These Highlights Do Not Include All The Information Needed To Use Oseltamivir Phosphate For Oral Suspension Safely And Effectively. See Full Prescribing Information For Oseltamivir Phosphate For Oral Suspension.
005b2784-e410-a50c-e063-6394a90a881c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Oseltamivir phosphate for oral suspension is an influenza neuraminidase inhibitor (NAI) indicated for: Treatment of acute, uncomplicated influenza A and B in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours. ( 1.1 ) Prophylaxis of influenza A and B in patients 1 year and older. ( 1.2 ) Limitations of Use : Not a substitute for annual influenza vaccination. ( 1.3 ) Consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use. ( 1.3 ) Not recommended for patients with end-stage renal disease not undergoing dialysis. ( 1.3 )
Indications and Usage
Oseltamivir phosphate for oral suspension is an influenza neuraminidase inhibitor (NAI) indicated for: Treatment of acute, uncomplicated influenza A and B in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours. ( 1.1 ) Prophylaxis of influenza A and B in patients 1 year and older. ( 1.2 ) Limitations of Use : Not a substitute for annual influenza vaccination. ( 1.3 ) Consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use. ( 1.3 ) Not recommended for patients with end-stage renal disease not undergoing dialysis. ( 1.3 )
Dosage and Administration
Administer oseltamivir phosphate for oral suspension for the treatment of influenza in patients 2 weeks of age or older [see Dosage and Administration (2.2) ] or for prophylaxis of influenza in patients 1 year and older [see Dosage and Administration (2.3) ] using: Oseltamivir phosphate for oral suspension (supplied as a powder). This is the preferred formulation (6 mg per mL) for patients who cannot swallow capsules. Prior to use, the supplied oseltamivir phosphate for oral suspension powder must be constituted with water by the pharmacist to produce the oral suspension [see Dosage and Administration ( 2.5 )]. The oral suspension may be taken with or without food; however, tolerability may be enhanced if oseltamivir phosphate for oral suspension is taken with food. Adjust the oseltamivir phosphate for oral suspension dosage in patients with moderate or severe renal impairment [see Dosage and Administration (2.4) ].
Warnings and Precautions
Serious skin/hypersensitivity reactions such as Stevens-Johnson Syndrome, toxic epidermal necrolysis and erythema multiforme: Discontinue oseltamivir phosphate for oral suspension and initiate appropriate treatment if allergic-like reactions occur or are suspected. ( 5.1 ) Neuropsychiatric events: Patients with influenza, including those receiving oseltamivir phosphate for oral suspension, particularly pediatric patients, may be at an increased risk of confusion or abnormal behavior early in their illness. Monitor for signs of abnormal behavior. ( 5.2 )
Contraindications
Oseltamivir phosphate for oral suspension is contraindicated in patients with known serious hypersensitivity to oseltamivir or any component of the product. Severe allergic reactions have included anaphylaxis and serious skin reactions including toxic epidermal necrolysis, Stevens-Johnson Syndrome, and erythema multiforme [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following serious adverse reactions are discussed below and elsewhere in the labeling: Serious skin and hypersensitivity reactions [see Warnings and Precautions (5.1) ] Neuropsychiatric events [see Warnings and Precautions (5.2) ]
Drug Interactions
Live attenuated influenza vaccine (LAIV), intranasal: Avoid administration of LAIV within 2 weeks before or 48 hours after oseltamivir phosphate for oral suspension use, unless medically indicated. ( 7 )
Medication Information
Warnings and Precautions
Serious skin/hypersensitivity reactions such as Stevens-Johnson Syndrome, toxic epidermal necrolysis and erythema multiforme: Discontinue oseltamivir phosphate for oral suspension and initiate appropriate treatment if allergic-like reactions occur or are suspected. ( 5.1 ) Neuropsychiatric events: Patients with influenza, including those receiving oseltamivir phosphate for oral suspension, particularly pediatric patients, may be at an increased risk of confusion or abnormal behavior early in their illness. Monitor for signs of abnormal behavior. ( 5.2 )
Indications and Usage
Oseltamivir phosphate for oral suspension is an influenza neuraminidase inhibitor (NAI) indicated for: Treatment of acute, uncomplicated influenza A and B in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours. ( 1.1 ) Prophylaxis of influenza A and B in patients 1 year and older. ( 1.2 ) Limitations of Use : Not a substitute for annual influenza vaccination. ( 1.3 ) Consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use. ( 1.3 ) Not recommended for patients with end-stage renal disease not undergoing dialysis. ( 1.3 )
Dosage and Administration
Administer oseltamivir phosphate for oral suspension for the treatment of influenza in patients 2 weeks of age or older [see Dosage and Administration (2.2) ] or for prophylaxis of influenza in patients 1 year and older [see Dosage and Administration (2.3) ] using: Oseltamivir phosphate for oral suspension (supplied as a powder). This is the preferred formulation (6 mg per mL) for patients who cannot swallow capsules. Prior to use, the supplied oseltamivir phosphate for oral suspension powder must be constituted with water by the pharmacist to produce the oral suspension [see Dosage and Administration ( 2.5 )]. The oral suspension may be taken with or without food; however, tolerability may be enhanced if oseltamivir phosphate for oral suspension is taken with food. Adjust the oseltamivir phosphate for oral suspension dosage in patients with moderate or severe renal impairment [see Dosage and Administration (2.4) ].
Contraindications
Oseltamivir phosphate for oral suspension is contraindicated in patients with known serious hypersensitivity to oseltamivir or any component of the product. Severe allergic reactions have included anaphylaxis and serious skin reactions including toxic epidermal necrolysis, Stevens-Johnson Syndrome, and erythema multiforme [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following serious adverse reactions are discussed below and elsewhere in the labeling: Serious skin and hypersensitivity reactions [see Warnings and Precautions (5.1) ] Neuropsychiatric events [see Warnings and Precautions (5.2) ]
Drug Interactions
Live attenuated influenza vaccine (LAIV), intranasal: Avoid administration of LAIV within 2 weeks before or 48 hours after oseltamivir phosphate for oral suspension use, unless medically indicated. ( 7 )
Description
Oseltamivir phosphate for oral suspension is an influenza neuraminidase inhibitor (NAI) indicated for: Treatment of acute, uncomplicated influenza A and B in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours. ( 1.1 ) Prophylaxis of influenza A and B in patients 1 year and older. ( 1.2 ) Limitations of Use : Not a substitute for annual influenza vaccination. ( 1.3 ) Consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use. ( 1.3 ) Not recommended for patients with end-stage renal disease not undergoing dialysis. ( 1.3 )
Section 42229-5
Adults and Adolescents (13 years of age and older)
The recommended oral dosage of oseltamivir phosphate for oral suspension for treatment of influenza in adults and adolescents 13 years and older is 75 mg twice daily (12.5 mL of oral suspension twice daily) for 5 days.
