision of the affected area and skin grafting. Extravasation should be considered if a patient experiences a burning or stinging sensation or shows other evidence indicating peri-venous infiltration or extravasation; however, extravasation may be present in patients who do not experience a stinging or burning sensation or when blood return is present on aspiration of the infusion needle.
When given via a peripheral venous line, infuse Doxorubicin Hydrochloride Injection over 10 minutes or less to minimize the risk of thrombosis or perivenous extravasation.
If extravasation is suspected, immediately discontinue the intravenous injection or continuous intravenous infusion [see Dosage and Administration (2.5)]. Apply ice to the site intermittently for 15 minutes, 4 times a day for 3 days. In adults, if appropriate, administer dexrazoxane at the site of extravasation as soon as possible and within the first 6 hours after extravasation.
7.4 Concomitant Use of 6 Mercaptopurine (7.4 Concomitant Use of 6-Mercaptopurine)
Doxorubicin hydrochloride may potentiate 6-mercaptopurine-induced hepatotoxicity. In 11 patients with refractory leukemia treated with 6-mercaptopurine (500 mg/m2 intravenously daily for 5 days per cycle every 2–3 weeks) and doxorubicin hydrochloride (50 mg/m2 intravenous once per cycle every 2–3 weeks) alone or with vincristine and prednisone, all developed hepatic dysfunction manifested by increased total serum bilirubin, alkaline phosphatase and aspartate aminotransferase.
2.2 Recommended Dosage for Other Cancers
-
•The recommended dosage of Doxorubicin Hydrochloride Injection when used as a single agent is 60 mg/m2 to 75 mg/m2 intravenously every 21 days.
-
•The recommended dosage of Doxorubicin Hydrochloride Injection, when administered in combination with other chemotherapy drugs, is 40 mg/m2 to 75 mg/m2 intravenously every 21 to 28 days.
-
•Consider use of the lower Doxorubicin Hydrochloride Injection dose in the recommended dosage range or longer intervals between cycles for heavily pretreated patients, elderly patients, or obese patients.
-
•Cumulative doses above 550 mg/m2 are associated with an increased risk of cardiomyopathy [see Warnings and Precautions (5.1)].
5.5 Use in Patients With Hepatic Impairment (5.5 Use in Patients with Hepatic Impairment)
The clearance of doxorubicin is decreased in patients with elevated serum bilirubin with an increased risk of toxicity [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)]. Doxorubicin Hydrochloride Injection is contraindicated in patients with severe hepatic impairment (defined as Child Pugh Class C or serum bilirubin level greater than 5 mg/dL) [see Contraindications (4)]. Reduce the dose of Doxorubicin Hydrochloride Injection in patients with serum bilirubin levels of 1.2 to 5 mg/dL [see Dosage and Administration (2.4)]. Obtain liver tests including ALT, AST, alkaline phosphatase, and bilirubin prior to and during therapy.
2.4 Dosage Modifications for Hepatic Impairment
Doxorubicin Hydrochloride Injection is contraindicated in patients with severe hepatic impairment (Child-Pugh Class C or serum bilirubin greater than 5 mg/dL) [see Contraindications (4)].
Dosage modifications for Doxorubicin Hydrochloride Injection in patients with elevated serum total bilirubin concentrations [see Warnings and Precautions (5.5), Use in Specific Populations (8.6)] are provided in Table 1.
| Serum Total Bilirubin Concentration | Dosage Modification |
|---|---|
|
1.2–3 mg/dL |
50% |
|
3.1–5 mg/dL |
75% |
|
Greater than 5 mg/dL |
Do not initiate Doxorubicin Hydrochloride Injection; |
Principal Display Panel 10 Mg/5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 10 mg/5 mL Vial Label)
NDC 0069-4004-05
Single-dose
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
10 mg/5 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
Principal Display Panel 10 Mg/5 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 10 mg/5 mL Vial Carton)
NDC 0069-4004-05
Rx only
Single-dose: Discard unused portion
DOXOrubicin
Hydrochloride
Injection
10 mg/5 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 20 Mg/10 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 20 mg/10 mL Vial Label)
NDC 0069-4015-10
Single-dose
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
Principal Display Panel 50 Mg/25 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 50 mg/25 mL Vial Label)
NDC 0069-4026-25
Single-dose
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
50 mg/25 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
2.1 Recommended Dosage for Adjuvant Breast Cancer
The recommended dosage of Doxorubicin Hydrochloride Injection is 60 mg/m2 administered as an intravenous bolus on day 1 of each 21-day treatment cycle, in combination with cyclophosphamide, for a total of four cycles.