Section 42230-3
| PATIENT INFORMATION | |
|---|---|
|
Oseltamivir Phosphate (oh'' sel tam' i vir fos' fate) for oral suspension |
|
|
What is oseltamivir phosphate for oral suspension? Oseltamivir phosphate for oral suspension is a prescription medicine used to:
It is not known if oseltamivir phosphate for oral suspension is:
Oseltamivir phosphate for oral suspension does not treat or prevent illness that is caused by infections other than the influenza virus. Oseltamivir phosphate for oral suspension does not prevent bacterial infections that may happen with the flu. Oseltamivir phosphate for oral suspension is not recommended for people with end-stage renal disease (ESRD) who are not receiving dialysis. Oseltamivir phosphate for oral suspension does not take the place of receiving a flu vaccination. Talk to your healthcare provider about when you should receive an annual flu vaccination. |
|
|
Who should not take oseltamivir phosphate for oral suspension? Do not take oseltamivir phosphate for oral suspension if you are allergic to oseltamivir phosphate or any of the ingredients in oseltamivir phosphate for oral suspension. See the end of this leaflet for a complete list of ingredients in oseltamivir phosphate for oral suspension. |
|
|
What should I tell my healthcare provider before taking oseltamivir phosphate for oral suspension? Before you take oseltamivir phosphate for oral suspension, tell your healthcare provider if you:
Tell your healthcare provider about all the medicines you take,including prescription or over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
|
How should I take oseltamivir phosphate for oral suspension?
If your healthcare provider or pharmacist has instructed you to take oseltamivir phosphate for oral suspension, read the detailed Instructions for Use at the end of this leaflet. Ask your pharmacist if you have any questions. |
|
|
What are the possible side effects of oseltamivir phosphate for oral suspension? Oseltamivir phosphate for oral suspension may cause serious side effects, including:
|
|
|
○ skin rash or hives ○ your skin blisters and peels ○ blisters or sores in your mouth ○ itching |
○ swelling of your face, eyes, lips, tongue, or throat ○ trouble breathing ○ chest pain or tightness |
The most common side effects of oseltamivir phosphate for oral suspension when used for treatment of the flu include nausea, vomiting, and headache. The most common side effect of oseltamivir phosphate for oral suspension when used for prevention of the flu include nausea, vomiting, headache, and pain. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of oseltamivir phosphate. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
How should I store oseltamivir phosphate for oral suspension?
|
|
|
General information about the safe and effective use of oseltamivir phosphate for oral suspension. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use oseltamivir phosphate for oral suspension for a condition for which it was not prescribed. Do not give oseltamivir phosphate for oral suspension to other people, even if they have the same symptoms you have. It may harm them. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about oseltamivir phosphate for oral suspension that is written for health professionals. For more information, contact Lannett Company, Inc. at 1-844-834-0530. |
|
|
What are the ingredients in oseltamivir phosphate for oral suspension? Active ingredient:oseltamivir phosphate USP Inactive ingredients:
Oseltamivir phosphate for oral suspension:mannitol, saccharin sodium, sodium benzoate, sodium dihydrogen citrate,
Manufactured by: Sunshine Lake Pharma Co., Ltd., No. 1, Northern Industry Road, Northern Industry Park of Song Shan Lake, Dongguan 523808, Guangdong, China Distributed by: Lannett Company, Inc.
|
|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised 5/2023 |
Section 44425-7
Storage
Store dry powder at 20º to 25ºC (68º to 77ºF); excursions permitted to 15º to 30ºC (59º to 86ºF) [See USP Controlled Room Temperature]. Preserved in well closed container.
Store constituted oral suspension under refrigeration for up to 17 days at 2º to 8ºC (36º to 46ºF). Do not freeze. Alternatively, store constituted oral suspension for up to 10 days at 20º to 25ºC (68º to 77ºF); excursions permitted to 15º to 30ºC (59º to 86ºF) [See USP Controlled Room Temperature]. Keep the bottle in the outer carton in order to protect from light.
Section 59845-8
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised 5/2023 | |
| INSTRUCTIONS FOR USE | ||
| Oseltamivir Phosphate(oh'' sel tam' i vir fos' fate) for oral suspension | ||
|
How do I give a dose of oseltamivir phosphate for oral suspension? You will need: Step 1. Shake the oseltamivir phosphate for oral suspension bottle well before each use. Step 2. Open the bottle by pushing downward on the child resistant bottle cap and twisting it in the direction of the arrow. Step 3. Measure the oral suspension with an appropriate oral dosing dispenser to be sure you get the correct dose. Contact your pharmacist if you do not have an appropriate oral dosing dispenser. Step 4. Give the full contents of oral dosing dispenser directly into the mouth. Step 5. Close the bottle with the child-resistant bottle cap and put the bottle back into the outer carton to protect from light after each use. Step 6. Rinse oral dosing dispenser under running tap water and allow to air dry after each use. Manufactured by: Sunshine Lake Pharma Co., Ltd. No. 1, Northern Industry Road, Northern Industry Park of Song Shan Lake, Dongguan, Guangdong 523808, China Distributed by: Lannett Company, Inc.
|
10 Overdosage
Reports of overdoses with oseltamivir phosphate have been received from clinical trials and during postmarketing experience. In the majority of cases reporting overdose, no adverse reactions were reported. Adverse reactions reported following overdose were similar in nature to those observed with therapeutic doses of oseltamivir phosphate for oral suspension [see Adverse Reactions (6)] .
11 Description
Oseltamivir phosphate, USP, an influenza neuraminidase inhibitor (NAI), is available as:
A powder for oral suspension, which when constituted with water as directed contains 6 mg per mL oseltamivir base.
In addition to the active ingredient, the powder for oral suspension contains mannitol, saccharin sodium, sodium benzoate, sodium dihydrogen citrate, sorbitol, sour cherry flavor, titanium dioxide, and xanthan gum.
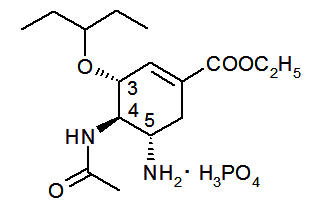
Oseltamivir phosphate, USP is a white crystalline solid with the chemical name (3R,4R,5S)‐4‐acetylamino‐5‐amino‐3(1‐ ethylpropoxy)‐1‐cyclohexene‐1‐carboxylic acid, ethyl ester, phosphate (1:1). The chemical formula is C 16H 28N 2O 4(free base). The molecular weight is 312.4 for oseltamivir free base and 410.4 for oseltamivir phosphate salt. The structural formula is as follows:
4 Contraindications
Oseltamivir phosphate for oral suspension is contraindicated in patients with known serious hypersensitivity to oseltamivir or any component of the product. Severe allergic reactions have included anaphylaxis and serious skin reactions including toxic epidermal necrolysis, Stevens-Johnson Syndrome, and erythema multiforme [see Warnings and Precautions (5.1)] .
6 Adverse Reactions
The following serious adverse reactions are discussed below and elsewhere in the labeling:
- Serious skin and hypersensitivity reactions [see Warnings and Precautions (5.1)]
- Neuropsychiatric events [see Warnings and Precautions (5.2)]
7 Drug Interactions
Live attenuated influenza vaccine (LAIV), intranasal:
Avoid administration of LAIV within 2 weeks before or 48 hours after oseltamivir phosphate for oral suspension use, unless medically indicated. ( 7)
8.6 Renal Impairment
Patients with renal impairment had higher blood levels of oseltamivir carboxylate compared to patients with normal renal function which may increase the risk of oseltamivir phosphate -associated adverse reactions. Therefore, dosage adjustment is recommended for patients with a serum creatinine clearance between 10 and 60 mL/minute and for patients with end-stage renal disease (ESRD) undergoing routine hemodialysis or continuous peritoneal dialysis treatment [see Dosage and Administration (2.4)]. Oseltamivir phosphate is not recommended for patients with ESRD not undergoing dialysis [see Indications and Usage (1.3)and Clinical Pharmacology (12.3)] .
1.3 Limitations of Use
- Oseltamivir phosphate for oral suspension is not a substitute for early influenza vaccination on an annual basis as recommended by the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices.
- Influenza viruses change over time. Emergence of resistance substitutions could decrease drug effectiveness. Other factors (for example, changes in viral virulence) might also diminish clinical benefit of antiviral drugs. Prescribers should consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use oseltamivir phosphate for oral suspension [see Microbiology (12.4)] .
- Oseltamivir phosphate for oral suspension is not recommended for patients with end-stage renal disease not undergoing dialysis [see Dosage and Administration (2.4)and Use in Specific Populations (8.6)].
8.7 Hepatic Impairment
No dosage adjustment is required in patients with mild to moderate hepatic impairment. The safety and pharmacokinetics in patients with severe hepatic impairment have not been evaluated [see Clinical Pharmacology (12.3)] .
1 Indications and Usage
Oseltamivir phosphate for oral suspension is an influenza neuraminidase inhibitor (NAI) indicated for:
- Treatment of acute, uncomplicated influenza A and B in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours. ( 1.1)
- Prophylaxis of influenza A and B in patients 1 year and older. ( 1.2)
Limitations of Use:
12.1 Mechanism of Action
Oseltamivir is an antiviral drug with activity against influenza virus [see Microbiology (12.4)].
1.1 Treatment of Influenza
Oseltamivir phosphate for oral suspension is indicated for the treatment of acute, uncomplicated illness due to influenza A and B infection in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours.
5 Warnings and Precautions
- Serious skin/hypersensitivity reactions such as Stevens-Johnson Syndrome, toxic epidermal necrolysis and erythema multiforme: Discontinue oseltamivir phosphate for oral suspension and initiate appropriate treatment if allergic-like reactions occur or are suspected. ( 5.1)
- Neuropsychiatric events: Patients with influenza, including those receiving oseltamivir phosphate for oral suspension, particularly pediatric patients, may be at an increased risk of confusion or abnormal behavior early in their illness. Monitor for signs of abnormal behavior. ( 5.2)
2 Dosage and Administration
Treatment of influenza
- Adults and adolescents (13 years and older): 75 mg twice daily for 5 days ( 2.2)
- Pediatric patients 1 to 12 years of age: Based on weight twice daily for 5 days ( 2.2)
- Pediatric patients 2 weeks to less than 1 year of age: 3mg/kg twice daily for 5 days ( 2.2)
- Renally impaired adult patients (creatinine clearance >30 to 60 mL/min): Reduce to 30 mg twice daily for 5 days ( 2.4)
- Renally impaired adult patients (creatinine clearance >10 to 30 mL/min): Reduce to 30 mg once daily for 5 days ( 2.4)
- ESRD patients on hemodialysis: Reduce to 30 mg immediately and then 30 mg after every hemodialysis cycle. Treatment duration not to exceed 5 days ( 2.4)
- ESRD patients on CAPD: Reduce to a single 30 mg dose immediately ( 2.4)
Prophylaxis of influenza
- Adults and adolescents (13 years and older): 75 mg once daily for at least 10 days (
2.3)
- Community outbreak: 75 mg once daily for up to 6 weeks ( 2.3)
- Pediatric patients 1 to 12 years of age: Based on weight once daily for 10 days (
2.3)
- Community outbreak: Based on weight once daily for up to 6 weeks ( 2.3)
- Renally impaired adult patients (creatinine clearance >30 to 60 mL/min): Reduce to 30 mg once daily ( 2.4)
- Renally impaired adult patients (creatinine clearance >10 to 30 mL/min): Reduce to 30 mg once every other day ( 2.4)
- ESRD patients on hemodialysis: Reduce to 30 mg immediately and then 30 mg after alternate hemodialysis cycles for the recommended duration of prophylaxis ( 2.4)
- ESRD patients on CAPD: Reduce to 30 mg immediately and then 30 mg once weekly for the recommended duration of prophylaxis ( 2.4)
5.2 Neuropsychiatric Events
There have been postmarketing reports of delirium and abnormal behavior leading to injury, and in some cases resulting in fatal outcomes, in patients with influenza who were receiving oseltamivir phosphate for oral suspension [see Adverse Reactions (6.2)] . Because these events were reported voluntarily during clinical practice, estimates of frequency cannot be made but they appear to be uncommon based on oseltamivir phosphate for oral suspension usage data. These events were reported primarily among pediatric patients and often had an abrupt onset and rapid resolution. The contribution of oseltamivir phosphate for oral suspension to these events has not been established. Influenza can be associated with a variety of neurologic and behavioral symptoms that can include events such as hallucinations, delirium, and abnormal behavior, in some cases resulting in fatal outcomes. These events may occur in the setting of encephalitis or encephalopathy but can occur without obvious severe disease. Closely monitor oseltamivir phosphate for oral suspension treated patients with influenza for signs of abnormal behavior. If neuropsychiatric symptoms occur, evaluate the risks and benefits of continuing oseltamivir phosphate for oral suspension for each patient.
1.2 Prophylaxis of Influenza
Oseltamivir phosphate for oral suspension is indicated for the prophylaxis of influenza A and B in patients 1 year and older.
3 Dosage Forms and Strengths
Oseltamivir Phosphate for Oral Suspension: 6 mg per mL (final concentration when constituted)
- White to light yellow powder blend for constitution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of oseltamivir phosphate. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to oseltamivir phosphate exposure.