Principal Display Panel 20 Mg/10 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 20 mg/10 mL Vial Carton)
NDC 0069-4015-10
Rx only
Single-dose: Discard unused portion
DOXOrubicin
Hydrochloride
Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 50 Mg/25 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 50 mg/25 mL Vial Carton)
NDC 0069-4026-25
Rx only
Single-dose: Discard unused portion
DOXOrubicin
Hydrochloride
Injection
50 mg/25 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 200 Mg/100 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 200 mg/100 mL Vial Label)
NDC 0069-4037-01
Multiple-dose Vial
Rx only
DOXOrubicin
Hydrochloride Injection
200 mg/100 mL
(2 mg/mL)
For Intravenous Use Only
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel 200 Mg/100 Ml Vial Carton (PRINCIPAL DISPLAY PANEL - 200 mg/100 mL Vial Carton)
NDC 0069-4037-01
Rx only
Multiple-dose Vial
DOXOrubicin
Hydrochloride
Injection
200 mg/100 mL
(2 mg/mL)
For Intravenous Use Only
Cytosafe® Vial
Caution: Cytotoxic Agent
PREMIERProRx®
Principal Display Panel – 10 Mg/5 Ml Vial Onco Tain® (PRINCIPAL DISPLAY PANEL – 10 mg/5 mL Vial ONCO-TAIN®)
NDC 0069-4205-05
Single-dose vial
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
10 mg/5 mL
(2 mg/mL)
For Intravenous Use Only
Principal Display Panel – 20 Mg/10 Ml Vial Onco Tain® (PRINCIPAL DISPLAY PANEL – 20 mg/10 mL Vial ONCO-TAIN®)
NDC 0069-0255-10
Single-dose vial
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
20 mg/10 mL
(2 mg/mL)
For Intravenous Use Only
Warning: Hazardous Drug
Principal Display Panel – 50 Mg/25 Ml Vial Onco Tain® (PRINCIPAL DISPLAY PANEL – 50 mg/25 mL Vial ONCO-TAIN®)
NDC 0069-3358-25
Single-dose vial
Discard unused portion
Rx only
DOXOrubicin
Hydrochloride Injection
50 mg/25 mL
(2 mg/mL)
For Intravenous Use Only
Warning: Hazardous Drug
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Doxorubicin hydrochloride treatment can increase the risk of secondary malignancies based on postmarketing reports [see Warnings and Precautions (5.2)]. Doxorubicin hydrochloride was mutagenic in the in vitro Ames assay, and clastogenic in multiple in vitro assays (CHO cell, V79 hamster cell, human lymphoblast, and SCE assays) and the in vivo mouse micronucleus assay.
Doxorubicin hydrochloride decreased fertility in female rats at the doses of 0.05 and 0.2 mg/kg/day (approximately 0.005 and 0.02 times the recommended human dose, based on body surface area).
A single intravenous dose of 0.1 mg/kg doxorubicin hydrochloride (approximately 0.01 times the recommended human dose based on body surface area) was toxic to male reproductive organs in animal studies, producing testicular atrophy, diffuse degeneration of the seminiferous tubules, and oligospermia/hypospermia in rats. Doxorubicin hydrochloride induces DNA damage in rabbit spermatozoa and dominant lethal mutations in mice.
Principal Display Panel – 200 Mg/100 Ml Vial Onco Tain® (PRINCIPAL DISPLAY PANEL – 200 mg/100 mL Vial ONCO-TAIN®)
NDC 0069-1442-04
Multiple-dose Vial
Rx only
DOXOrubicin
Hydrochloride Injection
200 mg/100 mL
(2 mg/mL)
For Intravenous Use Only
Warning: Hazardous Drug
5.7 Potentiation of Radiation Toxicity and Radiation Recall
Doxorubicin hydrochloride can increase radiation-induced toxicity to the myocardium, mucosa, skin, and liver. Radiation recall, including but not limited to cutaneous and pulmonary toxicity, can occur in patients who receive doxorubicin hydrochloride after prior radiation therapy.
Warning: Cardiomyopathy, Secondary Malignancies, Extravasation and Tissue Necrosis, and Severe Myelosuppression (WARNING: CARDIOMYOPATHY, SECONDARY MALIGNANCIES, EXTRAVASATION AND TISSUE NECROSIS, and SEVERE MYELOSUPPRESSION)
-
•Cardiomyopathy: Myocardial damage, including acute left ventricular failure, can occur with doxorubicin hydrochloride. The risk of cardiomyopathy is proportional to the cumulative exposure with incidence rates from 1%–20% for cumulative doses ranging from 300 mg/m2 to 500 mg/m2 when doxorubicin hydrochloride is administered every 3 weeks. The risk of cardiomyopathy is further increased with concomitant cardiotoxic therapy. Assess left ventricular ejection fraction (LVEF) before and regularly during and after treatment with doxorubicin hydrochloride [see Warnings and Precautions (5.1)].
-
•Secondary Malignancies: Secondary acute myelogenous leukemia (AML) and myelodysplastic syndrome (MDS) occur at a higher incidence in patients treated with anthracyclines, including doxorubicin hydrochloride [see Warnings and Precautions (5.2)].
-
•Extravasation and Tissue Necrosis: Extravasation of doxorubicin hydrochloride can result in severe local tissue injury and necrosis requiring wide excision of the affected area and skin grafting. Immediately terminate the drug and apply ice to the affected area [see Warnings and Precautions (5.3)].
-
•Severe myelosuppression resulting in serious infection, septic shock, requirement for transfusions, hospitalization, and death may occur [see Warnings and Precautions (5.4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:38:13.568170 · Updated: 2026-03-14T22:17:00.667686