General disorders and administration site conditions:Swelling of the face or tongue, allergy, anaphylactic/anaphylactoid reactions, hypothermia
Skin and subcutaneous tissue disorders:Rash, dermatitis, urticaria, eczema, toxic epidermal necrolysis, Stevens-Johnson Syndrome, erythema multiforme [see Warnings and Precautions (5.1)]
Gastrointestinal Disorders:Gastrointestinal bleeding, hemorrhagic colitis
Cardiac Disorders:Arrhythmia
Hepatobiliary Disorders:Hepatitis, abnormal liver function tests
Nervous System Disorders:Seizure
Metabolism and Nutrition Disorders:Aggravation of diabetes
Psychiatric Disorders:Abnormal behavior, delirium, including symptoms such as hallucinations, agitation, anxiety, altered level of consciousness, confusion, nightmares, delusions [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
8.9 Immunocompromised Patients
Efficacy of oseltamivir phosphate for the treatment or prophylaxis of influenza has not been established in immunocompromised patients [see Clinical Studies (14.2)] . Safety of oseltamivir phosphate has been demonstrated for up to 12 weeks for prophylaxis of influenza in immunocompromised patients [see Adverse Reactions (6.1)].
5.3 Risk of Bacterial Infections
There is no evidence for efficacy of oseltamivir phosphate for oral suspension in any illness caused by pathogens other than influenza viruses. Serious bacterial infections may begin with influenza-like symptoms or may coexist with or occur as complications during the course of influenza. Oseltamivir phosphate for oral suspension has not been shown to prevent such complications. Prescribers should be alert to the potential for secondary bacterial infections and treat them as appropriate.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
2.1 Dosage and Administration Overview
Administer oseltamivir phosphate for oral suspension for the treatment of influenza in patients 2 weeks of age or older [see Dosage and Administration (2.2)] or for prophylaxis of influenza in patients 1 year and older [see Dosage and Administration (2.3)] using:
- Oseltamivir phosphate for oral suspension (supplied as a powder). This is the preferred formulation (6 mg per mL) for
patients who cannot swallow capsules. Prior to use, the supplied oseltamivir phosphate for oral suspension powder must
be constituted with water by the pharmacist to produce the oral suspension [see Dosage and Administration ( 2.5)].
The oral suspension may be taken with or without food; however, tolerability may be enhanced if oseltamivir phosphate for oral suspension is taken with food.
Adjust the oseltamivir phosphate for oral suspension dosage in patients with moderate or severe renal impairment [see Dosage and Administration (2.4)].
5.1 Serious Skin/hypersensitivity Reactions
Cases of anaphylaxis and serious skin reactions including toxic epidermal necrolysis, Stevens-Johnson Syndrome, and erythema multiforme have been reported in postmarketing experience with oseltamivir phosphate for oral suspension. Stop oseltamivir phosphate for oral suspension and institute appropriate treatment if an allergic-like reaction occurs or is suspected. The use of oseltamivir phosphate for oral suspension is contraindicated in patients with known serious hypersensitivity to oseltamivir phosphate for oral suspension [see Contraindications (4)and Adverse Reactions (6.2)] .
8.8 Use in Patients With Chronic Conditions
Efficacy of oseltamivir phosphate in the treatment of influenza in patients with chronic cardiac disease and/or respiratory disease was evaluated in one randomized, placebo-controlled clinical trial. Efficacy in this population, as measured by time to alleviation of all symptoms, was not established, but no new safety signals were identified [ see Clinical Studies (14.1) ].
No clinical trial data are available regarding treatment of influenza in patients with any medical condition sufficiently severe or unstable to be considered at imminent risk of requiring hospitalization.
2.4 Dosage in Patients With Renal Impairment
Table 2displays the dosage recommendations for the treatment and prophylaxis of influenza in adults with various stages of renal impairment (estimated creatinine clearance of less than or equal to 90 mL per minute). Dosage modifications are recommended in adults with an estimated creatinine clearance less than or equal to 60 mL per minute [see Use in Specific Population (8.6)and Clinical Pharmacology (12.3)] .
| Renal Impairment
(Creatinine Clearance) |
Recommended Treatment Regimen
Oral suspension can be used for 30 mg dosing.
|
Recommended Prophylaxis Regimen
The recommended duration for post-exposure prophylaxis is at least 10 days and the recommended duration for community outbreak (seasonal/pre-exposure) prophylaxis is up to 6 weeks (or up to 12 weeks in immunocompromised patients).
|
|---|---|---|
| Mild
(>60 to 90 mL/minute) |
75 mg twice daily for 5 days | 75 mg once daily |
| Moderate
(>30 to 60 mL/minute) |
30 mg twice daily for 5 days | 30 mg once daily |
| Severe
(>10 to 30 mL/minute) |
30 mg once daily for 5 days | 30 mg every other day |
| ESRD Patients on Hemodialysis
(≤ 10 mL/minute) |
30 mg immediately and then 30 mg after every hemodialysis cycle
(treatment duration not to exceed 5 days) |
30 mg immediately and then 30 mg after alternate hemodialysis cycles |
| ESRD Patients on Continuous Ambulatory Peritoneal Dialysis
Data derived from studies in continuous ambulatory peritoneal dialysis (CAPD) patients.
(≤10 mL/minute) |
A single 30 mg dose administered immediately | 30 mg immediately and then 30 mg once weekly |
| ESRD Patients not on Dialysis | Oseltamivir phosphate for oral suspension is not recommended | Oseltamivir phosphate for oral suspension is are not recommended |
2.2 Recommended Dosage for Treatment of Influenza
Initiate treatment with oseltamivir phosphate for oral suspension within 48 hours of influenza symptom onset.
2.3 Recommended Dosage for Prophylaxis of Influenza
Initiate post-exposure prophylaxis with oseltamivir phosphate for oral suspension within 48 hours following close contact with an infected individual. Initiate seasonal prophylaxis with oseltamivir phosphate for oral suspension during a community outbreak.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In 2-year carcinogenicity studies in mice and rats given daily oral doses of the prodrug oseltamivir phosphate up to 400 mg/kg and 500 mg/kg, respectively, the prodrug and the active form oseltamivir carboxylate induced no statistically significant increases in tumors over controls. The mean maximum daily exposures to the prodrug in mice and rats were approximately 130- and 320-fold, respectively, greater than those in humans at the recommended clinical dose based on AUC comparisons. The respective safety margins of the exposures to the active oseltamivir carboxylate were 15- and 50-fold.
Oseltamivir was found to be non-mutagenic in the Ames test and the human lymphocyte chromosome assay with and without enzymatic activation and negative in the mouse micronucleus test. It was found to be positive in a Syrian Hamster Embryo (SHE) cell transformation test. Oseltamivir carboxylate was non-mutagenic in the Ames test and the L5178Y mouse lymphoma assay with and without enzymatic activation and negative in the SHE cell transformation test.
In a fertility and early embryonic development study in rats, doses of oseltamivir at 50, 250, and 1500 mg/kg/day were administered to females for 2 weeks before mating, during mating and until day 6 of pregnancy. Males were dosed for 4 weeks before mating, during mating, and for 2 weeks after mating. There were no effects on fertility, mating performance or early embryonic development at any dose level. The highest dose in this study was approximately 115 times the human systemic exposure (AUC 0-24h) of oseltamivir carboxylate that occurs after administration of the maximum recommended human dose.
Principal Display Panel 60 Ml Bottle Carton Ndc 0527 5137 62
NDC 0527-5137-62
Oseltamivir Phosphate for Oral Suspension
6 mg/mL
Each mL contains 6 mg oseltamivir base after constitution.
60 mL (usable volume after constitution)
Rx only
Sunshine Lake Pharma Co., Ltd.
5.4 Fructose Intolerance in Patients With Hereditary Fructose Intolerance
Fructose can be harmful to patients with hereditary fructose intolerance. One dose of 75 mg oseltamivir phosphate for oral suspension delivers 2 grams of sorbitol. This is above the daily maximum limit of sorbitol for patients with hereditary fructose intolerance and may cause dyspepsia and diarrhea.
2.5 Preparation and Storage of Constituted Oseltamivir Phosphate for Oral Suspension
Prior to dispensing to the patient, constitute oseltamivir phosphate for oral suspension (supplied as powder):
a) Tap the closed bottle containing the supplied oseltamivir phosphate for oral suspension white to light yellow powder several times to loosen the powder.
b) Tear off the foil seal after loosening the powder.
c) Measure 55 mL of water in a graduated cylinder.
d) Add the total amount of water for constitution to the bottle.
e) Close bottle with child‐resistant cap tightly and shake the closed bottle well for 15 seconds.
f) Label the bottle with instructions to “Shake Well Before Use”.
g) The constituted oral suspension contains 360 mg of oseltamivir base per 60 mL of volume (6 mg per mL) and is white to light yellow, sour cherry-flavored). Use the constituted oral suspension within 17 days of preparation when stored under refrigeration, 2º to 8ºC (36º to 46ºF), or within 10 days if stored at controlled room temperature, 20º to 25ºC (68º to 77ºF). Keep the bottle in the outer carton in order to protect from light. Write the expiration date of the constituted oral suspension on the bottle label.
h) Ensure patients have an oral dosing dispenser that measures the appropriate volume in milliliters. Counsel patients on how to utilize the oral dosing dispenser and correctly measure the oral suspension as prescribed (see Tables 1 and 2).
7.2 Drugs Without Clinically Significant Drug Interaction With Oseltamivir Phosphate for Oral Suspension
No dose adjustments are needed for either oseltamivir or the concomitant drug when coadministering oseltamivir with amoxicillin, acetaminophen, aspirin, cimetidine, antacids (magnesium and aluminum hydroxides and calcium carbonates), rimantadine, amantadine, or warfarin [see Clinical Pharmacology (12.3)] .
Structured Label Content
Section 42229-5 (42229-5)
Adults and Adolescents (13 years of age and older)
The recommended oral dosage of oseltamivir phosphate for oral suspension for treatment of influenza in adults and adolescents 13 years and older is 75 mg twice daily (12.5 mL of oral suspension twice daily) for 5 days.
Section 42230-3 (42230-3)
| PATIENT INFORMATION | |
|---|---|
|
Oseltamivir Phosphate (oh'' sel tam' i vir fos' fate) for oral suspension |
|
|
What is oseltamivir phosphate for oral suspension? Oseltamivir phosphate for oral suspension is a prescription medicine used to:
It is not known if oseltamivir phosphate for oral suspension is:
Oseltamivir phosphate for oral suspension does not treat or prevent illness that is caused by infections other than the influenza virus. Oseltamivir phosphate for oral suspension does not prevent bacterial infections that may happen with the flu. Oseltamivir phosphate for oral suspension is not recommended for people with end-stage renal disease (ESRD) who are not receiving dialysis. Oseltamivir phosphate for oral suspension does not take the place of receiving a flu vaccination. Talk to your healthcare provider about when you should receive an annual flu vaccination. |
|
|
Who should not take oseltamivir phosphate for oral suspension? Do not take oseltamivir phosphate for oral suspension if you are allergic to oseltamivir phosphate or any of the ingredients in oseltamivir phosphate for oral suspension. See the end of this leaflet for a complete list of ingredients in oseltamivir phosphate for oral suspension. |
|
|
What should I tell my healthcare provider before taking oseltamivir phosphate for oral suspension? Before you take oseltamivir phosphate for oral suspension, tell your healthcare provider if you:
Tell your healthcare provider about all the medicines you take,including prescription or over-the-counter medicines, vitamins, and herbal supplements. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|
|
How should I take oseltamivir phosphate for oral suspension?
If your healthcare provider or pharmacist has instructed you to take oseltamivir phosphate for oral suspension, read the detailed Instructions for Use at the end of this leaflet. Ask your pharmacist if you have any questions. |
|
|
What are the possible side effects of oseltamivir phosphate for oral suspension? Oseltamivir phosphate for oral suspension may cause serious side effects, including:
|
|
|
○ skin rash or hives ○ your skin blisters and peels ○ blisters or sores in your mouth ○ itching |
○ swelling of your face, eyes, lips, tongue, or throat ○ trouble breathing ○ chest pain or tightness |
The most common side effects of oseltamivir phosphate for oral suspension when used for treatment of the flu include nausea, vomiting, and headache. The most common side effect of oseltamivir phosphate for oral suspension when used for prevention of the flu include nausea, vomiting, headache, and pain. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of oseltamivir phosphate. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
|
How should I store oseltamivir phosphate for oral suspension?
|
|
|
General information about the safe and effective use of oseltamivir phosphate for oral suspension. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use oseltamivir phosphate for oral suspension for a condition for which it was not prescribed. Do not give oseltamivir phosphate for oral suspension to other people, even if they have the same symptoms you have. It may harm them. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about oseltamivir phosphate for oral suspension that is written for health professionals. For more information, contact Lannett Company, Inc. at 1-844-834-0530. |
|
|
What are the ingredients in oseltamivir phosphate for oral suspension? Active ingredient:oseltamivir phosphate USP Inactive ingredients:
Oseltamivir phosphate for oral suspension:mannitol, saccharin sodium, sodium benzoate, sodium dihydrogen citrate,
Manufactured by: Sunshine Lake Pharma Co., Ltd., No. 1, Northern Industry Road, Northern Industry Park of Song Shan Lake, Dongguan 523808, Guangdong, China Distributed by: Lannett Company, Inc.
|
|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised 5/2023 |
Section 44425-7 (44425-7)
Storage
Store dry powder at 20º to 25ºC (68º to 77ºF); excursions permitted to 15º to 30ºC (59º to 86ºF) [See USP Controlled Room Temperature]. Preserved in well closed container.
Store constituted oral suspension under refrigeration for up to 17 days at 2º to 8ºC (36º to 46ºF). Do not freeze. Alternatively, store constituted oral suspension for up to 10 days at 20º to 25ºC (68º to 77ºF); excursions permitted to 15º to 30ºC (59º to 86ºF) [See USP Controlled Room Temperature]. Keep the bottle in the outer carton in order to protect from light.
Section 59845-8 (59845-8)
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised 5/2023 | |
| INSTRUCTIONS FOR USE | ||
| Oseltamivir Phosphate(oh'' sel tam' i vir fos' fate) for oral suspension | ||
|
How do I give a dose of oseltamivir phosphate for oral suspension? You will need: Step 1. Shake the oseltamivir phosphate for oral suspension bottle well before each use. Step 2. Open the bottle by pushing downward on the child resistant bottle cap and twisting it in the direction of the arrow. Step 3. Measure the oral suspension with an appropriate oral dosing dispenser to be sure you get the correct dose. Contact your pharmacist if you do not have an appropriate oral dosing dispenser. Step 4. Give the full contents of oral dosing dispenser directly into the mouth. Step 5. Close the bottle with the child-resistant bottle cap and put the bottle back into the outer carton to protect from light after each use. Step 6. Rinse oral dosing dispenser under running tap water and allow to air dry after each use. Manufactured by: Sunshine Lake Pharma Co., Ltd. No. 1, Northern Industry Road, Northern Industry Park of Song Shan Lake, Dongguan, Guangdong 523808, China Distributed by: Lannett Company, Inc.
|
10 Overdosage (10 OVERDOSAGE)
Reports of overdoses with oseltamivir phosphate have been received from clinical trials and during postmarketing experience. In the majority of cases reporting overdose, no adverse reactions were reported. Adverse reactions reported following overdose were similar in nature to those observed with therapeutic doses of oseltamivir phosphate for oral suspension [see Adverse Reactions (6)] .
11 Description (11 DESCRIPTION)
Oseltamivir phosphate, USP, an influenza neuraminidase inhibitor (NAI), is available as:
A powder for oral suspension, which when constituted with water as directed contains 6 mg per mL oseltamivir base.
In addition to the active ingredient, the powder for oral suspension contains mannitol, saccharin sodium, sodium benzoate, sodium dihydrogen citrate, sorbitol, sour cherry flavor, titanium dioxide, and xanthan gum.
Oseltamivir phosphate, USP is a white crystalline solid with the chemical name (3R,4R,5S)‐4‐acetylamino‐5‐amino‐3(1‐ ethylpropoxy)‐1‐cyclohexene‐1‐carboxylic acid, ethyl ester, phosphate (1:1). The chemical formula is C 16H 28N 2O 4(free base). The molecular weight is 312.4 for oseltamivir free base and 410.4 for oseltamivir phosphate salt. The structural formula is as follows:
4 Contraindications (4 CONTRAINDICATIONS)
Oseltamivir phosphate for oral suspension is contraindicated in patients with known serious hypersensitivity to oseltamivir or any component of the product. Severe allergic reactions have included anaphylaxis and serious skin reactions including toxic epidermal necrolysis, Stevens-Johnson Syndrome, and erythema multiforme [see Warnings and Precautions (5.1)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed below and elsewhere in the labeling:
- Serious skin and hypersensitivity reactions [see Warnings and Precautions (5.1)]
- Neuropsychiatric events [see Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Live attenuated influenza vaccine (LAIV), intranasal:
Avoid administration of LAIV within 2 weeks before or 48 hours after oseltamivir phosphate for oral suspension use, unless medically indicated. ( 7)
8.6 Renal Impairment
Patients with renal impairment had higher blood levels of oseltamivir carboxylate compared to patients with normal renal function which may increase the risk of oseltamivir phosphate -associated adverse reactions. Therefore, dosage adjustment is recommended for patients with a serum creatinine clearance between 10 and 60 mL/minute and for patients with end-stage renal disease (ESRD) undergoing routine hemodialysis or continuous peritoneal dialysis treatment [see Dosage and Administration (2.4)]. Oseltamivir phosphate is not recommended for patients with ESRD not undergoing dialysis [see Indications and Usage (1.3)and Clinical Pharmacology (12.3)] .
1.3 Limitations of Use
- Oseltamivir phosphate for oral suspension is not a substitute for early influenza vaccination on an annual basis as recommended by the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices.
- Influenza viruses change over time. Emergence of resistance substitutions could decrease drug effectiveness. Other factors (for example, changes in viral virulence) might also diminish clinical benefit of antiviral drugs. Prescribers should consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use oseltamivir phosphate for oral suspension [see Microbiology (12.4)] .
- Oseltamivir phosphate for oral suspension is not recommended for patients with end-stage renal disease not undergoing dialysis [see Dosage and Administration (2.4)and Use in Specific Populations (8.6)].
8.7 Hepatic Impairment
No dosage adjustment is required in patients with mild to moderate hepatic impairment. The safety and pharmacokinetics in patients with severe hepatic impairment have not been evaluated [see Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
Oseltamivir phosphate for oral suspension is an influenza neuraminidase inhibitor (NAI) indicated for:
- Treatment of acute, uncomplicated influenza A and B in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours. ( 1.1)
- Prophylaxis of influenza A and B in patients 1 year and older. ( 1.2)
Limitations of Use:
12.1 Mechanism of Action
Oseltamivir is an antiviral drug with activity against influenza virus [see Microbiology (12.4)].
1.1 Treatment of Influenza
Oseltamivir phosphate for oral suspension is indicated for the treatment of acute, uncomplicated illness due to influenza A and B infection in patients 2 weeks of age and older who have been symptomatic for no more than 48 hours.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serious skin/hypersensitivity reactions such as Stevens-Johnson Syndrome, toxic epidermal necrolysis and erythema multiforme: Discontinue oseltamivir phosphate for oral suspension and initiate appropriate treatment if allergic-like reactions occur or are suspected. ( 5.1)
- Neuropsychiatric events: Patients with influenza, including those receiving oseltamivir phosphate for oral suspension, particularly pediatric patients, may be at an increased risk of confusion or abnormal behavior early in their illness. Monitor for signs of abnormal behavior. ( 5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Treatment of influenza
- Adults and adolescents (13 years and older): 75 mg twice daily for 5 days ( 2.2)
- Pediatric patients 1 to 12 years of age: Based on weight twice daily for 5 days ( 2.2)
- Pediatric patients 2 weeks to less than 1 year of age: 3mg/kg twice daily for 5 days ( 2.2)
- Renally impaired adult patients (creatinine clearance >30 to 60 mL/min): Reduce to 30 mg twice daily for 5 days ( 2.4)
- Renally impaired adult patients (creatinine clearance >10 to 30 mL/min): Reduce to 30 mg once daily for 5 days ( 2.4)
- ESRD patients on hemodialysis: Reduce to 30 mg immediately and then 30 mg after every hemodialysis cycle. Treatment duration not to exceed 5 days ( 2.4)
- ESRD patients on CAPD: Reduce to a single 30 mg dose immediately ( 2.4)
Prophylaxis of influenza
- Adults and adolescents (13 years and older): 75 mg once daily for at least 10 days (
2.3)
- Community outbreak: 75 mg once daily for up to 6 weeks ( 2.3)
- Pediatric patients 1 to 12 years of age: Based on weight once daily for 10 days (
2.3)
- Community outbreak: Based on weight once daily for up to 6 weeks ( 2.3)
- Renally impaired adult patients (creatinine clearance >30 to 60 mL/min): Reduce to 30 mg once daily ( 2.4)
- Renally impaired adult patients (creatinine clearance >10 to 30 mL/min): Reduce to 30 mg once every other day ( 2.4)
- ESRD patients on hemodialysis: Reduce to 30 mg immediately and then 30 mg after alternate hemodialysis cycles for the recommended duration of prophylaxis ( 2.4)
- ESRD patients on CAPD: Reduce to 30 mg immediately and then 30 mg once weekly for the recommended duration of prophylaxis ( 2.4)
5.2 Neuropsychiatric Events
There have been postmarketing reports of delirium and abnormal behavior leading to injury, and in some cases resulting in fatal outcomes, in patients with influenza who were receiving oseltamivir phosphate for oral suspension [see Adverse Reactions (6.2)] . Because these events were reported voluntarily during clinical practice, estimates of frequency cannot be made but they appear to be uncommon based on oseltamivir phosphate for oral suspension usage data. These events were reported primarily among pediatric patients and often had an abrupt onset and rapid resolution. The contribution of oseltamivir phosphate for oral suspension to these events has not been established. Influenza can be associated with a variety of neurologic and behavioral symptoms that can include events such as hallucinations, delirium, and abnormal behavior, in some cases resulting in fatal outcomes. These events may occur in the setting of encephalitis or encephalopathy but can occur without obvious severe disease. Closely monitor oseltamivir phosphate for oral suspension treated patients with influenza for signs of abnormal behavior. If neuropsychiatric symptoms occur, evaluate the risks and benefits of continuing oseltamivir phosphate for oral suspension for each patient.
1.2 Prophylaxis of Influenza
Oseltamivir phosphate for oral suspension is indicated for the prophylaxis of influenza A and B in patients 1 year and older.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Oseltamivir Phosphate for Oral Suspension: 6 mg per mL (final concentration when constituted)
- White to light yellow powder blend for constitution.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of oseltamivir phosphate. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to oseltamivir phosphate exposure.
General disorders and administration site conditions:Swelling of the face or tongue, allergy, anaphylactic/anaphylactoid reactions, hypothermia
Skin and subcutaneous tissue disorders:Rash, dermatitis, urticaria, eczema, toxic epidermal necrolysis, Stevens-Johnson Syndrome, erythema multiforme [see Warnings and Precautions (5.1)]
Gastrointestinal Disorders:Gastrointestinal bleeding, hemorrhagic colitis
Cardiac Disorders:Arrhythmia
Hepatobiliary Disorders:Hepatitis, abnormal liver function tests
Nervous System Disorders:Seizure
Metabolism and Nutrition Disorders:Aggravation of diabetes
Psychiatric Disorders:Abnormal behavior, delirium, including symptoms such as hallucinations, agitation, anxiety, altered level of consciousness, confusion, nightmares, delusions [see Warnings and Precautions (5.2)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
8.9 Immunocompromised Patients
Efficacy of oseltamivir phosphate for the treatment or prophylaxis of influenza has not been established in immunocompromised patients [see Clinical Studies (14.2)] . Safety of oseltamivir phosphate has been demonstrated for up to 12 weeks for prophylaxis of influenza in immunocompromised patients [see Adverse Reactions (6.1)].
5.3 Risk of Bacterial Infections
There is no evidence for efficacy of oseltamivir phosphate for oral suspension in any illness caused by pathogens other than influenza viruses. Serious bacterial infections may begin with influenza-like symptoms or may coexist with or occur as complications during the course of influenza. Oseltamivir phosphate for oral suspension has not been shown to prevent such complications. Prescribers should be alert to the potential for secondary bacterial infections and treat them as appropriate.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
2.1 Dosage and Administration Overview
Administer oseltamivir phosphate for oral suspension for the treatment of influenza in patients 2 weeks of age or older [see Dosage and Administration (2.2)] or for prophylaxis of influenza in patients 1 year and older [see Dosage and Administration (2.3)] using:
- Oseltamivir phosphate for oral suspension (supplied as a powder). This is the preferred formulation (6 mg per mL) for
patients who cannot swallow capsules. Prior to use, the supplied oseltamivir phosphate for oral suspension powder must
be constituted with water by the pharmacist to produce the oral suspension [see Dosage and Administration ( 2.5)].
The oral suspension may be taken with or without food; however, tolerability may be enhanced if oseltamivir phosphate for oral suspension is taken with food.
Adjust the oseltamivir phosphate for oral suspension dosage in patients with moderate or severe renal impairment [see Dosage and Administration (2.4)].
5.1 Serious Skin/hypersensitivity Reactions (5.1 Serious Skin/Hypersensitivity Reactions)
Cases of anaphylaxis and serious skin reactions including toxic epidermal necrolysis, Stevens-Johnson Syndrome, and erythema multiforme have been reported in postmarketing experience with oseltamivir phosphate for oral suspension. Stop oseltamivir phosphate for oral suspension and institute appropriate treatment if an allergic-like reaction occurs or is suspected. The use of oseltamivir phosphate for oral suspension is contraindicated in patients with known serious hypersensitivity to oseltamivir phosphate for oral suspension [see Contraindications (4)and Adverse Reactions (6.2)] .
8.8 Use in Patients With Chronic Conditions (8.8 Use in Patients with Chronic Conditions)
Efficacy of oseltamivir phosphate in the treatment of influenza in patients with chronic cardiac disease and/or respiratory disease was evaluated in one randomized, placebo-controlled clinical trial. Efficacy in this population, as measured by time to alleviation of all symptoms, was not established, but no new safety signals were identified [ see Clinical Studies (14.1) ].
No clinical trial data are available regarding treatment of influenza in patients with any medical condition sufficiently severe or unstable to be considered at imminent risk of requiring hospitalization.
2.4 Dosage in Patients With Renal Impairment (2.4 Dosage in Patients with Renal Impairment)
Table 2displays the dosage recommendations for the treatment and prophylaxis of influenza in adults with various stages of renal impairment (estimated creatinine clearance of less than or equal to 90 mL per minute). Dosage modifications are recommended in adults with an estimated creatinine clearance less than or equal to 60 mL per minute [see Use in Specific Population (8.6)and Clinical Pharmacology (12.3)] .
| Renal Impairment
(Creatinine Clearance) |
Recommended Treatment Regimen
Oral suspension can be used for 30 mg dosing.
|
Recommended Prophylaxis Regimen
The recommended duration for post-exposure prophylaxis is at least 10 days and the recommended duration for community outbreak (seasonal/pre-exposure) prophylaxis is up to 6 weeks (or up to 12 weeks in immunocompromised patients).
|
|---|---|---|
| Mild
(>60 to 90 mL/minute) |
75 mg twice daily for 5 days | 75 mg once daily |
| Moderate
(>30 to 60 mL/minute) |
30 mg twice daily for 5 days | 30 mg once daily |
| Severe
(>10 to 30 mL/minute) |
30 mg once daily for 5 days | 30 mg every other day |
| ESRD Patients on Hemodialysis
(≤ 10 mL/minute) |
30 mg immediately and then 30 mg after every hemodialysis cycle
(treatment duration not to exceed 5 days) |
30 mg immediately and then 30 mg after alternate hemodialysis cycles |
| ESRD Patients on Continuous Ambulatory Peritoneal Dialysis
Data derived from studies in continuous ambulatory peritoneal dialysis (CAPD) patients.
(≤10 mL/minute) |
A single 30 mg dose administered immediately | 30 mg immediately and then 30 mg once weekly |
| ESRD Patients not on Dialysis | Oseltamivir phosphate for oral suspension is not recommended | Oseltamivir phosphate for oral suspension is are not recommended |
2.2 Recommended Dosage for Treatment of Influenza
Initiate treatment with oseltamivir phosphate for oral suspension within 48 hours of influenza symptom onset.
2.3 Recommended Dosage for Prophylaxis of Influenza
Initiate post-exposure prophylaxis with oseltamivir phosphate for oral suspension within 48 hours following close contact with an infected individual. Initiate seasonal prophylaxis with oseltamivir phosphate for oral suspension during a community outbreak.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In 2-year carcinogenicity studies in mice and rats given daily oral doses of the prodrug oseltamivir phosphate up to 400 mg/kg and 500 mg/kg, respectively, the prodrug and the active form oseltamivir carboxylate induced no statistically significant increases in tumors over controls. The mean maximum daily exposures to the prodrug in mice and rats were approximately 130- and 320-fold, respectively, greater than those in humans at the recommended clinical dose based on AUC comparisons. The respective safety margins of the exposures to the active oseltamivir carboxylate were 15- and 50-fold.
Oseltamivir was found to be non-mutagenic in the Ames test and the human lymphocyte chromosome assay with and without enzymatic activation and negative in the mouse micronucleus test. It was found to be positive in a Syrian Hamster Embryo (SHE) cell transformation test. Oseltamivir carboxylate was non-mutagenic in the Ames test and the L5178Y mouse lymphoma assay with and without enzymatic activation and negative in the SHE cell transformation test.
In a fertility and early embryonic development study in rats, doses of oseltamivir at 50, 250, and 1500 mg/kg/day were administered to females for 2 weeks before mating, during mating and until day 6 of pregnancy. Males were dosed for 4 weeks before mating, during mating, and for 2 weeks after mating. There were no effects on fertility, mating performance or early embryonic development at any dose level. The highest dose in this study was approximately 115 times the human systemic exposure (AUC 0-24h) of oseltamivir carboxylate that occurs after administration of the maximum recommended human dose.
Principal Display Panel 60 Ml Bottle Carton Ndc 0527 5137 62 (PRINCIPAL DISPLAY PANEL - 60 mL Bottle Carton - NDC 0527-5137-62)
NDC 0527-5137-62
Oseltamivir Phosphate for Oral Suspension
6 mg/mL
Each mL contains 6 mg oseltamivir base after constitution.
60 mL (usable volume after constitution)
Rx only
Sunshine Lake Pharma Co., Ltd.
5.4 Fructose Intolerance in Patients With Hereditary Fructose Intolerance (5.4 Fructose Intolerance in Patients with Hereditary Fructose Intolerance)
Fructose can be harmful to patients with hereditary fructose intolerance. One dose of 75 mg oseltamivir phosphate for oral suspension delivers 2 grams of sorbitol. This is above the daily maximum limit of sorbitol for patients with hereditary fructose intolerance and may cause dyspepsia and diarrhea.
2.5 Preparation and Storage of Constituted Oseltamivir Phosphate for Oral Suspension
Prior to dispensing to the patient, constitute oseltamivir phosphate for oral suspension (supplied as powder):
a) Tap the closed bottle containing the supplied oseltamivir phosphate for oral suspension white to light yellow powder several times to loosen the powder.
b) Tear off the foil seal after loosening the powder.
c) Measure 55 mL of water in a graduated cylinder.
d) Add the total amount of water for constitution to the bottle.
e) Close bottle with child‐resistant cap tightly and shake the closed bottle well for 15 seconds.
f) Label the bottle with instructions to “Shake Well Before Use”.
g) The constituted oral suspension contains 360 mg of oseltamivir base per 60 mL of volume (6 mg per mL) and is white to light yellow, sour cherry-flavored). Use the constituted oral suspension within 17 days of preparation when stored under refrigeration, 2º to 8ºC (36º to 46ºF), or within 10 days if stored at controlled room temperature, 20º to 25ºC (68º to 77ºF). Keep the bottle in the outer carton in order to protect from light. Write the expiration date of the constituted oral suspension on the bottle label.
h) Ensure patients have an oral dosing dispenser that measures the appropriate volume in milliliters. Counsel patients on how to utilize the oral dosing dispenser and correctly measure the oral suspension as prescribed (see Tables 1 and 2).
7.2 Drugs Without Clinically Significant Drug Interaction With Oseltamivir Phosphate for Oral Suspension (7.2 Drugs Without Clinically Significant Drug Interaction with Oseltamivir Phosphate for Oral Suspension)
No dose adjustments are needed for either oseltamivir or the concomitant drug when coadministering oseltamivir with amoxicillin, acetaminophen, aspirin, cimetidine, antacids (magnesium and aluminum hydroxides and calcium carbonates), rimantadine, amantadine, or warfarin [see Clinical Pharmacology (12.3)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:12.963750 · Updated: 2026-03-14T22:05:10.914388